1 Name of Medicine
Burosumab.
2 Qualitative and Quantitative Composition
Crysvita 10 mg/mL solution for injection contains 10 mg burosumab in a single use vial.
Crysvita 20 mg/mL solution for injection contains 20 mg burosumab in a single use vial.
Crysvita 30 mg/mL solution for injection contains 30 mg burosumab in a single use vial.
Burosumab is a recombinant human monoclonal IgG1 antibody for FGF23 and is produced by recombinant DNA technology using Chinese hamster ovary mammalian cell culture.
Excipient with known effect.
Each vial contains 45.91 mg sorbitol.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Solution for injection.
Sterile, single-use, preservative-free, clear to slightly opalescent, colourless to pale brownish-yellowish solution.
4.1 Therapeutic Indications
Crysvita (burosumab) is indicated for the treatment of X-linked hypophosphataemia (XLH) in adults, adolescents and children 1 year of age or older.
Crysvita (burosumab) is indicated for the treatment of FGF23-related hypophosphataemia in tumour-induced osteomalacia associated with phosphaturic mesenchymal tumours that cannot be curatively resected or localised in adults, adolescents and children 1 year of age or older.
4.2 Dose and Method of Administration
Treatment should be initiated and monitored by specialist medical practitioners experienced in the management of patients with metabolic bone disease.
Important dosage and administration information.
Discontinue oral phosphate and active vitamin D analogues (e.g. calcitriol, paricalcitol, doxercalciferol, calcifediol) 1 week prior to initiation of treatment (see Section 4.3 Contraindications). As burosumab however increases active vitamin D synthesis (see Section 5.1), patients' requirements for vitamin D replacement or supplementation should be assessed. Vitamin D replacement or supplementation with inactive forms of vitamin D may be continued as per local guidelines (see 25-Hydroxy vitamin D supplementation).
Fasting serum phosphate level should be below the reference range for age prior to initiation of treatment (see Section 4.3 Contraindications).
Burosumab is administered by subcutaneous injection and should be administered by a healthcare provider.
The maximum volume of burosumab per injection site is 1.5 mL. If multiple injections are required, administer at different injection sites.
X-linked hypophosphataemia.
Paediatric patients with X-linked hypophosphataemia (children aged 1-11 years and adolescents aged 12-17 years of age). The recommended starting dose regimen is 0.8 mg/kg of body weight, rounded to the nearest 10 mg, every 2 weeks. The minimum starting dose is 10 mg up to a maximum dose of 90 mg. All doses should be rounded to the nearest 10 mg.
After initiation of treatment with burosumab, fasting serum phosphate should be measured every 4 weeks for the first 3 months of treatment, and thereafter as appropriate. If serum phosphate is within the reference range for age, continue with the same dose. Follow dose adjustment schedule in Tables 1 and 2 to maintain serum phosphate within the reference range for age.
Dose adjustment.
Reassess fasting serum phosphate level 4 weeks after dose adjustment.
Do not adjust burosumab more frequently than every 4 weeks.
Dose increase.
If serum phosphate is below the reference range for age, the dose may be increased stepwise up to a maximum of 2.0 mg/kg, rounded to the nearest 10 mg, administered every 2 weeks (maximum dose of 90 mg).

Dose decrease.
If serum phosphate is above the reference range for age, withhold the next dose and reassess the serum phosphate level within 4 weeks. The patient must have serum phosphate below the reference range for age to restart burosumab. Once serum phosphate is below the reference range for age, treatment may be restarted as per Table 2. Reassess serum phosphate level 4 weeks after dose adjustment. If serum phosphate is below the reference range for age 4 weeks after dose adjustment, the dose can be restarted at 0.8 mg/kg every 2 weeks.
 Adult patients with X-linked hypophosphataemia (18 years of age and older). The recommended dose regimen in adults is 1 mg/kg of body weight, rounded to the nearest 10 mg up to a maximum dose of 90 mg, administered every 4 weeks.
Adult patients with X-linked hypophosphataemia (18 years of age and older). The recommended dose regimen in adults is 1 mg/kg of body weight, rounded to the nearest 10 mg up to a maximum dose of 90 mg, administered every 4 weeks.
After initiation of treatment with burosumab, measure fasting serum phosphate every 4 weeks, measured 2 weeks post-dose, for the first 3 months of treatment, and thereafter as appropriate. If serum phosphate is within the normal range, continue with the same dose.
Dose decrease.
Reassess fasting serum phosphate level 2 weeks after dose adjustment.
Do not adjust burosumab more frequently than every 4 weeks.
If serum phosphate is above the normal range, withhold the next dose and reassess the serum phosphate level within 4 weeks. The patient must have serum phosphate below the normal range to be able to restart burosumab. Once serum phosphate is below the normal range, treatment may be restarted at half the initial starting dose up to a maximum dose of 40 mg every 4 weeks according to the dose schedule shown in Table 3. Reassess serum phosphate 2 weeks after any change in dose.

Tumour-induced osteomalacia (TIO).
The posology in children and adolescents with TIO aged 1 to 17 years has been simulated using extrapolation of adult TIO population PK and PK/PD models to children aged 1-12 years and adolescents aged 13-17 years (see Section 5.2).
Children with TIO aged 1 to 12 years. The recommended starting dose for children aged 1 to 12 years is 0.4 mg/kg of body weight, given every 2 weeks. Doses should be rounded to the nearest 10 mg. The maximum dose is 90 mg.
Dose increase.
If serum phosphate is below the reference range for age, the dose may be increased in a stepwise manner. Doses should be increased by an initial increment of 0.6 mg/kg with subsequent increments, depending on patient's response to treatment, of 0.5 mg/kg (up to a maximum dose of 2.0 mg/kg), rounding the amount as described above, up to a maximum dose of 90 mg, given every 2 weeks.
Adolescents with TIO aged 13 to 17 years. The recommended starting dose for adolescents aged 13 to 17 years is 0.3 mg/kg of body weight, given every 2 weeks. Doses should be rounded to the nearest 10 mg. The maximum dose is 180 mg.
Dose increase.
If serum phosphate is below the reference range for age, the dose may be increased in a stepwise manner. Doses should be increased by an initial increment of 0.3 mg/kg with subsequent increments of between 0.2 mg/kg - 0.5 mg/kg (dose increment dependent on the patient's serum phosphate response to treatment), rounding the amount as described above, up to a maximum dose of 2.0 mg/kg (maximum dose 180 mg), given every 2 weeks.
Children and adolescents with TIO aged 1 to 17 years. After initiation of treatment with burosumab, fasting serum phosphate should be measured every 4 weeks, measured 2 weeks post-dose, for the first 3 months and thereafter as appropriate. If fasting serum phosphate is within the reference range for age, the same dose should be maintained. If fasting serum phosphate is not within the reference range, dose adjustment (dose increase/dose decrease) may be required. 4 weeks after any dose adjustment, fasting serum phosphate should be remeasured, and then either dose maintained if within reference range or further dose adjustment considered.
Dose adjustment.
Fasting serum phosphate should be measured 4 weeks after dose adjustment. Burosumab should not be adjusted more frequently than every 4 weeks.
Dose decrease.
If serum phosphate is above the reference range for age, the next dose should be withheld and the fasting serum phosphate level reassessed in 2 weeks. Once serum phosphate is below the reference range for age, treatment may be restarted at half the previous dose in rounding the amount as described above. The fasting serum phosphate level should be assessed 4 weeks after the dose adjustment. If the level remains below the reference range for age after the re-started dose, the dose can be further adjusted.
Dose conversion at age 18 years.
At 18 years of age the patient should convert to the adult dose and dosing regimen as outlined below.
Adults with TIO. The recommended starting dose for adults is 0.3 mg/kg body weight, rounded to the nearest 10 mg, given every 4 weeks.
After initiation of treatment with burosumab, fasting serum phosphate should be measured every 4 weeks, measured 2 weeks after each dose for the first 3 months of treatment, and thereafter as appropriate. If serum phosphate is within the reference range, the same dose should be maintained.
Dose increase.
If serum phosphate is below the reference range, the dose may be increased in a stepwise manner. Doses should be increased by an initial increment of 0.3 mg/kg, with subsequent increments of between 0.2 mg/kg - 0.5 mg/kg (dose dependent on the patient's response to treatment), up to a maximum dose of 2.0 mg/kg (maximum dose 180 mg), given every 4 weeks. Fasting serum phosphate should be measured 2 weeks after dose adjustment.
For patients whose serum phosphate still remains below the reference range, despite providing the maximum dose every 4 weeks, the previous dose may be divided and given every 2 weeks, with incremental increases as required, as outlined above, up to a maximum dose of 2.0 mg/kg every 2 weeks (maximum dose 180 mg).
Dose decrease.
If serum phosphate is above the reference range, the next dose should be withheld and the fasting serum phosphate level reassessed in 2 weeks. The patient must have serum phosphate below the reference range before restarting burosumab. Once serum phosphate is below the reference range, treatment may be restarted at approximately half the previous dose, administered every:
4 weeks (for patients dosed every 4 weeks before dose interruption);
2 weeks (for patients dosed every 2 weeks before dose interruption).
Serum phosphate should be reassessed 2 weeks after any change in dose.
If the level remains below the reference range after the re-started dose, the dose can be further adjusted.
Dose interruption in paediatric and adult patients with TIO.
If a patient undergoes treatment of the underlying tumour (i.e. surgical excision or radiation therapy) burosumab treatment should be interrupted.
Following completion of the treatment of the underlying tumour, serum phosphate should be reassessed before reinitiating treatment with burosumab. Burosumab treatment should be resumed at the patient's original starting dose if serum phosphate level remains below the lower limit of the normal reference range. Follow the recommended dose adjustment outlined above to maintain serum phosphate level within the normal reference range for age.
For all patients with TIO, treatment should be discontinued if the treating physician considers that no meaningful improvement in biochemical or clinical markers of response are observed, despite the maximum dose being administered.
All patients.
Missed or late dosing.
If a patient misses a dose, resume burosumab as soon as possible at prescribed dose. To avoid missed doses, treatments may be administered 3 days either side of the scheduled treatment date.
25-Hydroxy vitamin D supplementation.
Monitor 25-hydroxy vitamin D levels. Supplement with cholecalciferol or ergocalciferol to maintain 25-hydroxy vitamin D levels in the normal range for age. Do not administer active vitamin D analogues during burosumab treatment (see Section 4.3 Contraindications).
Special populations.
Renal impairment.
Limited data are available in patients with renal impairment. Burosumab must not be given to patients with severe or end stage renal disease (see Section 5.2 Pharmacokinetic Properties).
Method of administration.
For subcutaneous use.
Burosumab should be injected in the upper arm, abdomen, buttock or thigh. Injection sites should be rotated with each injection administered at a different anatomic location than the previous injection. Do not inject into moles, scars, or areas where the skin is tender, bruised, red, hard, or not intact. The maximum volume of medicinal product per injection site is 1.5 mL. If more than 1.5 mL is required on a given dosing day, the total volume of medicinal product should be split and should be administered at different injection sites. Injection sites should be rotated and carefully monitored for signs of potential reactions (see Section 4.4 Special Warnings and Precautions for Use).4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in Section 6.1.
Concurrent administration with oral phosphate and/or active vitamin D analogues.
Serum phosphate level within or above the normal range for age at initiation of treatment.
Severe renal impairment or end stage renal disease (see Section 5.2 Pharmacokinetic Properties).
4.4 Special Warnings and Precautions for Use
Traceability.
In order to improve the traceability of biological medicinal products, the name and batch number of administered product should be clearly recorded with the patient's records.
Ectopic mineralisation.
Ectopic mineralisation, as manifested by nephrocalcinosis, has been observed in patients with XLH treated with oral phosphate and active vitamin D analogues; these medicinal products should be stopped at least 1 week prior to initiating burosumab treatment (see Section 4.2).
Monitoring for signs and symptoms of nephrocalcinosis, e.g. by renal ultrasonography, is recommended at the start of treatment and every 6 months for the first 12 months of treatment, and annually thereafter. Monitoring of plasma alkaline phosphatase, calcium, parathyroid hormone (PTH) and creatinine is recommended every 6 months (every 3 months for children 1-2 years) or as indicated.
Monitoring of urine calcium and phosphate is suggested every 3 months.
Hyperphosphataemia.
Levels of fasting serum phosphate should be monitored due to the risk of hyperphosphataemia. To decrease the risk for ectopic mineralisation, it is recommended that fasting serum phosphate does not exceed the upper limit of the normal reference range for age. Dose interruption and/or dose reduction may be required (see Section 4.2). Periodic measurement of post prandial serum phosphate is advised.
To prevent hyperphosphataemia, treatment with burosumab should be interrupted in patients with tumour-induced osteomalacia who undergo treatment of the underlying tumour. Burosumab treatment should be reinitiated only if the patient's serum phosphate level remains below the lower limit of the normal reference range (see Section 4.2).
Hypercalcaemia and hyperparathyroidism.
Increases in serum calcium or parathyroid hormone have been reported in patients treated with burosumab. In patients with tertiary hyperparathyroidism, risk factors such as prolonged immobilisation, dehydration, hypervitaminosis D, or renal impairment may increase the risk of moderate to severe hypercalcaemia. Monitor serum calcium and parathyroid hormone levels before and during burosumab treatment. In patients with moderate to severe hypercalcaemia, burosumab should not be administered until hypercalcaemia is adequately managed.
Injection site reactions.
Administration may result in local injection site reactions. Administration should be interrupted in any patient experiencing severe injection site reactions.
Hypersensitivity.
Discontinue burosumab if serious hypersensitivity reactions occur.
Use in the elderly.
No conclusion can be made regarding any differences in safety or efficacy between patients over 65 years of age and those under 65 years of age.
Paediatric use.
X-linked hypophosphataemia.
The safety and efficacy of burosumab in children with XLH under 1 year of age has not been established in clinical studies.
Tumour-induced osteomalacia (TIO).
The safety and efficacy of burosumab in paediatric patients with TIO have not been established in clinical studies.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No pharmacokinetic interaction studies have been performed for burosumab.
Concurrent administration of burosumab with oral phosphate and active vitamin D analogues is contraindicated as it may cause an increased risk of hyperphosphataemia and hypercalcaemia (see Section 4.3).
Caution should be exercised when combining burosumab with calcimimetic medicinal products (i.e. agents that mimic the effect of calcium on tissues by activating the calcium receptor). Co-administration of these medicinal products has not been studied in clinical trials, therefore close monitoring of serum calcium levels is recommended.
4.6 Fertility, Pregnancy and Lactation
Women of childbearing potential.
Women of childbearing potential should use effective contraception during treatment with burosumab and for at least fourteen (14) weeks after stopping treatment.
Effects on fertility.
There are no clinical data available on the effect of burosumab on human fertility. The healthcare provider should discuss the use of appropriate contraception with sexually active women and adolescent girls taking burosumab to assist in weighing up the individual risks and benefits. No specific fertility studies in animals with burosumab were conducted, but relevant surrogate endpoints were examined in a general toxicity study in cynomolgus monkeys. Animals were treated with burosumab at doses up to 30 mg/kg every 2 weeks (yielding up to 56 or 14 times the exposure in adults at the maximum recommended clinical dose of 1 mg/kg every 4 weeks or 2 mg/kg every 2 weeks, for XLH or TIO treatments, respectively). Male monkeys showed mineralisation of the rete testis and seminiferous tubules (associated with hyperphosphataemia), but no changes in semen analysis. No adverse effects on reproductive organs to suggest impairment of fertility were observed in female monkeys.
(Category B3)
There are no available data on burosumab use in pregnant women to inform a drug-associated risk of adverse developmental outcomes. Adverse effects on embryofetal development were observed with burosumab in animals. Burosumab is not recommended for use during pregnancy. Fetal loss and pre-term births were increased and the gestation period was shortened in pregnant cynomolgus monkeys given burosumab at 30 mg/kg once every 2 weeks (yielding 64 or 15 times the exposure in adults at the maximum recommended clinical dose of 1 mg/kg every 4 weeks or 2 mg/kg every 2 weeks, for XLH or TIO treatments, respectively). This occurred in conjunction with maternal hyperphosphataemia and placental mineralisation. Burosumab was shown to cross the placenta. Ectopic mineralisation was not observed in fetuses or offspring, and treatment did not produce malformations, or affect growth, development or survival of the offspring.
There are no data on the use of burosumab in breastfeeding women. It is unknown whether burosumab is excreted in human milk, although the presence of the maternal IgG in milk is recognised. A decision must be made whether to discontinue breast feeding or to discontinue/abstain from burosumab therapy taking into account the benefit of breast feeding for the child, the benefit of the therapy for the woman and the potential risk to the child from exposure to burosumab.4.7 Effects on Ability to Drive and Use Machines
Burosumab may cause dizziness and, as a result, may influence the ability to drive or operate machinery. Patients should be cautioned about operating hazardous machines, including motor vehicles, until they are reasonably certain that burosumab does not affect them adversely.
4.8 Adverse Effects (Undesirable Effects)
The adverse reactions are presented by system organ class and frequency categories, defined using the following convention: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1000 to < 1/100); rare (≥ 1/10,000 to < 1/1000); very rare (< 1/10,000), not known (cannot be estimated from the available data). Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness.
Adverse reactions reported in paediatric patients with XLH ≥ 1 year of age.
The most common adverse reactions (> 10%) reported in paediatric patients ≥ 1 year of age during clinical trials were injection site reactions (56%), cough (56%), headache (50%), pyrexia (43%), pain in extremity (40%), vomiting (39%), tooth abscess (35%), vitamin D decreased (32%), diarrhoea (25%), rash (24%), nausea (15%), constipation (11%), dental caries (11%) and myalgia (11%). This data is based the integrated safety assessment of 94 subjects who received at least one dose of burosumab and were included in studies UX023-CL201, UX023-CL205 and UX023-CL301.
An overview of adverse reactions observed from burosumab clinical trials and post marketing sources in paediatrics is presented in Table 4.
 Description of selected adverse reactions in children with XLH ≥ 1 year of age.
Description of selected adverse reactions in children with XLH ≥ 1 year of age. Injection site reactions.
Approximately 56% of the patients had an injection site reaction. The injection site reactions were generally mild in severity, occurred within 1 day of medicinal product injection, lasted approximately 1 to 3 days, required no treatment, and resolved in almost all instances.
Hypersensitivity reactions.
The most frequent potential hypersensitivity reaction was rash (18%). The events were mild or moderate in severity.
Adverse events reported in paediatric patients with XLH ≥ 1 year of age in study UX023-CL301.
The most common adverse events in paediatric patients ≥ 1 year of age (reported in ≥ 5% of patients and having a higher incidence with burosumab than with active control) in Study UX023-CL301 are listed in Table 5.

Adverse reactions reported in adult patients with XLH.
The most common adverse reactions reported in adult patients during clinical trials were back pain (23%), headache (21%), tooth infection (19%), restless legs syndrome (13%), muscle spasms (12%), vitamin D decrease (15%) and dizziness (11%). This data is based on the integrated safety assessment of 176 subjects who received at least 1 dose of burosumab and were included in studies UX023-CL203, UX023-CL303, UX023-CL304, KRN23-INT-001 and KRN23-INT-002.
An overview of adverse reactions observed from burosumab clinical trials and post marketing sources in adults is presented in Table 6.
 Description of selected adverse reactions in adults with XLH.
Description of selected adverse reactions in adults with XLH. Injection site reactions.
The frequency of injection site reactions was 12% in both burosumab and placebo treatment groups (injection site reaction, erythema, rash, bruising, pain, pruritus and haematoma). The injection site reactions were generally mild in severity, occurred within 1 day of medicinal product injection, lasted approximately 1 to 3 days, required no treatment, and resolved in almost all instances.
Hypersensitivity reactions.
The frequency of hypersensitivity reactions was 6% in both the burosumab and placebo groups. The hypersensitivity reactions were mild to moderate in severity.
Hyperphosphataemia.
In the double-blind period of Study UX023-CL303, 9 of 134 (7%) patients in the burosumab treatment group experienced hyperphosphataemia meeting the protocol-specified criteria for dose reduction. The hyperphosphataemia was managed with dose reduction.
Restless legs syndrome.
Approximately 12% of the burosumab treatment group and 8% in the placebo group had a worsening of baseline restless legs syndrome or new onset restless legs syndrome of mild to moderate severity.
Adverse events reported in adult patients with XLH in study UX023-CL303.
The most common adverse events in adult patients (reported in ≥ 5% of patients and having a higher incidence with burosumab than with placebo) in Study UX023-CL303 are listed in Table 7.

Immunogenicity.
Overall, the incidence of anti-drug antibodies (ADA) to burosumab was < 10% in adults and paediatric subjects administered burosumab. The incidence of neutralising ADA was 3.2% and neutralising ADA were only found in paediatric subjects. No adverse events, loss of efficacy, or changes in pharmacokinetics profile were associated with these findings.
Tumour-induced osteomalacia (TIO).
Adverse reactions reported in adult patients with TIO. The most common adverse reactions (≥ 20%) reported in adult patients with TIO during clinical studies (up to week 300 in study UX023T-CL201 and week 220 in study KRN23-002) were constipation (22%), muscle spasms (22%) and rash (22%). Table 8 includes the core safety profile of burosumab in adult patients with TIO, derived from clinical studies (up to week 300 in study UX023T-CL201 and week 220 in study KRN23-002) and post marketing experience.
 Description of selected adverse reactions in adults with TIO.
Description of selected adverse reactions in adults with TIO. Hypersensitivity.
In patients with TIO, the frequency of hypersensitivity reactions (rash, drug eruption, and hypersensitivity) based on data from completed long term clinical studies was 30%.
Restless legs syndrome.
In patients with TIO, based on data from completed long term clinical studies, 11% of patients experienced events of restless legs syndrome of mild to moderate severity.
Injection site reactions.
Up to week 300, the frequency of injection site reactions was 11.1%. Injection site reactions were generally mild in severity, required no treatment and resolved in all cases.
Hyperphosphataemia.
Up to week 300, 7.4% of patients experienced events of hyperphosphataemia, which were managed with dose reduction.
Adverse events reported in adult patients with TIO in studies UX023T-CL201 and KRN23-002. The most common adverse events in adult patients (reported in ≥ 10% of patients) in studies UX023T-CL201 (up to week 300) and KRN23-002 (up to week 220) are listed in Table 9. Out of 27 adult patients with TIO, there are 26 patients (96.3%) with any treatment emergent adverse event (TEAE).

Immunogenicity.
In adults completing long-term clinical studies, up to week 300 (see Section 5.1), ADAs were identified in two (2) patients. None of these patients developed neutralising ADAs. No adverse events, loss of efficacy, or changes in the pharmacokinetic profile of burosumab were associated with these findings.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
There is no experience with overdose of burosumab. In case of overdose, it is recommended to stop burosumab and to monitor biochemical response. For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Burosumab is a recombinant human monoclonal IgG1 antibody that binds to and inhibits the biological activity of fibroblast growth factor 23 (FGF23), present in excess in X-linked hypophosphataemia. Neutralisation of FGF23 by burosumab increases renal tubular reabsorption of phosphate and the serum concentration of 1, 25 dihydroxy-Vitamin D.
Clinical trials.
Paediatric X-linked hypophosphataemia.
Burosumab has been evaluated in 3 studies enrolling a total of 126 paediatric patients with XLH.
Study UX023-CL301. Paediatric Study UX023-CL301 is a randomised, open-label study in 61 paediatric XLH patients, 1 to 12 years old, that compared treatment with burosumab to active control (oral phosphate and active vitamin D therapy). All patients had radiographic evidence of rickets at baseline, with an RSS score of ≥ 2 and had received oral phosphate and active vitamin D analogues for a mean (SD) duration of 3.8 (3.1) years. Oral phosphate and active vitamin D analogues were discontinued prior to study enrolment for a 7-day washout period and then restarted for patients in the active control group. Patients were randomised to receive either burosumab at a starting dose of 0.8 mg/kg every 2 weeks or oral phosphate and active vitamin D. Patients randomised to active control received a mean oral phosphate dose of approximately 41 mg/kg/day (18 to 110 mg/kg/day) at Week 40 and approximately 46 mg/kg/day (18 to 166 mg/kg/day) at Week 64. They also received either a mean oral calcitriol dose of 26 nanogram/kg/day at Week 40 and 27 nanogram/kg/day at Week 64 or a therapeutically equivalent amount of alfacalcidol. Eight patients in the burosumab group titrated up to 1.2 mg/kg based on serum phosphate measurements.
Study UX023-CL201. Paediatric Study UX023-CL201, is a randomised, open-label study in 52 prepubescent XLH patients, 5 to 12 years old, which compared treatment with burosumab administered every 2 weeks versus every 4 weeks. Following an initial 16-week dose titration phase, patients completed 48 weeks of treatment with burosumab every 2 weeks or every 4 weeks. Burosumab dose was adjusted to target a fasting serum phosphate concentration of 1.13 to 1.62 mmol/L [3.5 to 5.0 mg/dL] based on the fasting phosphate level on the day of dosing.
Twenty-six of 52 patients received burosumab every 2 weeks up to a maximum dose of 2 mg/kg. The average dose was 0.73 mg/kg (range: 0.3, 1.5) at Week 16, 0.98 mg/kg (range: 0.4, 2.0) at Week 40 and 1.04 mg/kg (range: 0.4, 2.0) at Week 60. The remaining 26 patients received burosumab every 4 weeks. At study entry, the mean age of patients was 8.5 years and 46% were male. Ninety-six percent had received oral phosphate and active vitamin D analogues for a mean (SD) duration of 6.9 (2.4) years. Oral phosphate and active vitamin D analogues were discontinued prior to study enrolment. Ninety-four percent of patients had radiographic evidence of rickets at baseline.
Study UX023-CL205. Paediatric Study UX023-CL205 is a randomised, open-label study in 13 paediatric XLH patients, aged 1 to 4 years. Patients received burosumab at a dose of 0.8 mg/kg every 2 weeks with 5 patients titrating up to 1.2 mg/kg based on serum phosphate measurements. All patients completed at least 40 weeks on study; no patients discontinued. At study entry, the mean age of patients was 2.9 years and 69% were male. All patients had radiographic evidence of rickets at baseline and had received oral phosphate and active vitamin D analogues for a mean (SD) duration of 16.4 (13.8) months. Oral phosphate and active vitamin D analogues were discontinued prior to study enrolment.
Serum phosphate.
In Study UX023-CL301, burosumab increased mean (SD) serum phosphate levels from 2.4 (0.24) mg/dL at baseline to 3.3 (0.43) mg/dL at Week 40. In the active control group, mean (SD) serum phosphate concentrations increased from 2.3 (0.26) mg/dL at baseline to 2.5 (0.34) mg/dL at Week 40 (Figure 1). The renal phosphate reabsorption assessed by TmP/GFR increased in the burosumab treated patients from a mean (SD) of 2.2 (0.37) at baseline to 3.4 (0.67) mg/dL at Week 40. In the active control group, mean (SD) TmP/GFR decreased from 2.0 (0.33) mg/dL at baseline to 1.8 (0.35) mg/dL at Week 40.
In Study UX023-CL201, burosumab increased serum phosphate levels 2.4 (0.41) mg/dL at baseline to 3.3 (0.40) mg/dL and 3.4 (0.45) mg/dL at Week 40 and Week 64 in the patients who received burosumab every 2 weeks. The ratio of renal tubular maximum reabsorption rate of phosphate to glomerular filtration rate (TmP/GFR) increased in these patients from mean (SD) of 2.2 (0.49) at baseline to 3.3 (0.60) and 3.4 (0.53) mg/dL at Week 40 and Week 64.
In Study UX023-CL205, mean (SD) fasting serum phosphate levels increased from 2.5 (0.28) mg/dL at baseline to 3.5 (0.49) mg/dL at Week 40.

Radiographic evaluation of rickets.
Radiographs were examined to assess XLH-related rickets using the 10-point Thacher Rickets Severity Score (RSS) and the 7-point Radiographic Global Impression of Change (RGI-C). The RSS score is assigned based on images of the wrist and knee from a single time point, with higher scores indicating greater rickets severity. The RGI-C score is assigned based on side-by side comparisons of wrist and knee radiographs from 2 time points, with higher scores indicating greater improvement in radiographic evidence of rickets. An RGI-C score of +2.0 was defined as radiographic evidence of substantial healing.
In study UX023-CL301, burosumab healed and reduced the severity of rickets to a greater extent than active control after 40 weeks of treatment, as assessed by decreasing RSS total scores and increasing RGI-C global score, shown in Table 10.
 In Study UX023-CL201 and study UX023-CL205, RSS total score decreased, and RGI-C global score demonstrated healing after 40 weeks of treatment with burosumab every 2 weeks as shown in Table 11.
In Study UX023-CL201 and study UX023-CL205, RSS total score decreased, and RGI-C global score demonstrated healing after 40 weeks of treatment with burosumab every 2 weeks as shown in Table 11.

Lower extremity skeletal abnormality.
In Study UX023-CL301, lower extremity skeletal abnormalities were assessed by RGI-C in standing long leg radiographs. At Week 64, the burosumab group maintained greater improvement compared with the active control group (LS mean [SE]: +1.25 [0.17] versus +0.29 [0.12]; difference of +0.97 [95% CI: +0.57, +1.37, GEE model]).
Serum alkaline phosphatase activity.
In Study UX023-CL301, mean (SD) serum alkaline phosphatase (ALP) activity decreased from 511 (125) U/L at baseline to 337 (86) U/L in the burosumab group (mean change: -33%), and from 523 (154) U/L at baseline to 495 (182) U/L in the active control group (mean change: -5%) at Week 64.
In Study UX023-CL201, mean (SD) serum total ALP activity decreased from 462 (110) U/L at baseline to 354 (73) U/L at Week 64 (mean change: -23%) in patients who received burosumab every 2 weeks.
In Study UX023-CL205, mean (SD) serum total ALP activity decreased from 549 (194) U/L at baseline to 335 (88) U/L at Week 40 (mean change: -36%).
Growth.
In Study UX023-CL301, burosumab treatment for 64 weeks increased standing mean (SD) height Z score from -2.32 (1.17) at baseline to -2.11 (1.11) at Week 64 (LS mean change [SE] of +0.17 [0.07]). In the active control group, the mean (SD) height Z score increased from -2.05 (0.87) at baseline to -2.03 (0.83) at Week 64 (LS mean change [SE] of +0.02 [0.04]).
In Study UX023-CL201, burosumab treatment for 64 weeks increased standing mean (SD) height Z score from -1.72 (1.03) at baseline to -1.54 (1.13) in the patients who received burosumab every 2 weeks (LS mean change of +0.19 [95% CI: 0.09 to 0.29]).
Adult X-linked hypophosphataemia.
Study UX023-CL303 is a randomised, double-blind, placebo-controlled study in 134 adult XLH patients. The study comprises a 24-week, double-blind, placebo-controlled treatment phase followed by a 24-week open-label treatment period in which all patients received burosumab. Subjects then continued burosumab treatment for an additional 48 weeks in a Treatment Extension Period I (until Week 96) and Treatment Extension Period II (End of Treatment). Burosumab was administered at a dose of 1 mg/kg every 4 weeks. At study entry, the mean age of patients was 40 years (range 19 to 66 years) and 35% were male. All patients had skeletal pain associated with XLH/osteomalacia at baseline. The baseline mean (SE) serum phosphate concentration was below the lower limit of normal at 0.64 (0.10) mmol/L [1.98 (0.314) mg/dL]. Oral phosphate and active vitamin D analogues were not allowed during the study. Of the 134 subjects who enrolled in the study, one patient in the burosumab group discontinued treatment during the 24-week placebo-controlled treatment period, and 7 patients discontinued Burosumab during the open-label treatment period. During Treatment Extension Period I, 7 patients discontinued treatment, and during Treatment Extension Period II, 1 patient discontinued treatment.
Study UX023-CL304 is an open-label, single-arm study in 14 adult XLH patients to assess the effects of burosumab on improvement of osteomalacia as determined by histologic and histomorphometric evaluation of iliac crest bone biopsies. Patients received 1.0 mg/kg burosumab every 4 weeks. At study entry, the mean age of patients was 40 years (range 25 to 52 years) and 43% were male. Oral phosphate and active vitamin D analogues were not allowed during the study.
Serum phosphate.
In Study UX023-CL303 at baseline, mean (SD) serum phosphate was 1.9 (0.32) mg/dL and 2.0 (0.30) mg/dL in the placebo and burosumab groups, respectively. During the initial 24-week double-blind, placebo-controlled period, mean (SD) serum phosphate across the midpoints of dose intervals (2 weeks post dose) was 2.1 (0.30) mg/dL and 3.2 (0.53) mg/dL in the placebo and burosumab groups, and mean (SD) serum phosphate across the ends of dose intervals was 2.1(0.30) mg/dL and 2.7 (0.45) mg/dL in the placebo and burosumab groups.
A total of 93% of patients treated with burosumab achieved a serum phosphate level above the lower limit of normal (LLN) compared to 8% in the placebo group through Week 24 (Table 12).
 During the open-label treatment period, serum phosphate was maintained during continued burosumab therapy (Figure 2), with no evidence of loss of effect through Week 48. Similar results were seen during Treatment Extension Period I and Extension Treatment Period II.
During the open-label treatment period, serum phosphate was maintained during continued burosumab therapy (Figure 2), with no evidence of loss of effect through Week 48. Similar results were seen during Treatment Extension Period I and Extension Treatment Period II.
 At baseline, the mean (SD) ratio of TmP/GFR was 1.60 (0.37) and 1.68 (0.40) mg/dL in the placebo and burosumab groups, respectively. At Week 22 (midpoint of a dose interval), mean (SD) TmP/GFR was 1.69 (0.37) and 2.73 (0.75) mg/dL in the placebo and burosumab groups. At Week 24 (end of a dose interval), mean (SD) TmP/GFR was 1.73 (0.42) and 2.22 (0.49) mg/dL in the placebo and burosumab groups. During the open-label treatment period, TmP/GFR remained stable during continued burosumab therapy through Week 48. Similar results were seen during Treatment Extension Period I and Treatment Extension Period II.
At baseline, the mean (SD) ratio of TmP/GFR was 1.60 (0.37) and 1.68 (0.40) mg/dL in the placebo and burosumab groups, respectively. At Week 22 (midpoint of a dose interval), mean (SD) TmP/GFR was 1.69 (0.37) and 2.73 (0.75) mg/dL in the placebo and burosumab groups. At Week 24 (end of a dose interval), mean (SD) TmP/GFR was 1.73 (0.42) and 2.22 (0.49) mg/dL in the placebo and burosumab groups. During the open-label treatment period, TmP/GFR remained stable during continued burosumab therapy through Week 48. Similar results were seen during Treatment Extension Period I and Treatment Extension Period II.
Bone histomorphometry.
In Study UX023-CL304, after 48 weeks of treatment, healing of osteomalacia was observed in 10 patients as demonstrated by decreases in osteoid volume/bone volume (OV/BV) from a mean (SD) score of 26% (12.4) at baseline to 12% (6.6). Osteoid thickness (O.Th) declined in 11 patients from a mean (SD) of 17 (4.1) micrometres to 12 (3.1) micrometres.
Patient reported outcomes.
Study UX023-CL303 evaluated stiffness and physical function using the Western Ontario and McMaster Universities Osteoarthritis (WOMAC) Index and pain using the Brief Pain Inventory (BPI).
WOMAC Index: The WOMAC Index comprises three domains: stiffness, pain and physical function; each domain ranges from 0 (best health) to 100 (worst health). Changes from baseline in WOMAC stiffness and physical function domain scores were key secondary endpoints. Burosumab treatment for 24 weeks resulted in a statistically significant favourable change relative to placebo in WOMAC Stiffness and trends favouring burosumab treatment in WOMAC Physical Function as shown in Table 13.

Clinical efficacy in adult patients with tumour-induced osteomalacia.
Burosumab has been evaluated in 2 studies enrolling a total of 27 patients with TIO.
Study UX023T-CL201. Study UX023T-CL201 is a 48-week, open-label, single arm study in 14 adult patients with a confirmed diagnosis of FGF23-related hypophosphataemia produced by an underlying tumour that was not amenable to surgical excision or could not be located. After completion of week 48, 11 patients enrolled in a treatment extension period, up to week 300.
Of the 14 TIO patients enrolled, 8 were male, and ranged from 33 years to 68 years of age (median 59.5 years). Oral phosphate and active vitamin D analogues were discontinued 2 weeks prior to study enrolment. Patients received burosumab every 4 weeks at a weight based starting dose of 0.3 mg/kg that was titrated to achieve a fasting serum phosphate level of 2.5 to 4.0 mg/dL. The maximum dose allowed was 2.0 mg/kg every 2 weeks. The mean dose was 0.87 mg/kg at week 48 (n=12), 0.67 mg/kg at week 144 (n=10), 0.66 mg/kg at week 240 (n=5), 0.80 mg/kg at week 248 (n=3) and 0.60 mg/kg at week 296 (n=1). During the study period, one patient received every 2 week dosing between week 136 and 154.
Study KRN23-002. Study KRN23-002 is a 48-week open-label, single-arm study in 13 adult patients with a confirmed diagnosis of TIO. After completion of week 48, 6 patients enrolled in a treatment extension period up to marketing authorisation approval (up to week 230).
Of the 13 TIO patients who received treatment, 6 were male, and ranged from 41 years to 73 years of age (median 58.0 years). Oral phosphate and active vitamin D analogues were discontinued 2 weeks prior to study enrolment. Patients received burosumab every 4 weeks at a weight based starting dose of 0.3 mg/kg to achieve a fasting serum phosphate level of 2.5 to 4.0 mg/dL. The mean (SD) dose was 0.91 (0.59) mg/kg at week 48 (n=12), 0.96 (0.70) mg/kg at week 88 (n=12), 1.14 (0.56) mg/kg at week 184 (n=5), and 1.30 (0.71) mg/kg at week 220 (n=2).
Serum phosphate.
In study UX023T-CL201, burosumab increased mean (SD) serum phosphate levels from 1.60 (0.47) mg/dL at baseline to 2.64 (0.76) mg/dL averaged across the midpoint of dose intervals through week 24 with 50% of patients (7/14) achieving a mean serum phosphate level above the LLN averaged across the midpoint of dose intervals through week 24. At week 48 (a visit coinciding with the end of the dose interval), the mean (SD) serum phosphate level was 2.53 (0.62) mg/dL; the LS mean (SE) change (GEE model) from baseline to week 48 was 0.89 (0.17) mg/dL (p < 0.001). Increases were sustained during the study period. The mean (SD) serum phosphate level was 2.40 (0.70) mg/dL at week 96, 2.59 (0.60) mg/dL at week 144, 2.48 (0.55) mg/dL at week 192, and 2.57 (0.38) mg/dL at week 240. The LS mean (SE) change (GEE model) from baseline to week 240 was 0.92 (0.13) mg/dL (p < 0.001). Increases in the mean serum phosphate levels were sustained near or above the LLN through week 240 (Figure 3). The ratio of TmP/GFR increased in these patients from a mean (SD) of 1.12 (0.54) mg/dL at baseline to 2.12 (0.64) mg/dL at week 48 and remained stable through week 144.
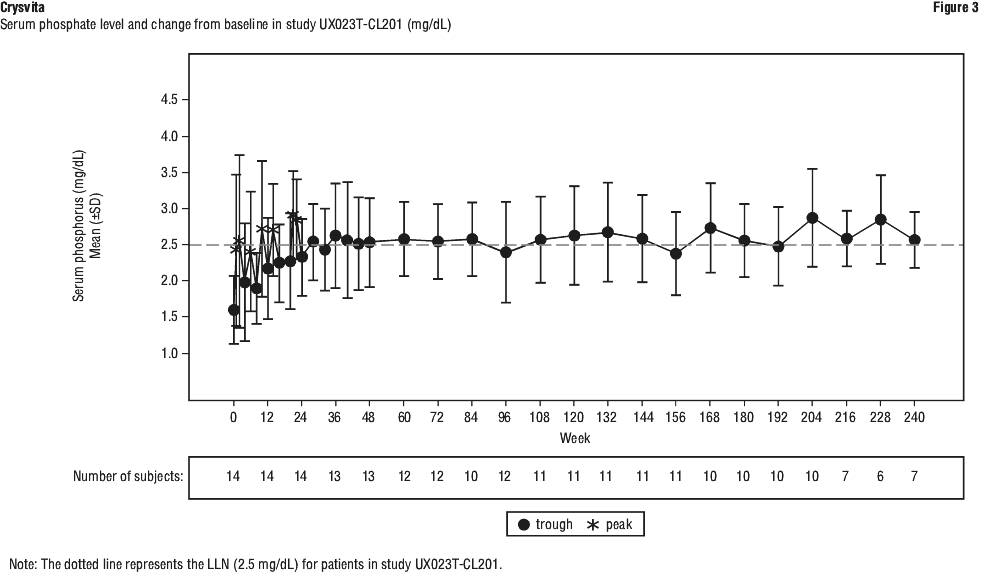 In study KRN23-002, burosumab increased mean (SD) serum phosphate level from 1.62 (0.49) mg/dL at baseline to 2.63 (0.87) mg/dL averaged across the midpoint of dose intervals through week 24 with 69% percent of patients (9/13) achieving a mean serum phosphate level above the LLN averaged across the midpoint on dose interval through week 24.
In study KRN23-002, burosumab increased mean (SD) serum phosphate level from 1.62 (0.49) mg/dL at baseline to 2.63 (0.87) mg/dL averaged across the midpoint of dose intervals through week 24 with 69% percent of patients (9/13) achieving a mean serum phosphate level above the LLN averaged across the midpoint on dose interval through week 24.
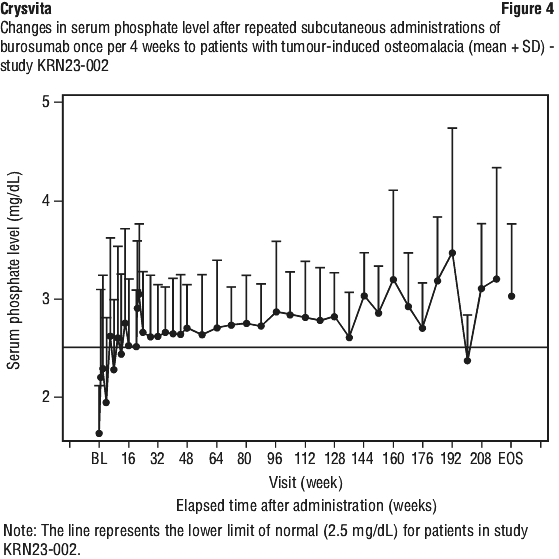
Thereafter, serum phosphate levels were 2.70 ± 0.44 mg/dL at week 48, 2.73 ± 0.39 mg/dL at week 72, 2.72 ± 0.43 mg/dL at week 88, and 3.02 ± 0.74 mg/dL at EOS which remained in the normal range. The renal phosphate reabsorptive capacity, as assessed by TmP/GFR, increased from a mean (SD) of 1.15 (0.43) mg/dL at baseline to 2.30 mg/dL (0.48) mg/dL at week 48, 2.30 mg/dL (0.83) at week 72, 2.48 mg/dL (0.81) at week 96, and 2.63 mg/dL (0.83) at EOS (Figure 4).
Bone histomorphometry.
In study UX023T-CL201, osteomalacia was present at baseline in 9 of 11 patients with paired bone biopsies, and healing was assessed after 48 weeks of treatment. In these 9 patients with osteomalacia at baseline, OV/BV decreased from a mean (SD) score of 17.6% (19.5) at baseline to 12.1% (15.4), a change of -31%. O.Th declined from a mean (SD) of 16.5 (12.0) micrometres to 11.3 (9.2) micrometres, a change of -31%. MLt declined in 3 patients from a mean (SD) of 667 (414) days to 331 (396) days, a change of -50%.
In study KRN23-002, 3 patients had bone biopsies performed prior to start of burosumab and after 48 weeks of treatment and results were consistent with those observed in study UX023T-CL201. Mean (SD) O.Th was 16.03 (13.67) micrometres at baseline and 13.50 (7.05) micrometres at week 48. Mean (SD) OS/BS was 47.7 (23.1)% at baseline and 43.0 (5.6)% at week 48. Mean (SD) OV/BV was 14.03 (15.23)% at baseline and 9.23 (5.48)% at week 48.
Radiological evaluation of osteomalacia.
In study UX023T-CL201, 99mtechnetium-labelled whole body bone scans were performed at baseline and subsequent timepoints up to week 144 in all 14 TIO patients. In baseline bone scans, 100% of patients had areas of tracer uptake, presumed to be nontraumatic fractures or pseudofractures. Baseline bone scans were compared by a central reader, blinded to subject data, to subsequent bone scans. The number of areas of tracer uptake decreased from week 48 through week 144, suggesting healing of fractures and pseudofractures.
Paediatric patients with TIO.
There are no clinical trials with burosumab in paediatric patients of any age with TIO. The posology of burosumab in paediatric TIO patients has been determined from pharmacokinetic modelling and simulation (see Section 5.2)
5.2 Pharmacokinetic Properties
The following pharmacokinetic parameters were observed in patients with XLH administered the approved recommended starting dosage based on a 70 kg patient, unless otherwise specified.
Burosumab exhibited linear pharmacokinetics following subcutaneous injections within the dose range of 0.1 to 1 mg/kg (0.08 to 0.8 times the maximum approved recommended dosage based on a 70 kg patient). The steady-state trough mean (±SD) concentration of burosumab was 5.8 (± 3.4) microgram/mL in adult patients, 15.8 (± 9.4) microgram/mL in patients aged 5 to 12 years, and 14.2 (± 4.0) microgram/mL in patients aged 1 to 4 years.
Absorption.
The burosumab mean Tmax values ranged from 8 to 11 days. The median time to reach maximum serum concentrations (Tmax) of burosumab is approximately 7 to 13 days.
Distribution.
The apparent volume of distribution of burosumab is 7.35 L.
Biotransformation.
The exact pathway for burosumab metabolism has not been characterised. Burosumab is expected to be degraded into small peptides and amino acids via catabolic pathways.
Elimination.
The clearance of burosumab is dependent on body weight and estimated to be 0.278 L/day in a typical adult (70 kg) XLH patient with corresponding disposition half-life (t1/2) in the serum of approximately 18 days.
Linearity/non-linearity.
Burosumab displays pharmacokinetics that are linear to dose over the subcutaneous dose range of 0.1 to 1.0 mg/kg.
Paediatric pharmacokinetics/pharmacodynamics.
No significant difference has been observed in the Pharmacokinetics (PK) and Pharmacodynamics of the paediatric XLH population as compared with the adult XLH population. Burosumab clearance and volume of distribution are body weight dependent.
Paediatric patients with TIO.
The starting dose of burosumab for paediatric patients with TIO is based on population PK/PD modelling and simulations which indicate that a starting dose of 0.4 mg/kg every 2 weeks for children aged 1-12 years and 0.3 mg/kg every 2 weeks for adolescents aged 13-17 years is predicted to result in a proportion of paediatric patients with TIO reaching normal serum phosphate levels. These can be titrated up to a maximum of 2.0 mg/kg every 2 weeks (the highest dose simulated).
Special populations.
Population PK analyses using data from paediatric and adult subjects who have XLH and adult subjects with TIO indicated that age, sex, race, ethnicity, baseline serum albumin, baseline serum alkaline phosphate, baseline serum alanine aminotransferase, and baseline creatinine clearance ≥ 49.9 mL/min, were not significant predictors of burosumab PK. Based on the population PK analysis, the PK characteristics of burosumab were similar between XLH and TIO patients. The effect of renal impairment on the pharmacokinetics of burosumab is unknown. However, renal impairment can induce abnormal mineral metabolism which could increase phosphate concentrations greater than expected with burosumab alone. This increase may result in hyperphosphataemia which can induce nephrocalcinosis.
Burosumab must not be given to patients with severe renal impairment, defined as:
Paediatric patients with an estimated glomerular filtration rate (eGFR) of 15 mL/min/1.73 m3 to 29 mL/min/1.73 m3 or end stage renal disease (eGFR < 15 mL/min/1.73 m3);
Adult patients with a calculated eGFR of 15 mL/min/1.73 m3 to 29 mL/min/1.73 m3 or end stage renal disease (eGFR < 15 mL/min/1.73 m3).
5.3 Preclinical Safety Data
Genotoxicity.
Genotoxicity studies have not been conducted with burosumab. As a large protein molecule, burosumab is not expected to interact with DNA or other chromosomal material.
Carcinogenicity.
The carcinogenic potential of burosumab has not been investigated in long-term animal studies.6 Pharmaceutical Particulars
6.1 List of Excipients
L-histidine; D-sorbitol; polysorbate 80; L-methionine; hydrochloric acid, 10% (for pH adjustment); water for injection.
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
This medicinal product must not be mixed with other medicinal products.
6.4 Special Precautions for Storage
Store in a refrigerator (2°C to 8°C). Do not freeze.
Store in original package to protect from light.
6.5 Nature and Contents of Container
Each pack contains 1 mL solution in a clear glass vial with butyl rubber stopper, and aluminium seal.
6.6 Special Precautions for Disposal
Product is for single use in one patient only. Discard any residue.
Do not shake the vial before use.
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
 Heavy chains (HC) are shown in blue and Light chains are shown in red.
Heavy chains (HC) are shown in blue and Light chains are shown in red.
Black lines show the location of disulfide bonds.
N297 of HC: glycosylation site.
CAS number.
CAS registry number: 1610833-03-8.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes












 In Study UX023-CL201 and study UX023-CL205, RSS total score decreased, and RGI-C global score demonstrated healing after 40 weeks of treatment with burosumab every 2 weeks as shown in Table 11.
In Study UX023-CL201 and study UX023-CL205, RSS total score decreased, and RGI-C global score demonstrated healing after 40 weeks of treatment with burosumab every 2 weeks as shown in Table 11.
 During the open-label treatment period, serum phosphate was maintained during continued burosumab therapy (Figure 2), with no evidence of loss of effect through Week 48. Similar results were seen during Treatment Extension Period I and Extension Treatment Period II.
During the open-label treatment period, serum phosphate was maintained during continued burosumab therapy (Figure 2), with no evidence of loss of effect through Week 48. Similar results were seen during Treatment Extension Period I and Extension Treatment Period II. At baseline, the mean (SD) ratio of TmP/GFR was 1.60 (0.37) and 1.68 (0.40) mg/dL in the placebo and burosumab groups, respectively. At Week 22 (midpoint of a dose interval), mean (SD) TmP/GFR was 1.69 (0.37) and 2.73 (0.75) mg/dL in the placebo and burosumab groups. At Week 24 (end of a dose interval), mean (SD) TmP/GFR was 1.73 (0.42) and 2.22 (0.49) mg/dL in the placebo and burosumab groups. During the open-label treatment period, TmP/GFR remained stable during continued burosumab therapy through Week 48. Similar results were seen during Treatment Extension Period I and Treatment Extension Period II.
At baseline, the mean (SD) ratio of TmP/GFR was 1.60 (0.37) and 1.68 (0.40) mg/dL in the placebo and burosumab groups, respectively. At Week 22 (midpoint of a dose interval), mean (SD) TmP/GFR was 1.69 (0.37) and 2.73 (0.75) mg/dL in the placebo and burosumab groups. At Week 24 (end of a dose interval), mean (SD) TmP/GFR was 1.73 (0.42) and 2.22 (0.49) mg/dL in the placebo and burosumab groups. During the open-label treatment period, TmP/GFR remained stable during continued burosumab therapy through Week 48. Similar results were seen during Treatment Extension Period I and Treatment Extension Period II.
 In study KRN23-002, burosumab increased mean (SD) serum phosphate level from 1.62 (0.49) mg/dL at baseline to 2.63 (0.87) mg/dL averaged across the midpoint of dose intervals through week 24 with 69% percent of patients (9/13) achieving a mean serum phosphate level above the LLN averaged across the midpoint on dose interval through week 24.
In study KRN23-002, burosumab increased mean (SD) serum phosphate level from 1.62 (0.49) mg/dL at baseline to 2.63 (0.87) mg/dL averaged across the midpoint of dose intervals through week 24 with 69% percent of patients (9/13) achieving a mean serum phosphate level above the LLN averaged across the midpoint on dose interval through week 24.
 Heavy chains (HC) are shown in blue and Light chains are shown in red.
Heavy chains (HC) are shown in blue and Light chains are shown in red.