What is in this leaflet
This leaflet answers some common questions about DARZALEX Concentrate for Solution for Infusion. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you being given DARZALEX against the benefits this medicine is expected to have for you.
If you have any concerns about being given DARZALEX ask your doctor.
Keep this leaflet while you are being treated with DARZALEX. You may need to read it again.
What DARZALEX is used for
DARZALEX is an anti-cancer medicine and contains the active substance daratumumab. This belongs to a group of medicines called "monoclonal antibodies". One of the ways monoclonal antibodies work is by attaching themselves to specific cancer cells in your body, so your immune system can destroy them.
DARZALEX is used to treat adults with multiple myeloma (cancer of the bone marrow).
Your doctor may have prescribed DARZALEX for another reason.
Ask your doctor if you have any questions about why DARZALEX has been prescribed for you.
This medicine is available only with a doctor's prescription.
Before you are given DARZALEX
When you must not use it:
Do not use DARZALEX if:
- you know you are allergic (hypersensitive) to daratumumab or other ingredients of DARZALEX. See Product Description at the end of this leaflet for a list of ingredients.
Symptoms of an allergic reaction may include rash, itching or hives on the skin, shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body.
DARZALEX should not be given to children or young people below 18 years of age. This is because it is not known how the medicine will affect them.
Before you start to use it:
Tell your doctor if you have or have had any medical conditions, especially the following:
- blood disorder with a low level of white blood cells or platelets. This disorder may become worse during treatment with DARZALEX.
- Hepatitis B infection
DARZALEX could cause hepatitis B virus to become active again. Your doctor will check you for signs of this infection before, during and for some time after treatment with DARZALEX.
Tell your doctor if you are pregnant or intend to become pregnant. Like most medicines used to treat cancer, DARZALEX is not recommended for use during pregnancy.
Tell your doctor if you are trying to make your partner pregnant.
Both men and women receiving DARZALEX and their partners must use a reliable method of contraception during and for 3 months after ceasing treatment with DARZALEX.
Tell your doctor if you are breastfeeding or intend to breastfeed. It is not known whether DARZALEX passes into breast milk. Therefore there is a possibility that the breastfed baby may be affected.
If you wish to restart breastfeeding after your DARZALEX treatment, you must discuss this with your doctor or nurse, who will tell you when it is safe to do so.
If you have not told your doctor about any of the above, tell them before you start treatment with DARZALEX.
Taking other medicines:
Tell your doctor if you are taking any other medicines, including vitamins and herbal supplements and including medicines you can buy with or without a prescription from a pharmacy, supermarket or health food shop.
These medicines may be affected by DARZALEX or may affect how well DARZALEX works. Your doctor or pharmacist can tell you what to do if you are using any other medicines.
How DARZALEX is given
How much is given:
Your doctor will work out your dose and schedule of DARZALEX. The dose of DARZALEX will depend on your body weight. The usual starting dose of DARZALEX is 16 mg per kg of body weight.
DARZALEX may be given alone or together with other medicines used to treat multiple myeloma.
Ask your doctor if you want to know more about the dose of DARZALEX you receive.
How it is given:
DARZALEX will be given to you by a doctor or nurse.
DARZALEX will be diluted in sterile normal sodium chloride (salt) solution for injection.
It is given over several hours as a drip into a vein ("intravenous infusion").
When it is given:
Your doctor will tell you when DARZALEX will be given. The frequency depends on whether it is given alone or together with other medicines used to treat multiple myeloma.
Medicines given during treatment with DARZALEX:
DARZALEX may be given alone or together with other medicines used to treat multiple myeloma (with bortezomib, thalidomide and dexamethasone, or with bortezomib, melphalan and prednisone, or with lenalidomide and dexamethasone or with bortezomib and dexamethasone).
Read the Consumer Medicine Information for all medicines you take in combination with DARZALEX.
You may be given medicines to lower the chance of getting shingles.
Before each infusion of DARZALEX you will be given medicines which help to lower the chance of infusion-related reactions. These may include:
- medicines for an allergic reaction (anti-histamines)
- medicines for inflammation (corticosteroids)
- medicines for fever (such as paracetamol).
After each infusion of DARZALEX you will be given medicines by mouth (such as corticosteroids) to lower the chance of infusion-related reactions.
People with breathing problems:
If you have breathing problems, such as asthma or Chronic Obstructive Pulmonary Disease (COPD), you may be given medicines to inhale which help your breathing problems:
- medicines to help the airways in your lungs stay open (bronchodilators)
- medicines to lower swelling and irritation in your lungs (corticosteroids)
What do I do if I receive too much? (overdose):
This medicine will be given by your doctor or nurse. In the unlikely event that you are given too much (an overdose) your doctor will check you for side effects.
If you forget your appointment to have DARZALEX:
It is very important to go to all your appointments to make sure your treatment works. If you miss an appointment, make another one as soon as possible.
If you have any further questions on the use of this medicine, ask your doctor or nurse.
While you are using DARZALEX
Things you must do:
Be sure to keep all your doctor's appointments so your progress can be checked. Your doctor will want to do some blood, urine and other tests from time to time to check on your progress and detect any unwanted side effects.
Keep follow up appointments with your doctor. It is important to have your follow-up doses of DARZALEX at the appropriate times to get the best effects from your treatment.
Be sure to follow your doctor's instructions about other medicines you should take, and other things you should do.
Tell any other doctors, dentists and pharmacists who are treating you that you are having DARZALEX.
If you are about to be started on any new medicines, tell your doctor, dentist or pharmacist that you are having DARZALEX.
If you become pregnant or your partner becomes pregnant while being given DARZALEX, tell your doctor immediately.
If you need to have a blood test, tell the person doing the test that you are using DARZALEX.
Decreased blood cell counts
DARZALEX can decrease white blood cell counts which help fight infections, and blood cells called platelets which help to clot blood.
Tell your doctor if you develop fever or if you have signs of bruising or bleeding.
The following precautions should be taken to reduce your risk of infection or bleeding:
- Avoid people who have infections. Check with your doctor immediately if you think you may be getting an infection, or if you get a fever, chills, cough, hoarse throat, lower back or side pain or find it is painful or difficult to urinate.
- Be careful when using a toothbrush, toothpick or dental floss. Your doctor, dentist, nurse or pharmacist may recommend other ways to clean your teeth and gums. Check with your doctor before having any dental work.
- Be careful not to cut yourself when you are using sharp objects such as a razor or nail cutters.
Blood transfusions
If you need a blood transfusion, you will have a blood test first to match your blood type. DARZALEX can affect the results of this blood test. Tell the person doing the test that you are using DARZALEX.
Hepatitis B
If you have ever had a hepatitis B infection, DARZALEX could cause hepatitis B virus to become active again. Your doctor will check you for signs of this infection before, during and for some time after treatment with DARZALEX.
Tell your doctor right away if you get increasingly tired or get yellowing of your skin or white part of your eyes.
Infusion-related reactions
Before and after each infusion of DARZALEX, you will be given medicines which help to lower the chance of infusion-related reactions (see “Medicines given during treatment with DARZALEX”). Some serious allergic reactions and other severe infusion related reactions have resulted in death. These reactions can happen during the infusion or in the 3 days after the infusion.
Tell your doctor or nurse right away if you get any of the infusion-related reactions listed at the top of the Side Effects section.
If you get infusion-related reactions, you may need other medicines, or the infusion may need to be slowed down or stopped. When these reactions go away, or get better the infusion can be started again.
These reactions are most likely to happen with the first infusion. If you have had an infusion-related reaction once it is less likely to happen again. Your doctor may decide not to continue DARZALEX if you have a strong infusion-related reaction.
Things to be careful of
Be careful driving or operating machinery until you know how DARZALEX affects you. DARZALEX may cause tiredness and other effects in some people. Make sure you know how you react to DARZALEX before you drive a car, operate machinery, or do anything else that could be dangerous if you have side effects.
Side Effects
Like all medicines, DARZALEX can cause side effects, although not everybody gets them. Some of these effects may be serious. However, there may be ways to reduce the discomfort of these effects. You may need medical treatment if you get some of the side effects.
Tell your doctor, nurse or pharmacist as soon as possible if you do not feel well while you are being treated with DARZALEX.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Below is a list of the more common side effects that you could get while being treated with DARZALEX:
- Infusion-related reactions
Tell your doctor or nurse right away if you get any of the following signs of an infusion-related reaction during, or in the 3 days after, the infusion. You may need other medicines, or the infusion may need to be slowed down or stopped.
- chills
- sore throat, cough
- nausea
- vomiting
- itchy, runny or blocked nose
- feeling short of breath or other breathing problems
- chest discomfort
- dizziness or light-headedness (hypotension)
- itching
- wheezing
- chest pain
- eye pain
- blurred vision
If you get any of the infusion-related reactions above, tell your doctor or nurse right away.
- Other side effects
- fever
- chills
- feeling very tired
- feeling weak
- flu
- headache
- diarrhoea
- constipation
- nerve damage that may cause tingling, numbness or pain
- high blood pressure
- muscle spasms
- swollen hands, ankles or feet
- lung infection (pneumonia)
- bronchitis
- build-up of fluid in the lungs making you short of breath
- infections of the airways – such as nose, sinuses or throat
- low number of red blood cells which carry oxygen in the blood (anaemia)
- low number of white blood cells which help fight infections (neutropenia, lymphopenia, leukopenia)
- low number of a type of blood cell called platelets which help to clot blood (thrombocytopenia)
- high level of sugar in the blood
- low level of calcium in the blood
- low level of antibodies called 'immunoglobulins' in the blood which help fight infections (hypogammaglobulinemia).
- inflamed pancreas
- irregular heart beat (atrial fibrillation)
- urinary tract infection
- dehydration
- fainting
- chest pain
- other pain (eg back, joints)
- inflamed liver (hepatitis)
- small blisters in clusters on the skin (herpes)
- a type of herpes virus infection (cytomegalovirus infection)
- rash, itching
- unusual feeling in the skin (such as tingling or crawling feeling)
- redness of the skin or redness and pain at injection site
- decreased appetite
- severe infection throughout the body (sepsis).
- COVID-19 infection
If you think you are having an allergic reaction to DARZALEX, tell you doctor immediately or go to Accident and Emergency at your nearest hospital.
Symptoms usually include some or all of the following:
- rash, itching or hives on the skin
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
Other side effects not listed above may also occur in some people.
Tell your doctor, nurse or pharmacist if you notice any other effect that is making you feel unwell.
Product Description
Storage
Unopened vials:
DARZALEX should be stored in a refrigerator (2 degrees to 8 degrees Celsius).
Do not freeze.
Store in the original package to protect from light.
Keep this medicine out of the sight and reach of children.
What it looks like:
DARZALEX is available as a colourless to yellow, preservative free liquid concentrate for intravenous infusion after dilution.
DARZALEX is supplied as a carton pack containing one single-use glass vial.
Ingredients
Active ingredient:
- daratumumab 100 mg
(for DARZALEX 20mg/mL 5mL vials) OR - daratumumab 400 mg
(for DARZALEX 20mg/mL 20mL vials)
Other ingredients:
- glacial acetic acid
- mannitol
- polysorbate 20
- sodium acetate trihydrate
- sodium chloride
- water for injections.
Sponsor
JANSSEN-CILAG Pty Ltd
1-5 Khartoum Rd
Macquarie Park NSW 2113 Australia
Telephone: 1800 226 334
Registration numbers
DARZALEX 100 mg/5mL (AUST R 281842)
DARZALEX 400 mg/20mL (AUST R 281843)
This leaflet was prepared 28 October 2022.
Published by MIMS December 2022

 Bortezomib is administered by subcutaneous (SC) injection or intravenous (IV) injection at a dose of 1.3 mg/m2 body surface area twice weekly for two weeks (Days 1, 4, 8, and 11) of repeated 28-day (4-week) induction treatment cycles (Cycles 1-4) and two consolidation cycles (Cycles 5 and 6) following ASCT after Cycle 4.
Bortezomib is administered by subcutaneous (SC) injection or intravenous (IV) injection at a dose of 1.3 mg/m2 body surface area twice weekly for two weeks (Days 1, 4, 8, and 11) of repeated 28-day (4-week) induction treatment cycles (Cycles 1-4) and two consolidation cycles (Cycles 5 and 6) following ASCT after Cycle 4. Bortezomib is given twice weekly at Weeks 1, 2, 4 and 5 for the first 6-week cycle (8 doses), followed by once weekly at Weeks 1, 2, 4 and 5 for eight additional 6-week cycles (32 additional doses for a total of 40 doses). For information on the VMP dose and dosing schedule when administered with Darzalex, see Section 5.1 Pharmacodynamic Properties, Clinical trials.
Bortezomib is given twice weekly at Weeks 1, 2, 4 and 5 for the first 6-week cycle (8 doses), followed by once weekly at Weeks 1, 2, 4 and 5 for eight additional 6-week cycles (32 additional doses for a total of 40 doses). For information on the VMP dose and dosing schedule when administered with Darzalex, see Section 5.1 Pharmacodynamic Properties, Clinical trials. For dosing instructions for medicinal products administered with Darzalex see Section 5.1 Pharmacodynamic Properties, Clinical trials and manufacturer's Product Information.
For dosing instructions for medicinal products administered with Darzalex see Section 5.1 Pharmacodynamic Properties, Clinical trials and manufacturer's Product Information. For dosing instructions of medicinal products administered with Darzalex, see Section 5.1 Pharmacodynamic Properties, Clinical trials and manufacturer's Product Information. See Table 5.
For dosing instructions of medicinal products administered with Darzalex, see Section 5.1 Pharmacodynamic Properties, Clinical trials and manufacturer's Product Information. See Table 5.

 Laboratory abnormalities worsening during treatment from baseline listed in Table 8.
Laboratory abnormalities worsening during treatment from baseline listed in Table 8.
 Laboratory abnormalities worsening during treatment from baseline listed in Table 10.
Laboratory abnormalities worsening during treatment from baseline listed in Table 10.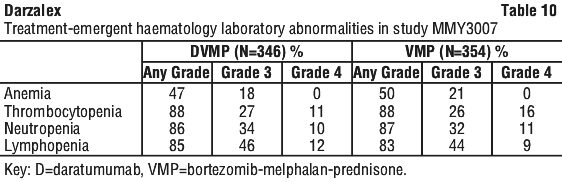
 Laboratory abnormalities worsening during treatment from baseline listed in Table 12.
Laboratory abnormalities worsening during treatment from baseline listed in Table 12.
 Laboratory abnormalities worsening during treatment are listed in Table 14.
Laboratory abnormalities worsening during treatment are listed in Table 14.
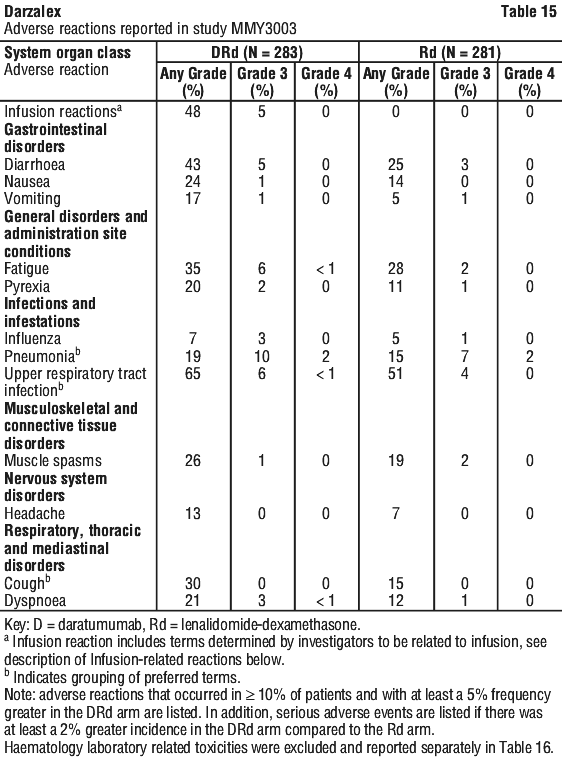 Laboratory abnormalities worsening during treatment from baseline are listed in Table 16.
Laboratory abnormalities worsening during treatment from baseline are listed in Table 16.
 Laboratory abnormalities worsening during treatment from baseline are listed in Table 18.
Laboratory abnormalities worsening during treatment from baseline are listed in Table 18.
 Laboratory abnormalities worsening during treatment from baseline listed in Table 20.
Laboratory abnormalities worsening during treatment from baseline listed in Table 20.



 A total of 1085 patients were randomised: 543 to the DVTd arm and 542 to the VTd arm. The baseline demographic and disease characteristics were similar between the two treatment groups. The median age was 58 (range: 22 to 65 years). The majority were male (59%), 48% had an Eastern Cooperative Oncology Group (ECOG) performance score of 0, 42% had an ECOG performance score of 1 and 10% had an ECOG performance score of 2. Forty percent had ISS Stage I, 45% had ISS Stage II and 15% had ISS Stage III disease.
A total of 1085 patients were randomised: 543 to the DVTd arm and 542 to the VTd arm. The baseline demographic and disease characteristics were similar between the two treatment groups. The median age was 58 (range: 22 to 65 years). The majority were male (59%), 48% had an Eastern Cooperative Oncology Group (ECOG) performance score of 0, 42% had an ECOG performance score of 1 and 10% had an ECOG performance score of 2. Forty percent had ISS Stage I, 45% had ISS Stage II and 15% had ISS Stage III disease. With a median follow-up of 18.8 months, the primary analysis of PFS in Study MMY3006 demonstrated an improvement in Progression Free Survival (PFS) in the DVTd arm as compared to the VTd arm; the median PFS had not been reached in either arm. Treatment with DVTd resulted in a reduction in the risk of progression or death by 53% compared to VTd alone (HR = 0.47; 95% CI: 0.33, 0.67; p < 0.0001). Results of an updated PFS analysis after a median follow-up of 44.5 months showed that median PFS was not reached in the DVTd arm and was 51.5 months in the VTd arm (HR = 0.58; 95% CI: 0.47, 0.71; p < 0.0001). See Figure 1.
With a median follow-up of 18.8 months, the primary analysis of PFS in Study MMY3006 demonstrated an improvement in Progression Free Survival (PFS) in the DVTd arm as compared to the VTd arm; the median PFS had not been reached in either arm. Treatment with DVTd resulted in a reduction in the risk of progression or death by 53% compared to VTd alone (HR = 0.47; 95% CI: 0.33, 0.67; p < 0.0001). Results of an updated PFS analysis after a median follow-up of 44.5 months showed that median PFS was not reached in the DVTd arm and was 51.5 months in the VTd arm (HR = 0.58; 95% CI: 0.47, 0.71; p < 0.0001). See Figure 1.
 A total of 706 patients were randomised; 350 to the DVMP arm and 356 to the VMP arm. The baseline demographic and disease characteristics were similar between the two treatment groups. The median age was 71 (range: 40-93) years, with 30% of the patients ≥ 75 years of age. The majority were White (85%), female (54%), 25% and had an ECOG performance score of 0, 50% had an ECOG performance score of 1 and 25% had an ECOG performance score of 2. Patients had IgG/IgA/light chain myeloma in 64%/22%/10% of instances, 19% had ISS Stage I, 42% had ISS Stage II and 38% had ISS Stage III disease. Efficacy was evaluated by PFS based on IMWG criteria.
A total of 706 patients were randomised; 350 to the DVMP arm and 356 to the VMP arm. The baseline demographic and disease characteristics were similar between the two treatment groups. The median age was 71 (range: 40-93) years, with 30% of the patients ≥ 75 years of age. The majority were White (85%), female (54%), 25% and had an ECOG performance score of 0, 50% had an ECOG performance score of 1 and 25% had an ECOG performance score of 2. Patients had IgG/IgA/light chain myeloma in 64%/22%/10% of instances, 19% had ISS Stage I, 42% had ISS Stage II and 38% had ISS Stage III disease. Efficacy was evaluated by PFS based on IMWG criteria. After a median follow-up of 40 months, an improvement in overall survival (OS) was demonstrated for the DVMP arm (83 deaths, 23.7%) as compared to the VMP arm (126 deaths, 35.6%) (HR = 0.60; 95% CI: 0.46, 0.80; p = 0.0003), representing a 40% reduction in the risk of death in patients treated in the DVMP arm. Median OS was not reached for either arm. See Figure 3.
After a median follow-up of 40 months, an improvement in overall survival (OS) was demonstrated for the DVMP arm (83 deaths, 23.7%) as compared to the VMP arm (126 deaths, 35.6%) (HR = 0.60; 95% CI: 0.46, 0.80; p = 0.0003), representing a 40% reduction in the risk of death in patients treated in the DVMP arm. Median OS was not reached for either arm. See Figure 3. Additional efficacy results from Study MMY3007 are presented in Table 27.
Additional efficacy results from Study MMY3007 are presented in Table 27. In responders, the median time to response was 0.79 months (range: 0.4 to 15.5 months) in the DVMP group and 0.82 months (range: 0.7 to 12.6 months) in the VMP group. The median duration of response had not been reached in the DVMP group and was 21.3 months (range: 18.4, not estimable) in the VMP group.
In responders, the median time to response was 0.79 months (range: 0.4 to 15.5 months) in the DVMP group and 0.82 months (range: 0.7 to 12.6 months) in the VMP group. The median duration of response had not been reached in the DVMP group and was 21.3 months (range: 18.4, not estimable) in the VMP group. A total of 737 patients were randomised: 368 to the DRd arm and 369 to the Rd arm. The baseline demographic and disease characteristics were similar between the two treatment groups. The median age was 73 (range: 45-90) years, with 44% of the patients ≥ 75 years of age. The majority were White (92%), male (52%), 34% had an ECOG performance score of 0, 50% had an ECOG performance score of 1 and 17% had an ECOG performance score of ≥ 2. Twenty-seven percent had International Staging System (ISS) Stage I, 43% had ISS Stage II and 29% had ISS Stage III disease. Efficacy was evaluated by progression free survival (PFS) based on IMWG criteria.
A total of 737 patients were randomised: 368 to the DRd arm and 369 to the Rd arm. The baseline demographic and disease characteristics were similar between the two treatment groups. The median age was 73 (range: 45-90) years, with 44% of the patients ≥ 75 years of age. The majority were White (92%), male (52%), 34% had an ECOG performance score of 0, 50% had an ECOG performance score of 1 and 17% had an ECOG performance score of ≥ 2. Twenty-seven percent had International Staging System (ISS) Stage I, 43% had ISS Stage II and 29% had ISS Stage III disease. Efficacy was evaluated by progression free survival (PFS) based on IMWG criteria. After a median follow-up of 56 months, an improvement in OS was demonstrated for the DRd arm (117 deaths, 31.8%) as compared to the Rd arm (156 deaths, 42.3%) (HR = 0.68; 95% CI: 0.53, 0.86; p = 0.0013), representing a 32% reduction in the risk of death in patients treated in the DRd arm. Median OS was not reached for either arm. The 60 month survival rate was 66% (95% CI: 61, 71) in the DRd arm and was 53% (95% CI: 47, 59) in the Rd arm. See Figure 5.
After a median follow-up of 56 months, an improvement in OS was demonstrated for the DRd arm (117 deaths, 31.8%) as compared to the Rd arm (156 deaths, 42.3%) (HR = 0.68; 95% CI: 0.53, 0.86; p = 0.0013), representing a 32% reduction in the risk of death in patients treated in the DRd arm. Median OS was not reached for either arm. The 60 month survival rate was 66% (95% CI: 61, 71) in the DRd arm and was 53% (95% CI: 47, 59) in the Rd arm. See Figure 5. Additional efficacy results from Study MMY3008 are presented in Table 29.
Additional efficacy results from Study MMY3008 are presented in Table 29. In responders, the median time to response was 1.05 months (range: 0.2 to 12.1 months) in the DRd group and 1.05 months (range: 0.3 to 15.3 months) in the Rd group. The median duration of response had not been reached in the DRd group and was 34.7 months (95% CI: 30.8, not estimable) in the Rd group.
In responders, the median time to response was 1.05 months (range: 0.2 to 12.1 months) in the DRd group and 1.05 months (range: 0.3 to 15.3 months) in the Rd group. The median duration of response had not been reached in the DRd group and was 34.7 months (95% CI: 30.8, not estimable) in the Rd group. A total of 498 patients were randomised; 251 to the DVd arm and 247 to the Vd arm. The baseline demographic and disease characteristics were similar between the Darzalex and the control arm. The median patient age was 64 years (range 30 to 88 years); 12% were ≥ 75 years, 57% were male; 87% Caucasian, 5% Asian and 4% African American. Patients had received a median of 2 prior lines of therapy and 61% of patients had received prior autologous stem cell transplantation (ASCT). Sixty-nine percent (69%) of patients had received a prior PI (66% received bortezomib) and 76% of patients received an IMiD (42% received lenalidomide). At baseline, 32% of patients were refractory to the last line of treatment and the proportions of patients refractory to any specific prior therapy were well balanced between the treatment groups. Thirty-three percent (33%) of patients were refractory to an IMiD only, and 28% were refractory to lenalidomide. Efficacy was evaluated by PFS based on IMWG criteria.
A total of 498 patients were randomised; 251 to the DVd arm and 247 to the Vd arm. The baseline demographic and disease characteristics were similar between the Darzalex and the control arm. The median patient age was 64 years (range 30 to 88 years); 12% were ≥ 75 years, 57% were male; 87% Caucasian, 5% Asian and 4% African American. Patients had received a median of 2 prior lines of therapy and 61% of patients had received prior autologous stem cell transplantation (ASCT). Sixty-nine percent (69%) of patients had received a prior PI (66% received bortezomib) and 76% of patients received an IMiD (42% received lenalidomide). At baseline, 32% of patients were refractory to the last line of treatment and the proportions of patients refractory to any specific prior therapy were well balanced between the treatment groups. Thirty-three percent (33%) of patients were refractory to an IMiD only, and 28% were refractory to lenalidomide. Efficacy was evaluated by PFS based on IMWG criteria. After a median follow-up of 73 months, an improvement in OS was demonstrated for the DVd arm (148 deaths, 59.0%) as compared to the Vd arm (171 deaths, 69.2%) (HR = 0.74; 95% CI: 0.59, 0.92; p = 0.0075), representing a 26% reduction in the risk of death in patients treated in the DVd arm. The median OS was 49.6 months (95% CI: 42.2, 62.3) in the DVd arm and 38.5 months (95% CI: 31.2, 46.2) in the Vd arm. The 72-month survival rate was 39% (95% CI: 33, 45) in the DVd arm and was 25% (95% CI: 20, 31) in the Vd arm. See Figure 7.
After a median follow-up of 73 months, an improvement in OS was demonstrated for the DVd arm (148 deaths, 59.0%) as compared to the Vd arm (171 deaths, 69.2%) (HR = 0.74; 95% CI: 0.59, 0.92; p = 0.0075), representing a 26% reduction in the risk of death in patients treated in the DVd arm. The median OS was 49.6 months (95% CI: 42.2, 62.3) in the DVd arm and 38.5 months (95% CI: 31.2, 46.2) in the Vd arm. The 72-month survival rate was 39% (95% CI: 33, 45) in the DVd arm and was 25% (95% CI: 20, 31) in the Vd arm. See Figure 7. Additional efficacy results from Study MMY3004 are presented in Table 31.
Additional efficacy results from Study MMY3004 are presented in Table 31.
 A total of 569 patients were randomised; 286 to the DRd arm and 283 to the Rd arm. The baseline demographic and disease characteristics were similar between the Darzalex and the control arm. The median patient age was 65 years (range 34 to 89 years), 11% were ≥ 75 years, 59% were male; 69% Caucasian, 18% Asian, and 3% African American. Patients had received a median of 1 prior line of therapy. Sixty-three percent (63%) of patients had received prior autologous stem cell transplantation (ASCT). The majority of patients (86%) received a prior proteasome inhibitor (PI), 55% of patients had received a prior immunomodulatory agent (IMiD), including 18% of patients who had received prior lenalidomide, and 44% of patients had received both a prior PI and IMiD. At baseline, 27% of patients were refractory to the last line of treatment. Eighteen percent (18%) of patients were refractory to a PI only, and 21% were refractory to bortezomib. Efficacy was evaluated by PFS based on IMWG criteria.
A total of 569 patients were randomised; 286 to the DRd arm and 283 to the Rd arm. The baseline demographic and disease characteristics were similar between the Darzalex and the control arm. The median patient age was 65 years (range 34 to 89 years), 11% were ≥ 75 years, 59% were male; 69% Caucasian, 18% Asian, and 3% African American. Patients had received a median of 1 prior line of therapy. Sixty-three percent (63%) of patients had received prior autologous stem cell transplantation (ASCT). The majority of patients (86%) received a prior proteasome inhibitor (PI), 55% of patients had received a prior immunomodulatory agent (IMiD), including 18% of patients who had received prior lenalidomide, and 44% of patients had received both a prior PI and IMiD. At baseline, 27% of patients were refractory to the last line of treatment. Eighteen percent (18%) of patients were refractory to a PI only, and 21% were refractory to bortezomib. Efficacy was evaluated by PFS based on IMWG criteria. After a median follow-up of 80 months, an improvement in OS was demonstrated for the DRd arm (153 deaths, 53.5%) as compared to the Rd arm (175 deaths, 61.8%) (HR = 0.73; 95% CI: 0.58, 0.91; p = 0.0044), representing a 27% reduction in the risk of death in patients treated in the DRd arm. The median OS was 67.6 months (95% CI: 53.1, 80.5) in the DRd arm and 51.8 months (95% CI: 44.0, 60.0) in the Rd arm. The 78-month survival rate was 47% (95% CI: 41, 52) in the DRd arm and was 35% (95% CI: 30, 41) in the Rd arm. See Figure 9.
After a median follow-up of 80 months, an improvement in OS was demonstrated for the DRd arm (153 deaths, 53.5%) as compared to the Rd arm (175 deaths, 61.8%) (HR = 0.73; 95% CI: 0.58, 0.91; p = 0.0044), representing a 27% reduction in the risk of death in patients treated in the DRd arm. The median OS was 67.6 months (95% CI: 53.1, 80.5) in the DRd arm and 51.8 months (95% CI: 44.0, 60.0) in the Rd arm. The 78-month survival rate was 47% (95% CI: 41, 52) in the DRd arm and was 35% (95% CI: 30, 41) in the Rd arm. See Figure 9. Additional efficacy results from Study MMY3003 are presented in Table 33.
Additional efficacy results from Study MMY3003 are presented in Table 33.
 After a median follow-up of 17.1 months, 95 deaths were observed [N=59 (19%) in the DKd group and N=36 (23%) in the Kd group]. Overall survival (OS) data were not mature, however, there was a trend toward longer OS in the DKd arm compared with the Kd arm (HR=0.745; 95% CI: 0.491, 1.131; p=0.0836).
After a median follow-up of 17.1 months, 95 deaths were observed [N=59 (19%) in the DKd group and N=36 (23%) in the Kd group]. Overall survival (OS) data were not mature, however, there was a trend toward longer OS in the DKd arm compared with the Kd arm (HR=0.745; 95% CI: 0.491, 1.131; p=0.0836). In responders, the median time to response was 1 month (range: 1 to 14 months) in the DKd group and 1 month (range: 1 to 10 months) in the Kd group. The median duration of response had not been reached in the DKd group and was 16.6 months (95% CI: 13.9, not estimable) in the Kd group.
In responders, the median time to response was 1 month (range: 1 to 14 months) in the DKd group and 1 month (range: 1 to 10 months) in the Kd group. The median duration of response had not been reached in the DKd group and was 16.6 months (95% CI: 13.9, not estimable) in the Kd group.
 Overall response rate (ORR) in MMY2002 was similar regardless of type of prior anti-myeloma therapy. At a survival update with a median duration of follow up of 14.7 months, median OS was 17.5 months (95% CI: 13.7, not estimable).
Overall response rate (ORR) in MMY2002 was similar regardless of type of prior anti-myeloma therapy. At a survival update with a median duration of follow up of 14.7 months, median OS was 17.5 months (95% CI: 13.7, not estimable).
