What is in this leaflet
This leaflet answers some common questions about DBL Bortezomib Powder for Injection. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking DBL Bortezomib Powder for Injection against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What DBL Bortezomib Powder for Injection is used for
DBL Bortezomib Powder for Injection is used to treat
- multiple myeloma (cancer of the bone marrow)
- mantle cell lymphoma (a type of cancer affecting the lymph nodes)
This medicine belongs to a group of drugs called antineoplastic or cytotoxic medicines.
These medicines are used to kill cancer cells.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
This medicine is not addictive and is available only with a doctor's prescription.
Before you are given DBL Bortezomib Powder for Injection
When you must not be given it
Do not use DBL Bortezomib Powder for Injection if you have an allergy to:
- bortezomib, boron or mannitol.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Tell your doctor if you are pregnant or intend to become pregnant. It may affect your developing baby if you use DBL Bortezomib Powder for Injection during pregnancy. It is not recommended for use during pregnancy.
Tell your doctor if you are breastfeeding or intend to breastfeed. It is not known whether DBL Bortezomib Powder for Injection passes into breast milk. Therefore there is a possibility that your baby may be affected.
If you wish to restart breast-feeding after your DBL Bortezomib Powder for Injection treatment, you must discuss this with your doctor or nurse, who will tell you when it is safe to do so.
Before you start to use it
Tell your doctor if you have or have had any of the following medical conditions:
- blood disorder with a low level of red or white blood cells or platelets. This disorder may become worse during treatment with DBL Bortezomib Powder for Injection.
- if you are suffering from diarrhoea or vomiting as this may become worse during treatment with DBL Bortezomib Powder for Injection.
- a history of fainting, dizziness or light-headedness
- kidney problems
- liver problems, including hepatitis infection
- problems with numbness, tingling or pain in the hands or feet (neuropathy). This effect may be worsened by treatment with DBL Bortezomib Powder for Injection
- seizures
- any bleeding problems
- problems with your heart
- lung or breathing problems
Tell your doctor if you are pregnant or plan to become pregnant.
Tell your doctor if you are trying to make your partner pregnant. Both men and women receiving DBL Bortezomib Powder for Injection and their partners must use a reliable method of contraception while receiving DBL Bortezomib Powder for Injection.
If you have not told your doctor about any of the above, tell him/ her before you start your treatment with DBL Bortezomib Powder for Injection.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and DBL Bortezomib Powder for Injection may interfere with each other. These include:
- amiodarone, a medicine used to treat irregular heart beat
- medicines used to treat viral infections such as flu, herpes and HIV
- isoniazid, a medicine used to treat tuberculosis
- nitrofurantoin, a medicine used to treat urinary tract infections
- ketoconazole, a medicine used to treat fungal infections
- ritonavir, a medicine used to treat HIV infection
- rifampicin, a medicine used to treat infections such as tuberculosis
- medicines used to treat high cholesterol levels in the blood
- medicines used to treat diabetes
- medicines that may lower blood pressure
- medicine used to treat epilepsy such as carbamazepine and phenobarbital
- phenytoin, a medicine used in preventing seizures
- St John's Wort (Hypericum perforatum).
These medicines may be affected by DBL Bortezomib Powder for Injection or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How DBL Bortezomib Powder for Injection is given
Your treatment with DBL Bortezomib Powder for Injection will be done under the supervision of a doctor. This medicine will be given by a healthcare professional (eg doctor or nurse) experienced in giving this type of 'oncology' medicine.
How much is given:
Your doctor will decide how much you will receive (the dose). This dose will be calculated from your height and weight. It will also depend on how other parts of your body are functioning, such as kidney and liver and what other medicines you are taking.
Your doctor may change the dose during treatment depending on your response.
Ask your doctor or pharmacist if you want to know more about the dose of DBL Bortezomib Powder for Injection you are receiving.
How it is given:
DBL Bortezomib Powder for Injection will be dissolved in a sterile solution for injection. The solution is given as an injection into your vein (intravenously) over 3 to 5 seconds.
The solution can also be given subcutaneously as an injection into your thighs or stomach.
When it is given:
Multiple Myeloma
One cycle of treatment with DBL Bortezomib Powder for Injection may consist of a total of 4 doses given over 3 weeks. Doses are given on days 1, 4, 8 and 11 followed by a ten day break from the treatment.
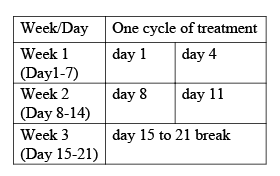
Note day 1 is the first dose you will receive your DBL Bortezomib Powder for Injection in a treatment cycle. Subsequent doses for the same treatment cycle will be counted from day 1, e.g. your 3rd dose will be the 8th day (day 8).
When DBL Bortezomib Powder for Injection is given with thalidomide and dexamethasone, the treatment consists of a total of 3 cycles (9 weeks) for the induction stage. During the induction stage, DBL Bortezomib Powder for Injection is administered twice weekly (days 1, 4, 8 and 11).
When DBL Bortezomib Powder for Injection is given with dexamethasone; the treatment consists of a total of 4 cycles (12 weeks). DBL Bortezomib Powder for Injection will be administered twice weekly (days 1, 4, 8 and 11).
When DBL Bortezomib Powder for Injection is given with melphalan and prednisone, one cycle of treatment is 6 weeks and the treatment consists of a total of 9 cycles (54 weeks). In Cycles 1-4, DBL Bortezomib Powder for Injection is administered twice weekly (days 1, 4, 8, 11, 22, 25, 29and 32). In Cycles 5-9, DBL Bortezomib Powder for Injection is administered once weekly (days (1, 8, 22 and 29).
Mantle Cell Lymphoma
When DBL Bortezomib Powder for Injection is given with rituximab, cyclophosphamide, doxorubicin and prednisone, one cycle is 3 weeks and the treatment consists of a total of up to 8 cycles (24 weeks). For each cycle, DBL Bortezomib Powder for Injection is given on days 1, 4, 8 and 11, followed by a ten day break from the treatment.
Your doctor will decide on the number of cycles of DBL Bortezomib Powder for Injection needed. This will depend on how you respond to treatment.
If you receive too much (overdose)
As DBL Bortezomib Powder for Injection is given to you under the supervision of your doctor; it is very unlikely that you will receive too much.
However if you experience any side effects after being given DBL Bortezomib Powder for Injection, tell your doctor or nurse immediately or go to Accident and Emergency at your nearest hospital. You may need urgent medical attention.
While you are receiving DBL Bortezomib Powder for Injection
Things you must do
It is important to receive your doses of DBL Bortezomib Powder for Injection at the appropriate times according to your treatment cycle to get the best effects from your treatment. This will be explained to you by your doctor or nurse.
Please ensure that you follow your doctor's instructions about other medicines you should take, and other things you should do.
You may need to take other medicines to help prevent unwanted effects of DBL Bortezomib Powder for Injection. You may also need to drink extra fluids if you experience vomiting and/or diarrhoea.
Ask your doctor or pharmacist if you have any questions.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are receiving DBL Bortezomib Powder for Injection.
Tell any other doctors, dentists, and pharmacists who treat you that you are receiving this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests (such as blood or urine) from time to time to make sure the medicine is working and to prevent unwanted side effects.
DBL Bortezomib Powder for Injection can lower the number of white blood cells and platelets in your blood. This means that you have an increased chance of getting an infection or bleeding. The following precautions should be taken to reduce your risk of infection or bleeding:
- Avoid people who have infections. Check with your doctor immediately if you think you may be getting an infection, or if you get a fever, chills, cough, hoarse throat, lower back or side pain or find it’s painful or difficult to urinate.
- Be careful when using a toothbrush, toothpick or dental floss. Your doctor, dentist, nurse or pharmacist may recommend other ways to clean your teeth and gums. Check with your doctor before having any dental work.
- Be careful not to cut yourself when you are using sharp objects such as a razor or nail cutters.
- Avoid contact sports or other situations where you may bruise or get injured.
Things to be careful of
Be careful driving or operating machinery until you know how DBL Bortezomib Powder for Injection affects you. This medicine may cause dizziness, light-headedness, tiredness, fainting, double or blurred vision in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous. Make sure you know how you react to DBL Bortezomib Powder for Injection before you drive a car or operate machinery.
Be careful when drinking alcohol while you are taking this medicine. If you drink alcohol, dizziness or light-headedness) may be worse.
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Side effects
Like other medicines that treat cancer, DBL Bortezomib Powder for Injection may have unwanted side effects, some of which may be serious. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor, nurse or pharmacist as soon as possible if you do not feel well while you are receiving DBL Bortezomib Powder for Injection
Below is a list of side effects that you may experience while receiving DBL Bortezomib Powder for Injection;
- tiredness, generally feeling unwell, weakness
- feeling sick (nausea) or vomiting
- diarrhoea
- constipation
- loss of appetite, and/or weight, fear of gaining weight
- bleeding or bruising more easily than normal
- sensitivity, numbness, tingling or burning sensation of the skin, or pain in the hands or feet
- fever, chills
- anaemia (a condition in which there is a decreased number of red blood cells) e.g. tiredness, headaches, being short of breath when exercising, dizziness and looking pale
- frequent infections such as fever, severe chills, sore throat or mouth ulcers
- herpes virus or hepatitis infections
- headache, trouble sleeping, sweating, anxiety, mood swings, confusion or depression
- painful, swollen joints
- pain in your limbs, back pain, bone pain, muscle cramps
- temporary inflammation of the nerves, causing pain, weakness, and paralysis in the extremities and often progressing to the chest and face
- swelling (around the eyes or in the ankles, wrists, arms, legs or face)
- pins and needles and unpleasant sensations
- difficulty in breathing
- dizziness
- dehydration
- cough
- aching muscles, muscle tenderness or weakness not caused by exercise
- uncomfortable feeling in the stomach or belching after eating
- stomach pain
- blockage in the intestine
- bad taste in the mouth
- low blood pressure (dizziness, light headedness or fainting)
- high blood pressure
- chest pain
- small blisters in clusters on the skin (herpes)
- rash, itching
- redness of the skin or redness and pain at injection site
- hair loss
- blurred vision
- pneumonia
- allergic reaction.
If you think you are having an allergic reaction to DBL Bortezomib Powder for Injection, tell you doctor immediately or go to Accident and Emergency at your nearest hospital.
Symptoms usually include some or all of the following:
- rash, itching or hives on the skin
- shortness of breath, wheezing or difficulty breathing swelling of the face, lips, tongue or other parts of the body.
Other side effects not listed above may also occur in some people.
Tell your doctor, nurse or pharmacist if you notice any other effect that is making you feel unwell.
After using DBL Bortezomib Powder for Injection
Storage
DBL Bortezomib Powder for Injection vials are kept in the outer carton, protected from light and in a cool dry place below 25 °C and will be stored in the pharmacy or on the ward.
Product description
What it looks like
DBL Bortezomib Powder for Injection is a white to off-white powder in a clear glass vial.
Each pack contains one single-use vial.
Before injection, DBL Bortezomib Powder for Injection powder is dissolved in a small quantity of sterile, sodium chloride solution. The solution for injection is clear and colourless.
Ingredients
DBL Bortezomib Powder for Injection contains:
- bortezomib 1 mg
(for DBL Bortezomib Powder for Injection 1 mg Vials)
OR
- bortezomib 2.5 mg
(for DBL Bortezomib Powder for Injection 2.5 mg Vials)
OR
- bortezomib 3 mg
(for DBL Bortezomib Powder for Injection 3 mg Vials)
OR
- bortezomib 3.5 mg
(for DBL Bortezomib Powder for Injection 3.5 mg Vials) as the active ingredient.
Other ingredients:
- mannitol
- nitrogen
Supplier
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
Australian Registration Numbers
DBL Bortezomib Powder for Injection 1 mg vial
(AUST R 313359)
DBL Bortezomib Powder for Injection 2.5 mg vial
(AUST R 275710)
DBL Bortezomib Powder for Injection 3 mg vial
(AUST R 275706)
DBL Bortezomib Powder for Injection 3.5 mg vial
(AUST R 275708).
Date of Preparation
This leaflet was prepared in February 2023.
™ = Trademark
© Pfizer Australia Pty Ltd
Published by MIMS August 2025




 For additional information concerning melphalan and prednisone, see manufacturer's Product Information documents.
For additional information concerning melphalan and prednisone, see manufacturer's Product Information documents.
 In addition, when bortezomib is given in combination with other chemotherapeutic medicinal products, appropriate dose reductions for these medicinal products should be considered in the event of toxicities, according to the recommendations in the respective Product Information documents.
In addition, when bortezomib is given in combination with other chemotherapeutic medicinal products, appropriate dose reductions for these medicinal products should be considered in the event of toxicities, according to the recommendations in the respective Product Information documents.

 In the combination study of bortezomib with rituximab, cyclophosphamide, doxorubicin and prednisone (Bortezomib-R-CAP) in previously untreated mantle cell lymphoma patients, the incidence of thrombocytopenia adverse events (≥ Grade 4) was 32% versus 2% for the rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) arm. The incidence of bleeding adverse events (≥ Grade 3) was 1.7% (4 patients) in the Bortezomib-R-CAP arm and was 1.2% (3 patients) in the R-CHOP arm.
In the combination study of bortezomib with rituximab, cyclophosphamide, doxorubicin and prednisone (Bortezomib-R-CAP) in previously untreated mantle cell lymphoma patients, the incidence of thrombocytopenia adverse events (≥ Grade 4) was 32% versus 2% for the rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) arm. The incidence of bleeding adverse events (≥ Grade 3) was 1.7% (4 patients) in the Bortezomib-R-CAP arm and was 1.2% (3 patients) in the R-CHOP arm. During consolidation therapy of the GIMEMA study, grade 3-4 adverse events were similar to those reported during induction, although rates were much lower. Notably, the rate of grade 3-4 peripheral neuropathy was 1.2% with Bortezomib-TD consolidation compared to 0% with TD consolidation.
During consolidation therapy of the GIMEMA study, grade 3-4 adverse events were similar to those reported during induction, although rates were much lower. Notably, the rate of grade 3-4 peripheral neuropathy was 1.2% with Bortezomib-TD consolidation compared to 0% with TD consolidation.
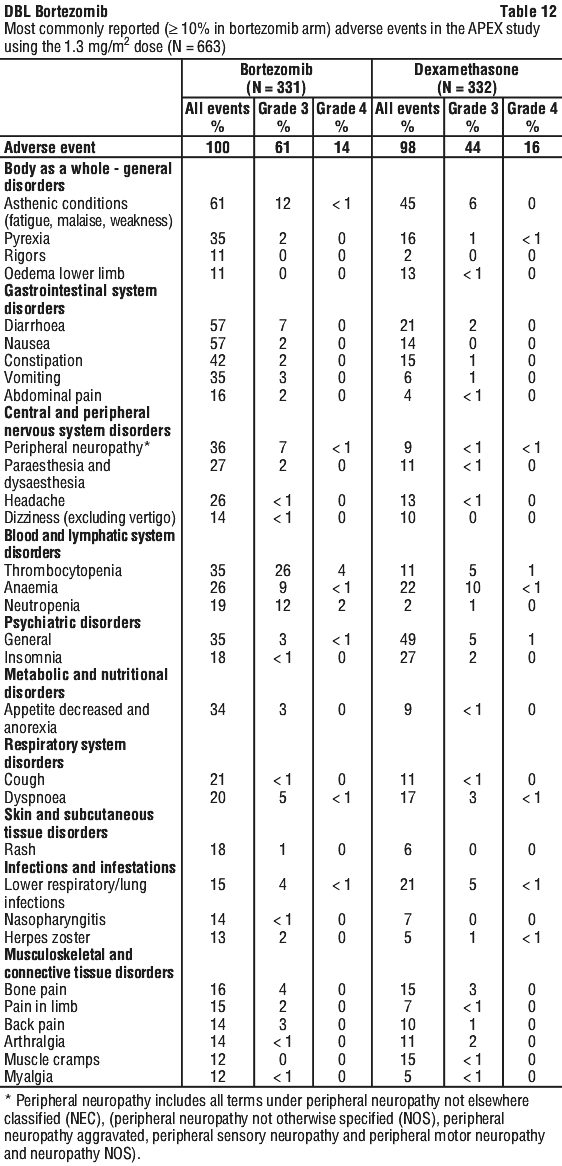
 Although, in general safety data were similar for the IV and SC treatment groups, Table 14 highlights differences larger than 10% in the overall incidence of adverse drug reactions between the two treatment arms.
Although, in general safety data were similar for the IV and SC treatment groups, Table 14 highlights differences larger than 10% in the overall incidence of adverse drug reactions between the two treatment arms. Patients who received bortezomib subcutaneously compared to intravenous administration had 13% lower overall incidence of treatment emergent adverse drug reactions that were grade 3 or higher in toxicity (57% vs. 70% respectively; p-value is 0.0784), and a 5% lower incidence of discontinuation of bortezomib (22% vs. 27%; p-value is 0.5052). The overall incidence of diarrhoea (24% for the SC arm vs. 36% for the IV arm; p-value is 0.0572), gastrointestinal and abdominal pain (6% for the SC arm vs. 19% for the IV arm; p-value is 0.0049), asthenic conditions (27% for SC arm vs. 39% for IV arm), upper respiratory tract infections (14% SC arm vs. 26% IV arm; p-value is 0.0903) and peripheral neuropathy NEC (38% SC arm vs. 53% IV arm; p-value is 0.0444) were 12%-15% lower in the subcutaneous group than the intravenous group. In addition, the incidence of peripheral neuropathies that were grade 3 or higher in toxicity was 10% lower (6% for SC vs. 16% for IV; p-value is 0.0264), and the discontinuation rate due to peripheral neuropathies was 8% lower for the subcutaneous group (5%) as compared to the intravenous group (14%); p-value is 0.0771.
Patients who received bortezomib subcutaneously compared to intravenous administration had 13% lower overall incidence of treatment emergent adverse drug reactions that were grade 3 or higher in toxicity (57% vs. 70% respectively; p-value is 0.0784), and a 5% lower incidence of discontinuation of bortezomib (22% vs. 27%; p-value is 0.5052). The overall incidence of diarrhoea (24% for the SC arm vs. 36% for the IV arm; p-value is 0.0572), gastrointestinal and abdominal pain (6% for the SC arm vs. 19% for the IV arm; p-value is 0.0049), asthenic conditions (27% for SC arm vs. 39% for IV arm), upper respiratory tract infections (14% SC arm vs. 26% IV arm; p-value is 0.0903) and peripheral neuropathy NEC (38% SC arm vs. 53% IV arm; p-value is 0.0444) were 12%-15% lower in the subcutaneous group than the intravenous group. In addition, the incidence of peripheral neuropathies that were grade 3 or higher in toxicity was 10% lower (6% for SC vs. 16% for IV; p-value is 0.0264), and the discontinuation rate due to peripheral neuropathies was 8% lower for the subcutaneous group (5%) as compared to the intravenous group (14%); p-value is 0.0771.
 In addition, compared with the TD arm, Progression Free Survival (PFS) was also significantly longer for patients randomised to the Bortezomib-TD arm (HR, 0.629 [CI: 0.451-0.878], p=0.0061). The estimated 3-year PFS rate was 68% in the Bortezomib-TD arm and 56% in TD (p=0.0057) (see Figure 1). 58 (24.5%) and 86 (36%) patients progressed or died, respectively. The estimated 3-year probability of progression or relapse was 29% in the Bortezomib-TD versus 39% in the TD arm (HR, 0.609 [CI: 0.425-0.873], p=0.0073; p=0.0061 by Kaplan-Meier analysis) (see Figure 2).
In addition, compared with the TD arm, Progression Free Survival (PFS) was also significantly longer for patients randomised to the Bortezomib-TD arm (HR, 0.629 [CI: 0.451-0.878], p=0.0061). The estimated 3-year PFS rate was 68% in the Bortezomib-TD arm and 56% in TD (p=0.0057) (see Figure 1). 58 (24.5%) and 86 (36%) patients progressed or died, respectively. The estimated 3-year probability of progression or relapse was 29% in the Bortezomib-TD versus 39% in the TD arm (HR, 0.609 [CI: 0.425-0.873], p=0.0073; p=0.0061 by Kaplan-Meier analysis) (see Figure 2).
 The IFM-2005, Phase III, randomised (1:1:1:1), multi-centre, open-label study was conducted to compare the efficacy and safety of bortezomib-dexamethasone (Bortezomib-Dex) and vincristine doxorubicin-dexamethasone (VAD) as induction therapy prior to HDT-ASCT, and to evaluate the impact of post-induction consolidation therapy. Patients in this study were randomised to receive VAD plus no consolidation (arm A1), VAD plus dexamethasone, cyclophosphamide, etoposide, cis-platin (DCEP) consolidation (arm A2), Bortezomib-Dex plus no consolidation (arm B1), or Bortezomib-Dex plus DCEP consolidation (arm B2).
The IFM-2005, Phase III, randomised (1:1:1:1), multi-centre, open-label study was conducted to compare the efficacy and safety of bortezomib-dexamethasone (Bortezomib-Dex) and vincristine doxorubicin-dexamethasone (VAD) as induction therapy prior to HDT-ASCT, and to evaluate the impact of post-induction consolidation therapy. Patients in this study were randomised to receive VAD plus no consolidation (arm A1), VAD plus dexamethasone, cyclophosphamide, etoposide, cis-platin (DCEP) consolidation (arm A2), Bortezomib-Dex plus no consolidation (arm B1), or Bortezomib-Dex plus DCEP consolidation (arm B2). A total of 184/218 (84.4%) and 197/223 (88.3%) evaluable patients who received VAD and Bortezomib-Dex induction, respectively, underwent autologus stem cell transplantation. The number of patients who received a second transplantation was 41 (20.8%) in the Bortezomib-Dex arm, compared to 50 (27.2%) for patients in the VAD arm. Post-transplant response rates are shown in Table 18.
A total of 184/218 (84.4%) and 197/223 (88.3%) evaluable patients who received VAD and Bortezomib-Dex induction, respectively, underwent autologus stem cell transplantation. The number of patients who received a second transplantation was 41 (20.8%) in the Bortezomib-Dex arm, compared to 50 (27.2%) for patients in the VAD arm. Post-transplant response rates are shown in Table 18. In addition, the median PFS was 29.7 months among patients who received VAD versus 36.0 months among patients who received Bortezomib-Dex induction, with 128 (52.9%) of 242 and 110 (45.8%) of 240 patients, respectively, having progressed (p=0.064, or p=0.057 if adjusted for initial stratification factors) after median follow-up of 31.2 months.
In addition, the median PFS was 29.7 months among patients who received VAD versus 36.0 months among patients who received Bortezomib-Dex induction, with 128 (52.9%) of 242 and 110 (45.8%) of 240 patients, respectively, having progressed (p=0.064, or p=0.057 if adjusted for initial stratification factors) after median follow-up of 31.2 months. At the time of a pre-specified interim analysis, the primary endpoint, time to progression, was met and patients in the MP arm were offered Bortezomib-MP treatment. Survival continued to be followed after the interim analysis. Median follow-up in the initial analysis (Table 20 and Figure 3) was 16.3 months. Median follow-up in the last survival analysis (Figure 4) was 36.7 months. Median overall survival in the MP arm was 43.1 months and was not reached in the Bortezomib-MP arm. Fifty percent of subjects in the MP arm subsequently received bortezomib.
At the time of a pre-specified interim analysis, the primary endpoint, time to progression, was met and patients in the MP arm were offered Bortezomib-MP treatment. Survival continued to be followed after the interim analysis. Median follow-up in the initial analysis (Table 20 and Figure 3) was 16.3 months. Median follow-up in the last survival analysis (Figure 4) was 36.7 months. Median overall survival in the MP arm was 43.1 months and was not reached in the Bortezomib-MP arm. Fifty percent of subjects in the MP arm subsequently received bortezomib. The time to progression (TTP) was significantly longer on the bortezomib arm (see Figure 3).
The time to progression (TTP) was significantly longer on the bortezomib arm (see Figure 3). A significant survival advantage is shown with bortezomib (see Figure 4).
A significant survival advantage is shown with bortezomib (see Figure 4).


 For the 121 patients achieving a response (CR or PR) on the bortezomib arm, the median duration was 8.0 months (95% CI: 6.9, 11.5 months) compared to 5.6 months (95% CI: 4.8, 9.2 months) for the 56 responders on the dexamethasone arm.
For the 121 patients achieving a response (CR or PR) on the bortezomib arm, the median duration was 8.0 months (95% CI: 6.9, 11.5 months) compared to 5.6 months (95% CI: 4.8, 9.2 months) for the 56 responders on the dexamethasone arm. As shown in Figure 6, bortezomib had a significant survival advantage relative to dexamethasone (p < 0.05). The median follow-up was 8.3 months.
As shown in Figure 6, bortezomib had a significant survival advantage relative to dexamethasone (p < 0.05). The median follow-up was 8.3 months.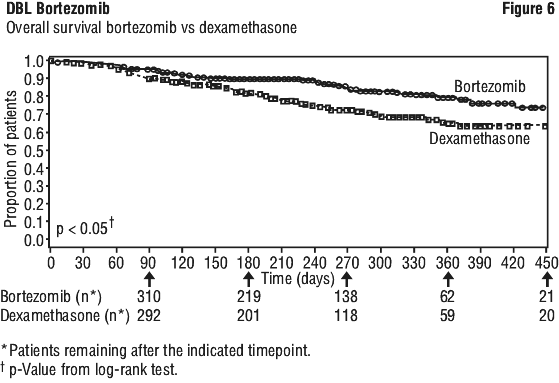
 Table 25 presents a cross-tabulation summary of best response by algorithm after 4 cycles versus after 8 cycles for patients who received dexamethasone. Eighty-two subjects in the SC treatment group and 39 subjects in the IV treatment group received dexamethasone after cycle 4.
Table 25 presents a cross-tabulation summary of best response by algorithm after 4 cycles versus after 8 cycles for patients who received dexamethasone. Eighty-two subjects in the SC treatment group and 39 subjects in the IV treatment group received dexamethasone after cycle 4. Relative to previously reported outcomes, the ORR after 8 cycles of treatment (52% in both treatment groups) and time to progression (median 10.4 months and 9.4 months in SC and IV treatment groups, respectively), including the effect of the addition of dexamethasone from cycle 5 onwards, were higher than observed in prior registration study with single agent IV bortezomib, APEX, (38% ORR and median TTP of 6.2 months for the bortezomib arm). Time to Progression and ORR was also higher compared to the subgroup of patients on APEX that received only 1 prior line of therapy (43% ORR and median TTP of 7.0 months) (Table 23).
Relative to previously reported outcomes, the ORR after 8 cycles of treatment (52% in both treatment groups) and time to progression (median 10.4 months and 9.4 months in SC and IV treatment groups, respectively), including the effect of the addition of dexamethasone from cycle 5 onwards, were higher than observed in prior registration study with single agent IV bortezomib, APEX, (38% ORR and median TTP of 6.2 months for the bortezomib arm). Time to Progression and ORR was also higher compared to the subgroup of patients on APEX that received only 1 prior line of therapy (43% ORR and median TTP of 7.0 months) (Table 23). Patients who did not obtain an optimal response to therapy with bortezomib alone were able to receive high-dose dexamethasone in conjunction with bortezomib (i.e. 40 mg dexamethasone with each dose of bortezomib administered orally as 20 mg on the day of and 20 mg the day after bortezomib administration, (i.e. Days 1, 2, 4, 5, 8, 9, 11, and 12), thus 160 mg over 3 weeks. Eighteen percent (13/74) of patients achieved or had an improved response (CR 11% or PR 7%) with combination treatment.
Patients who did not obtain an optimal response to therapy with bortezomib alone were able to receive high-dose dexamethasone in conjunction with bortezomib (i.e. 40 mg dexamethasone with each dose of bortezomib administered orally as 20 mg on the day of and 20 mg the day after bortezomib administration, (i.e. Days 1, 2, 4, 5, 8, 9, 11, and 12), thus 160 mg over 3 weeks. Eighteen percent (13/74) of patients achieved or had an improved response (CR 11% or PR 7%) with combination treatment.
 Molecular formula: C19H25BN4O4.
Molecular formula: C19H25BN4O4.