What is in this leaflet
This leaflet answers some common questions about Dexmedetomidine Accord.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Dexmedetomidine Accord against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor, nurse or pharmacist.
Keep this leaflet. You may need to read it again.
What Dexmedetomidine Accord is used for
Intensive Care Sedation
Dexmedetomidine Accord can be used as a sedative (calming agent) if adults need to be calm or sleepy in the Intensive Care Unit whilst they are being ventilated (on a breathing machine). It may be given as an infusion up to 24 hours.
Procedural Sedation
Dexmedetomidine Accord can be given to adults prior to an operation if they are not on a ventilator (breathing machine) if it is required for the procedure or surgery that they be sleepy and calm.
This medicine belongs to a group of medicines called alpha-2-receptor agonists. This medicine works by its actions on brain chemicals.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
Dexmedetomidine Accord is only available with a doctor’s prescription.
Before you are given Dexmedetomidine Accord
When you must not be given it
Do not take Dexmedetomidine Accord if you have an allergy to:
- any medicine containing dexmedetomidine hydrochloride
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not give this medicine to a child under the age of 18 years. Safety and effectiveness in children younger than 18 years have not been established.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you are given it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- heart problems
- high or low blood pressure
- diabetes
- kidney or liver problems
Elderly patients greater than 65 years old may be more prone to the blood pressure lowering effects of Dexmedetomidine Accord.
Tell your doctor if you are pregnant or plan to become pregnant or are breast-feeding. The active ingredient in Dexmedetomidine Accord may affect your developing baby if you take it during pregnancy.
It also passes into breast milk and there is a possibility that your baby may be affected.
Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor about any of the above, tell him/her before you start taking Dexmedetomidine Accord.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Dexmedetomidine Accord may interfere with each other. These include:
- medicines used to produce calmness or to help you sleep, such as sevoflurane, isoflurane, propofol, alfentanil and midazolam
- strong pain relievers
These medicines may be affected by Dexmedetomidine Accord or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How Dexmedetomidine Accord is given
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
How much is given
Your doctor will decide what dose you will receive. This depends on your condition and other factors such as your weight. The dose will be adjusted to keep you at the right depth of sleep or sedation.
How it is given
Dexmedetomidine Accord is given by a slow injection (drip) into a vein.
Dexmedetomidine Accord should only be given by a doctor or nurse.
If you are given too much (overdose)
As Dexmedetomidine Accord is given to you under the supervision of your doctor, it is very unlikely that you will receive too much.
Symptoms of an overdose may include extreme drowsiness, confusion, dizziness, weakness or becoming unconscious.
While you are given Dexmedetomidine Accord
Things you must do
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
If you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
Things you must not do
Do not take Dexmedetomidine Accord to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful driving or operating machinery until you know how Dexmedetomidine Accord affects you. This medicine may cause dizziness, light-headedness, tiredness, drowsiness, and therefore affect alertness in some people. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous. Ask your doctor when you can return to work involving driving or operating machinery or heavy equipment.
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Side Effects
Tell your doctor, nurse or pharmacist as soon as possible if you do not feel well while you are taking Dexmedetomidine Accord.
This medicine helps provide sedation for most people, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
If you are over 65 years of age you may have an increased chance of getting side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor, nurse or pharmacist to answer any questions you may have.
Tell your doctor, nurse or pharmacist if you notice any of the following and they worry you:
- dizziness
- light-headedness
- nausea and/or vomiting
- high temperature
- dry mouth
- constipation or diarrhoea
- feelings of agitation, confusion or tiredness
- fluid retention or swelling in the arms or legs
- changes in your blood sugar levels
- increased sweating
- changes to your vision
- reduced urine output or feeling thirsty
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Tell your doctor or nurse as soon as possible if you notice any of the following:
- changes in heart rate including slowing or quickening of heart beat
- shortness of breath, rapid breathing or breathing difficulties
The above list includes serious side effects that may require medical attention.
If any of the following happen, tell your doctor or nurse immediately or go to Accident and Emergency at your nearest hospital.
- pressure, tightness or pain in your chest or arms that may spread to your neck, jaw or back
- unusual bruising
- overheating of your body that you can’t control by normal cooling methods
- excessive thirst, extreme fatigue, lack of energy, confusion, muscle twitching or spasms, restlessness, seizures
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation, and your medicine may need to be stopped.
Tell your doctor, nurse or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Storage
Dexmedetomidine Accord will be stored in the pharmacy or kept on the ward. It is kept in a cool dry place where the temperature stays below 25ºC.
Product Description
What it looks like
Dexmedetomidine Accord is a clear, colourless solution. It is available in 2 mL glass vials in packs of 1, 5 or 25.
Not all pack sizes may be marketed.
Ingredients
Active ingredient:
Dexmedetomidine (as hydrochloride)
Inactive ingredients:
sodium chloride
water for injections
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Name and Address of the Sponsor
Accord Healthcare Pty Ltd
Level 24, 570 Bourke Street
Melbourne, VIC, 3000
Australia
Australian Registration Numbers
200 micrograms/2 mL: AUST R 307241
Date of Preparation
This leaflet was prepared on 18 March 2020.
Published by MIMS October 2020

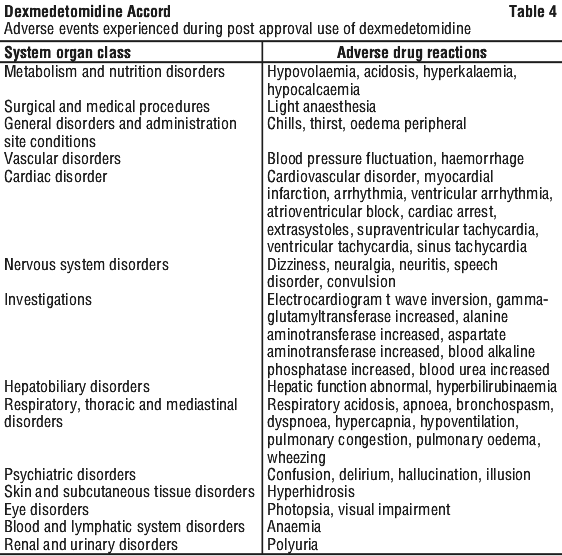
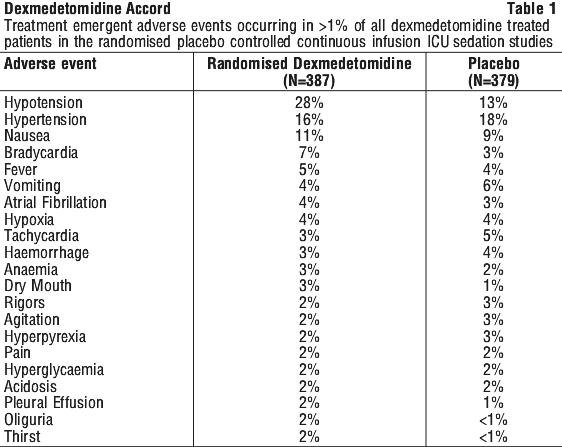 Adverse event information derived from the midazolam-controlled, continuous infusion trial of dexmedetomidine for sedation in a predominantly medical ICU setting in which 244 patients received dexmedetomidine for long-term sedation. Treatment-emergent adverse events occurring at an incidence of > 5% are provided in Table 2. The mean total dose was 72.5 microgram/kg (range: 0.1 to 489.9), mean dose per hour was 0.83 microgram/kg/hr (range: 0.18 to 1.54) and the mean duration of infusion of 3.4 days (range: 0.02 to 15.6). The population was between 18 to 89 years of age, 46% > 65 years of age, 51% male and 79% Caucasian. The most frequent adverse events for this population were hypotension, tachycardia, bradycardia and systolic hypertension (see Section 4.4 Special Warnings and Precautions for Use).
Adverse event information derived from the midazolam-controlled, continuous infusion trial of dexmedetomidine for sedation in a predominantly medical ICU setting in which 244 patients received dexmedetomidine for long-term sedation. Treatment-emergent adverse events occurring at an incidence of > 5% are provided in Table 2. The mean total dose was 72.5 microgram/kg (range: 0.1 to 489.9), mean dose per hour was 0.83 microgram/kg/hr (range: 0.18 to 1.54) and the mean duration of infusion of 3.4 days (range: 0.02 to 15.6). The population was between 18 to 89 years of age, 46% > 65 years of age, 51% male and 79% Caucasian. The most frequent adverse events for this population were hypotension, tachycardia, bradycardia and systolic hypertension (see Section 4.4 Special Warnings and Precautions for Use).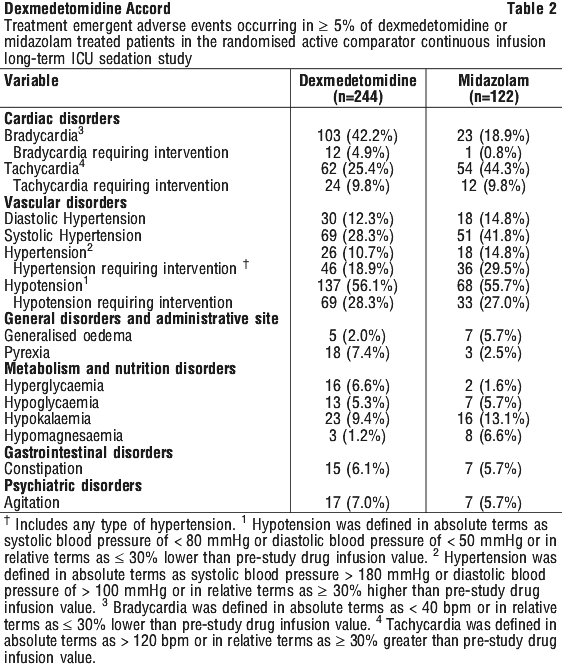 The following adverse events occurred between 2 and 5% for dexmedetomidine and midazolam, respectively: anaemia (2.9%, 4.1%), thrombocytopaenia (0.8%, 2.5%), atrial fibrillation (2.0%, 3.3%), abdominal distension (4.1%, 1.6%), abdominal pain (1.2%, 3.3%), diarrhoea (4.9%, 4.1%), nausea (4.1%, 1.6%), vomiting (2.0%, 4.9%), peripheral oedema (4.1%, 4.9%), pneumonia (1.2%, 4.9%), sepsis (2.5%, 2.5%), septic shock (1.6%, 2.5%), urinary tract infection (0, 3.3%), haemoglobin decreased (0, 2.5%), urine output decreased (2.0%, 3.3%), electrolyte imbalance (0.8%, 2.5%), fluid overload (1.6%, 4.1%), hypernatraemia (2.5%, 1.6%), hypophosphataemia (2.5%, 1.6%), headache (2.0%, 0.8%), anxiety (2.5%, 0), oliguria (0.4%, 2.5%), renal failure acute (2.5%, 0.8%), acute respiratory distress syndrome (2.5%, 0.8%), pharyngolaryngeal pain (2.5%, 4.9%), pleural effusion (2.9%, 2.5%), respiratory failure (4.5%, 3.3%), decubitus ulcer (1.2%, 4.9%) and rash (0.8%, 2.5%).
The following adverse events occurred between 2 and 5% for dexmedetomidine and midazolam, respectively: anaemia (2.9%, 4.1%), thrombocytopaenia (0.8%, 2.5%), atrial fibrillation (2.0%, 3.3%), abdominal distension (4.1%, 1.6%), abdominal pain (1.2%, 3.3%), diarrhoea (4.9%, 4.1%), nausea (4.1%, 1.6%), vomiting (2.0%, 4.9%), peripheral oedema (4.1%, 4.9%), pneumonia (1.2%, 4.9%), sepsis (2.5%, 2.5%), septic shock (1.6%, 2.5%), urinary tract infection (0, 3.3%), haemoglobin decreased (0, 2.5%), urine output decreased (2.0%, 3.3%), electrolyte imbalance (0.8%, 2.5%), fluid overload (1.6%, 4.1%), hypernatraemia (2.5%, 1.6%), hypophosphataemia (2.5%, 1.6%), headache (2.0%, 0.8%), anxiety (2.5%, 0), oliguria (0.4%, 2.5%), renal failure acute (2.5%, 0.8%), acute respiratory distress syndrome (2.5%, 0.8%), pharyngolaryngeal pain (2.5%, 4.9%), pleural effusion (2.9%, 2.5%), respiratory failure (4.5%, 3.3%), decubitus ulcer (1.2%, 4.9%) and rash (0.8%, 2.5%).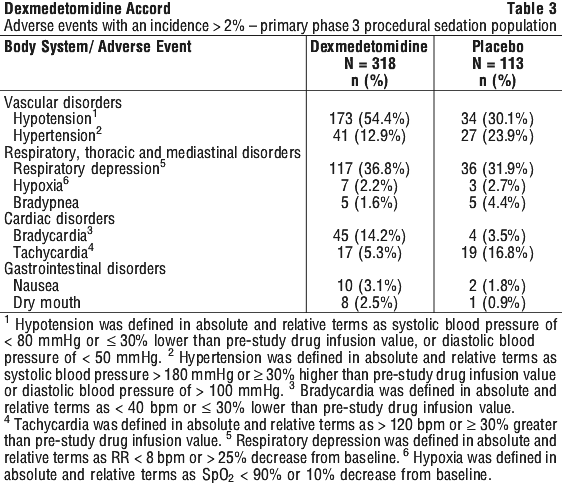
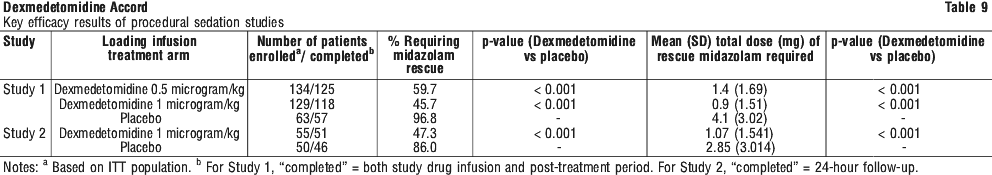
 In the first study, 175 adult patients were randomised to receive placebo and 178 to receive dexmedetomidine by intravenous infusion at a dose of 0.4 microgram/kg/hr (with allowed adjustment between 0.2 and 0.7 microgram/kg/hr) following an initial loading infusion of 1 (one) microgram/kg IV over 10 minutes. The study drug infusion rate was adjusted to maintain a Ramsay sedation score of ≥ 3. Patients were allowed to receive "rescue" midazolam as needed to augment the study drug infusion. In addition, morphine sulfate was administered for pain as needed. The primary outcome measure for this study was the total amount of rescue medication (midazolam) needed to maintain sedation as specified while intubated. Patients randomised to placebo received significantly more midazolam than patients randomised to dexmedetomidine (see Table 6).
In the first study, 175 adult patients were randomised to receive placebo and 178 to receive dexmedetomidine by intravenous infusion at a dose of 0.4 microgram/kg/hr (with allowed adjustment between 0.2 and 0.7 microgram/kg/hr) following an initial loading infusion of 1 (one) microgram/kg IV over 10 minutes. The study drug infusion rate was adjusted to maintain a Ramsay sedation score of ≥ 3. Patients were allowed to receive "rescue" midazolam as needed to augment the study drug infusion. In addition, morphine sulfate was administered for pain as needed. The primary outcome measure for this study was the total amount of rescue medication (midazolam) needed to maintain sedation as specified while intubated. Patients randomised to placebo received significantly more midazolam than patients randomised to dexmedetomidine (see Table 6).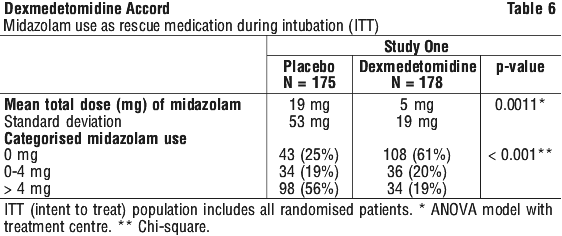 A prospective secondary analysis assessed the dose of morphine sulfate administered to patients in the dexmedetomidine and placebo groups. On average, dexmedetomidine-treated patients received less morphine sulfate for pain than placebo-treated patients (0.47 versus 0.83 mg/h). In addition, 44% (79 of 178 patients) of dexmedetomidine patients received no morphine sulfate for pain versus 19% (33 of 175 patients) in the placebo group.
A prospective secondary analysis assessed the dose of morphine sulfate administered to patients in the dexmedetomidine and placebo groups. On average, dexmedetomidine-treated patients received less morphine sulfate for pain than placebo-treated patients (0.47 versus 0.83 mg/h). In addition, 44% (79 of 178 patients) of dexmedetomidine patients received no morphine sulfate for pain versus 19% (33 of 175 patients) in the placebo group.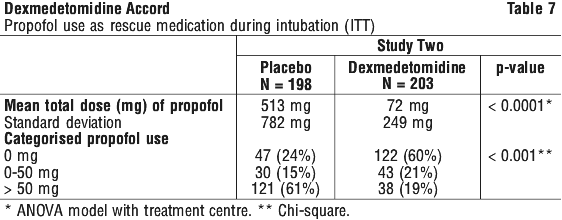 A prospective secondary analysis assessed the dose of morphine sulfate administered to patients in the dexmedetomidine and placebo groups. On average, dexmedetomidine-treated patients received less morphine sulfate for pain than placebo-treated patients (0.43 versus 0.89 mg/h). In addition, 41% (83 of 203 patients) of dexmedetomidine patients received no morphine sulfate for pain versus 15% (30 of 198 patients) in the placebo group.
A prospective secondary analysis assessed the dose of morphine sulfate administered to patients in the dexmedetomidine and placebo groups. On average, dexmedetomidine-treated patients received less morphine sulfate for pain than placebo-treated patients (0.43 versus 0.89 mg/h). In addition, 41% (83 of 203 patients) of dexmedetomidine patients received no morphine sulfate for pain versus 15% (30 of 198 patients) in the placebo group.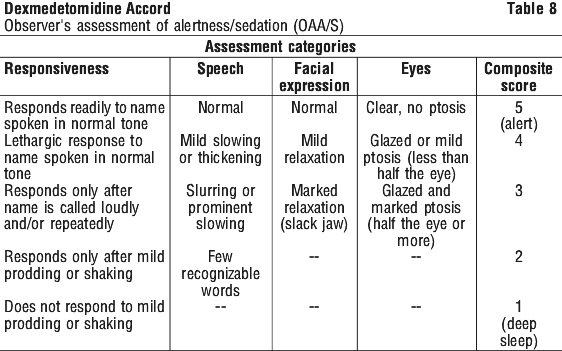 Patients were randomised to receive a loading infusion of either dexmedetomidine 1 microgram/kg or dexmedetomidine 0.5 microgram/kg, or placebo (normal saline) given over 10 minutes and followed by a maintenance infusion started at 0.6 microgram/kg/hr. The maintenance infusion of study drug could be titrated from 0.2 microgram/kg/hr to 1 microgram/kg/hr to achieve the targeted sedation score (OAA/S ≤ 4). Patients were allowed to receive rescue midazolam as needed to achieve and/or maintain an OAA/S < 4. After achieving the desired level of sedation, a local or regional anaesthetic block was performed. Demographic characteristics were similar between the dexmedetomidine and placebo groups. Efficacy results showed that dexmedetomidine was significantly more effective than placebo when used to sedate non-intubated patients requiring monitored anaesthesia care during surgical and other procedures (Table 9).
Patients were randomised to receive a loading infusion of either dexmedetomidine 1 microgram/kg or dexmedetomidine 0.5 microgram/kg, or placebo (normal saline) given over 10 minutes and followed by a maintenance infusion started at 0.6 microgram/kg/hr. The maintenance infusion of study drug could be titrated from 0.2 microgram/kg/hr to 1 microgram/kg/hr to achieve the targeted sedation score (OAA/S ≤ 4). Patients were allowed to receive rescue midazolam as needed to achieve and/or maintain an OAA/S < 4. After achieving the desired level of sedation, a local or regional anaesthetic block was performed. Demographic characteristics were similar between the dexmedetomidine and placebo groups. Efficacy results showed that dexmedetomidine was significantly more effective than placebo when used to sedate non-intubated patients requiring monitored anaesthesia care during surgical and other procedures (Table 9).