1 Name of Medicine
Metformin hydrochloride.
2 Qualitative and Quantitative Composition
Each Diaformin Viatris 500 tablet contains 500 mg of metformin hydrochloride as the active ingredient.
Each Diaformin Viatris 850 tablet contains 850 mg of metformin hydrochloride as the active ingredient.
Each Diaformin Viatris 1000 tablet contains 1000 mg of metformin hydrochloride as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Diaformin Viatris 500 mg tablet.
White, round, normal convex, film coated tablets plain on both the sides. Diameter about 12 mm.
Diaformin Viatris 850 mg tablet.
White, round, normal convex, film coated tablets plain on both the sides. Diameter about 13.5 mm.
Diaformin Viatris 1000 mg tablet.
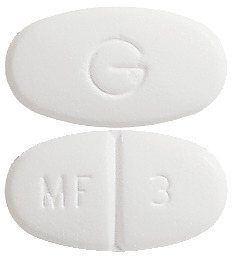
White, oval, film coated tablet, debossed "MF" and "3" on either side of the break line and "G" on the other side.4.1 Therapeutic Indications
Metformin is indicated in the treatment of type 2 diabetes mellitus in adults, children from 10 years of age and adolescents, particularly in overweight patients, when dietary management and exercise alone does not result in adequate glycaemic control.
For adult patients, metformin may be used as initial treatment or in sulfonylurea failures either alone or in combination with a sulfonylurea and other oral agents or as adjuvant therapy in insulin requiring type 2 diabetes.
4.2 Dose and Method of Administration
Life threatening lactic acidosis can occur due to accumulation of metformin. Risk factors include renal impairment, old age and the use of high doses of metformin above 2 g per day.
It is important that the tablets are taken in divided doses with meals.
Half tablet doses of the 1000 mg tablet may be administered, if care is taken not to crush or chew the tablets.
Initially 500 mg should be taken once or twice a day and if necessary increased over a few weeks up to 1 g three times per day.
The dose should be titrated with gradual dose increments until the desired effect is obtained. 500 mg three times a day is often sufficient to obtain diabetic control. If necessary, the dose can be increased to 1 g three times daily, which is the maximum recommended daily dose. Control may be attained within a few days but occasionally requires up to two weeks. Once control has been obtained, the dosage should be reviewed and reduced to the lowest maintenance level consistent with good diabetic control.
Metformin dosage should be frequently reviewed in patients stabilised on metformin, especially if they develop an illness, as they may tolerate the drug less well, particularly if the illness is accompanied by a decrease in renal function. If necessary, metformin should be ceased for a few days during an illness and then restarted at low dosage, as for initial therapy.
The action of metformin is progressive and no final assessment of the patient's real response should be made before the 21st day of treatment. Blood sugar estimations are recommended during the initial 15 days of stabilisation. Metformin will not produce a hypoglycaemic state when used alone; however, due to its action in increasing insulin effectiveness, care must be taken when metformin is initially administered with parenteral doses of insulin.
Elderly.
The initial and maintenance dosing of metformin should be conservative in elderly patients, due to the potential for decreased renal function in this population. Any dosage adjustment should be based on a careful assessment of renal function. Generally, elderly patients should not be titrated to the maximum dose of metformin.
Use in children and adolescents.
Metformin can be used as monotherapy in children from 10 years of age and adolescents. The usual starting dose is one tablet of 500 mg or 850 mg once daily, given during meals or after meals.
After 10 to 15 days the dose should be adjusted on the basis of blood glucose measurements. A slow increase of dose may improve gastrointestinal tolerability.
The maximum recommended dose of metformin is 2 g daily, taken as two or three divided doses.
In debilitated or malnourished patients.
The dosing should be conservative and based on a careful assessment of renal function.4.3 Contraindications
Juvenile diabetes mellitus that is uncomplicated and well regulated on insulin.
Diabetes mellitus regulated by diet alone.
During or immediately following surgery where insulin is essential.
Hypersensitivity to metformin hydrochloride or to any of the excipients.
Any type of metabolic acidosis (such as lactic acidosis, diabetic ketoacidosis).
Diabetic pre-coma.
Renal failure or renal dysfunction (creatinine clearance < 60 mL/min).
Acute conditions with the potential to alter renal function such as: dehydration, severe infection, shock, intravascular administration of iodinated contrast materials (see Section 4.4 Special Warnings and Precautions for Use).
Acute or chronic disease which may cause tissue hypoxia such as: cardiac failure, recent myocardial infarction, respiratory failure, pulmonary embolism, shock, acute significant blood loss, sepsis, gangrene, pancreatitis.
Elective major surgery (see Section 4.4 Special Warnings and Precautions for Use).
Severe hepatic insufficiency, acute alcohol intoxication, alcoholism.
Lactation.
4.4 Special Warnings and Precautions for Use
Lactic acidosis.
Lactic acidosis is a rare but serious (high mortality in the absence of prompt treatment), metabolic complication that can occur due to metformin accumulation. Reported cases of lactic acidosis in patients on metformin have occurred primarily in diabetic patients with significant renal failure. The incidence of lactic acidosis can and should be reduced by assessing other associated risk factors such as poorly controlled diabetes, ketosis, prolonged fasting, excessive alcohol intake, hepatic insufficiency and any condition associated with hypoxia.
Diagnosis.
The risk of lactic acidosis must be considered in the event of non-specific signs such as muscle cramps with digestive disorders such as abdominal pain and severe asthenia.
Lactic acidosis is characterised by acidotic dyspnea, abdominal pain and hypothermia followed by coma. Diagnostic laboratory findings are decreased blood pH, plasma lactate levels above 5 mmol/L, and an increased anion gap and lactate/pyruvate ratio. If metabolic acidosis is suspected, metformin should be discontinued and the patient should be hospitalised immediately (see Section 4.9 Overdose, Treatment).
Renal function.
As metformin hydrochloride is excreted by the kidney, it is recommended that creatinine clearance and/or serum creatinine levels be determined before initiating treatment and regularly thereafter:
At least annually in patients with normal renal function.
At least two to four times a year in patients with serum creatinine levels at the upper limit of normal and in elderly subjects.
Decreased renal function in elderly subjects is frequent and asymptomatic. Special caution should be exercised in situations where renal function may become impaired, for example when initiating antihypertensive therapy or diuretic therapy and when starting therapy with a non-steroidal anti-inflammatory drug (NSAID).
Administration of iodinated contrast materials.
The intravascular administration of iodinated contrast materials in radiologic studies can lead to renal failure. This may induce metformin accumulation and may expose to lactic acidosis. Therefore, metformin must be discontinued either 48 hours before the test when renal function is known to be impaired or from the time of the test when renal function is known to be normal. Metformin may not be reinstituted until 48 hours afterwards, and only after renal function has been re-evaluated and found to be normal (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Surgery.
Metformin hydrochloride must be discontinued 48 hours before elective major surgery. Therapy may be restarted no earlier than 48 hours following surgery and only after renal function has been re-evaluated and found to be normal.
Other precautions.
All patients should continue their diet with a regular distribution of carbohydrate intake during the day. Overweight patients should continue their energy-restricted diet.
The usual laboratory tests for diabetes monitoring should be performed regularly.
Metformin hydrochloride alone does not cause hypoglycaemia; however, caution is advised when it is used in combination with other antidiabetic agents (sulphonylureas, glinides, insulin).
Patients receiving continuous metformin therapy: it is recommended that serum vitamin B12 levels be measured prior to initiation treatment with metformin, after 6 months treatment and thereafter annually because of reports of decreased vitamin B12 absorption associated with metformin administration.
Use in the elderly.
The risk of lactic acidosis, in association with metformin, is increased in elderly patients on long-term therapy due to the physiological alteration of the renal function and the possible accumulation of metformin. Metformin may be used in the elderly if Contraindications and Precautions are respected, the dosage is frequently reviewed and renal function monitored.
Decreased renal function in elderly subjects is frequent and asymptomatic. Special caution should be exercised in situations where renal function may become impaired.
Paediatric use.
Metformin is not recommended for use in children under 10 years of age.
The diagnosis of type 2 diabetes mellitus should be confirmed before treatment with metformin is initiated.
No effect of metformin on growth and puberty has been detected during controlled clinical studies of one-year duration but clinical data in relation to the long-term effect of metformin on the development of skeletal and reproductive system in children and adolescents are not available. Therefore, a careful follow-up of the effect of metformin on these parameters in metformin-treated children, especially pre-pubescent children, is recommended.
Effects on laboratory tests.
No information is available.4.5 Interactions with Other Medicines and Other Forms of Interactions
Contraindicated combinations.
Iodinated contrast materials.
Metformin must be discontinued either 48 hours before the test when renal function is known to be impaired, or from the time of the test when renal function is known to be normal (see Section 4.4 Special Warnings and Precautions for Use, Administration of iodinated contrast materials).
Inadvisable combinations.
Alcohol.
Increased risk of lactic acidosis in acute alcohol intoxication, particularly in case of fasting or malnutrition; hepatic insufficiency.
Avoid consumption of alcohol and alcohol-containing medications. Alcohol may make the signs of hypoglycaemia less clear, and delayed hypoglycaemia can occur. The CNS depressant effects of alcohol plus hypoglycaemia can make driving or the operation of dangerous machinery much more hazardous.
Combinations requiring precautions for use.
Medicinal products with intrinsic hyperglycaemic activity (e.g. glucocorticoids and tetracosactides (systemic and local routes), beta-2-agonists, danazol, chlorpromazine at high dosages of 100 mg per day and diuretics.
More frequent blood glucose monitoring may be required, especially at the beginning of treatment. If necessary, adjust the metformin dosage during therapy with the respective medicinal product and upon discontinuation.
Diuretics, especially loop diuretics.
May increase the risk of lactic acidosis due to their potential to decrease renal function.
ACE-inhibitors.
ACE-inhibitors may decrease the blood glucose levels. Therefore, dose adjustment of metformin hydrochloride may be necessary when such medicinal products are added or discontinued.
Calcium channel blockers.
Calcium channel blockers may affect glucose control in diabetic patients; regular monitoring of glycaemic control is recommended.
Beta-blockers.
Co-administration of metformin and beta-blockers may result in a potentiation of the anti-hyperglycaemic action. In addition, some of the premonitory signs of hypoglycaemia, in particular tachycardia, may be masked. Monitoring of blood glucose should be undertaken during dosage adjustment of either agent.
Cimetidine.
Reduced clearance of metformin has been reported during cimetidine therapy, so a dose reduction should be considered.
Anticoagulants.
Metformin increases the elimination rate of vitamin K antagonists. Consequently, the prothrombin time should be closely monitored in patients in whom metformin and vitamin K antagonists are being co-administered. Cessation of metformin in patients receiving vitamin K antagonists can cause marked increases in the prothrombin time.
Nifedipine.
A single-dose, metformin-nifedipine drug interaction study in normal healthy volunteers demonstrated that co-administration of metformin and nifedipine increased plasma metformin Cmax and AUC by 20% and 9%, respectively, and increased the amount of metformin excreted in the urine. Tmax and half-life of metformin were unaffected. Nifedipine appears to enhance the absorption of metformin. Metformin had minimal effects on the pharmacokinetics of nifedipine.
Organic cation transporters (OCT).
Metformin is a substrate of both transporters OCT1 and OCT2.
Co-administration of metformin with:
Substrates/inhibitors of OCT1 (such as verapamil) may reduce efficacy of metformin.
Inducers of OCT1 (such as rifampicin) may increase gastrointestinal absorption and efficacy.
Substrates/ inhibitors of OCT2 (such as cimetidine, dolutegravir, crizotinib, olaparib, daclatasvir, vandetanib) may decrease the renal elimination of metformin and thus lead to an increase metformin plasma concentration.
Carbonic anhydrase inhibitors.
Topiramate or other carbonic anhydrase inhibitors (e.g. zonisamide, acetazolamide or dichlorphenamide) frequently cause a decrease in serum bicarbonate and induce non-anion gap, hyperchloremic metabolic acidosis. Concomitant use of these drugs with Metformin hydrochloride tablet may increase the risk for lactic acidosis. Consider more frequent monitoring of these patients.
NSAID.
May increase the risk of lactic acidosis and adversely affect renal function.
Therefore, caution is advised when these drugs are co-administered with metformin and a dose adjustment may be considered, particularly in patients with renal impairment.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Fertility of male or female rats was unaffected by metformin administration at doses up to 600 mg/kg/day, or approximately twice the maximum recommended daily dose on a body surface area basis.
(Category C)
To date, no relevant epidemiological data is available. Animal studies do not indicate harmful effects with respect to pregnancy, embryonal or foetal development, parturition or postnatal development.
Metformin was not teratogenic in rats and rabbits at doses up to 600 mg/kg/day, or about two times the maximum recommended human daily dose on a body surface area basis. Determination of foetal concentrations demonstrated a partial placental barrier to metformin. Because animal reproduction studies are not always predictive of human response, any decision to use this drug should be balanced against the benefits and risks. The safety of metformin in pregnant women has not been established.
When the patient plans to become pregnant and during pregnancy, it is recommended that diabetes should not be treated with metformin but insulin should be used to maintain blood glucose levels as close to normal as possible in order to lower the risk of foetal malformations associated with abnormal blood glucose levels.
Australian Categorisation Definition of Category C: Drugs which, owing to their pharmacological effects, have caused or may be suspected of causing, harmful effects on the human fetus or neonate without causing malformations. These effects may be reversible. Accompanying texts should be consulted for further details.
Metformin is excreted into milk in lactating rats. Similar data are not available in humans and a decision should be made whether to discontinue breastfeeding or to discontinue metformin, taking into account the importance of the drug to the mother.4.7 Effects on Ability to Drive and Use Machines
Metformin monotherapy does not cause hypoglycaemia and therefore has no effect on the ability to drive or to use machinery.
However, patients should be alerted to the risk of hypoglycaemia when metformin is used in combination with other antidiabetic agents (sulphonylureas, glinides, insulin).
4.8 Adverse Effects (Undesirable Effects)
The following undesirable effects may occur under treatment with metformin hydrochloride. Frequencies are defined as follows: very common: ≥ 1/10; common: ≥ 1/100, < 1/10; uncommon: ≥ 1/1,000, < 1/100; rare: ≥ 1/10,000, < 1/1,000; very rare: < 1/10,000; not known (cannot be estimated from the available data).
Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
Nervous system disorders.
Common: taste disturbance.
Gastrointestinal disorders.
Very common: gastrointestinal disorders such as diarrhoea, nausea, vomiting, abdominal pain and loss of appetite. These undesirable effects occur most frequently during initiation of therapy and resolve spontaneously in most cases. To prevent them, it is recommended that metformin be taken in 2 or 3 daily doses during or after meals. A slow increase of the dose may also improve gastrointestinal tolerability.
Skin and subcutaneous tissue disorders.
Very rare: skin reactions such as erythema, pruritus and urticaria.
Metabolism and nutrition disorders.
Very rare: lactic acidosis (see Section 4.4 Special Warnings and Precautions for Use).
Decrease of vitamin B12 absorption with a decrease in serum levels has been observed in patients treated long-term with metformin. Consideration of such an aetiology is recommended if a patient presents with megaloblastic anaemia. Therefore, serum B12 levels should be appropriately monitored or periodic parenteral B12 supplementation should be considered (see Section 4.4 Special Warnings and Precautions for Use, Other precautions).
Hepatobiliary disorders.
Very rare: isolated reports of liver function test abnormalities or hepatitis resolving upon metformin discontinuation.
In clinical trials in children and adolescents with type 2 diabetes, the profile of adverse reactions was similar to that observed in adults.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Symptoms.
Hypoglycaemia has not been seen with ingestion of up to 85 g of metformin alone, although lactic acidosis has occurred in such circumstances. This disorder is a medical emergency and must be treated in hospital. The onset of lactic acidosis is often subtle and accompanied only by non-specific symptoms such as malaise, myalgia, respiratory distress, increasing somnolence and non-specific abdominal distress. There may be associated hypothermia, hypotension and resistant bradyarrhythmias with more marked acidosis. Lactic acidosis should be suspected in any diabetic patient with metabolic acidosis lacking evidence of ketoacidosis (ketonuria and ketonaemia).
Treatment.
Lactic acidosis may develop in diabetic metformin-treated patients with overdose. Lactic acidosis is diagnosed and monitored by measurement of serum electrolytes, arterial pH and pCO2 and arterial lactate plasma level.
The aim of treatment is to manage any underlying disorder and in some cases this will be sufficient to enable the body's homeostatic mechanism to correct the acid-base imbalance. The advantages of more active treatment of the acidosis must be balanced against the risks, including over-alkalinisation with sodium bicarbonate. Because metformin hydrochloride is dialysable (with a clearance of up to 170 mL/min under good haemodynamic conditions), prompt haemodialysis is recommended to correct the acidosis and remove the accumulated metformin.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Metformin is a biguanide with antihyperglycaemic effects, lowering both basal and postprandial plasma glucose. It does not stimulate insulin secretion and therefore does not produce hypoglycaemia.
Metformin may act via 3 mechanisms:
1. Reduction of hepatic glucose production by inhibiting gluconeogenesis and glycogenolysis.
2. In muscle, by increasing insulin sensitivity, improving peripheral glucose uptake and utilisation.
3. Delay of intestinal glucose absorption.
Metformin stimulates intracellular glycogen synthesis by acting on glycogen synthase.
Metformin increases the transport capacity of all types of membrane glucose transporters (GLUT).
In humans, independently of its action on glycaemia, metformin has favourable effects on lipid metabolism. This has been shown at therapeutic doses in controlled, medium-term or long-term clinical studies: metformin reduces total cholesterol, LDL cholesterol and triglyceride levels.
Clinical trials.
The prospective randomised (UKPDS) study has established the long term benefit of intensive blood glucose control in type 2 diabetes.
Analysis of the results for overweight patients treated with metformin after failure of diet alone showed:
a significant reduction of the absolute risk of any diabetes related complication in the metformin group (29.8 events/ 1000 patient years) versus diet alone (43.3 events/ 1000 patient years), p = 0.0023, and versus the combined sulfonylurea and insulin monotherapy groups (40.1 events/1000 patient years), p = 0.0034;
a significant reduction of the absolute risk of diabetes related mortality: metformin 7.5 events/1000 patient years, diet alone 12.7 events/1000 patient years, p = 0.017;
a significant reduction of the absolute risk of overall mortality: metformin 13.5 events/1000 patient years versus diet alone 20.6 events/ 1000 patient years (p = 0.011), and versus the combined sulfonylurea and insulin monotherapy groups 18.9 events/ 1000 patient years (p = 0.021);
a significant reduction in the absolute risk of myocardial infarction: metformin 11 events/1000 patient years, diet alone 18 events/ 1000 patient years (p = 0.01).
Benefit regarding clinical outcome has not been shown for metformin hydrochloride used as second line therapy, in combination with a sulfonylurea.
In type 1 diabetes, the combination of metformin and insulin has been used in selected patients, but the clinical benefit of this combination has not been formally established.
Paediatrics.
In a double blind, placebo-controlled study in 82 paediatric patients aged 10 to 16 years with type 2 diabetes (mean FPG 10.1 mmol/L), treatment with metformin (up to 2000 mg/day) for up to 16 weeks (mean duration of treatment 11 weeks) resulted in a significant mean net reduction in FPG of 3.6 mmol/L, compared with placebo.
5.2 Pharmacokinetic Properties
Absorption.
After oral administration, metformin hydrochloride is absorbed along the entire gastrointestinal mucosa. Studies using single oral doses of metformin tablets indicate that there is a lack of dose proportionality with increasing doses, which is due to decreased absorption rather than an increase in elimination. After oral administration, metformin absorption is saturable and incomplete. It is assumed that the pharmacokinetics of metformin absorption are non-linear.
At usual clinical doses and dosing schedules of metformin tablets, steady-state plasma concentrations are reached in 24 to 48 hours and are generally less than 1 microgram/mL. During controlled clinical trials, maximum metformin plasma levels did not generally exceed 5 microgram/mL, even at maximum doses.
Distribution.
Metformin is not bound to plasma proteins.
Metabolism.
Metformin is excreted unchanged in the urine and does not undergo hepatic metabolism.
Excretion.
In patients with decreased renal function (based on measured creatinine clearance), the plasma half-life of metformin is prolonged and renal clearance is decreased in proportion to the decrease in creatinine clearance, e.g. if creatinine clearance is 10-30 mL/min, renal clearance is reduced to 20% of normal.
A randomised, open-label bioequivalence study in 16 healthy volunteers showed, on the basis of Cmax and AUC determinations, that one tablet containing 1000 mg of metformin can replace two tablets containing 500 mg of metformin. The relative bioavailability of one tablet containing 1000 mg of metformin compared to two tablets containing 500 mg of metformin is 92.6% for AUC and 88% for Cmax.
Paediatrics.
Following an oral dose, children 12 years and older, have shown similar pharmacokinetic profile of metformin to that observed in adults. Pharmacokinetic data in children between 10 and 12 years are not available.
5.3 Preclinical Safety Data
Genotoxicity.
No evidence of a mutagenic potential of metformin was found in the Ames test (S. typhimurium), gene mutation test (mouse lymphoma cells), chromosomal aberrations test (human lymphocytes), or in vivo micronuclei formation test (mouse bone marrow).
Carcinogenicity.
Long term carcinogenicity studies have been performed in rats (dosing duration of 104 weeks) and mice (dosing duration of 91 weeks) at doses up to and including 900 mg/kg/day and 1500 mg/kg/day, respectively. These doses are both approximately two to three times the recommended human daily dose on a body surface area basis. No evidence of carcinogenicity with metformin was found in either male or female mice. Similarly, there was no tumourigenic potential observed with metformin in male rats. However, an increased incidence of benign stromal uterine polyps was seen in female rats treated with 900 mg/kg/day.6 Pharmaceutical Particulars
6.1 List of Excipients
The tablet contains povidone, magnesium stearate, and are film-coated with a proprietary ingredient, Opadry complete film coating system 20C59060 Clear (ARTG PI No. 106044).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Store in the original container.
6.5 Nature and Contents of Container
Diaformin Viatris 500 tablet.
Available in PVC/Al blister packs - pack size of 10, 20 and 100 tablets or HDPE bottles with a child resistant closure - pack size of 100 tablets.
Diaformin Viatris 850 tablet.
Available in PVC/Al blister packs - pack size of 10, 20 and 60 tablets or HDPE bottles with a child resistant closure - pack size of 60 tablets.
Diaformin Viatris 1000 tablet.
Available in PVC/Al blister packs - pack size of 10, 30, 60 and 90 tablets or HDPE bottles with a child resistant closure - pack size of 10, 30, 60 and 90 tablets.
Some strengths, pack sizes and/or pack types may not be marketed.
Australian register of therapeutic goods (ARTG).
AUST R 206739.
Diaformin Viatris metformin hydrochloride 500 mg film-coated tablet bottle.
AUST R 206740.
Diaformin Viatris metformin hydrochloride 500 mg film-coated tablet blister pack.
AUST R 206738.
Diaformin Viatris metformin hydrochloride 850 mg film-coated tablet bottle.
AUST R 206737.
Diaformin Viatris metformin hydrochloride 850 mg film-coated tablet blister pack.
AUST R 206736.
Diaformin Viatris metformin hydrochloride 1000 mg film-coated tablet bottle.
AUST R 206735.
Diaformin Viatris metformin hydrochloride 1000 mg film-coated tablet blister pack.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
Metformin hydrochloride is a white, crystalline powder which is odourless or almost odourless and hygroscopic. It is freely soluble in water, slightly soluble in ethanol (96%), and practically insoluble in chloroform and in ether.
Chemical name: 1,1-dimethylbiguanide hydrochloride.
Structural formula:
 Molecular formula: C4H11N5, HCl.
Molecular formula: C4H11N5, HCl.
Molecular weight: 165.6.
CAS number.
1115-70-4.7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Summary Table of Changes


 Molecular formula: C4H11N5, HCl.
Molecular formula: C4H11N5, HCl.