What is in this leaflet
This leaflet answers some common questions about Dolased Forte It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor or pharmacist has weighed the risks of you taking Dolased Forte against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again
What Dolased Forte is used for
Dolased Forte has pain relieving, fever reducing and calmative effects.
Dolased Forte is used for the short-term management of severe pain for which other treatment options have failed, are contraindicated, not tolerated or are otherwise inappropriate to provide sufficient management of pain
It is suitable for people who cannot take aspirin for pain relief.
Dolased Forte is available only with a doctor's prescription.
Dolased Forte is not recommended for use in children under the age of 12 years.
Before you take DOLASED FORTE
When you must not take it
Do not take Dolased Forte if you have an allergy to paracetamol, codeine phosphate, doxylamine succinate or any of the ingredients listed at the end of this leaflet.
Do not take this medicine if you have or have had any of the following medical conditions:
- Acute breathing difficulties such as bronchitis, unstable asthma or emphysema
- Glucose-6-phosphate-dehydrogenase deficiency (an enzyme deficiency)
- Diarrhoea caused by antibiotics or poisoning
Do not take Dolased Forte if you have alcohol dependence.
Do not take Dolased Forte during the third trimester of pregnancy.
Do not take Dolased Forte during labour, especially if the baby is premature. This medicine contains codeine, which may produce withdrawal effects in the newborn baby.
Do not take Dolased Forte if you are breastfeeding or planning to breastfeed. Dolased Forte passes into breast milk and there is a possibility your baby may be affected.
Do not take Dolased Forte after the expiry date (EXP) printed on the pack. If you take this medicine after the expiry date has passed, it may not work as well.
Do not take Dolased Forte if the packaging is torn, shows signs of tampering, or if it doesn't look quite right.
If you are not sure whether you should start taking Dolased Forte, contact your doctor or pharmacist.
Before you start to take Dolased Forte
Tell your doctor or pharmacist if you have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes
Tell your doctor or pharmacist if you have or have had any medical conditions, especially the following:
- Liver problems
- Kidney problems
- Difficulty breathing, wheezing, chronic cough, asthma, or other chronic breathing conditions
- A history of drug dependence, including alcohol dependence
- Recent stomach, intestine or urinary tract surgery
- Prostate problems
- Underactive thyroid
- Head injury
- Fits or seizures
Tell your pharmacist or doctor if you are pregnant or plan to become pregnant. Your pharmacist or doctor will discuss the benefits and possible risks of taking the medicine during pregnancy.
If you have not told your doctor about any of the above, tell him/her before you start taking Dolased Forte.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Tell your doctor or pharmacist if you are taking any other medicines, which contain paracetamol, codeine or doxylamine.
Some medicines can interfere with Dolased Forte including:
- Any medicine causing sleepiness or drowsiness
- Medicines used to treat depression
- Some types of antihistamines
- Warfarin, a medicine used for blood clots
- Medicines used to treat diarrhoea, nausea or vomiting
- Propantheline, a drug used to treat stomach ulcers
If you are not sure whether you are taking any of these medicines, check with your doctor or pharmacist. Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking Dolased Forte.
How to take DOLASED FORTE
Follow all directions given to you by your doctor or pharmacist carefully.
If you do not understand the instructions in this leaflet, ask your doctor or pharmacist for help.
How much to take
The usual adult dose is one or two tablets every 4 to 6 hours as needed for relief. Do not exceed 8 tablets in a 24 hour period.
Dolased Forte is not recommended for children under 12 years of age.
Follow all directions given to you by your doctor and pharmacist carefully. Their directions may differ from the information contained in this leaflet.
How to take DOLASED FORTE
Swallow the prescribed dose of Dolased Forte whole with a glass of water. It can be taken with or without food.
If you forget to take Dolased Forte
The pain is likely to return. Take it as soon as you remember, and then go back to taking it as before.
Do not take a double dose to make up for the dose you missed.
If you are not sure what to do, ask your doctor or pharmacist.
How long to take Dolased Forte for
Withdrawal
Continue taking your medicine for as long as your doctor tells you. If you stop having this medicine suddenly, your pain may worsen and you may experience some or all of the following withdrawal symptoms:
- nervousness, restlessness, agitation, trouble sleeping or anxiety
- body aches, weakness or stomach cramps
- loss of appetite, nausea, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- watery eyes, runny nose, chills or yawning
- increased sweating.
Dolased Forte given to the mother during labour can cause breathing problems and signs of withdrawal in the newborn.
If you take too much Dolased Forte (overdose)
If you or someone else receive too much (overdose), and experience one or more of the symptoms below, immediately call triple zero (000) for an ambulance. Keep the person awake by talking to them or gently shaking them every now and then. You should follow the above steps even if someone other than you have accidentally used Dolased Forte that was prescribed for you. If someone takes an overdose they may experience one or more of the following symptoms:
- Slow, unusual or difficult breathing
- Drowsiness, dizziness or unconsciousness
- Slow or weak heartbeat
- Nausea or vomiting
- Convulsions or fits
If you think you or someone else may have used too much Dolased Forte, you should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
When seeking medical attention, take this leaflet and remaining medicine with you to show the doctor. Also tell them about any other medicines or alcohol which have been taken.
Depending on your body’s individual ability to break down codeine, you may experience signs of overdose even when you take Dolased Forte as recommended by your doctor. If overdose symptoms occur, seek immediate medical advice.
While you are taking Dolased Forte
Things you must do
Before starting any new medicine, tell your doctor or pharmacist that you are taking Dolased Forte.
If you become pregnant while taking Dolased Forte tell your doctor.
Keep all your doctor’s appointments so that your progress can be checked.
Tell your doctor if you feel the dose you are taking is too strong or too weak.
If the dose is too strong you may feel very drowsy or have difficulty breathing.
If the dose is too weak you may continue to feel some pain between doses.
Things you must not do
Do not stop taking Dolased Forte, or change the dose, without checking with your doctor. On rare occasions, especially if you have been taking Dolased Forte for a while stopping treatment abruptly can make you feel unwell.
Do not take Dolased Forte to treat any other conditions unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful driving or operating machinery until you know how Dolased Forte affects you. It may cause dizziness and drowsiness in some people. Make sure you know how you react to Dolased Forte before you drive a car, or do anything else that could be dangerous if you are dizzy.
This effect may be made worse by drinking alcohol or some other medicines that act on the central nervous system.
Addiction
You can become addicted to Dolased Forte even if you take it exactly as prescribed. Dolased Forte may become habit forming causing mental and physical dependence. If abused it may become less able to reduce pain.
Dependence
As with all other opioid containing products, your body may become used to you taking Dolased Forte. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking Dolased Forte suddenly, so it is important to take it exactly as directed by your doctor.
Tolerance
Tolerance to Dolased Forte may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
Dolased Forte may be dangerous if you take large amounts over a long period of time.
Do not take high doses of the medicine for long periods of time unless your doctor tells you to. This medicine contains codeine, which may be habit forming.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking DOLASED FORTE.
This medicine helps most people with your condition, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Tell your doctor if you notice any of the following and they worry you:
- Skin rash
- Constipation
- Nausea and vomiting
- Drowsiness
- Sweating
These are the more common side effects of Dolased Forte. They are usually mild.
Tell your doctor as soon as possible if you notice any of the following:
- Shortness of breath
- Mouth ulcers, fever and sore throat
- Bleeding, bruising more easily
- Unusual or extreme mood swings
- Dizziness, light-headedness
- Flushing of the face
- Painful red areas with blisters and peeling layers of skin which may be accompanied by fever and/or chills
- Severe blisters and bleeding in the lips, eyes, mouth, nose and genitals
- Hepatitis (symptoms include loss of appetite, itching, yellowing of the skin and eyes, light coloured bowel motions, dark coloured urine)
The above list includes serious side effects that may require medical attention. These side effects are rare.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- Wheezing or difficulty breathing
- Swelling of the face, lips, tongue or other parts of the body
- Rash, itching or hives on the skin
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Other side effects not listed above may occur in some patients. Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
After taking DOLASED FORTE
Storage
Keep your tablets in the pack until it is time to take them. If you take the tablets out of the pack they will not keep well.
Keep your tablets in a cool dry dark place where the temperature stays below 25°C.
Do not store Dolased Forte or any other medicine in the bathroom or near a sink.
Do not leave it in the car on hot days or on window sills. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and- a-half metres above the ground is a good place to store medicines.
Disposal
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
Product description
What it looks like
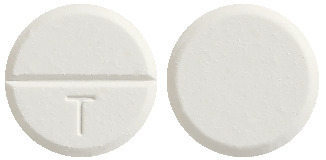
Dolased Forte (Paracetamol, codeine phosphate and doxylamine succinate) is a round, white tablet with “T” on one side of a break line. Dolased Forte is available in a blister pack in cartons of 2, 20 and 40 tablets.
Not all pack sizes may be marketed.
Ingredients
The active substances are
Paracetamol 450mg
Codeine phosphate 30mg
Doxylamine succinate 5mg
The other ingredients are:
Crospovidone
Pregelatinised maize starch
Lactose monohydrate
Povidone
Glyceryl monostearate
Stearic acid
Magnesium stearate
Dolased Forte does not contain tartrazine or any other azo dyes.
Sponsor
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australian Registration Number:
AUST R 203987
This leaflet was prepared in September 2020
Published by MIMS October 2025