1 Name of Medicine
Gadoteric acid.
2 Qualitative and Quantitative Composition
Dotagraf solution for injection contains gadoteric acid 0.5 mM/mL.
Each vial or bottle contains the active ingredient gadoteric acid 279.32 mg/mL (0.5 M). Gadoteric acid is a complex of the paramagnetic ion gadolinium oxide 90.62 mg/mL with tetraxetan (DOTA; 1,4,7,10-tetraazacyclododecane-N,N',N",N'"-tetraacetic acid) 202.46 mg/mL.
Dotagraf has the following chemical physical properties.
Osmolality: 1350 mOsmol/kg.
Viscosity at 20°C: 3.2 mPa.s.
Viscosity at 37°C: 2.0 mPa.s.
pH: 6.5 to 8.0.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Solution for injection.
Dotagraf is a sterile, preservative-free, clear, colourless to yellow solution for injection free of visible particles and packaged in glass vials or bottles for intravenous injection.
4.1 Therapeutic Indications
Dotagraf is indicated, in adults and children, for use with magnetic resonance imaging to provide contrast enhancement for intracranial and spinal lesions with abnormal blood brain barrier or abnormal vascularity, and for whole body imaging (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
4.2 Dose and Method of Administration
Dosage.
The lowest effective dose should be used. The dose should be calculated based on the patient's body weight and should not exceed the recommended dose per kilogram of body weight detailed in this section.
Adults (≥ 18 years).
The maximum recommended dose is 0.1 mmol/kg, i.e. 0.2 mL/kg for adults.
Paediatrics (0 to 18 years).
The maximum recommended dose is 0.1 mmol/kg, i.e. 0.2 mL/kg for children and infants.
Method of administration.
Dotagraf is intended for intravenous administration only.
Volumes required for doses of 0.1 mmol/kg are shown in Table 1.
 The product is intended for intravenous administration only.
The product is intended for intravenous administration only.
Intravascular administration of contrast media should be, if possible, done with the patient lying down. After administration, the patient must be kept under observation for at least 30 minutes, as the majority of serious adverse reactions occur during this period.
Use of Dotagraf in a single-use setting.
Vials or bottles containing contrast medium solutions are not intended for the withdrawal of multiple doses.
The rubber stopper of the vial or bottle should never be pierced more than once.
Each vial or bottle should be used in one patient on one occasion only and any residue should be discarded. Dotagraf contains no preservatives.
If a single-use injector system is used, Instructions For Use (IFU) of the device manufacturer must be followed, including connecting tubes and all disposable parts.
Ensure the single-use disposables are replaced after each patient.
Use of Dotagraf with a multi-patient use injector system and/or large volume containers (100 mL).
The transmission of viral infections and bacterial contamination are recognised but rare potential complications of multi-use containers and administration devices. The following measures should be strictly adhered to when using Dotagraf:
the multiple withdrawal and administration of contrast medium must be done utilising a device and disposables system validated and approved for multiple-patient use, such as an automatic injector system;
the rubber stopper of the bottle should never be pierced more than once;
Instructions For Use (IFU) of the device manufacturer must be followed, including connecting tubes and all disposable parts of the injector system. Ensure the single-use disposables are replaced after each patient.
Dosage adjustment.
Paediatrics (0-18 years).
Depending on the amount of gadoteric acid to be given to the child, it is preferable to use gadoteric acid vials with a single use syringe of a volume adapted to this amount in order to have a better precision of the injected volume.
In neonates and infants the required dose should be administered by hand.
Due to immature renal function in neonates up to 4 weeks of age and infants up to 1 year of age, Dotagraf should only be used in these patients after careful consideration, at a dose not exceeding 0.1 mmol/kg body weight. Due to the lack of information on repeated administration, Dotagraf injections should not be repeated unless the interval between injections is at least 7 days.
Renal impairment and liver transplant patients.
The adult dose applied to patients with mild to moderate renal impairment (GFR ≥ 30 mL/min/1.73 m2). Dotagraf should only be used in patients with severe renal impairment (GFR < 30 mL/min/1.73 m2) and in patients in the perioperative liver transplantation period after careful risk/benefit assessment and if the diagnostic information is essential and not available with non-contrast enhanced MRI (see Section 4.4 Special Warnings and Precautions for Use). If it is necessary to use Dotagraf, the dose should not exceed 0.1 mmol/kg body weight. More than one dose should not be used during a scan. Because of the lack of information on repeated administration, Dotagraf injections should not be repeated unless the interval between injections is at least 7 days.
Elderly.
No dosage adjustment is considered necessary. Caution should be exercised in elderly patients (see Section 4.4 Special Warnings and Precautions for Use).
Hepatic impairment.
The adult dose applies to these patients. Caution is recommended, especially in the case of perioperative liver transplantation period.4.3 Contraindications
Dotagraf is contraindicated in patients with hypersensitivity to gadoteric acid, meglumine or any products containing gadolinium.
Contraindications related to magnetic resonance imaging include patients with pacemakers, patients with ferromagnetic vascular clips, infusion pumps, nerve stimulators, cochlear implants, or suspected intracorporal metallic foreign bodies, particularly in the eye.
4.4 Special Warnings and Precautions for Use
Warning: nephrogenic systemic fibrosis.
Gadolinium based contrast agents increase the risk for nephrogenic systemic fibrosis (NSF) in patients with:
acute or chronic severe renal insufficiency (glomerular filtration rate < 30 mL/min/1.73 m2); or
acute renal insufficiency of any severity due to the hepatorenal syndrome or in the perioperative liver transplantation period.
Severe renal impairment and liver transplant patients.
There have been reports of nephrogenic systemic fibrosis (NSF) associated with use of some gadolinium-based contrast agents (GBCAs) in patients with acute or chronic severe renal impairment (a glomerular filtration rate < 30 mL/min/1.73 m2) and patients with acute renal insufficiency of any severity due to the hepatorenal syndrome or in the perioperative liver transplantation period. As there is a possibility that NSF may occur with gadoteric acid, it should only be used in patients with severe renal impairment and in patients with perioperative liver transplantation after careful risk/benefit assessment and if the diagnostic information is essential and not available with non-contrast enhanced MRI.
Haemodialysis shortly after gadoteric acid administration may be useful at removing gadoteric acid from the body.
NSF is a debilitating and sometimes fatal disease affecting the skin, muscle, and internal organs.
Screen all patients for renal dysfunction by obtaining a history and/or laboratory tests.
When administering a GBCA, do not exceed the dose recommended in the product labelling. Allow sufficient time for elimination of the GBCA prior to any re-administration.
Warnings.
Caution should be exercised in patients suffering from severe renal failure. There is no clinical data concerning the elimination of gadoteric acid in patients with renal failure requiring dialysis.
Do not use by intrathecal route. For intravenous injection only.
Gadoteric acid must not be used intrathecally. Serious, life-threatening and fatal cases, primarily with neurological reactions (e.g. coma, encephalopathy, seizures), have been reported with intrathecal use. Gadoteric acid should be strictly administered via intravenous injection.
In the event of extravasation, local intolerance reactions may be observed necessitating short term local treatment.
Diagnostic procedures involving the use of MRI contrast agents should be conducted under supervision of a physician with the prerequisite training and a thorough knowledge of the procedure to be performed. Appropriate facilities should be available for coping with any complication of the procedure, as well as for emergency treatment of severe reaction to the contrast itself.
The possibility of a reaction, including serious, life-threatening, fatal anaphylactoid or cardiovascular reactions or other idiosyncratic reactions should always be considered (see Section 4.8 Adverse Effects (Undesirable Effects)) especially in patients with a history of hypersensitivity.
Accumulation of gadolinium in the brain.
The current evidence suggests that gadolinium accumulates in the brain after multiple administrations of GBCAs. Increased signal intensity on non-contrast T1 weighted images of the brain has been observed after multiple administrations of GBCAs in patients with normal renal function. Gadolinium has been detected in brain tissue after multiple exposures to GBCAs, particularly in the dentate nucleus and globus pallidus. The evidence suggests that gadolinium accumulation is higher after repeat administration of linear than after repeat administration of macrocyclic agents.
The clinical significance of gadolinium accumulation in the brain is presently unknown. In order to minimise potential risks associated with gadolinium accumulation in the brain, it is recommended to use the lowest effective dose and perform a careful benefit risk assessment before administering repeated doses.
Central nervous system disorders.
Like with other GBCAs special precaution is necessary in patients with a low threshold for seizures. Precautionary measures should be taken, e.g. close monitoring. All equipment and therapies necessary to counter any convulsions which may occur must be made ready for use beforehand.
Hypersensitivity.
As with other GBCAs, hypersensitivity reactions can occur including life-threatening events (see Section 4.8 Adverse Effects (Undesirable Effects)). Hypersensitivity reactions may be either allergic (described as anaphylactic reactions when serious) or non-allergic. They can be either immediate (less than 60 minutes), or delayed (up to 7 days). However, as with other contrast media of this class, the occurrence of delayed reactions up to several days cannot be excluded. Anaphylactic reactions occur immediately and can be fatal. They are independent of the dose, can occur after even the first dose of the product and are often unpredictable.
There is always a risk of hypersensitivity regardless of the dose injected.
Patients with hypersensitivity or who have already experienced a reaction during previous administration of a gadolinium-containing MRI contrast media present an increased risk of experiencing another reaction on subsequent administration of the same product, or possibly other products, and are therefore considered to be at high risk.
As known from the use of iodinated contrast media, hypersensitivity reactions can be aggravated in patients on beta-blockers, and particularly in the presence of bronchial asthma. These patients may be refractory to standard treatment of hypersensitivity reactions with beta-agonists.
The injection of Dotagraf may aggravate symptoms of an existing asthma. In patients with asthma unbalanced by the treatment, the decision to use Dotagraf must be made after careful evaluation of the risk/benefit ratio.
Before any contrast media is injected, the patient should be questioned for a history of allergy (e.g. seafood allergy, hay fever, hives), sensitivity to contrast media and bronchial asthma as the reported incidence of adverse reactions to contrast media is higher in patients with these conditions and premedication with antihistamines and/or glucocorticoids may be considered. In such patients, the decision to use Dotagraf must be made after careful evaluation of the risk-benefit ratio.
During the examination, supervision by a physician is necessary. If hypersensitivity reactions occur, administration of Dotagraf must be discontinued immediately and, if necessary, specific therapy instituted. A venous access should thus be kept during the entire examination.
To permit immediate emergency countermeasures, appropriate therapies (e.g. epinephrine and antihistamines), an endotracheal tube and a respirator should be ready at hand.
Use in the elderly.
As the renal clearance of gadoteric acid may be impaired in the elderly, it is particularly important to screen patients aged 65 years and older for renal dysfunction.
Paediatric use.
Neonates and infants.
Due to immature renal function in neonates up to 4 weeks of age and infants up to 1 year of age, gadoteric acid should only be used in these patients after careful consideration.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
No interactions with other medicinal products have been observed. Formal interaction studies have not been carried out.
Concomitant medications to be taken into account.
Beta-blockers, vasoactive substances, angiotensin-converting enzyme inhibitors, angiotensin II receptor antagonists.
These medicinal products decrease the efficacy of the mechanisms of cardiovascular compensation for blood pressure disorders. The radiologist must be informed before injection of gadolinium complexes and resuscitation equipment must be at hand.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Gadoteric acid had no effects on the reproduction and fertility of male or female rats at repeat doses up to 1.6 mmol/kg/day, or approximately 2.6 times the clinical dose, adjusted for body surface area. No peri-postnatal toxicity studies have been conducted with gadoteric acid in animals.
Data on the use of gadolinium-based contrast agents, including gadoteric acid, in pregnant women is limited. Gadolinium can cross the placenta. It is unknown whether exposure to gadolinium is associated with adverse effects in the foetus. Animal studies do not indicate direct or indirect harmful effects with respect to reproductive toxicity. Gadoteric acid should not be used during pregnancy unless the clinical condition of the woman requires the use of gadoteric acid.
Following administration of an intravenous dose of gadoteric acid to rats on gestation day 18, the maximum total fetal concentration of gadolinium was 5% of the maternal plasma concentration, and the maximum quantity of gadolinium in the whole litter was 0.07% of the total dose.
Teratology studies in which rats and rabbits were administered gadoteric acid at an intravenous dose up to 0.8 mmol/kg/day, or approximately 1.3 times (rats) or 2.4 times (rabbits) the clinical dose, adjusted for body surface area, showed no evidence of embryotoxicity or teratogenicity. In the rat study, bodyweights of high-dose offspring were reduced up to mid-lactation, but subsequent mating performance and fertility were unaffected.
No studies have been carried out concerning the passage of gadoteric acid into human breast milk. If an investigation is indicated during lactation, it is advisable that lactation woman discard their milk for the 24 hours following administration.
In goats administered an intravenous dose of gadoteric acid, excretion in milk accounted for 0.02% of the total dose, and gadoteric acid excretion was only detectable during the first 24 hours.4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed. Ambulant patients while driving vehicles or operating machinery should take into account that nausea may incidentally occur.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials.
Table 2 shows the adverse experiences reported among patients in controlled clinical trials of intravenous gadoteric acid, in the management of brain, spine and whole body pathologies.
Adverse reactions are presented in Table 2 by system organ class and by frequency according to the following categories: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1000 to < 1/100), rare (≥ 1/10,000 to < 1/1000), very rare (< 1/10,000) not known (cannot be estimated from the available data).
 Table 3 shows the adverse event experience reported in a study which compared gadoteric acid and dimeglumine gadopentetate in imaging of brain and spine pathologies. The adverse events reported in subjects who received gadoteric acid are also included in the adverse experiences reported in Table 2.
Table 3 shows the adverse event experience reported in a study which compared gadoteric acid and dimeglumine gadopentetate in imaging of brain and spine pathologies. The adverse events reported in subjects who received gadoteric acid are also included in the adverse experiences reported in Table 2.

Post-marketing experience.
Since post-marketing, the most commonly reported adverse reactions following administration of gadoteric acid are nausea, vomiting, pruritus and hypersensitivity reactions.
In hypersensitivity reactions, the reactions most frequently observed are skin reactions, which can be localised, extended or generalised. These reactions are usually immediate (during the injection or within one hour after the start of injection) or sometimes delayed (one hour to several days after injection), presenting as skin reactions.
Immediate reactions include one or more effects which appear simultaneously or sequentially, which are most often cutaneous, respiratory, gastrointestinal, articular and/or cardiovascular reactions.
Each sign may be a warning sign of a shock starting, however, it is very rarely fatal.
Rare anaphylactoid reactions have been reported that may be very rarely severe, life-threatening or have a fatal outcome, particularly in patients with a history of allergy. These allergic reactions can occur irrespective of the amount administered and the mode of administration and may take the form of one or more of the following symptoms: Angioedema, anaphylactic shock, circulatory and cardiac arrest, hypotension, larynx oedema, bronchospasm, laryngospasm, pulmonary oedema, dyspnoea, stridor, coughing, pruritus, rhinitis, sneezing, conjunctivitis, urticaria and rash. Some of these symptoms may be the first signs of incipient state of anaphylactic shock.
Isolated cases of nephrogenic systemic fibrosis (NSF) have been reported with gadoteric acid, most of which were in patients co-administered other GBCAs (see Section 4.4 Special Warnings and Precautions for Use). See Table 4.
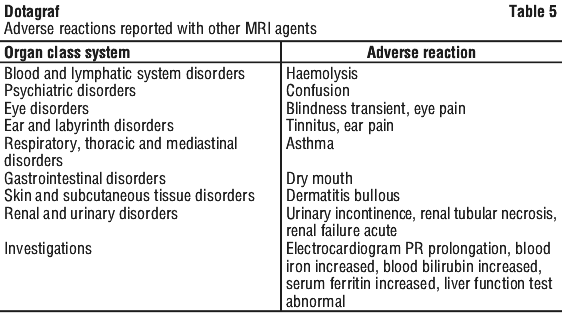
 The following adverse reactions were reported with other intravenous contrast agents for MRI. See Table 5.
The following adverse reactions were reported with other intravenous contrast agents for MRI. See Table 5.
Adverse reaction in paediatrics.
Safety of paediatric patients was considered in clinical trials and post-marketing studies. As compared to adult, the safety profile of gadoteric acid did not show any specificity in children. Most reactions are gastrointestinal symptoms or signs of hypersensitivity.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
There is no experience to date of overdose with gadoteric acid.
Gadoteric acid can be removed by haemodialysis. However, there is no evidence that haemodialysis is suitable for prevention of nephrogenic systemic fibrosis (NSF).
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Gadoteric acid has paramagnetic properties which increase contrast enhancement in MRI. The presence of seven (7) unpaired electrons in the gadolinium outer shell explains that this ion has a high magnetic moment and thus a strong paramagnetic effect. Gadoteric acid shortens the longitudinal relaxation time (T1) and the transverse relaxation time (T2) of water protons in tissues where it is distributed. At clinical doses the major effect is on T1 relaxation time. This results in an increased signal intensity in T1-weighted sequences and allows a better visualisation of abnormal structures or lesions.
Gadoteric acid has no specific pharmacodynamic activity and is biologically inert.
Clinical trials.
Two randomised, double blind studies compared diagnostic information from gadoteric acid and dimeglumine gadopentetate, both administered at 0.1 mmol/kg, in enhancement of MRI of intracranial and spinal lesions. A total of 319 adult patients with CNS or spinal pathology were enrolled in the two studies. Efficacy was evaluated by comparison of pre and post contrast MRI by investigators. Diagnostic efficacy scores (diagnostic quality, diagnostic consequences and management consequences) were similar in the two treatment groups.
Open label clinical studies evaluated the pre and post contrast diagnostic efficacy of gadoteric acid in MRI of histologically confirmed primary and secondary liver tumours, bone and soft tissue tumours, uterine and ovarian tumours and local recurrence of breast cancers.
Three open label studies evaluated the efficacy and safety of 0.1 mmol/kg gadoteric acid in paediatric populations. A total of 99 children, in the age range 0.04 to 17 years, were evaluated who required neurological examination by MRI. Only one child aged less than one month received gadoteric acid. There is limited information in children (n=9) aged less than 2 years.
5.2 Pharmacokinetic Properties
After intravenous injection, gadoteric acid is distributed in the extracellular fluid of the body. It does not bind with plasma albumin.
Current evidence suggests that gadolinium accumulates in the brain after repeated administrations of GBCAs although the exact mechanism of gadolinium passage into the brain has not been established.
In patients with normal renal function, the plasma half-life is approximately 90 minutes. The volume of distribution is approximately 0.25 L/kg, the plasma clearance rate is approximately 0.1 L/h/kg and about 90% of the product is excreted in urine in 24 hours. It is eliminated by glomerular filtration in unchanged form. Plasma clearance is decreased to 0.036 L/h/kg in moderate renal failure and to 0.012 L/h/kg in severe renal failure.
5.3 Preclinical Safety Data
Animal toxicity.
The acute toxicity of gadoteric acid injected intravenously (2 mL min-1) was studied in mice (at doses between 8 and 13 mmol/kg) and in rats (at a dose of 12.6 mmol/kg). Clinical signs of toxicity were convulsions and transient respiratory disorders. Deaths occurred in the two studies, in mice from a dose of 9 mmol/kg upwards, or approximately 7 times the clinical dose (20 times in rats), adjusted for body surface area. Necropsy revealed a haemorrhagic appearance in the lungs and sometimes in the kidney. In studies of the proconvulsive potential of gadoteric acid, and intravenous dose up to 8 mmol/kg had no convulsive effects in mice, but an intravenous dose of 4 mmol/kg in mice, or an intracisternal dose of 1 or 2 micromol in rats, slightly potentiated the convulsive effects of a subthreshold dose of picrotoxin. In toxicity studies, rats and dogs administered gadoteric acid for 28 days at daily intravenous doses up to 1.5 mmol/kg, or approximately 2.4 times and 8 times, respectively, the clinical dose, adjusted for body surface area, caused vacuolisation of the proximal tubular cells of the kidney. This effect was fully reversible in dogs, and partly reversible in rats, over a 28-day recovery period.
Genotoxicity.
Gadoteric acid was not mutagenic in Salmonella typhimurium, nor in Chinese hamster V79 cells, and did not induce chromosomal aberrations in Chinese hamster ovary cells in vitro, with or without metabolic activation. Gadoteric acid did not induce micronuclei in mice following an intravenous dose of up to 3.5 mmol/kg.
Carcinogenicity.
The carcinogenic potential of gadoteric acid has not been investigated in long-term animal studies.6 Pharmaceutical Particulars
6.1 List of Excipients
Dotagraf also contains meglumine, tetraxetan (DOTA) and water for injections. It contains no antimicrobial preservatives.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.4 Special Precautions for Storage
Store below 30°C.
In-use.
Unused Dotagraf in opened containers must be discarded immediately.
Unused Dotagraf in a 100 mL bottle and used with a multi-patient injector system must be discarded within 24 hours after first opening the container.
6.5 Nature and Contents of Container
Dotagraf is a solution containing 0.5 mM of gadoteric acid and is registered in packs of 10 x 10 mL, 10 x 15 mL, 10 x 20 mL type II clear glass vials or 10 x 60 mL and 10 x 100 mL type II clear glass bottles. Each vial or bottle contains a synthetic latex-free bromobutyl rubber stopper with an aluminium snap cap and a plastic flip off lid.
Not all presentations are marketed.
6.6 Special Precautions for Disposal
Any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
Chemically, gadoteric acid is 1,4,7,10-tetraazacyclododecane-N, N', N'', N'''-tetraacetic acid gadolinium complex and has the following structural formula.
 Molecular weight: 558.64.
Molecular weight: 558.64.
CAS number.
72573-82-1.
Chemical formula: C16H25GdN4O8.7 Medicine Schedule (Poisons Standard)
Not Scheduled.
Summary Table of Changes


 The product is intended for intravenous administration only.
The product is intended for intravenous administration only. Table 3 shows the adverse event experience reported in a study which compared gadoteric acid and dimeglumine gadopentetate in imaging of brain and spine pathologies. The adverse events reported in subjects who received gadoteric acid are also included in the adverse experiences reported in Table 2.
Table 3 shows the adverse event experience reported in a study which compared gadoteric acid and dimeglumine gadopentetate in imaging of brain and spine pathologies. The adverse events reported in subjects who received gadoteric acid are also included in the adverse experiences reported in Table 2.
 The following adverse reactions were reported with other intravenous contrast agents for MRI. See Table 5.
The following adverse reactions were reported with other intravenous contrast agents for MRI. See Table 5.
 Molecular weight: 558.64.
Molecular weight: 558.64.