Notes
Distributed by Sandoz Pty Ltd
1 Name of Medicine
Doxylamine succinate.
2 Qualitative and Quantitative Composition
Each Dozavit tablet contains doxylamine succinate 25 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Dozavit tablets are a white, uncoated tablet with break-bar on one side.
4.1 Therapeutic Indications
For the temporary relief of sleeplessness.
4.2 Dose and Method of Administration
Adults and children over 12 years.
1 or 2 tablets, with water, 20 minutes before retiring.
Do not exceed the recommended dose.4.3 Contraindications
Dozavit is contraindicated in patients with:
Epilepsy.
Narrow angle glaucoma.
Known hypersensitivity to doxylamine succinate.
Known hypersensitivity to any other ingredients in the product (see Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
Caution should be exercised for people with medical conditions such as:
Prostatic enlargement.
Urinary retention.
Glaucoma.
Pyloroduodenal obstruction.
Cardiovascular disorders.
Use in the elderly.
No data available.
Paediatric use.
Not recommended for the use in children under 12 years of age except on medical advice.
Effects on laboratory tests.
Skin tests.
Antihistamines should be discontinued approximately 48 hours prior to any skin tests since these drugs may suppress positive reactions to dermal reactivity indicators.4.5 Interactions with Other Medicines and Other Forms of Interactions
Doxylamine succinate may enhance the sedative effect of other central nervous system (CNS) depressants such as opioid analgesics, neuroleptics, alcohol, hypnotics and psychotherapeutic drugs.
Monoamine oxidase inhibitors (MAOIs) may enhance anti-muscarinic effects.
Additive anti-muscarinic action with other anti-muscarinic drugs such as atropine and tricyclic antidepressants.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category A)
In the first trimester of pregnancy, doxylamine succinate tablets should be used only when the medical practitioner considers it essential for the welfare of the patient.
Doxylamine succinate should not be given to breast-feeding women, as it may inhibit lactation. Doxylamine succinate may be excreted in human milk.4.7 Effects on Ability to Drive and Use Machines
Doxylamine succinate may cause drowsiness in some patients. Such patients should avoid operating vehicles or machinery or engaging in activities which require them to be fully alert. Avoid alcohol.
4.8 Adverse Effects (Undesirable Effects)
Generally, antihistamine may cause:
Dry mouth, dizziness, gastrointestinal disturbances, blurred vision, urinary difficulty and retention, paradoxical CNS stimulation (insomnia, nervousness, euphoria, irritability, tremors, nightmares, hallucinations and convulsions), tachycardia.
Also, the following have been reported:
Epigastric distress, thickening of bronchial secretions, hypersensitivity reactions.
Rarely, the following have been reported:
Blood disorders, hypotension, tinnitus, headache, paraesthesia.
Doxylamine succinate may cause excessive drowsiness in some individuals.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Doxylamine succinate ingested in large amounts may cause impaired consciousness and less commonly cause psychotic behaviour, seizures, dilated pupils, tachycardia, and in one case, rhabdomyolysis had been reported.
Treatment.
Supportive care, decontamination and enhancing elimination are useful in the treatment of overdose with doxylamine.
Supportive care.
Assessment of airway, breathing and circulation should be performed.
Decontamination.
Administration of activated charcoal in a dose of 1-2 g/kg of body weight for children and 50-100 g for adults. Each gram of charcoal should be diluted with 6-10 mL of water. Proprietary products such as Carbosorb and Carobosorb S are available. In irrational patients, 1-2 mg IV of physiostigmine may be useful to achieve cooperation with gastrointestinal decontamination.
Enhancing elimination.
The clearance of the drug can be achieved by repeated doses of activated charcoal. Activated charcoal should not be used in patients in whom bowel sounds are absent.
(Reference "Hypotox on line" suggested by Poison Information Centre and APF 16th Ed.).
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Doxylamine succinate is an antihistamine of the ethanolamine class with pronounced sedative and anti-muscarinic effects.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Doxylamine succinate has a peak plasma level 99 nanogram/mL and time of peak 2.4 hours.1
Doxylamine succinate has an elimination half-life of 10.1 hours and apparent oral clearance of 217 mL/minute.1
Elderly and young women did not differ significantly in peak plasma, time to Cmax, elimination half-life, volume of distribution or clearance. Cmax and tmax. Also, these parameters did not differ between elderly and young men. However, elderly men had reduced doxylamine clearance and prolonged half-life in comparison to elderly women.2
1 Friedman H; Greenblatt DJ. The pharmacokinetics of doxylamine: use of automated gas chromatography with nitrogen phosphorus detection. J Clin Pharmacol 1985:25: 448-51.
2 Friedman H, et al. Clearance of the antihistamine doxylamine: reduced in elderly men but not in elderly women. Clin Pharmacokinetic 1989; 16; 312-16.
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
No data available.6 Pharmaceutical Particulars
6.1 List of Excipients
Calcium hydrogen phosphate dihydrate, povidone, pregelatinised maize starch, microcrystalline cellulose, croscarmellose sodium, magnesium stearate, purified talc.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
2 years.
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Container type.
PVC/PVDC-Aluminium blister packs in a carton.
Pack sizes.
10 tablets (blister pack of 10 tablets in a carton).
12 tablets (blister pack of 12 tablets in a carton).
20 tablets (2 blister packs of 10 tablets in a carton).
Not all presentations may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Doxylamine succinate is a white or creamy white powder. Soluble in water (1 in 1), soluble in alcohol and in chloroform (1 in 2), slightly soluble in ether (1 in 370) (USP).
Chemical structure.
Chemical structure of doxylamine succinate.
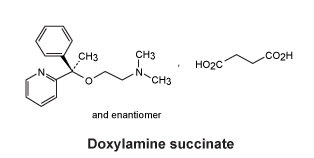 Chemical name: N,N-Dimethyl-2-[α-methyl-α-(2-pyridyl)benzyloxy]ethylamine hydrogen succinate.
Chemical name: N,N-Dimethyl-2-[α-methyl-α-(2-pyridyl)benzyloxy]ethylamine hydrogen succinate.
Molecular formula: C21H28N2O5.
Molecular weight: 388.5.
CAS number.
562-10-7.7 Medicine Schedule (Poisons Standard)
S3 - Pharmacist Only Medicine.
Summary Table of Changes
