What is in this leaflet
This leaflet answers some common questions about Endometrin Pessaries.
The leaflet does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using Endometrin against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Endometrin is used for
Endometrin is provided as a pessary (or vaginal tablet) that contains the natural female hormone, progesterone.
Endometrin is for women who need extra progesterone while undergoing treatment in an Assisted Reproductive Technology (ART) programme (e.g. IVF).
Progesterone acts on the lining of the uterus (womb) and it helps you to become and to stay pregnant when you are treated for infertility.
Your doctor may have prescribed Endometrin for another purpose. Ask your doctor if you have any questions why this medicine has been prescribed for you.
Endometrin is available only with a doctor's prescription.
This medicine is not addictive.
Before you use Endometrin
When you must not use it
Do not use Endometrin if you have an allergy to:
- any medicine containing progesterone
- any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include:
- skin rash, itching or hives
- swelling of the face, lips, tongue or other parts of the body
- shortness of breath, wheezing or difficulty breathing.
Do not use Endometrin if you have or have had any of the following conditions:
- unusual vaginal bleeding that has not been evaluated by your doctor
- missed abortion or ectopic pregnancy (pregnancy outside of the womb)
- severe liver problems
- known or suspected cancer of the breast or genital tract
- blood clots in the legs, lungs, eyes or elsewhere in the body
- porphyria disorder (a blood disease).
Do not use Endometrin if you are breast-feeding. If you take this medicine after the expiry date has passed, it may not work.
Do not use Endometrin after the expiry date on the pack has passed.
Do not use Endometrin if the packaging is torn or shows signs of tampering or the product does not look quite right. If it has expired or is damaged, return it to your pharmacist or doctor for disposal.
If you are not sure whether you should start using Endometrin talk to your doctor.
Before you start to use it
Tell your doctor or pharmacist if you have allergies to:
- any other medicines
- any other substances, such as foods, preservatives or dyes.
You must tell your doctor or pharmacist if you have or have had any of the following problems:
- epilepsy
- migraine
- asthma
- heart, liver or kidney disease
- diabetes
- history of depression.
If you have not told your doctor about any of the above, tell them before you use Endometrin.
Taking other medicines
Tell your doctor if you are taking any other medicines, including medicines that you buy without a prescription from your pharmacy, supermarket or health food shop. Some medicines may affect the way other medicines work.
Some medicines may interfere with progesterone if taken at the same time. These include:
- rifampicin
- carbamazepine
- St John's Wort (herbal product)
- ketoconazole.
Endometrin should not be used at the same time as other vaginal preparations. Your doctor or pharmacist has more information on medicines to be careful with or avoid while using Endometrin.
How to use Endometrin
How much to use
Your doctor or pharmacist will tell you how many tablets you need to use each day. The usual dosage is one 100 mg tablet placed directly into the vagina three times daily starting on the day of egg retrieval.
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
Endometrin tablets are intended to be placed directly into the vagina. An applicator is provided in the pack to help administer the tablets.
Follow the directions on how to administer the tablet below:
- wash your hands thoroughly with soap and water
- unwrap the applicator and remove one vaginal tablet from the blister
- put one tablet in the space provided at the end of the applicator. The tablet should fit securely and not fall out
- the applicator with the tablet may be inserted into the vagina while you are standing, sitting, or when lying on your back with your knees bent. Gently insert the thin end of the applicator well into the vagina
- push the plunger to release the tablet
- remove the applicator and rinse it thoroughly in warm running water, wipe dry with a soft tissue and keep the applicator for subsequent use.
The directions on how to administer the tablet are also provided with each pack of Endometrin.
If you do not understand the instructions, ask your doctor or pharmacist for help.
How long to use it
The use of Endometrin may be continued for up to 10 weeks if pregnancy is confirmed (12 week of gestation).
If you forget to use it
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to, as usual.
Otherwise, use it as soon as you remember, and then go back to using your medicine as you would normally.
Do not use a double dose to make up for the dose that you missed.
If you are not sure what to do, talk to your doctor or pharmacist. If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
If you use too much (overdose):
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26), or go to accident and emergency at your nearest hospital, if you think that you or anyone else may have used too much Endometrin. Do this even if there are no signs of discomfort or poisoning.
While you are using Endometrin
Things you must do
Tell any other doctors or pharmacists who are treating you that you are using Endometrin.
If you are about to start taking any new medicines, tell your doctor or pharmacist that you are using Endometrin.
Take special care and tell your doctor straight away if you experience any of these symptoms during treatment or even a few days after the last dosage:
- pains in your calves or chest, a sudden shortness of breath or coughing blood indicating possible clots in the legs, heart or lungs
- severe headache or vomiting, dizziness, faintness or changes in vision or speech, weakness or numbness of an arm or leg indicating possible clots in the brain or eye
- worsening symptoms of depression.
Things you must not do
Do not give Endometrin to anyone else, even if they have the same condition as you.
Do not use Endometrin to treat any other complaints unless your doctor has told you to.
Do not stop using Endometrin or lower the dosage, without checking with your doctor or pharmacist.
Things to be careful of
Be careful driving or operating machinery until you know how Endometrin affects you. Some people may experience drowsiness or dizziness. Make sure you know how you react to Endometrin before you drive a car, operate machinery or do anything else that could be dangerous if you are dizzy or light-headed. If this occurs, do not drive.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using Endometrin. Endometrin helps most people with low progesterone levels, but it may have unwanted side effects in a few people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- headache
- vaginal disorders (such as discomfort, burning sensation, discharge, dryness and bleeding)
- uterine cramping
- abdominal pain or bloating (swelling)
- nausea.
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Tell your doctor as soon as possible if you notice any of the following:
- dizziness
- insomnia
- diarrhoea
- constipation
- urticaria (allergic rash)
- rash
- vaginal disorders (e.g. vaginal discomfort, burning sensation, discharge, dryness and bleeding)
- fungal infections in the vagina (e.g. vaginal thrush)
- breast disorders (e.g. breast pain, breast swelling and breast tenderness)
- itching in the genital area
- swelling of the limbs.
The above list includes serious side effects which may require medical attention. Serious side effects are rare.
Other side effects not listed above may also occur in some patients. Tell your doctor if you notice anything that is making you feel unwell.
Ask your doctor or pharmacist if you do not understand anything in this list.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After using Endometrin
Storage
Keep this medicine in its original packaging until it is time to use them.
Keep this medicine in a cool dry place where the temperature stays below 25°C.
Do not store it or any other medicine in the bathroom or near a sink.
Do not leave it in the car or on window sills.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines
Disposal
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that are left over.
Product description
What it looks like
Endometrin Pessaries are white to off-white, convex and oblong tablets with the inscriptions "FPI" on one side and "100" on the other side. Endometrin Pessaries are supplied in blister strips packaged in an outer carton. Each carton contains 21 tablets and one vaginal applicator.
Ingredients
Active ingredient:
- progesterone.
Inactive ingredients:
- colloidal anhydrous silica
- lactose monohydrate
- pregelatinised maize starch
- povidone
- adipic acid
- sodium bicarbonate
- sodium lauryl sulphate
- magnesium stearate.
Sponsor
Endometrin Pessaries are supplied in Australia by:
Ferring Pharmaceuticals Pty Ltd
Suite 2, Level 1, Building 1,
20 Bridge Street
Pymble, NSW 2073
This leaflet was prepared in September 2019.
Endometrin Pessaries 100 mg
AUST R 189948
DOCS#30117v2A
Published by MIMS December 2019



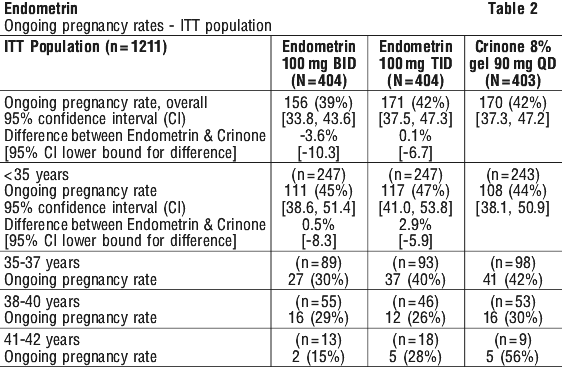 Endometrin 100 mg TID met the noninferiority criterion relative to Crinone 8%, as Endometrin TID was well within the 10% lower bound to demonstrate noninferiority in ongoing pregnancy rate to Crinone 8%. Endometrin BID was just above the 10% lower bound in ongoing pregnancy rate.
Endometrin 100 mg TID met the noninferiority criterion relative to Crinone 8%, as Endometrin TID was well within the 10% lower bound to demonstrate noninferiority in ongoing pregnancy rate to Crinone 8%. Endometrin BID was just above the 10% lower bound in ongoing pregnancy rate.

