What is in this leaflet?
This leaflet answers some common questions about using the medicine Enstilar®. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using Enstilar® against the benefits it is expected to have for you.
If you have any concerns about using Enstilar®, talk to your doctor or pharmacist.
What Enstilar® is used for
Enstilar® is a topical treatment for psoriasis. Psoriasis is a skin disorder in which skin cells are overactive. This causes redness, scaling and thickness of your skin. Enstilar® contains two active ingredients:
- calcipotriol as monohydrate (related to Vitamin D);
- betamethasone dipropionate (a corticosteroid).
The calcipotriol in Enstilar® treats the overactive skin cells. The betamethasone dipropionate relieves redness, swelling, itching and irritation of the skin.
Enstilar® is available only with a doctor’s prescription.
This medicine is not expected to affect your ability to drive a car or operate machinery.
This medicine is not addictive.
Before you use Enstilar®
When you must not use Enstilar®
Do not use Enstilar® if you:
- are allergic to calcipotriol, betamethasone dipropionate or any of the other ingredients listed at the end of this leaflet (see Product description) or any other similar medicine, such as topical corticosteroids
- have high blood calcium levels (ask your doctor)
- have other types of psoriasis known as generalised pustular or erythrodermic exfoliative psoriasis (your doctor will identify these conditions for you)
Some of the symptoms of an allergic reaction include shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or “hives” on the skin.
Do not use Enstilar® on skin affected by:
- viral, fungal, bacterial or parasitic skin infections
- tuberculosis (TB)
- perioral dermatitis (red rash around the mouth)
- thin skin, easily damaged skin, stretch marks
- ichthyosis (dry skin with fish like scales)
- acne
- rosacea
- ulcers or broken skin
Inform your doctor if you are suffering from any illness before using Enstilar®.
Do not use Enstilar®
- after the expiry date printed on the pack OR
- if the can has been opened for more than 6 months OR
- if the packaging is torn, doesn’t look quite right or shows signs of tampering.
If any of these apply, return the medicine to your pharmacist for disposal.
If you are not sure whether you should start using Enstilar®, talk to your doctor.
Before you start to use it
Tell your doctor if you have a known allergy to Enstilar® calcipotriol, betamethasone dipropionate or any of the ingredients listed at the end of this leaflet.
Tell your doctor if you
- are pregnant
- are breast-feeding
- intend to breast feed.
Do not use Enstilar® in children. Safety and effectiveness of Enstilar® in those below 18 years of age has not been established.
If you have not told your doctor about any of the above, tell him/her before you start using this medicine.
Other medicines
Tell your doctor or pharmacist if you are using any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Your doctor and pharmacist have more information on medicines you should be careful with or avoid whilst using Enstilar®.
How to use Enstilar®
Caution: Extremely flammable. Please read important information under ‘Storage’.
Follow carefully all directions given to you by your doctor or pharmacist. They may differ from the information contained in this leaflet.
If you do not understand the instructions in this leaflet, ask your doctor or pharmacist for help.
Enstilar® is for topical use (on the skin). Before use, read the patient information, even if you have used Enstilar® before. Enstilar® is designed for direct application (spray on) to your skin, where it is affected by psoriasis.
Important: Do not use more than 15g in one day. 15g corresponds to the amount sprayed from the can when the actuator is fully pressed down for 1 minute. Do not use more than 100g in one week.
For example, no more than 2 cans in one week. One 60g can should last you at least 4 days.
If you also use other calcipotriol containing products (e.g.: Daivobet® gel or ointment, Daivonex®) for your psoriasis, then the total amount of calcipotriol products must not exceed 15g per day or 100 grams per week.
NOTE: If you use more than the recommended amount of Enstilar®, the levels of calcium in your blood may rise. This is known as “hypercalcaemia”.
Hypercalcaemia:
- is measured by a blood test
- may be harmful
- may mean that Enstilar® or any other product containing calcipotriol (e.g.: Daivobet® gel or ointment, Daivonex®) should not be used until the calcium level has become normal again.
Blood calcium levels will quickly normalise when your doctor discontinues Enstilar® treatment.
How to use Enstilar®
Enstilar® should be applied to dry skin on the affected areas of the body or scalp.
For body psoriasis
- Shake the can for a few seconds before use and remove the cap.
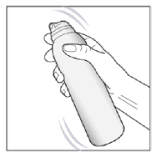
- Apply the medication by holding the can at least 3cm (approximately 1.5 inches) from the skin and spray onto the affected area.
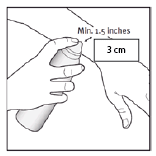
- The foam can be sprayed by holding the can in any orientation except horizontally. Apply a layer of foam only to the psoriasis plaques. Spray directly onto each affected skin area and gently rub in. If you accidentally put some of the foam on normal skin, wipe it off as soon as possible.
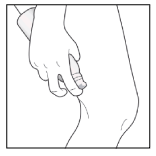
- Gently rub the foam into your affected skin areas
You will need to repeat steps 1 to 4 until all of the affected areas are covered.
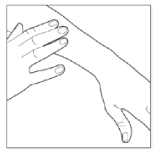
- After applying your medication, put the cap back on the can to prevent accidental pressing of the can when not in use.
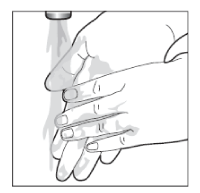
- Wash your hands well after using Enstilar® (unless you are using the foam to treat your hands). This will avoid accidentally spreading the foam to other parts of your body (especially face, mouth and eyes).
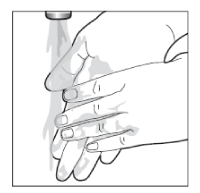
- Take care not to get the foam spray in your eyes. If this happens, rinse your eyes with clean water and seek medical advice.
- If you accidentally put some of the foam onto skin not affected by psoriasis, wipe it off as soon as possible.
- Do not bandage, tightly cover or wrap the treated area
For best results leave on during the day or night. The treated area does not need to be covered. You may wear your usual clothes however avoid contact with fabric that is easily stained by grease (e.g. silk).
For scalp psoriasis
- Comb the hair to remove any loose scales.
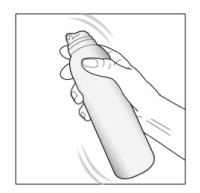
- Shake the can for a few seconds before use and remove the cap.
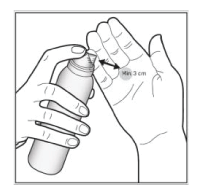
- Hold the can at least 3 cm (1.5 inches) away. Spray directly into the palm of your hand. The foam can be sprayed holding the can in any orientation except horizontally.
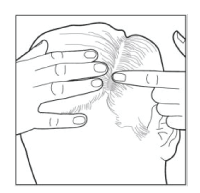
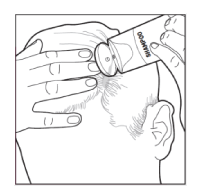
- Scoop the foam onto your finger and apply it directly to the affected areas of the scalp. Minimize applying into the hair to make it easier to wash it out. Gently rub the foam into the scalp.
- After applying your medication, put the cap back on the can to prevent accidental pressing of the can when not in use
- Wash your hands well after using Enstilar®. This will avoid accidentally spreading the foam to other parts of your body (especially the face, mouth and eyes).
- Take care not to get the foam in your eyes. If this happens, rinse your eyes with clean water and tell your doctor
- If you accidentally put some of the foam onto skin not affected by psoriasis, wipe it off as soon as possible
- Do not bandage, tightly cover or wrap the treated area
For best results, do not wash your hair immediately after applying. Leave Enstilar® on during the day or night. The treated area does not need to be covered.
After applying to the scalp, the following instructions when washing your hair, might be useful.
- Apply a mild, non-medicated shampoo to the dry hair, focusing on the areas where the foam was applied.
It is easier to remove Enstilar® when the shampoo is applied to dry hair as water dilutes the cleansing effect of the shampoo.
- Massage the shampoo into the dry hair/scalp. Leave the shampoo on the scalp for a couple of minutes before washing.
- Rinse thoroughly with water.
- Repeat normal shampooing if necessary.
Do not use Enstilar®
- on large areas of damaged skin;
- in skin folds (eg. groin, underarm);
- on the genitalia;
- inside the mouth, nose or in the eyes;
- on the face (an itchy red rash may develop).
- under occlusive dressings (such as a bandage).
Wash your hands thoroughly after applying Enstilar® on any part of your body, unless your hands are being treated.
Thoroughly wash your hands prior to handling infants or children.
If any skin irritation develops, wash off the Enstilar®.
When to use Enstilar®
Apply Enstilar® once a day at the most convenient time for you, trying to applying it about the same time of the day each day (for example, morning, afternoon or evening).
As a guide on how much Enstilar® to use: A short 2 second spray of Enstilar® provides 0.5g of the medication and this amount should be sufficient to cover an area of skin roughly corresponding to the area of an adult hand.
How long to use it
Each treatment course would normally last for up to four weeks.
Your doctor may decide to stop treatment with Enstilar® once your psoriasis has improved, and to recommence it as needed. Your doctor may decide on a different treatment period and/or repeated treatment.
If you forget to use it
If you forget to use Enstilar®, use it as soon as you remember, then continue as before. Do not use an increased dose of the foam to make up for the forgotten doses.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to use your medicine, ask your doctor or pharmacist for some hints.
If you use too much or use it other than directed (Overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26 in Australia) for advice, or go to the Emergency Department at the nearest hospital, if you think that you or anyone else may have used too much or swallowed some of this medicine. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using Enstilar®
Things you must do
While you are using Enstilar®, protect the treated skin areas from sunlight and Ultraviolet (UV) rays, for example, by wearing protective clothing. This is particularly important if your job or lifestyle means you spend time outdoors
If you are going to start any new medicine, tell your doctor and pharmacist that you are using Enstilar®.
If you become pregnant while using Enstilar®, tell your doctor immediately.
Keep all appointments with your doctor so that your progress can be checked. Your doctor may do blood tests whilst you are using Enstilar® (or any other product containing calcipotriol, for example Daivobet® gel or ointment, Daivonex®). These tests check the levels of calcium in your blood and how well your kidneys are working (“renal function”).
Things you must not do
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not use more than the recommended daily or weekly dose (see How to use Enstilar®).
Do not use other topical corticosteroids (e.g. Advantan®, Diprosone®, Elocon®) on areas treated with Enstilar®.
Side effects
As with all medicines, Enstilar® can cause some side effects.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist:
- if you have any unpleasant effects while you are using Enstilar®
or - if you notice any of the following and they worry you:
- skin irritation (including itching, redness, dryness, swelling or peeling)
- skin pain (including burning)
- a rash on your face or body
- a change in the colour of your skin
- your skin is more sensitive to light
- change in control of diabetes
Enstilar® contains butylated hydroxytoluene. This may cause local skin reactions (e.g. contact dermatitis), or irritation to the eyes and mucous membranes.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using Enstilar®
Keep Enstilar® in the box until required.
Storage
Keep Enstilar® in a cool dry place, store below 25°C.
CAUTION: Extremely flammable Pressurised container: May burst if heated. Protect from sunlight and do not expose to temperatures above 50°C. Do not pierce or burn, even after use. Do not spray on an open flame or other ignition source. Keep away from sparks, open flames and other ignition sources. Do not smoke while applying Enstilar®.
Keep Enstilar® in the box to protect from light.
Keep Enstilar® where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
Ask your pharmacist what to do with any Enstilar® that is left over if:
- your doctor tells you to stop using Enstilar®, or
- the can has been opened for more than 6 months or
- the expiry date has passed.
Product description
What it looks like
Enstilar® is in a pressurised can. After spraying, a white to off-white foam is formed on the skin. The foam is a non-expanding foam, which gradually collapses after spraying.
Ingredients
Enstilar® contains the active ingredients:
- calcipotriol (50 microgram/g) [as monohydrate]
- betamethasone dipropionate (500 microgram/g).
It also contains:
- liquid paraffin
- polyoxypropylene-11 stearyl ether
- dl-alpha-tocopherol
- White soft paraffin
- Butylated hydroxytoluene (included as an antioxidant in polyoxypropylene-11 stearyl ether)
- Propellants: butane and methyl ether
The Australian Registration Number is AUST R 263292.
This is not all the information available on Enstilar®. If you have any more questions or are unsure about anything, ask your doctor or pharmacist.
Supplier
Enstilar® is supplied in Australia by:
LEO Pharma Pty Ltd
Brisbane, QLD Australia
Toll free no: 1800 991 778
Web: www.leo-pharma.com.au
Enstilar, Daivobet, Daivonex, LEO and LEO/lion device are registered trademarks of LEO Pharma A/S.
This leaflet was prepared in July 2020.
Published by MIMS January 2021

 The following adverse reactions are considered to be related to the pharmacological classes of calcipotriol and betamethasone, respectively.
The following adverse reactions are considered to be related to the pharmacological classes of calcipotriol and betamethasone, respectively.
 Results for the primary endpoint 'treatment success' (PGA) of body at week 4 showed Enstilar to be statistically significantly more effective than all the comparators included and responses were observed in all categories of baseline disease severity.
Results for the primary endpoint 'treatment success' (PGA) of body at week 4 showed Enstilar to be statistically significantly more effective than all the comparators included and responses were observed in all categories of baseline disease severity. Enstilar was statistically significantly more effective compared to calcipotriol and also associated with a higher rate of treatment success than BDP but this comparison did not reach statistical significance.
Enstilar was statistically significantly more effective compared to calcipotriol and also associated with a higher rate of treatment success than BDP but this comparison did not reach statistical significance.
 Statistically significant differences of at least 70% reduction in both itch and itch related sleep loss were observed in favour of subjects receiving Enstilar compared to those receiving foam vehicle from day 3 and throughout the treatment period.
Statistically significant differences of at least 70% reduction in both itch and itch related sleep loss were observed in favour of subjects receiving Enstilar compared to those receiving foam vehicle from day 3 and throughout the treatment period. Subjects on long-term maintenance treatment with Enstilar had longer time to first relapse, greater proportion of days in remission during the trial, and fewer relapses than subjects using foam vehicle. Table 6 presents an overview of the effect on relapse in this trial.
Subjects on long-term maintenance treatment with Enstilar had longer time to first relapse, greater proportion of days in remission during the trial, and fewer relapses than subjects using foam vehicle. Table 6 presents an overview of the effect on relapse in this trial.
 Calcipotriol monohydrate is (5Z, 7E, 22E, 24S) -24-Cyclopropyl-9,10-secochola- 5,7,10(19),22-tetraene-1α,3β,24-triol monohydrate. The molecular weight of calcipotriol monohydrate is 430.6.
Calcipotriol monohydrate is (5Z, 7E, 22E, 24S) -24-Cyclopropyl-9,10-secochola- 5,7,10(19),22-tetraene-1α,3β,24-triol monohydrate. The molecular weight of calcipotriol monohydrate is 430.6. Betamethasone dipropionate is 9-fluoro-11 β, 17, 21-trihydroxy-16 β-methylpregna-1,4- diene-3,20-dione 17,21-dipropionate. The empirical formula is C28H37FO7. The molecular weight of betamethasone dipropionate is 504.6.
Betamethasone dipropionate is 9-fluoro-11 β, 17, 21-trihydroxy-16 β-methylpregna-1,4- diene-3,20-dione 17,21-dipropionate. The empirical formula is C28H37FO7. The molecular weight of betamethasone dipropionate is 504.6.