1. Why am I using Entyvio?
Entyvio pre-filled pen contains the active ingredient vedolizumab.
Vedolizumab is a monoclonal antibody. Monoclonal antibodies are proteins that recognise and bind to certain special proteins in the body.
Entyvio specifically binds to a protein called integrin α4β7 present on certain white blood cells. Integrin α4β7 can act to increase inflammation seen in ulcerative colitis and Crohn’s disease. Entyvio works by blocking α4β7 integrins and so reduces inflammation.
Ulcerative colitis
Ulcerative colitis is an inflammatory disease of the large bowel. Entyvio is used to treat the signs and symptoms of moderate to severe ulcerative colitis in adults who have not responded well enough or are intolerant to other treatments.
Crohn’s disease
Crohn’s disease is an inflammatory disease of the bowel. It may also affect any part of the gut. Entyvio is used to treat the signs and symptoms of moderate to severe Crohn’s disease in adults who have not responded well enough or are intolerant to other treatments.
2. What should I know before I use Entyvio?
Warnings
Do not use Entyvio if:
- You are allergic to vedolizumab, or any of the ingredients listed at the end of this leaflet.
- Symptoms of an allergic reaction may include wheezing or difficulty breathing, hives, itching of the skin, swelling or dizziness.
- You have an active severe infection, such as for example tuberculosis, blood poisoning, serious abscesses.
- After the expiry date printed on the pack or if the packaging is damaged or shows signs of tampering. If it has expired or is damaged return it to your pharmacist for disposal.
Check with your doctor if you:
- Have an infection, or think you have an infection.
- Are going to receive any vaccination or have recently had a vaccination. Entyvio may affect the way you respond to a vaccination.
- Have any allergies to any other medicines, foods, preservatives or dyes.
- Take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor if you are pregnant or intend to become pregnant. The effects of Entyvio in pregnant women are not known. Therefore, this medicine is not recommended for use during pregnancy unless you and your doctor decide that the benefit to you clearly outweighs the potential risk to your baby. Your doctor can discuss the risks and benefits involved.
If you are a woman of childbearing potential, you should use adequate contraception to avoid falling pregnant and continue to use it for at least 4.5 months after the last treatment with Entyvio.
Tell your doctor if you are breastfeeding or intend to breastfeed. Entyvio passes into breast milk and it is not known what effect this may have on your baby. If you are breastfeeding, your doctor may advise you to stop breastfeeding while you are using this medicine.
Use in Children
There is not enough information to recommend the use of this medicine for children under the age of 18 years.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Entyvio should not be given with other biologic medicines that suppress your immune system as this combination has not been studied in clinical trials.
Tell your doctor if you have previously taken natalizumab (a medicine for multiple sclerosis) or rituximab (a medicine for certain types of cancer and rheumatoid arthritis). Your doctor will decide if you can use Entyvio.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Entyvio.
Published by MIMS September 2021

 No clinically relevant differences in the overall safety profile and adverse events were observed in patients who received vedolizumab in the EARNEST trial (pouchitis). Rectal bleeding, asthenia and chest discomfort were reported in vedolizumab group (3%).
No clinically relevant differences in the overall safety profile and adverse events were observed in patients who received vedolizumab in the EARNEST trial (pouchitis). Rectal bleeding, asthenia and chest discomfort were reported in vedolizumab group (3%). In exploratory analyses, the beneficial effect of intravenous vedolizumab on clinical response, remission and mucosal healing was observed both in patients with no prior TNFα antagonist exposure and in those who had failed prior TNFα antagonist therapy.
In exploratory analyses, the beneficial effect of intravenous vedolizumab on clinical response, remission and mucosal healing was observed both in patients with no prior TNFα antagonist exposure and in those who had failed prior TNFα antagonist therapy. In the GEMINI I study, the induction regimen was administered at weeks 0 and 2 and maintenance dosing started at week 6. However exploratory analyses suggest a higher rate of long term clinical response and remission will be achieved with a 0, 2 and 6 week induction regimen followed by maintenance treatment every 8 weeks for patients who demonstrate a clinical response (reduction in complete Mayo score of ≥ 3 points and ≥ 30% from baseline with an accompanying decrease in rectal bleeding subscore of ≥ 1 point or absolute rectal bleeding subscore of ≤ 1 point or reduction in partial Mayo score of ≥ 2 points and ≥ 25% from baseline with an accompanying decrease in rectal bleeding subscore of ≥ 1 point or absolute rectal bleeding subscore of ≤ 1 point) 6 to 8 weeks after completion of the induction regimen.
In the GEMINI I study, the induction regimen was administered at weeks 0 and 2 and maintenance dosing started at week 6. However exploratory analyses suggest a higher rate of long term clinical response and remission will be achieved with a 0, 2 and 6 week induction regimen followed by maintenance treatment every 8 weeks for patients who demonstrate a clinical response (reduction in complete Mayo score of ≥ 3 points and ≥ 30% from baseline with an accompanying decrease in rectal bleeding subscore of ≥ 1 point or absolute rectal bleeding subscore of ≤ 1 point or reduction in partial Mayo score of ≥ 2 points and ≥ 25% from baseline with an accompanying decrease in rectal bleeding subscore of ≥ 1 point or absolute rectal bleeding subscore of ≤ 1 point) 6 to 8 weeks after completion of the induction regimen.

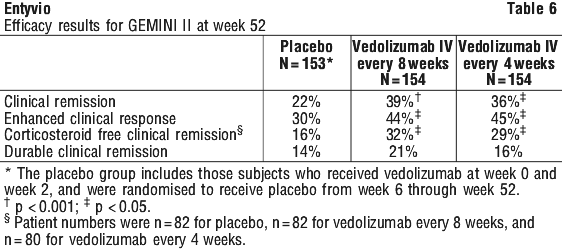 In the GEMINI II study, the induction regimen was administered intravenously at weeks 0 and 2 and maintenance dosing started at week 6. However exploratory analyses suggest a higher rate of long-term clinical response with a 0, 2 and 6 week induction regimen followed by maintenance treatment every 8 weeks for patients who demonstrate a clinical response (≥ 70-point decrease in CDAI score from baseline of induction) 6 to 8 weeks after completion of the induction regimen.
In the GEMINI II study, the induction regimen was administered intravenously at weeks 0 and 2 and maintenance dosing started at week 6. However exploratory analyses suggest a higher rate of long-term clinical response with a 0, 2 and 6 week induction regimen followed by maintenance treatment every 8 weeks for patients who demonstrate a clinical response (≥ 70-point decrease in CDAI score from baseline of induction) 6 to 8 weeks after completion of the induction regimen.

 Approximately two-thirds of patients had received prior (for UC or pouchitis) TNFα antagonist therapy (33 in vedolizumab and 31 in placebo treatment groups). Among these patients, 33.3% in the vedolizumab group achieved clinical remission at week 14 compared with 9.7% in the placebo group.
Approximately two-thirds of patients had received prior (for UC or pouchitis) TNFα antagonist therapy (33 in vedolizumab and 31 in placebo treatment groups). Among these patients, 33.3% in the vedolizumab group achieved clinical remission at week 14 compared with 9.7% in the placebo group. Do not store any unused portion of the reconstituted solution or infusion solution for reuse. Each vial is for single-use only.
Do not store any unused portion of the reconstituted solution or infusion solution for reuse. Each vial is for single-use only.