1 Name of Medicine
Epcoritamab.
2 Qualitative and Quantitative Composition
Epkinly 4 mg/0.8 mL concentrate solution for injection: each 0.8 mL single-dose vial contains 4 mg of epcoritamab.
Epkinly 48 mg/0.8 mL solution for injection: each 0.8 mL single-dose vial contains 48 mg of epcoritamab.
Epcoritamab is a humanised bispecific antibody that specifically binds to CD3+ T cells and CD20+ B cells. Epcoritamab is manufactured from two biological intermediates, which are produced in Chinese hamster ovary (CHO) cells using recombinant DNA technology.
Excipient with known effect.
Each vial of Epkinly contains 21.84 mg of sorbitol.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Epkinly is a sterile, preservative free, clear to slightly opalescent, colourless to slightly yellow solution, practically free of visible particles.
4.1 Therapeutic Indications
Epkinly is indicated for the treatment of adult patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) after two or more lines of systemic therapy. Epkinly is not indicated for the treatment of patients with primary central nervous system lymphoma.
This medicine has provisional approval in Australia for the treatment of adult patients with relapsed or refractory DLBCL after two or more lines of systemic therapy. The decision to approve this indication has been made on the basis of overall response and duration of response from an uncontrolled, open label phase I/II study. Continued approval of this indication depends on verification and description of benefit in confirmatory trials.
4.2 Dose and Method of Administration
Recommended dosage.
Epkinly is for subcutaneous (SC) injection only. Epkinly should be administered by an appropriately qualified healthcare professional. Administer Epkinly according to the following schedule in 28-day cycles (see Table 1). The Epkinly dosing schedule includes an initial priming dose of 0.16 mg on Cycle 1 Day 1, an intermediate dose of 0.8 mg on Cycle 1 Day 8, and a full dose of 48 mg administered from Cycle 1 Day 15 and onwards, according to Table 1.
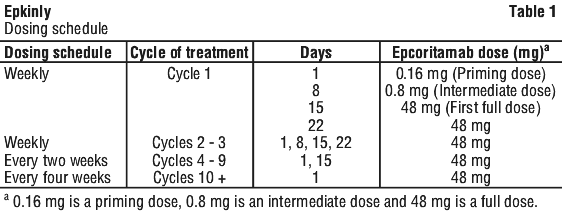 Administer Epkinly until disease progression or unacceptable toxicity.
Administer Epkinly until disease progression or unacceptable toxicity.
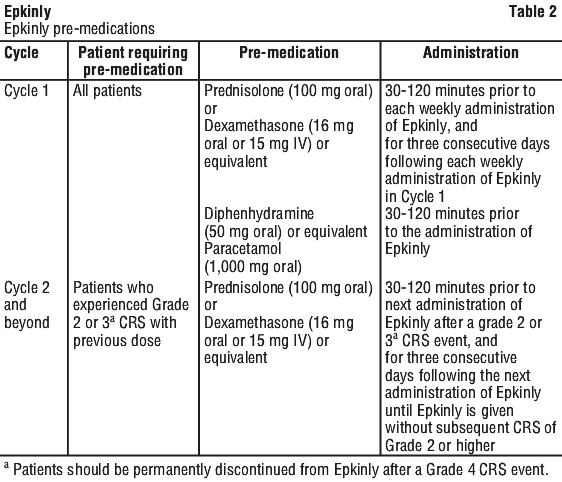
Pre-medications and prophylaxis.
Epkinly should be administered to adequately hydrated patients. Details on recommended pre-medication for cytokine release syndrome (CRS) is shown in Table 2.
 Prophylaxis against Pneumocystis jirovecii pneumonia (PCP) and herpes virus infections is strongly recommended especially during concurrent use of steroids.
Prophylaxis against Pneumocystis jirovecii pneumonia (PCP) and herpes virus infections is strongly recommended especially during concurrent use of steroids.
Monitor patients for potential CRS and immune effector cell-associated neurotoxicity syndrome (ICANS) following Epkinly administrations during cycle 1 and in subsequent cycles as needed at the physician's discretion. Following administration of the first full dose, patients should remain within close proximity to a healthcare facility that can assess and manage potential CRS and/or ICANS for at least 24 hours. Counsel patients on the signs and symptoms associated with CRS and ICANS and on seeking immediate medical attention should signs or symptoms occur at any time (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome, Immune effector cell-associated neurotoxicity syndrome).
Missed or delayed dose.
A re-priming cycle (identical to Cycle 1 with standard CRS prophylaxis) is required:
If there are more than 8 days between the priming dose (0.16 mg) and intermediate dose (0.8 mg), or
If there are more than 14 days between the intermediate dose (0.8 mg) and first full dose (48 mg), or
If there are more than 6 weeks between full doses (48 mg).
After the re-priming cycle, the patient should resume treatment with Day 1 of the next planned treatment cycle (subsequent to the cycle during which the dose was delayed).
Dosage modifications and management of adverse reactions.
Cytokine release syndrome (CRS).
Patients treated with Epkinly may develop CRS. Evaluate for and treat other causes of fever, hypoxia, and hypotension. If CRS is suspected, manage according to the recommendations in Table 3. Patients who experience CRS should be monitored more frequently during next scheduled Epkinly administrations.

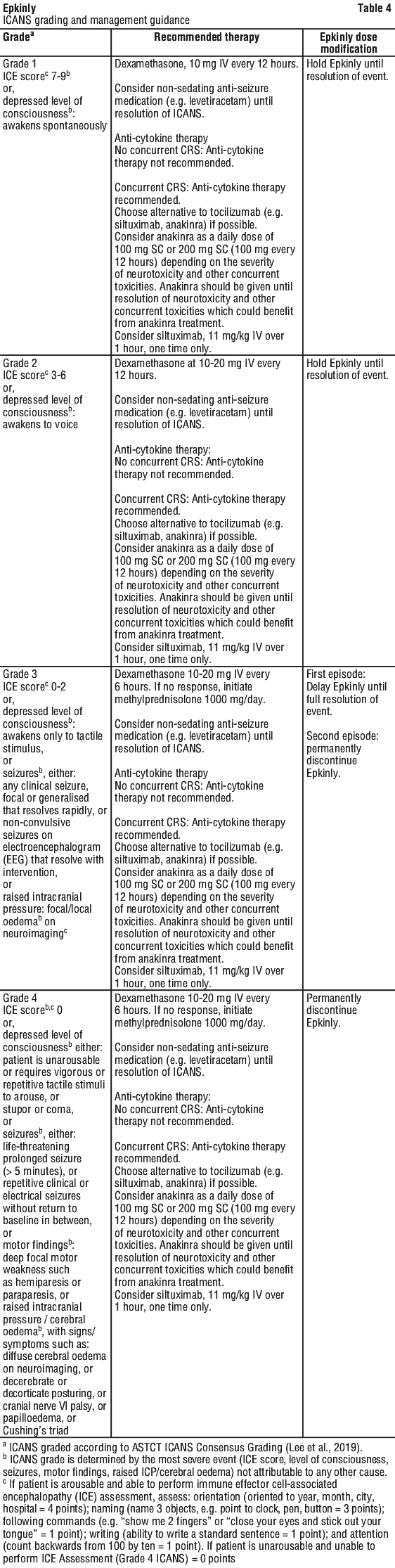
Immune effector cell-associated neurotoxicity syndrome (ICANS).
Monitor patients for signs and symptoms of ICANS. Rule out other causes of neurologic symptoms. If ICANS is suspected, manage according to the recommendations in Table 4.
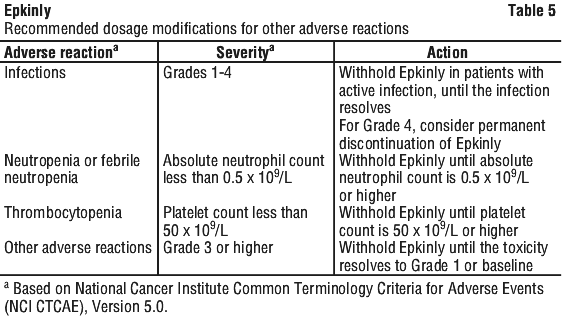
Other adverse reactions.
See Table 5 for recommended dosage modifications for other adverse reactions.
Preparation and administration.
Preparation of diluted Epkinly using the vial method. Read this entire section carefully before preparation of Epkinly. Certain doses of Epkinly require dilution prior to administration. Follow the preparation instructions provided below, as improper preparation may lead to improper dose. This section describes preparation of diluted Epkinly using the empty sterile vial method. For preparation using sterile syringe method, see Section 4.2 Dose and Method of Administration, Preparation of diluted Epkinly using the syringe method.
Epkinly should be prepared and administered by a healthcare provider as a subcutaneous injection (SC). Each vial of Epkinly is for single use in one patient only.
The administration of Epkinly takes place over the course of 28-day cycles, following the dosing scheduled in Section 4.2 Dose and Method of Administration, Recommended dosage. Inspect visually for particulate matter and discolouration prior to administration. Epkinly injection is a colourless to slightly yellow solution. Do not use if the solution is discoloured, or cloudy, or if foreign particles are present. Use aseptic technique to prepare Epkinly. Filtration of the diluted solution is not required.
0.16 mg priming dose preparation instructions (2 dilutions required) - empty sterile vial method.
Use an appropriately sized syringe, vial and needle for each transfer step.
Materials needed for preparation: 4 mg/0.8 mL Epkinly vial;
0.9% sodium chloride (NaCl) sterile solution;
two empty sterile vials between 10 and 20 mL (to be labelled dilution A and dilution B);
syringes to accommodate the below volumes: first dilution step - Epkinly 0.8 mL; first dilution step - sodium chloride 4.2 mL; second dilution step - dilution A 2 mL; second dilution step - sodium chloride 8 mL; dosing syringe 1 mL.
1. Prepare Epkinly vial.
a. Retrieve one 4 mg/0.8 mL Epkinly vial from the refrigerator.
b. Allow the vial to come to room temperature for no more than 1 hour.
c. Gently swirl the Epkinly vial.
Do not invert, vortex, or vigorously shake the vial.
2. Perform first dilution.
a. Label an appropriately sized empty vial as "dilution A".
b. Transfer 0.8 mL of Epkinly into the dilution A vial.
c. Transfer 4.2 mL of 0.9% sodium chloride, sterile solution, into the dilution A vial. The initially diluted solution contains 0.8 mg/mL of epcoritamab.
d. Gently swirl the dilution A vial for 30-45 seconds.
3. Perform second dilution.
a. Label an appropriately sized empty vial as "dilution B".
b. Transfer 2 mL of solution from the dilution A vial into the dilution B vial. The dilution A vial is no longer needed.
c. Transfer 8 mL of 0.9% sodium chloride, sterile solution into the dilution B vial to make a final concentration of 0.16 mg/mL.
d. Gently swirl the dilution B vial for 30-45 seconds.
4. Withdraw dose.
a. Withdraw 1 mL of the diluted Epkinly from the dilution B vial into a syringe to be used for patient dosing (dosing syringe).
5. Label syringe.
Label the dosing syringe with the drug name, dose strength (0.16 mg) and the time of day.
Discard the vial and any unused portion of Epkinly in accordance with local requirements.
0.8 mg intermediate dose preparation instructions (1 dilution required) - empty sterile vial method.
Use an appropriately sized syringe, vial and needle for each transfer step.
Materials needed for preparation: 4 mg/0.8 mL Epkinly vial;
0.9% sodium chloride (NaCl) sterile solution;
one empty sterile vial between 10 and 20 mL (to be labelled dilution A);
syringes to accommodate the below volumes: first dilution step - Epkinly 0.8 mL; first dilution step - sodium chloride 4.2 mL; dosing syringe 1 mL.
1. Prepare Epkinly vial.
a. Retrieve one 4 mg/0.8 mL Epkinly vial from the refrigerator.
b. Allow the vial to come to room temperature for no more than 1 hour.
c. Gently swirl the Epkinly vial.
Do not invert, vortex, or vigorously shake the vial.
2. Perform dilution.
a. Label an appropriately sized empty vial as "dilution A".
b. Transfer 0.8 mL of Epkinly into the dilution A vial.
c. Transfer 4.2 mL of 0.9% sodium chloride, sterile solution into the dilution A vial to make a final concentration of 0.8 mg/mL.
d. Gently swirl the dilution A vial for 30-45 seconds.
3. Withdraw dose.
a. Withdraw 1 mL of the diluted Epkinly from the dilution A vial into a syringe to be used for patient dosing (dosing syringe).
4. Label syringe. Label the dosing syringe with the drug name, dose strength (0.8 mg) and the time of day.
Discard the vial and any unused portion of Epkinly in accordance with local requirements.
Preparation of diluted Epkinly using the syringe method. Read this entire section carefully before preparation of Epkinly. Certain doses of Epkinly require dilution prior to administration. Follow the preparation instructions provided below, as improper preparation may lead to improper dose. This section describes preparation of diluted Epkinly using the empty sterile syringe method. For preparation using the sterile vial method, see Section 4.2 Dose and Method of Administration, Preparation of diluted Epkinly using the vial method.
Epkinly should be prepared and administered by a healthcare provider as a subcutaneous injection (SC). Each vial of Epkinly is for single use in one patient only. The administration of Epkinly takes place over the course of 28-day cycles, following the dosing scheduled in Section 4.2 Dose and Method of Administration, Recommended dosage.
Inspect visually for particulate matter and discolouration prior to administration. Epkinly injection is a colourless to slightly yellow solution. Do not use if the solution is discoloured, or cloudy, or if foreign particles are present. Use aseptic technique to prepare Epkinly. Filtration of the diluted solution is not required.
0.16 mg priming dose preparation instructions (2 dilutions required) - sterile syringe method.
Use an appropriately sized syringe and needle for each transfer step.
Materials needed for preparation: 4 mg/0.8 mL Epkinly vial;
0.9% sodium chloride (NaCl) sterile solution;
four suitable connectors;
syringes to accommodate the below volumes: syringe 1 - 0.8 mL; syringe 2 - 2 mL; dilution A - 5 mL; dilution B - 10 mL; dosing syringe - 1 mL.
1. Prepare Epkinly vial.
a. Retrieve one 4 mg/0.8 mL Epkinly vial from the refrigerator.
b. Allow the vial to come to room temperature for no more than 1 hour.
c. Gently swirl the Epkinly vial. Do not invert, vortex, or vigorously shake the vial.
2. Perform first dilution.
a. Label an appropriately sized syringe as "dilution A".
b. Withdraw 4.2 mL of 0.9% sodium chloride sterile solution into the dilution A syringe. Include approximately 0.2 mL air in the syringe.
c. In a new syringe labelled as "syringe 1", withdraw 0.8 mL of Epkinly.
d. Connect the two syringes (with a suitable connector) and push the 0.8 mL of Epkinly into the dilution A syringe. The initially diluted solution contains 0.8 mg/mL of epcoritamab.
e. Gently mix by inverting the connected syringes 180 degrees 5 times.
f. Disconnect the syringes and discard syringe 1.
3. Perform second dilution.
a. Label an appropriately sized syringe as "dilution B".
b. Withdraw 8 mL of 0.9% sodium chloride sterile solution into the dilution B syringe. Include approximately 0.2 mL air in the syringe.
c. Label another appropriately sized syringe as "syringe 2".
d. Connect syringe 2 to the dilution A syringe (with a suitable connector) and transfer 2 mL of solution into syringe 2. The dilution A syringe is no longer needed.
e. Connect syringe 2 to the dilution B syringe (with a suitable connector) and push the 2 mL of solution into the dilution B syringe to make a final concentration of 0.16 mg/mL.
f. Gently mix by inverting the connected syringes 180 degrees 5 times.
g. Disconnect the syringes and discard syringe 2.
4. Withdraw dose.
a. Connect and transfer 1 mL of the diluted Epkinly from the dilution B syringe (with a suitable connector) into a new syringe to be used for patient dosing (dosing syringe). The dilution B syringe is no longer needed.
5. Label syringe.
a. Label the dosing syringe with the drug name, dose strength (0.16 mg) and the time of day.
Discard the vial and any unused portion of Epkinly in accordance with local requirements.
0.8 mg intermediate dose preparation instructions (1 dilution required) - sterile syringe method.
Use an appropriately sized syringe and needle for each transfer step.
Materials needed for preparation: 4 mg/0.8 mL Epkinly vial;
0.9% sodium chloride (NaCl) sterile solution;
two suitable connectors;
syringes to accommodate the below volumes: syringe 1 - 0.8 mL; dilution A - 5 mL; dosing syringe -1 mL.
1. Prepare Epkinly vial.
a. Retrieve one 4 mg/0.8 mL Epkinly vial from the refrigerator.
b. Allow the vial to come to room temperature for no more than 1 hour.
c. Gently swirl the Epkinly vial.Do not invert, vortex, or vigorously shake the vial.
2. Perform dilution.
a. Label an appropriately sized syringe as "dilution A".
b. Withdraw 4.2 mL of 0.9% sodium chloride sterile solution into the dilution A syringe. Include approximately 0.2 mL air in the syringe.
c. In a new syringe labelled as "syringe 1", withdraw 0.8 mL of Epkinly.
d. Connect the two syringes (with a suitable connector) and push the 0.8 mL of Epkinly into the dilution A syringe to make a final concentration of 0.8 mg/mL.
e. Gently mix by inverting the connected syringes 180 degrees 5 times.
f. Disconnect the syringes and discard syringe 1.
3. Withdraw dose.
a. Connect and transfer 1 mL of the diluted Epkinly from the dilution A syringe (with a suitable connector) into a new syringe to be used for patient dosing (dosing syringe). The dilution A syringe is no longer needed.
4. Label syringe.
a. Label the dosing syringe with the drug name, dose strength (0.8 mg) and the time of day.
Discard the vial and any unused portion of Epkinly in accordance with local requirements.
48 mg full dose preparation instructions (no dilution required).
Epkinly should be prepared and administered by a healthcare provider as a subcutaneous injection (SC). Each vial of Epkinly is for single use in one patient only.
Inspect visually for particulate matter and discolouration prior to administration. Epkinly injection is a colourless to slightly yellow solution. Do not use if the solution is discoloured, or cloudy, or if foreign particles are present. Use aseptic technique to prepare Epkinly.
Epkinly 48 mg/0.8 mL vial is supplied as ready-to-use solution that does not need dilution prior to administration.
1. Prepare Epkinly vial.
a. Retrieve one 48 mg/0.8 mL Epkinly vial from the refrigerator.
b. Allow the vial to come to room temperature for no more than 1 hour.
c. Gently swirl the Epkinly vial.
Do not invert, vortex, or vigorously shake the vial.
2. Withdraw dose.
a. Withdraw 0.8 mL of the Epkinly into a syringe to be used for patient dosing (dosing syringe).
3. Label syringe.
a. Label the dosing syringe with the drug name, dose strength (48 mg) and the time of day.
Discard the vial and any unused portion of Epkinly in accordance with local requirements.
Storage for diluted and prepared Epkinly.
To reduce microbiological hazard, use as soon as practical after dilution. If storage is necessary, hold at 2-8°C for not more than 24 hours. Within these 24 hours, the Epkinly solution can be stored for up to 12 hours at room temperature from the start of dose preparation to administration. Minimise exposure to daylight. Allow Epkinly solution to equilibrate to room temperature before administration. Discard unused Epkinly solution beyond the allowable storage time.
Site administration.
The injection site should be preferably in the lower part of abdomen or the thigh. Change of injection site from left or right side or vice versa is recommended especially during the weekly administration (Cycles 1-3).4.3 Contraindications
Hypersensitivity to epcoritamab, or to any of the excipients listed in Section 6.1.
4.4 Special Warnings and Precautions for Use
Cytokine release syndrome.
Cytokine release syndrome (CRS), which may be life-threatening or fatal, occurred in patients receiving epcoritamab. The most common signs and symptoms of CRS include pyrexia, hypotension and hypoxia. Other signs and symptoms of CRS in greater than two patients includes chills, tachycardia, headache and dyspnoea.
The median time to onset of CRS from the most recent administered epcoritamab dose was 2 days (range: 1 to 11 days). The median time to onset after the first full dose was 20.6 hours (range: 0.2 days to 7 days). Most CRS events occurred in Cycle 1 and were associated with the first full dose of Epkinly. The median duration of CRS was 2 days (range: 1 to 27 days). Administer prophylactic corticosteroids to mitigate the risk of CRS (see Section 4.2 Dose and Method of Administration, Pre-medications and prophylaxis).
Monitor patients for potential CRS following epcoritamab administrations during Cycle 1 and in subsequent cycles as needed at the physician's discretion. For at least 24 hours following administration of the first full dose, patients should remain within close proximity to a healthcare facility that can assess and manage potential CRS. At the first signs or symptoms of CRS, institute treatment of supportive care with tocilizumab and/or corticosteroids as appropriate. Counsel patients on the signs and symptoms associated with CRS and instruct patients to contact their healthcare professional and seek immediate medical attention should signs or symptoms occur at any time. Management of CRS may require either temporary delay or discontinuation of epcoritamab based on the severity of CRS (see Section 4.2 Dose and Method of Administration, Dosage modifications and management of adverse reactions).
Haemophagocytic lymphohistiocytosis (HLH), including fatal cases, have been reported in patients receiving epcoritamab. HLH should be considered when the presentation of CRS is atypical or unresponsive and/or clinical manifestations of HLH such as prolonged fever, cytopenias, hyperferritinaemia, coagulopathy and elevated liver enzymes are present. If HLH is suspected, withhold Epkinly and initiate treatment for HLH per current practice guidelines.
Immune effector cell-associated neurotoxicity syndrome.
Immune effector cell-associated neurotoxicity syndrome (ICANS), including a fatal event, have occurred in patients receiving epcoritamab. ICANS may manifest as aphasia, altered level of consciousness, impairment of cognitive skills, motor weakness, seizures, and cerebral oedema.
The median time to onset of ICANS from the start of epcoritamab treatment (Cycle 1 Day 1) was 16.5 days (range: 8 to 141 days). The majority of cases of ICANS occurred within the Cycle 1 of epcoritamab treatment, however some occurred with delayed onset. The median duration of ICANS was 5 days (range: 1, 9 days). The onset of ICANS can be concurrent with CRS, following resolution of CRS or in the absence of CRS.
Monitor patients for signs and symptoms of ICANS following epcoritamab administrations during Cycle 1 and in subsequent cycles as needed at the physician's discretion. Following administration of the first full dose, patients should remain within close proximity to a healthcare facility that can assess and manage potential ICANS for at least 24 hours. At the first signs or symptoms of ICANS institute treatment with corticosteroids and non-sedating-anti-seizure medications as appropriate (see Section 4.2 Dose and Method of Administration, Dosage modifications and management of adverse reactions). Counsel patients on the signs and symptoms of ICANS and that the onset of events may be delayed. Instruct patients to contact their healthcare professional and seek immediate medical attention should signs or symptoms occur at any time. Delay or discontinue Epkinly as recommended (see Section 4.2 Dose and Method of Administration, Dosage modifications and management of adverse reactions).
Serious infections.
Treatment with Epkinly may lead to an increased risk of infections. Serious infections, including fatal infections were observed in patients treated with epcoritamab in clinical trials (see Section 4.8 Adverse Effects (Undesirable Effects)).
Avoid administration of Epkinly in patients with clinically significant active systemic infections. As appropriate, administer prophylactic antimicrobials (see Section 4.2 Dose and Method of Administration, Pre-medications and prophylaxis). Monitor patients for signs and symptoms of infections prior to and during treatment and treat according to standard/local guidelines and practice. In the event of febrile neutropenia, patients should be evaluated for infection and managed according to local guidelines.
Hypogammaglobulinaemia has also been reported in patients receiving epcoritamab (see Section 4.8 Adverse Effects (Undesirable Effects)). Immunoglobulin (Ig) levels should be monitored prior to and during treatment. Patients should be treated according to local guidelines, including infection precautions and antimicrobial prophylaxis.
Cases of progressive multifocal leukoencephalopathy (PML), including fatal cases, have been reported in patients treated with epcoritamab who have also received prior treatment with other immunosuppressive medications. If neurological symptoms suggestive of PML occur during epcoritamab therapy, treatment with Epkinly should be withheld and appropriate diagnostic measures initiated. Discontinue Epkinly if PML is confirmed.
Tumour lysis syndrome (TLS).
TLS has been reported in patients receiving epcoritamab (see Section 4.8 Adverse Effects (Undesirable Effects)). Patients at an increased risk for TLS are recommended to receive hydration and prophylactic treatment with a uric acid lowering agent. Patients should be monitored for signs or symptoms of TLS, especially patients with high tumour burden or rapidly proliferative tumours, and patients with reduced renal function. Patients should be monitored for blood chemistries and abnormalities should be managed promptly.
Tumour flare.
Tumour flare has been reported in patients treated with epcoritamab (see Section 4.8 Adverse Effects (Undesirable Effects)). Manifestations could include localised pain and swelling. Consistent with the mechanism of action of epcoritamab, tumour flare is likely due to the influx of T-cells into tumour sites following epcoritamab administration.
There are no specific risk factors for tumour flare that have been identified; however, there is a heightened risk of compromise and morbidity due to mass effect secondary to tumour flare in patients with bulky tumours located in close proximity to airways and/or a vital organ. Patients treated with epcoritamab should be monitored and evaluated for tumour flare at critical anatomical sites.
Immunisation.
Live and/or live-attenuated vaccines should not be given concurrently with Epkinly. Studies have not been conducted in patients who received live vaccines.
Patient card.
The doctor must inform the patient of the risk of CRS and ICANS and any signs and symptoms of CRS and ICANS. Patients must be instructed to seek immediate medical attention if they experience signs and symptoms of CRS and/or ICANS. Patients should be provided with a patient card and instructed to carry the card at all times. This card describes symptoms of CRS and ICANS which, if experienced, should prompt the patient to seek immediate medical attention.
Use in hepatic impairment.
Dose adjustments are not considered necessary in patients with mild hepatic impairment. No dose recommendations can be made for patients with moderate to severe hepatic impairment.
Use in renal impairment.
Dose adjustments are not considered necessary in patients with mild to moderate renal impairment. No dose recommendations can be made for patients with severe renal impairment to end-stage renal disease.
Use in the elderly.
In patients with DLBCL in EPCORE NHL-1, 44 (32%) were ≥ 65 to < 75 years of age and 29 (21%) were ≥ 75 years of age. No clinically meaningful differences in safety or efficacy were observed between patients ≥ 65 years of age compared with younger adult patients.
Paediatric use.
The safety and efficacy of Epkinly in children aged less than 18 years of age have not yet been established.
Effects on laboratory tests.
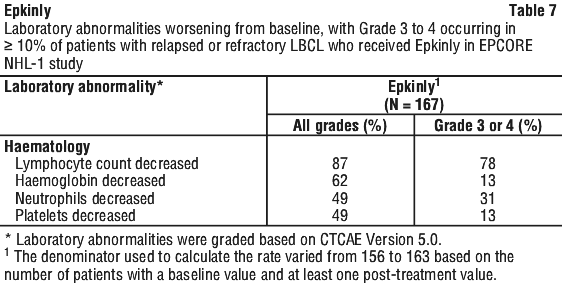
Grade 3 or 4 laboratory abnormalities worsening from baseline reported in at least 10% of patients with LBCL within the EPCORE NHL-1 study were lymphocyte count decreased (78%), neutrophil count decreased (31%), haemoglobin decreased (13%), and platelets decreased (13%).4.5 Interactions with Other Medicines and Other Forms of Interactions
No formal drug interaction studies have been conducted with Epkinly.
Elevation of certain proinflammatory cytokines by Epkinly may suppress CYP450 enzyme activities. On initiation of Epkinly therapy in patients being treated with CYP450 substrates with a narrow therapeutic index, therapeutic monitoring should be considered.
4.6 Fertility, Pregnancy and Lactation
Females of reproductive potential.
Verify pregnancy status in females of reproductive potential prior to initiating Epkinly treatment. Females of reproductive potential should use effective contraception during treatment with Epkinly and for at least 4 months after the last dose.
Effects on fertility.
No animal fertility studies have been conducted with epcoritamab. The effect of Epkinly on male and female fertility is unknown.
(Category C)
Female patients of reproductive potential must be advised to avoid pregnancy while receiving Epkinly and apprised of the potential risk to the fetus.
There are no data on the use of Epkinly in pregnant women. Embryofetal development studies have not been conducted with epcoritamab in animals.
Based on its mechanism of action, Epkinly may cause fetal harm, including B-cell lymphocytopenia and alterations in normal immune responses, when administered to pregnant women. IgG1 antibodies, such as epcoritamab, can cross the placenta resulting in fetal exposure. Fetal B-cell depletion poses a risk of opportunistic infections in the neonate. Epcoritamab-induced cytokine release may also pose a risk for embryofetal loss. The risk of malformations is considered to be low.
Postponing vaccination with live or live attenuated vaccines is recommended for neonates and infants who have been exposed to epcoritamab in utero until B-cell levels have recovered.
It is not known whether epcoritamab is excreted in human milk or its effect on milk production. Since IgGs are known to be present in milk, neonatal exposure to epcoritamab may occur via lactational transfer. Breast feeding should be discontinued during treatment with Epkinly and for at least 4 months after the last dose.4.7 Effects on Ability to Drive and Use Machines
No formal studies on the effect of Epkinly on the ability to drive and operate machines have been performed. Due to the potential for ICANS, patients should be advised to exercise caution while (or avoid if symptomatic) driving or using heavy or potentially dangerous machines.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at https://www.tga.gov.au/reporting-problems.
Clinical trial experience.
EPCORE NHL-1.
The safety of Epkinly was evaluated in a non-randomised, single-arm study in 167 patients with relapsed or refractory LBCL, including 148 patients with DLBCL, after two or more lines of systemic therapy and included all patients who enrolled to the 48 mg dose and received at least one dose of Epkinly.
The median duration of exposure to Epkinly was 3.7 months (range: 0 to 20 months).
Serious adverse reactions occurred in 40% of patients; the most frequent serious adverse reaction (≥ 10%) was CRS (31%). Two patients (1.2%) experienced a fatal adverse reaction; one each for ICANS and pneumonia.
Discontinuation due to adverse reactions occurred in 2.4% of patients. Discontinuation of epcoritamab due to pneumonia occurred in 2 patients and discontinuation due to CRS or ICANS occurred in 1 patient (each).
Dose delays due to adverse reactions occurred in 20% of patients. Adverse reactions leading to dose delays (≥ 3%) were CRS (7.2%), neutropenia (4.2%), pyrexia (3.0%), and thrombocytopenia (3.0%).
Table 6 provides adverse reactions reported in patients with relapsed or refractory LBCL. Adverse reactions are listed by MedDRA body system organ class, rate, and frequency.
Frequencies are defined as very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000), not known (cannot be estimated from available data). Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
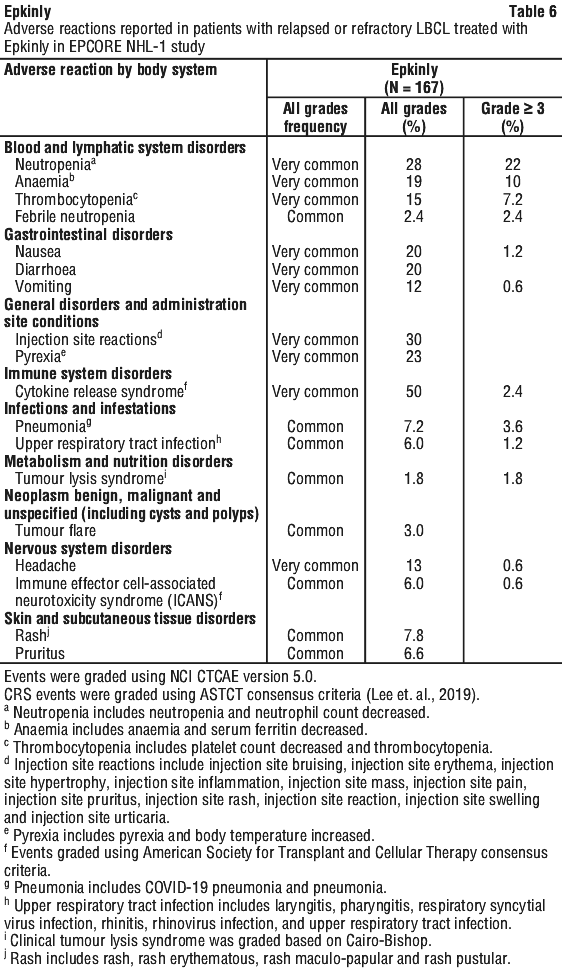 Table 7 summarises laboratory abnormalities in EPCORE NHL-1.
Table 7 summarises laboratory abnormalities in EPCORE NHL-1.
 Other adverse reactions: haemophagocytic lymphohistiocytosis (HLH) (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome), hypogammaglobulinaemia.
Other adverse reactions: haemophagocytic lymphohistiocytosis (HLH) (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome), hypogammaglobulinaemia.
Important adverse reactions.
Cytokine release syndrome (CRS).
CRS of any grade occurred in 50% (84/167) of patients treated with Epkinly. The incidence of Grade 1 was 31% (52/167), Grade 2 was 17% (28/167), and Grade 3 was 2.4% (4/167). The median time to onset of CRS from the most recent administered Epkinly dose was 2 days (range: 1 to 11 days). CRS resolved in 98.8% of patients, and the median duration of CRS events was 2 days (range 1 to 27 days).
The most common signs and symptoms of CRS included pyrexia 50% (83/167), hypotension 16% (26/167) and hypoxia 9.6% (16/167). Other signs and symptoms of CRS in greater than two patients included chills (4.8%), tachycardia (including sinus tachycardia [7.8%]), headache (13%) and dyspnoea (7.8%). In addition to corticosteroids use, tocilizumab was used to manage CRS event in 15% of patients.
Immune effector cell-associated neurotoxicity syndrome (ICANS).
ICANS occurred in 6% of patients treated with Epkinly; 4.2% experienced Grade 1 and 1.2% experienced Grade 2. One patient (0.6%) experienced an ICANS event of Grade 5 (fatal). The median time to first ICANS onset from the start of Epkinly treatment was 16.5 days (range: 8 to 141 days). ICANS resolved in 90% (9/10) of patients with supportive care. The median time to resolution of ICANS was 5 days (range: 1 to 9 days).
Serious infections.
Serious infections occurred in 16% of patients treated with Epkinly. The most frequent serious infections were pneumonia (2.4%), sepsis (2.4%), COVID-19 (1.8%), COVID-19 pneumonia (1.8%), bacteraemia (1.2%), septic shock (1.2%), and upper respiratory tract infection (1.2%). Fatal serious infections occurred in 4 (2.4%) patients.
Immunogenicity.
Epcoritamab has the potential to induce anti-drug-antibodies (ADA). The incidence of antibodies to epcoritamab was low and all the patients who were positive had low titres (≥ 1 in 0.6% (1/158)). Due to the low number of patients with ADAs, a meaningful analysis of the impact of ADAs on safety is limited (see Section 5.2 Pharmacokinetic Properties, Special populations).4.9 Overdose
For information on the management of overdose, contact the Poison Information Centre on 13 11 26 (Australia). In the event of overdose, monitor the patient for any signs or symptoms of adverse reactions and administer appropriate supportive treatment.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: other monoclonal antibodies and antibody-drug conjugates.
ATC code: L01FX27.
Epcoritamab induced depletion of circulating B cells (defined as CD19+ B-cell counts < 10 cell/microL in subjects who have detectable B cells at treatment initiation) after the first full dose (48 mg) which was sustained while patients remained on treatment. Subsequent treatment with epcoritamab induced expansion and activation of circulating T cells from baseline.
Following subcutaneous administration of epcoritamab, transient and modest elevations of circulating levels of selected cytokines (IFN-γ, TNFα, IL-6, IL-2, and IL-10) occurred, mostly after the first full dose (48 mg) with peak levels between 1 to 4 days. Levels returned to baseline prior to the subsequent full dose.
Mechanism of action.
Epcoritamab is a humanised IgG1-bispecific antibody that binds to a specific extracellular epitope of CD20 on B cells and to CD3 on T cells. CD20 is expressed on most human B-cell lymphomas and leukaemias and on B cells in peripheral blood, but not haematopoietic stem cells or plasma cells. The activity of epcoritamab is dependent upon simultaneous engagement of CD20-expressing cancer cells and CD3-expressing endogenous T cells by epcoritamab that induces specific T-cell activation and T-cell-mediated killing of CD20-expressing cells, as epcoritamab does not have direct immune effector mechanisms.
The Fc region of epcoritamab is silenced for direct immune effector mechanisms, such as antibody-dependent cellular cytotoxicity (ADCC), complement-dependent cellular cytotoxicity (CDC), and antibody-dependent cellular phagocytosis (ADCP).
Clinical trials.
EPCORE NHL-1.
Study EPCORE NHL-1 was an open-label, multi-cohort, multicentre, single-arm trial that evaluated epcoritamab as monotherapy in patients with relapsed or refractory large B-cell lymphoma (LBCL) after two or more lines of systemic therapy, including diffuse large B-cell lymphoma (DLBCL). The study includes a dose escalation part and an expansion part. The expansion part of the study included an aggressive non-Hodgkin lymphoma (aNHL) cohort, an indolent NHL (iNHL) cohort and a mantle-cell lymphoma (MCL) cohort. The pivotal cohort consisted of patients with DLBCL (N=139).
The study excluded patients with CNS involvement of lymphoma, allogeneic haematopoietic stem cell transplantation (HSCT) or solid organ transplant, ongoing active infectious diseases, any patients with known impaired T-cell immunity, a creatinine clearance of less than 45 mL/min, alanine aminotransferase > 3 times the upper limit of normal and cardiac ejection fraction less than 45%. Efficacy was evaluated in 139 patients with DLBCL who had received the 48 mg full dose of Epkinly subcutaneously (SC) in cycles of 4 weeks, i.e. 28 days. Epcoritamab was administered as a monotherapy as follows:
Cycle 1: Epkinly 0.16 mg on Day 1, 0.8 mg on Day 8, 48 mg on Day 15 and Day 22.
Cycles 2-3: Epkinly 48 mg on Days 1, 8, 15, and 22.
Cycles 4-9: Epkinly 48 mg on Days 1 and 15.
Cycles 10 and beyond: Epkinly 48 mg on Day 1.
Patients continued to receive Epkinly until disease progression or unacceptable toxicity.
The demographics and baseline characteristics are shown in Table 8.
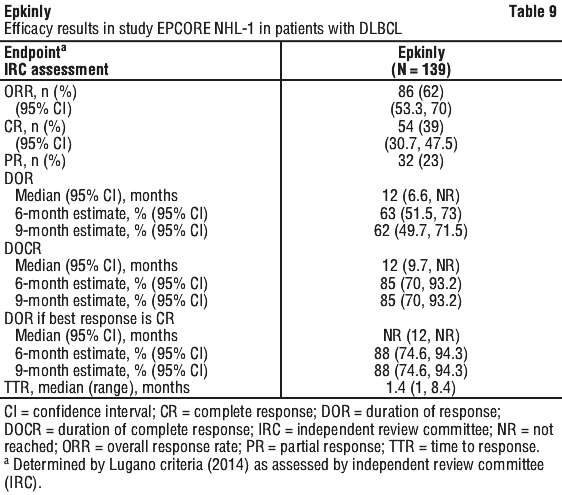
 Efficacy was established based on overall response rate (ORR) determined by Lugano criteria (2014) as assessed by Independent Review Committee (IRC). The median follow-up time was 11 months (range: 0.3 to 17.9 months).
Efficacy was established based on overall response rate (ORR) determined by Lugano criteria (2014) as assessed by Independent Review Committee (IRC). The median follow-up time was 11 months (range: 0.3 to 17.9 months).
 The median time to CR was 2.7 months (range: 1.2 to 11.1 months).
The median time to CR was 2.7 months (range: 1.2 to 11.1 months).
Response durations were longer in patients who achieved CR, as compared to patients with a best response of partial response (PR) (Table 9).
The overall response rates and complete response rates with Epkinly were consistent across the following subgroups: age, number of or response to prior lines of therapy, and prior CAR-T experience.
In subgroup analysis of patients (n = 53) who received CAR-T, the ORR was 53% (95% CI: 39, 67), and the CR rate was 34% (95% CI: 22, 48). Median duration of response for these patients was 9.7 months (95% CI: 5.4, NR), and the median progression-free survival was 2.7 months (95% CI: 1.4, 11).
In subgroup analysis of patients (n = 86) with no prior CAR-T, the ORR was 67% (95% CI: 56, 77), and the CR rate was 42% (95% CI: 31, 53). Median duration of response for these patients was 12 months (95% CI: 5.6, NR); and the median progression-free survival was 5.4 months (95% CI: 3.7, NR).
In a pre-specified subgroup analysis of patients (n = 82) who were primary refractory to anti-lymphoma therapy, the ORR was 54% (95% CI: 42, 65), and the CR rate was 30% (95% CI: 21, 42).
Median overall survival (OS) for patients on Epkinly was not reached.
5.2 Pharmacokinetic Properties
The population pharmacokinetics (PK) following subcutaneous administration of epcoritamab was described by a two-compartment model with first order subcutaneous absorption and target-mediated drug elimination. The moderate to high pharmacokinetic variability for epcoritamab was observed and characterised by inter-individual variability (IIV) ranging from 25.7% to 137.5% coefficient of variation (CV) for epcoritamab PK parameters.
Following the recommended SC dose of epcoritamab 48 mg, the geometric mean (% CV) Cmax of epcoritamab is 10.8 microgram/mL (41.7%) and AUC0-7d is 68.9 day*microgram/mL (45.1%) at the end of the weekly dosing schedule.
The geometric mean (% CV) Cmax of epcoritamab is 7.52 microgram/mL (41.1%) and AUC0-14d is 82.6 day*microgram/mL (49.3%) at the end of every two weeks dosing schedule.
The geometric mean (% CV) Cmax of epcoritamab is 4.76 microgram/mL (51.6%) and AUC0-28d is 74.3 day*microgram/mL (69.5%) at steady state during every 4-week dosing schedule.
Absorption.
The peak concentrations occurred around 3-4 days (Tmax) in patients receiving the 48 mg full dose.
Distribution.
The geometric mean (% CV) central volume of distribution is 8.27 L (27.5%) based on population PK modelling.
Metabolism.
The metabolic pathway of epcoritamab has not been directly studied. Like other protein therapeutics, epcoritamab is expected to be degraded into small peptides and amino acids via catabolic pathways.
Excretion.
Epcoritamab is expected to undergo saturable target mediated clearance. The geometric mean (% CV) clearance (L/day) is 0.441 (27.8%). The half-life of epcoritamab is concentration dependent. The population PK model-derived geometric mean half-life of full dose epcoritamab (48 mg) ranged from 22 to 25 days based on frequency of dosing.
Special populations.
No clinically important effects on the pharmacokinetics of epcoritamab were observed based on age (20 to 89 years), sex, or race/ethnicity (white, Asian, and other), mild to moderate renal impairment (CrCl ≥ 30 mL/min to CrCl < 90 mL/min), and mild hepatic impairment (total bilirubin ≤ ULN and AST > ULN, or total bilirubin 1 to 1.5 times ULN and any AST) after accounting for differences in bodyweight. No patients with severe to end-stage renal disease (CrCl < 30 mL/min) or severe hepatic impairment (total bilirubin > 3 times ULN and any AST) have been studied. There is very limited data in moderate hepatic impairment (total bilirubin > 1.5 to 3 times ULN and any AST). Therefore, the pharmacokinetics of epcoritamab are unknown in these populations.
Like other therapeutic proteins, body weight (39 to 144 kg) has a statistically significant effect on the pharmacokinetics of epcoritamab, however this effect is not clinically relevant across body weight categories (< 65 kg, 65 - < 85 kg, ≥ 85 kg).
Paediatric population (< 18 years).
The pharmacokinetics of epcoritamab in paediatric patients has not been established.
Immunogenicity.
In EPCORE clinical study, 4 of 158 (2.5%) patients who were treated with Epkinly at the full dose of 48 mg and evaluable for the presence of anti-drug antibodies (ADA) tested positive for anti-epcoritamab antibodies on treatment (two at cycle 2 day 22, one at cycle 1 day 22, and one at cycle 2 day 1) with titres of 1:320 or less. There was no evidence of an altered pharmacokinetic profile with anti-epcoritamab binding antibody development based on a population PK analysis. There are insufficient data to evaluate the effect of ADA on the safety or efficacy of epcoritamab.
5.3 Preclinical Safety Data
Genotoxicity.
Genotoxicity studies have not been conducted with epcoritamab. As a large protein molecule, epcoritamab is not expected to interact with DNA or other chromosomal material.
Carcinogenicity.
Carcinogenicity studies have not been conducted with epcoritamab.6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium acetate trihydrate, glacial acetic acid, sorbitol, polysorbate 80, water for injections.
6.2 Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products and/or diluents except those listed in Section 4.2 Dose and Method of Administration, Preparation and administration.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store and transport at 2°C-8°C (Refrigerate. Do not freeze).
Keep in the original carton to protect from light. Do not shake.
See Section 4.2 Dose and Method of Administration, Storage for diluted and prepared Epkinly for information on the storage for diluted and prepared Epkinly.
6.5 Nature and Contents of Container
Epkinly 4 mg/0.8 mL concentrate for solution for injection.
Type I glass vial with bromobutyl rubber stopper and aluminum seal with plastic flip off cap. The vial stopper is not made with natural rubber latex. Pack size of 1 vial.
Epkinly 48 mg/0.8 mL solution for injection.
Type I glass vial with bromobutyl rubber stopper and aluminum seal with plastic flip off cap. The vial stopper is not made with natural rubber latex. Pack size of 1 vial.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Epcoritamab has a regular IgG1 structure and biochemical characteristics typical of human IgG1 and has an approximate molecular weight of 149 kDa.
CAS number.
2134641-34-0.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes


 Administer Epkinly until disease progression or unacceptable toxicity.
Administer Epkinly until disease progression or unacceptable toxicity. Prophylaxis against Pneumocystis jirovecii pneumonia (PCP) and herpes virus infections is strongly recommended especially during concurrent use of steroids.
Prophylaxis against Pneumocystis jirovecii pneumonia (PCP) and herpes virus infections is strongly recommended especially during concurrent use of steroids.


 Table 7 summarises laboratory abnormalities in EPCORE NHL-1.
Table 7 summarises laboratory abnormalities in EPCORE NHL-1. Other adverse reactions: haemophagocytic lymphohistiocytosis (HLH) (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome), hypogammaglobulinaemia.
Other adverse reactions: haemophagocytic lymphohistiocytosis (HLH) (see Section 4.4 Special Warnings and Precautions for Use, Cytokine release syndrome), hypogammaglobulinaemia. Efficacy was established based on overall response rate (ORR) determined by Lugano criteria (2014) as assessed by Independent Review Committee (IRC). The median follow-up time was 11 months (range: 0.3 to 17.9 months).
Efficacy was established based on overall response rate (ORR) determined by Lugano criteria (2014) as assessed by Independent Review Committee (IRC). The median follow-up time was 11 months (range: 0.3 to 17.9 months). The median time to CR was 2.7 months (range: 1.2 to 11.1 months).
The median time to CR was 2.7 months (range: 1.2 to 11.1 months).