1 Name of Medicine
Erythromycin lactobionate.
2 Qualitative and Quantitative Composition
Erythromycin Panpharma is available as a sterile, lyophilized cake for reconstitution containing the equivalent of 1 g erythromycin activity.
Erythromycin is produced by a strain of Streptomyces erythraeus and belongs to the macrolide group of antibiotics. It is basic and readily forms salts with acids.
Erythromycin Panpharma (sterile erythromycin lactobionate) is a soluble salt of erythromycin suitable for intravenous administration. The main component is erythromycin A lactobionate.
3 Pharmaceutical Form
Powder for injection. For intravenous infusion following reconstitution and dilution.
4.1 Therapeutic Indications
Oral erythromycin is not considered to be the antibiotic of choice in severely ill patients.
Erythromycin Panpharma (sterile erythromycin lactobionate) is indicated in the treatment of infections caused by susceptible strains of the designated organisms in the diseases listed below when oral administration is not possible or when the severity of the infection requires immediate high serum levels of erythromycin. Intravenous therapy should be replaced by oral administration at the appropriate time.
Upper respiratory tract infections caused by Streptococcus pyogenes (Group A betahaemolytic streptococci); Streptococcus pneumoniae (Diplococcus pneumoniae); Haemophilus influenzae (many strains of H. influenzae are not susceptible to the erythromycin concentrations ordinarily achieved).
Lower respiratory tract infections caused by Streptococcus pyogenes (Group A betahaemolytic streptococci); Streptococcus pneumoniae (Diplococcus pneumoniae).
Respiratory tract infections due to Mycoplasma pneumoniae.
Skin and skin structure infections caused by Streptococcus pyogenes and Staphylococcus aureus (resistant staphylococci may emerge during treatment).
Diphtheria - as an adjunct to diphtheria antitoxin in infections due to Corynebacterium diphtheriae to prevent establishment of carriers and to eradicate the organism in carriers.
Acute pelvic inflammatory disease caused by Neisseria gonorrhoeae: Erythromycin Panpharma (sterile erythromycin lactobionate) followed by erythromycin stearate, base or ethyl succinate orally, as an alternative drug in treatment of acute pelvic inflammatory disease caused by N. gonorrhoeae in female patients with a history of sensitivity to penicillin.
Before treatment of gonorrhoea, patients who are suspected of also having syphilis should have microscopic examination for T. pallidum (by immuno-fluorescence or dark field) before receiving erythromycin and monthly serologic tests for a minimum of 4 months thereafter.
Legionnaires' disease caused by Legionella pneumophila. Although no controlled clinical efficacy studies have been conducted, in vitro and limited preliminary clinical data suggest that erythromycin may be effective in treating Legionnaires' disease.
4.2 Dose and Method of Administration
Note.
For I.V. administration only.
Do not administer as a bolus. Erythromycin Panpharma (sterile erythromycin lactobionate) must be administered by continuous intermittent intravenous infusion only. Due to the local irritative effects of erythromycin as well as reports of QT interval prolongation and ventricular arrhythmias (some of which have been fatal) being associated with elevated serum concentrations of erythromycin, the drug must not be administered rapidly by direct intravenous injection (IV push).
Continuous infusion of erythromycin lactobionate is preferable due to the slower infusion rate and its lower concentration of erythromycin; however, intermittent infusion at six hour intervals is also effective. Intravenous erythromycin should be replaced by oral erythromycin as soon as possible.
For slow continuous infusion.
The final diluted solution of erythromycin lactobionate is prepared to give a concentration of 1 g per litre (1 mg/mL).
For intermittent infusion.
Administer one-fourth the total daily dose of erythromycin lactobionate by intravenous infusion over a minimum of 60 minutes at intervals not greater than every six hours. A longer period of infusion should be used in patients with risk factors or previous evidence of arrhythmias. The final diluted solution of erythromycin lactobionate is prepared to give a concentration of 1 to 5 mg/mL. No less than 100 mL of intravenous diluent should be used. Infusion should be sufficiently slow to minimise pain along the vein.
For the treatment of severe infections in adults and children, the recommended intravenous dose of erythromycin lactobionate is 15 to 20 mg/kg/day. Higher doses, up to 4 g/day, may be given for severe infections.
For treatment of acute pelvic inflammatory disease caused by N. gonorrhoeae, in female patients hypersensitive to penicillins, administer 500 mg erythromycin lactobionate every six hours for three days, followed by oral administration of 250 mg erythromycin stearate or base, or 400 mg erythromycin ethyl succinate, every six hours for seven days.
For treatment of Legionnaires' disease.
Although optimal doses have not been established, doses utilised in reported clinical data were 1 to 4 grams daily in divided doses.
Preparation of solution.
1. Prepare the initial solution of Erythromycin Panpharma by adding 20 mL of sterile water for injection to the 1 g vial. Use only sterile water for injection, as other diluents may cause precipitation during reconstitution. Do not use diluents containing preservatives or inorganic salts. Ensure that the contents of the vial are fully dissolved before using the product. The volume after reconstitution contains an excess to ensure that the stated volume can be withdrawn.
After reconstitution, each mL contains 50 mg of erythromycin activity.
2. Add the initial dilution to one of the following diluents before administration: 0.9% Sodium Chloride Injection, Lactated Ringer's Injection, Normosol-R.
For slow continuous infusion.
Add 20 mL of reconstituted solution to enough diluent to make up to 1000 mL total solution. This is equivalent to a concentration of 1 g of erythromycin per litre (1 mg/mL) or 0.1% infusion.
For intermittent infusion.
Add 20 mL of reconstituted solution to enough diluent to make up to 1000 mL total solution. This is equivalent to a concentration of 1 g of erythromycin per litre (1 mg/mL) or 0.1% for an intermittent infusion.
Add 20 mL of reconstituted solution to enough diluent to make up to 200 mL total solution. This is equivalent to a concentration of 5 mg/mL or 0.5% for an intermittent infusion.
If it is decided to administer the daily dose as an intermittent infusion, then the erythromycin concentration should not exceed 5 mg/mL and the time of each infusion should be over a minimum of 60 minutes.
3. The following solutions may also be used provided they are first buffered with 8.4% Sodium Bicarbonate Solution (add 0.5 mL of 8.4% Sodium Bicarbonate Solution per 100 mL of solution): 5% glucose injection, 5% glucose and Lactated Ringer's Injection, 5% glucose and 0.9% Sodium Chloride Injection.
Note.
8.4% Sodium Bicarbonate Solution must be added to these solutions so that their pH is in the optimum range for erythromycin lactobionate stability. Acidic solutions of erythromycin lactobionate are unstable and lose their potency rapidly. A pH of at least 5.5 is desirable for the final diluted solution of erythromycin lactobionate.
No drug or chemical agent should be added to an erythromycin lactobionate IV fluid admixture unless its effect on the chemical and physical stability of the solution has first been determined.
The product is for single use in one patient only. Discard any residue.
Stability.
To reduce the microbiological hazard, use as soon as practicable after reconstitution. The final diluted solution of erythromycin lactobionate should be completely administered within 8 hours, since it is not suitable for storage. If storage is necessary, hold at 2-8°C for not more than 12 hours.
Parenteral drug products should be inspected visually for particulate matter and discolouration prior to administration, whenever solution and container permit.4.3 Contraindications
Erythromycin is contraindicated in the case of:
Hypersensitivity to erythromycin or any of excipients in the formulation (see Section 6.1 List of Excipients);
Hypersensitivity to other antibiotics from the macrolide family;
Severely impaired hepatic function;
Concurrent treatment with HMG-CoA reductase inhibitors (e.g. lovastatin or simvastatin), ergotamine or dihydroergotamine (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions);
Erythromycin should not be given to patients with a history of QT prolongation (congenital or documented acquired QT prolongation) or ventricular cardiac arrhythmia, including torsades de pointes (see Section 4.4 Special Warnings and Precautions for Use; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions);
Clinically relevant cardiac arrhythmias (e.g. ventricular arrhythmias) or in severe congestive heart failure (NYHA IV);
Concomitant intake of medicinal products, which can lead to prolongation of the QT interval and under some circumstances to life-threatening ventricular arrhythmia (torsade de pointes) e.g. terfenadine, astemizole, cisapride, pimozide, domperidone, class IA and III antiarrhythmics (e.g. disopyramide), certain neuroleptics, tri- and tetracyclic antidepressants, arsenic trioxide, methadone, budipine, certain fluoroquinolones, imidazole anti-mycotics and anti-malarials (e.g. pentamidine i.v.) (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions);
Disturbances of the electrolyte balance (especially in the case of hypokalaemia and hypomagnesaemia due to the risk of prolongation of QT interval);
Rapid administration by direct intravenous injection (IV push) (see Section 4.4 Special Warnings and Precautions for Use).
4.4 Special Warnings and Precautions for Use
Cardiovascular events.
Prolongation of the QT interval, reflecting effects on cardiac repolarisation imparting a risk of developing cardiac arrhythmia and torsades de pointes, have been seen in patients treated with macrolides including erythromycin [see Section 4.3 Contraindications; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 4.8 Adverse Effects (Undesirable Effects)]. Fatalities have been reported.
Erythromycin should be used with caution in the following: Patients with coronary artery disease, severe cardiac insufficiency, conduction disturbances or clinically relevant bradycardia. Patients concomitantly taking other medicinal products associated with QT prolongation (see Section 4.3 Contraindications; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Limited data suggest that these adverse effects may be associated with abnormally elevated serum erythromycin concentrations following rapid administration. Erythromycin therefore must not be administered rapidly by direct intravenous injection (IV push) (see Section 4.2 Dose and Method of Administration). Elderly patients may be more susceptible to drug-associated effects on the QT interval.
Epidemiological studies investigating the risk of adverse cardiovascular outcomes with macrolides have shown variable results. Some observational studies have identified a rare short term risk of arrhythmia, myocardial infarction and cardiovascular mortality associated with macrolides including erythromycin. Consideration of these findings should be balanced with treatment benefits when prescribing erythromycin.
If during therapy with erythromycin symptoms such as palpitations, dizziness or syncope occur which can be signs of arrhythmia, an investigation of the patient including Electrocardiogram and determination of the QT interval should be initiated immediately.
Electrolyte disturbances promote the probability of cardiac arrhythmia. In the case of risk factors for electrolyte disturbances (such as diuretic/laxative medication, vomiting, diarrhoea, use of insulin in emergency situations, renal diseases or anorectic conditions), adequate laboratory tests and if necessary an adequate electrolyte balance should be carried out.
Use in hepatic impairment.
There have been reports of hepatic dysfunction, including increased liver enzymes, hepatomegaly and hepatocellular and/or cholestatic hepatitis with or without jaundice in patients receiving erythromycin products (see Section 4.8 Adverse Effects (Undesirable Effects)). Since erythromycin is principally excreted by the liver, caution should be exercised when erythromycin is administered to patients with impaired hepatic function. Erythromycin is contraindicated in severe hepatic impairment (see Section 4.3 Contraindications).
Patients with existing liver damage and allergies may be at higher risk of intrahepatic cholestasis and cholestatic jaundice due to sensitisation, resulting in colicky abdominal pain, nausea, vomiting, urticaria, eosinophilia and fever. Although these reactions can occur after initial administration, the risk increases with repeated administration and therapy lasting longer than 10 days (see Section 4.8 Adverse Effects (Undesirable Effects)).
Clostridium difficile-associated diseases.
The use of erythromycin can lead to the development of severe colitis as a result of colonisation with C. difficile, a toxin-producing organism. Colitis, which may or may not be accompanied by the formation of a pseudomembrane in the colon, can range in severity from mild diarrhoea to fatal colitis. If significant diarrhoea occurs, erythromycin should be discontinued (diarrhoea however, may begin several weeks to over two months after cessation of antibiotic therapy). This may be sufficient treatment in the early stages although cholestyramine orally may help by binding the toxin in the colonic lumen. However, in moderate to severe cases, appropriate therapy with a suitable oral antibacterial agent effective against C. difficile should be considered. Treatment with bacitracin has also been reported to be successful.
Medicines which delay peristalsis, e.g. opiates and diphenoxylate with atropine (Lomotil), may prolong and/or worsen the condition and should not be used. Vancomycin is not effective if given parenterally. Fluids, electrolytes and protein replacement therapy should be provided when indicated.
Musculature and nervous system.
Rhabdomyolysis with or without renal impairment has been reported in seriously ill patients receiving erythromycin concomitantly with simvastatin, lovastatin or atorvastatin (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). The concomitant use of these medicines with erythromycin is contraindicated (see Section 4.3 Contraindications).
Concomitant use of erythromycin with other statins should be instructed by the physician to pay attention to signs of myopathy (e.g. inexplicable muscle pain or weakness or dark coloured urine). If myopathy occurs, the intake of the statin has to be stopped immediately.
Erythromycin may aggravate the weakness of patients with myasthenia gravis.
Allergic reactions.
With the administration of erythromycin, severe, life-threatening allergic reactions may occur, e.g. severe skin reactions such as erythema multiforme, Stevens-Johnson syndrome or toxic epidermal necrolysis (especially in children of all ages), as well as angioneurotic oedema or anaphylaxis. A cross allergy in patients with hypersensitivity to macrolide antibiotics can exist, so in patients with known hypersensitivity to macrolides or related substances (e.g. ketolides), special caution is recommended. At first signs of hypersensitivity, erythromycin has to be stopped immediately and necessary symptomatic emergency measures initiated.
As with other macrolides, rare serious allergic reactions, including acute generalised exanthematous pustulosis (AGEP) have been reported. If an allergic reaction occurs, the drug should be discontinued and appropriate therapy should be instituted. Physicians should be aware that reappearance of the allergic symptoms may occur when symptomatic therapy is discontinued.
Prolonged or repeated therapy.
Overgrowth of non-susceptible bacteria or fungi may occur during prolonged or repeated therapy. If superinfection occurs, erythromycin should be discontinued and appropriate therapy instituted.
In the case of a treatment duration longer than 3 weeks, it is recommended that whole blood count and hepatic and renal function tests be performed at regular intervals.
When indicated, incision and drainage or other surgical procedures should be performed in conjunction with antibiotic therapy.
Eye disorder.
There is a risk for developing visual impairments after exposure to erythromycin. For some patients, a pre-existing dysfunction in mitochondrial metabolism from genetic causes such as Leber's hereditary optic neuropathy (LHON) and autosomal dominant optic atrophy (ADOA) might play a contributing role.
Pneumonia.
Due to very common resistance of Streptococcus pneumoniae against macrolides, erythromycin is not the first-choice therapy in case of ambulant acquired pneumonia. In hospital acquired pneumonia, erythromycin should only be used in combination with other antibiotics.
Use in the elderly.
See Section 4.4 Special Warnings and Precautions for Use, Cardiovascular events.
Paediatric use.
To avoid liver damage due to overdose in infants and toddlers, dosing should be dependent on the clinical picture and the course of the disease.
There have been reports of infantile hypertrophic pyloric stenosis (IHPS) occurring in infants following erythromycin therapy. Epidemiological studies including data from meta-analyses suggest a 2-3-fold increase in the risk of IHPS following exposure to erythromycin in infancy. This risk is highest following exposure to erythromycin during the first 14 days of life. Available data suggests a risk of 2.6% (95% CI: 1.5-4.2%) following exposure to erythromycin during this time period. The risk of IHPS in the general population is 0.1-0.2%. Since erythromycin may be used in the treatment of conditions in infants which are associated with significant mortality or morbidity (such as pertussis or chlamydia), the benefit of erythromycin therapy needs to be weighed against the potential risk of developing IHPS. Parents should be informed to contact their physician if vomiting or irritability with feeding occurs.
Effects on laboratory tests.
Erythromycin interferes with the fluorimetric determination of urinary catecholamines.4.5 Interactions with Other Medicines and Other Forms of Interactions
Theophylline.
Recent data from studies of erythromycin reveal that its use in patients who are receiving high doses of theophylline may be associated with an increase of serum theophylline levels and potential theophylline toxicity. In case of theophylline toxicity and/or elevated serum theophylline levels, the dose of theophylline should be reduced while the patient is receiving concomitant erythromycin therapy.
There have been published reports suggesting that when oral erythromycin is given concurrently with theophylline there is a significant decrease in erythromycin serum concentrations. This decrease could result in subtherapeutic concentrations of erythromycin.
Carbamazepine.
Erythromycin administration in patients receiving carbamazepine has been reported to cause increased serum levels of carbamazepine with subsequent development of signs of carbamazepine toxicity.
Digoxin.
Concomitant administration of erythromycin and digoxin has been reported to result in elevated serum digoxin levels.
Oral anticoagulants.
There have been reports of increased anticoagulant effects when erythromycin and oral anticoagulants (e.g. warfarin, rivaroxaban) were used concomitantly.
Medicines that prolong the QTc interval.
Erythromycin has been shown to prolong the QTc interval and is associated with case reports of torsade de pointes in some patients. Patients with uncorrected electrolyte disorders particularly hypokalaemia, known prolongation of the QTc interval, or those concurrently receiving medicines that prolong the QTc interval, in particular Class IA (e.g. quinidine, procainamide) or Class III (amiodarone, sotalol) antiarrhythmics, certain neuroleptics, tri- and tetracyclic antidepressants, ebastine, arsenic trioxide, methadone, budipine, certain fluoroquinolones, imidazole antimycotics and anti-malarials (e.g. pentamidine i.v.), are at increased risk of ventricular arrhythmias. As these predisposing conditions may increase the risk for ventricular arrhythmias, erythromycin should not be used in patients with ongoing proarrhythmic conditions (see Section 4.3 Contraindications).
Hydroxychloroquine and chloroquine.
Erythromycin should be used with caution in patients receiving these medicines known to prolong the QT interval due to the potential to induce cardiac arrhythmia and serious adverse cardiovascular events.
Medicines metabolised by the cytochrome P450 system.
Erythromycin is a substrate and inhibitor of the 3A isoform subfamily of the cytochrome P450 system (CYP3A) and P-glycoprotein. Co-administration of erythromycin and a medicine primarily metabolised by CYP3A may be associated with elevations in drug concentrations that could increase or prolong both the therapeutic or adverse effects of the concomitant medicine e.g. ciclosporin, phenytoin, felodipine, hexobarbital, carbamazepine, alfentanil, quinidine, disopyramide, bromocriptine, methylprednisolone, vinblastine, sildenafil, cilostazol, valproate, tacrolimus, terfenadine, mizolastine, domperidone, astemizole, rifabutin, verapamil, diltiazem, acenocoumarol, digoxin, dihydroergotamine, ergotamine, midazolam, omeprazole, theophylline, triazolam and antifungals (e.g. fluconazole, ketoconazole and itraconazole). Dosage adjustments may be considered, and when possible, serum concentrations of medicines primarily metabolised by CYP3A4 should be monitored closely in patients receiving erythromycin.
Erythromycin has been shown to prolong the QTc interval and is associated with case reports of torsades de pointes in some patients. In one published study patients who used both oral erythromycin and strong CYP3A inhibitors (azole antifungal medicines [ketoconazole, itraconazole and fluconazole, all administered systemically], diltiazem, verapamil, troleandomycin, mibefradil, nefazodone) had a risk of sudden death from cardiac causes that was five times as great as that among patients who had not used these medicines. Many of the medicines that are known to block CYP3A4 also have direct effects on repolarisation, which may cause a dramatic lengthening of the QT interval. Given that there are alternatives to erythromycin and these listed CYP3A inhibitors, the use of these combinations should be avoided.
Hypotension, bradyarrhythmia and lactic acidosis have been observed in patients receiving concurrent verapamil.
Medicines that induce CYP3A (such as rifampicin, phenytoin, carbamazepine, phenobarbital (phenobarbitone), St John's Wort) may induce the metabolism of erythromycin. This may lead to sub-therapeutic levels of erythromycin and a decreased effect. The induction decreases gradually after discontinuing treatment with CYP3A4 inducers. Erythromycin should not be used during, or for two weeks after stopping treatment, with CYP3A4 inducers.
The following are examples of some clinically significant CYP3A based drug interactions. Interactions with other medicines metabolised by the CYP3A isoform are also possible. The following CYP3A based drug interactions have been observed with erythromycin products in postmarketing experience.
Corticosteroids.
Caution should be exercised in concomitant use of erythromycin with systemic and inhaled corticosteroids that are primarily metabolised by CYP3A due to the potential for increased systemic exposure to corticosteroids. If concomitant use occurs, patients should be closely monitored for systemic corticosteroid undesirable effects.
Triazolobenzodiazepines (triazolam, alprazolam and midazolam).
Triazolam, alprazolam and midazolam plasma concentrations may approximately double when erythromycin is co-administered, due to a reduction in clearance and increase in elimination half-life but drug accumulation has not been observed with repeated dosing. Therefore, consideration of dose reduction may be appropriate in patients treated concurrently with triazolam, alprazolam or midazolam and erythromycin.
Ergotamine/dihydroergotamine.
Post-marketing reports indicate that co-administration of erythromycin with ergotamine or dihydroergotamine has been associated with acute ergot toxicity characterized by vasospasm and ischemia of the extremities and other tissues including the central nervous system (see Section 4.3 Contraindications).
Sildenafil.
Erythromycin has been reported to increase the systemic exposure (AUC) of sildenafil. Reduction of sildenafil dosage should be considered.
HMG-CoA reductase inhibitors.
Erythromycin has been reported to increase concentrations of HMG-CoA reductase inhibitors (e.g. lovastatin and simvastatin). Rare reports of rhabdomyolysis have been reported in patients taking these medicines concomitantly (see Section 4.3 Contraindications).
Colchicine.
There have been post-marketing reports of colchicine toxicity with concomitant use of erythromycin and colchicine.
Astemizole and terfenadine.
Erythromycin significantly alters the metabolism of astemizole and terfenadine when taken concomitantly. Rare cases of serious cardiovascular adverse events including cardiac arrest, torsade de pointes and other ventricular arrhythmias have been observed (see Section 4.3 Contraindications; Section 4.8 Adverse Effects (Undesirable Effects)).
Cisapride.
Elevated cisapride levels have been reported in patients receiving erythromycin, and cisapride concomitantly. This may result in QT prolongation and cardiac arrhythmias including ventricular tachycardia, ventricular fibrillation and torsade de pointes. Similar effects have been observed in patients taking pimozide and clarithromycin, another macrolide antibiotic (see Section 4.3 Contraindications).
Zopiclone.
Erythromycin has been reported to decrease the clearance of zopiclone and thus may increase the pharmacodynamic effects of this medicine.
Anti-bacterial agents.
Antagonism has been demonstrated in vitro between erythromycin and clindamycin, lincomycin and chloramphenicol. Same interaction is applicable with streptomycin, tetracyclines, colistin and bactericidal beta-lactam antibiotics (e.g. penicillin, cephalosporin).
Cimetidine.
It may inhibit the metabolism of erythromycin which may lead to an increased plasma concentration.
Protease inhibitors.
Protease inhibitors (e.g. ritonavir) has been reported to increase the level of effect of erythromycin by altering drug metabolism.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There was no apparent effect on male or female fertility in rats treated with erythromycin base by oral gavage at 700 mg/kg/day (approximately 9 times the human dose).
(Category A)
Category A: "Drugs which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed."
No evidence of teratogenicity or embryotoxicity was observed when erythromycin base was given by oral gavage to pregnant rats and mice at 700 mg/kg/day (approximately 9 times the maximum human dose), and to pregnant rabbits at 125 mg/kg/day (approximately 1.5 times the maximum human dose).
A slight reduction in birth weights was noted when female rats were treated prior to mating, during mating, gestation and lactation at an oral dosage of 700 mg/kg/day of erythromycin base; weights of the offspring were comparable to those of the controls by weaning. No evidence of teratogenicity or effects on reproduction was noted at this dosage. When administered during late gestation and lactation periods, this dosage of 700 mg/kg/day (14 times the maximum human dose) did not result in any adverse effects on birth weight, growth and survival of offspring.
There are no adequate and well-controlled studies in pregnant women. However, observational studies in humans have reported cardiovascular malformations after exposure to medicinal products containing erythromycin during early pregnancy.
Erythromycin has been reported to cross the placental barrier in humans, but foetal plasma levels are generally low. There have been reports suggesting erythromycin does not reach the foetus in adequate concentration to prevent congenital syphilis. Infants born to women treated during pregnancy with oral erythromycin for early syphilis should be treated with an appropriate penicillin regimen.
There have been reports that maternal macrolide antibiotics exposure within 10 weeks of delivery may be associated with a higher risk of infantile hypertrophic pyloric stenosis (IHPS).
Erythromycin should be used by women during pregnancy only if clearly needed.
Erythromycin is concentrated in breast milk and adverse effects have been seen in breast-fed infants including gastrointestinal disturbances, pyloric stenosis (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use), sensitisation or colonisation with fungi. Caution should be exercised when erythromycin is administered to a nursing woman.4.7 Effects on Ability to Drive and Use Machines
Erythromycin has a negligible influence on the ability to concentrate and react. However, the occurrence of undesirable effects can negatively influence the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Adverse effects following the use of intravenous erythromycin are rare. Occasional venous irritation has been encountered, but if the infusion is given slowly, in dilute solution, preferably by continuous intravenous infusion or intermittent infusion in no less than 60 minutes, pain and vessel trauma are minimised.
The following adverse effects have been reported for erythromycin. The adverse effects are listed according to the frequency defined as:
Very common (≥ 1/10); Common (≥ 1/100 - < 1/10); Uncommon (≥ 1/1000 - < 1/100); Rare (> 1/10,000 - < 1/1000); Very rare (< 1/10,000); Not known (cannot be estimated from the available data). See Table 1.
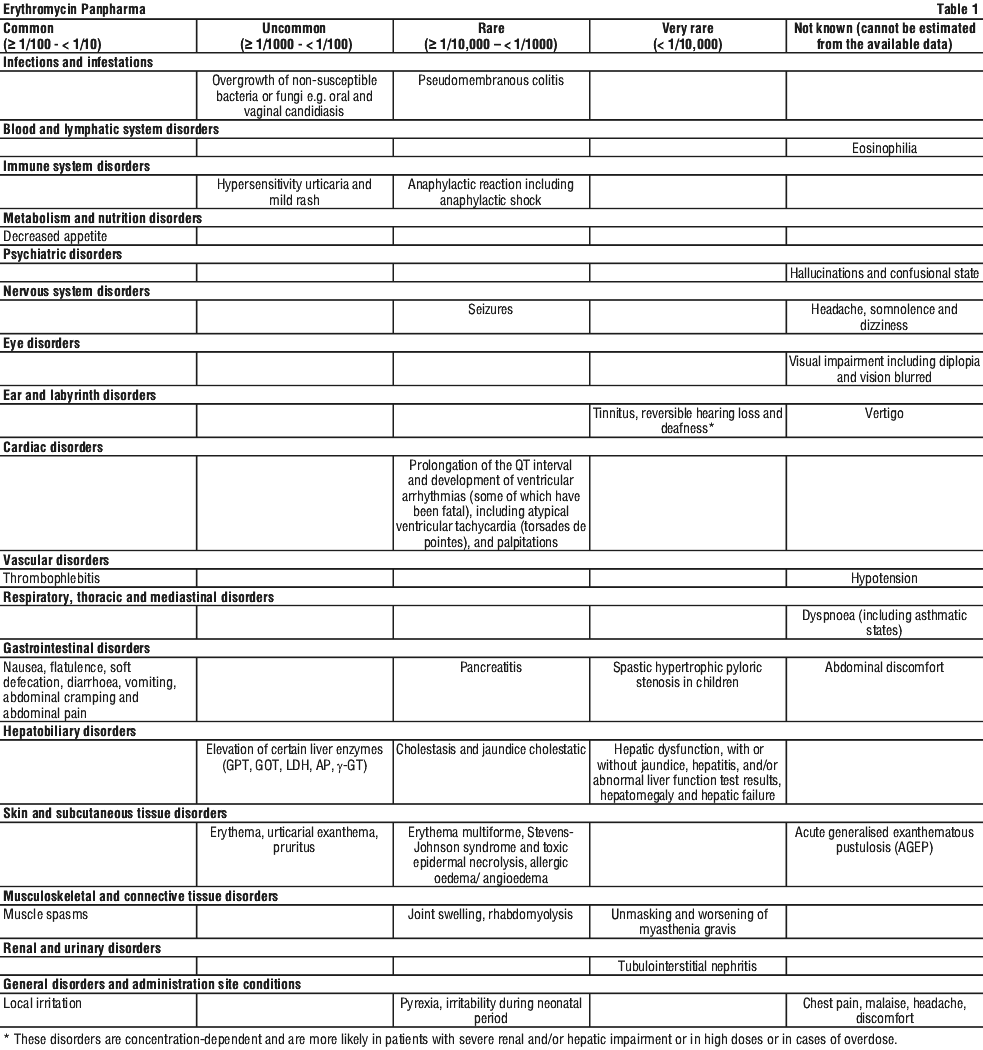
Infantile hypertrophic pyloric stenosis (IHPS).
7 out of 157 [5%] newborns developed severe non-bilious vomiting or irritability with feeding and IHPS who were given oral erythromycin for pertussis prophylaxis. The relative risk of IHPS was increased 6.8 fold (95% CI = 3-16) compared to a retrospective cohort of infants.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at https://www.tga.gov.au/safety/reporting-problems.4.9 Overdose
Reports indicate that the ingestion of large amounts of erythromycin can be expected to produce gastrointestinal distress, hearing problems and other adverse effects (see Section 4.8 Adverse Effects (Undesirable Effects)). Allergic reactions accompanying overdosage should be treated by the prompt elimination of unabsorbed drug and supportive measures. Erythromycin serum levels are not appreciably altered by haemodialysis or peritoneal dialysis.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Microbiology. Erythromycin binds to the 50S ribosomal subunits of susceptible bacteria and suppresses protein synthesis. The mode of action of erythromycin is by inhibition of the protein synthesis without affecting nucleic acid synthesis.
Erythromycin is usually active in vitro against the following Gram positive and Gram negative organisms.
Streptococcus pyogenes, alpha-haemolytic streptococcus (viridans group), Staphylococcus aureus, Streptococcus pneumoniae, Corynebacterium diphtheriae (as an adjunct to antitoxin), Corynebacterium minutissimum, Listeria monocytogenes, Clostridium tetani, Neisseria gonorrhoeae, Bordetella pertussis, Haemophilus influenzae (some strains are resistant), Legionella pneumophila, Treponema pallidum, Chlamydia trachomatis, Mycoplasma pneumoniae, Campylobacter jejuni (in severe or prolonged cases), Ureaplasma urealyticum.
Not all strains of the organism listed above are sensitive and culture and susceptibility testing should be done. Several strains of Haemophilus influenzae and staphylococci have been found to be resistant to erythromycin. Staphylococci resistant to erythromycin may emerge during a course of therapy.
Susceptibility testing. Dilution or diffusion techniques - either quantitative (MIC) or breakpoint, should be used following a regular updated, recognised and standardised method (e.g. CLSI).
Standardised susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures.
A report of "Susceptible" indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable.
A report of "Intermediate" indicates that the result should be considered equivocal, and if the microorganism is not fully susceptible to alternative clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone, which prevents small uncontrolled technical factors from causing major discrepancies in interpretation.
A report of "Resistant" indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable: other therapy should be selected.
Note 1.
The prevalence of resistance may vary geographically for selected species and local information on resistance is desirable, particularly when treating severe infections.
Note 2.
Many strains of Haemophilus influenzae are resistant to erythromycin alone, but are susceptible to erythromycin and sulfonamides together. Staphylococci resistant to erythromycin may emerge during a course of erythromycin therapy. Culture and susceptibility testing should be performed.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Absorption.
Intravenous infusion of 500 mg of erythromycin lactobionate at a constant rate over 1 hour in fasting adults produced a mean serum erythromycin level of approximately 7 microgram/mL at 20 minutes, 10 microgram/mL at 1 hour, 2.6 microgram/mL at 2.5 hours and 1 microgram/mL at 6 hours.
Distribution.
The extent of plasma protein binding has been variably reported but is probably of the order of 75%. Erythromycin diffuses readily into most body fluids with the exception of cerebrospinal fluid, synovial fluid and vitreous humor.
Erythromycin appears in breast milk at levels which are approximately 50% of the plasma concentration. It crosses the placenta and foetal plasma levels are usually 5%-20% of the maternal plasma concentration (see Section 4.4 Special Warnings and Precautions for Use).
Excretion.
In the presence of normal renal function, the plasma half-life is approximately 1.4 hours; this may increase to 6 hours in anuric patients but does not usually require dosage adjustment. Erythromycin is not removed by dialysis.
In the presence of normal hepatic function, erythromycin is concentrated in the liver and is excreted in the bile. However, only a small proportion of the administered dose appears in the bile. The effect of hepatic dysfunction on biliary excretion of erythromycin is not known. Approximately 12 to 15 percent of an intravenously administered dose of erythromycin is excreted in the urine unchanged. A substantial proportion of the administered dose remains unaccounted for and is presumably metabolised probably in the liver.
5.3 Preclinical Safety Data
Genotoxicity.
Erythromycin was not genotoxic in assays for bacterial and mammalian mutagenicity and for clastogenicity in vitro. The clastogenic potential of erythromycin has not been investigated in vivo.
Carcinogenicity.
Long-term (2 year) oral studies conducted in rats up to about 400 mg/kg/day and in mice up to about 500 mg/kg/day with erythromycin stearate did not provide evidence of tumourigenicity.6 Pharmaceutical Particulars
6.1 List of Excipients
Erythromycin Panpharma contains no excipients.
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from light.
6.5 Nature and Contents of Container
Erythromycin Panpharma (erythromycin lactobionate), powder for injection is supplied as a sterile, lyophilized cake for reconstitution in single clear, colourless glass vials with rubber stoppers and an aluminium seal and plastic flip-off cap. Each vial contains the equivalent of 1 g of erythromycin. Packs of 1, 10 or 25 vials.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
The structure formula of erythromycin lactobionate is shown:
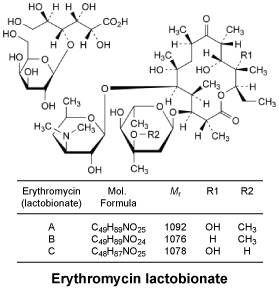 Active ingredient: erythromycin lactobionate.
Active ingredient: erythromycin lactobionate.
Chemical names: (3R,4S,5S,6R,7R,9R,11R,12R,13S,14R)-4-[(2,6-dideoxy-3-C-methyl-3-O-methyl-α-L-ribo-hexopyranosyl)oxy]-14-ethyl-7,12,13-trihydroxy-3,5,7,9,11,13-hexamethyl-6-[[3,4,6-trideoxy-3-(dimethylamino)-β-D-xylohexopyranosyl]oxy]oxacyclotetradecane-2,10-dione, 4-O-β-D-galactopyranosyl-D-gluconate erythromycin A lactobionate (main component).
Molecular weight: see Chemical structure.
Molecular formula: see Chemical structure.
The drug substance is a white or slightly yellow, hygroscopic powder and is soluble in water. A 2% to 5% solution in water has a pH of 6.5 to 7.5.
CAS number.
3847-29-8.7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (Schedule 4).
Summary Table of Changes



 Active ingredient: erythromycin lactobionate.
Active ingredient: erythromycin lactobionate.