What is in this leaflet
This leaflet answers some common questions about Evenity. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Evenity against the benefits they expect it will have for you.
Read this leaflet carefully before you start using Evenity. It contains important information for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Evenity is used for
Evenity is used to treat:
- osteoporosis in women after menopause
- osteoporosis in men
Bone is a living tissue and is renewed all the time. In women, the ovaries produce the hormone oestrogen which helps keep bones healthy. After menopause, the oestrogen level drops which affects the bone renewal cycle so that more bone is lost than made, resulting in a lower bone mass. This leaves bones thin and fragile and may eventually lead to a condition called osteoporosis.
Osteoporosis becomes more common, occurring in women and men with increasing age. Many people with osteoporosis have no symptoms but they are still at risk of breaking bones (developing fractures), especially in the spine, hips and wrists.
Evenity contains the active ingredient romosozumab, a medicine that helps build bone, which makes bones stronger and less likely to break.
Evenity inhibits a protein called sclerostin. Evenity binds to and prevents the activity of sclerostin. Evenity acts both to build new bone and also decrease break down of existing bone to strengthen bones, improve bone mass and structure, and lower the chance of breaking bones.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
This medicine is available only with a doctor's prescription.
Before you use Evenity
When you must not take it
Do not take Evenity if you have an allergy to:
- any medicine containing romosozumab
- any of the ingredients listed at the end of this leaflet.
- any medicines that are produced using Chinese Hamster Ovary cells.
Some of the symptoms of an allergic reaction may include:
- swelling usually of the face, lips, mouth, tongue or throat which may cause difficult in swallowing or breathing
- rash, itching or hives on the skin
Do not use Evenity if you have low levels of calcium in your blood (hypocalcaemia). Your doctor will be able to tell you if your calcium levels are too low.
Do not use Evenity in a child or adolescent. The use of Evenity in children or adolescents has not been studied.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you are using other medicines to treat osteoporosis, such as Prolia or bisphosphonates.
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have had calcium or vitamin D deficiency. Your doctor will do a blood test to check your calcium levels before you use Evenity if you have not had a test recently.
Tell your doctor if you have any problems with your mouth or teeth. Your doctor may ask you to undergo a dental examination.
Tell your doctor if you are pregnant, think you may be pregnant, or plan to become pregnant. Evenity has not been tested in pregnant women, and is not intended for use if you are pregnant. It is not known if Evenity may harm your unborn baby. Your doctor can discuss with you the risks and benefits involved.
Tell your doctor if you are breastfeeding or plan to. It is not known if Evenity is present in breast milk. Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor about any of the above, tell him/ her before you start taking Evenity.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
How to take Evenity
Follow all directions given to you by your doctor, nurse or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions, ask your doctor or pharmacist or nurse for help.
How much to take
The usual dose of Evenity is 210 mg.
To deliver a full dose, inject two 105 mg injections one after the other.
How to take it
Evenity is given as an injection under the skin. This is called a subcutaneous injection.
Your doctor may decide that it is best for you or your carer to inject Evenity. Your doctor, nurse or pharmacist will show you or your carer how to use Evenity. Do not use Evenity unless proper training has been given.
See the detailed "Instructions for Use" provided with the medicine for instructions about the right way to store, prepare, and give your Evenity injections.
When to take it
You will receive an Evenity dose (2 injections) once every month for 12 doses.
How long to take it
Continue taking your medicine for 12 months, unless your doctor tells you otherwise. When you have finished the 12 months course of romosozumab, you will need to continue treatment for osteoporosis with another medicine.
If you forget to take it
If you miss a dose it should be administered as soon as possible. From then on, it should be scheduled every month from the date of the last injection.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much Evenity. Do this even if there are no signs of discomfort or poisoning.
While you are using Evenity
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Evenity.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you become pregnant while taking this medicine, tell your doctor immediately.
Take calcium and vitamin D supplements if your doctor has told you to. Most people do not get enough calcium and vitamin D in their diets and supplements are needed.
Tell your doctor, nurse or pharmacist immediately if you notice spasms, twitches, or cramps in your muscles, and/or numbness or tingling in your fingers, toes or around your mouth. You may have low levels of calcium in your blood.
Tell your doctor, nurse or pharmacist immediately if you experience chest pain or pressure, shortness of breath, light-headedness, headache, numbness, or weakness in face, arm, or legs, difficulty talking, changes in vision, dizziness or loss of balance.
Tell your doctor if you have had a heart attack or stroke, or a history of other blood vessel problems. Serious cardiovascular (heart and blood vessel) problems have been reported in people using Evenity. Cardiovascular problems may include heart attack or stroke. It is not known whether Evenity caused these cardiovascular problems.
Tell your doctor and dentist immediately if you experience any problems with your mouth or teeth, such as loose teeth, pain or swelling, or non-healing of sores or discharge.
Among people who have used Evenity, a small number have developed severe jaw bone problems. It is not known whether Evenity caused these jaw bone problems.
You should maintain good oral hygiene and receive routine dental check-ups. If you wear dentures you should make sure these fit properly. If you are under dental treatment or will undergo dental surgery (e.g. tooth extractions), inform your doctor and tell your dentist that you are being treated with Evenity.
Tell your doctor if you experience any new or unusual pain in your hip, groin or thigh. Among people who have used EVENITY, a small number have developed unusual fractures in their thigh bone. It is not known whether EVENITY caused these unusual fractures.
Keep all of your doctor's appointments so that your progress can be checked.
Things you must not do
Do not take Evenity to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how Evenity affects you.
Evenity has no known effects on the ability to drive or use machines, but as a general precaution, avoid driving soon after you have an injection. Practice good dental hygiene.
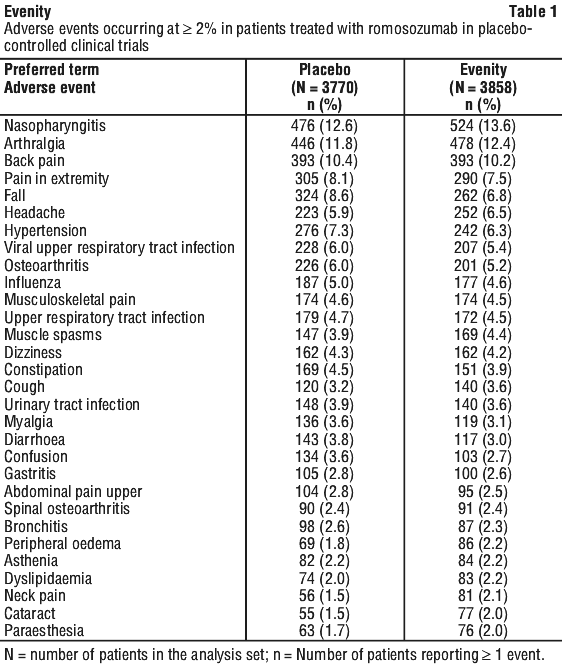
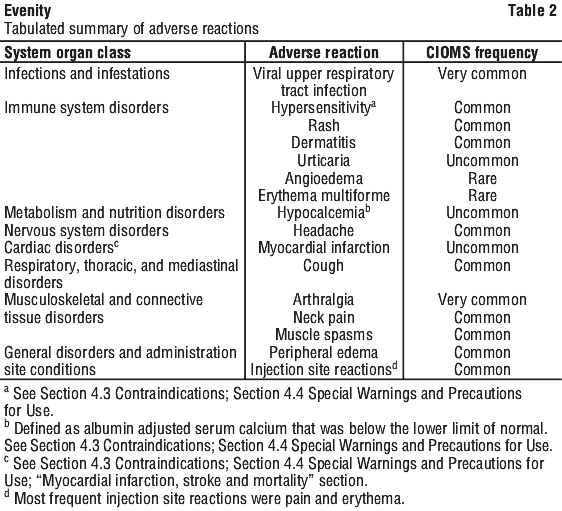
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Evenity.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor, nurse or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- redness, bruising or pain around the site where the injection was given (injection site reaction)
- common cold, such as runny nose, sore throat or cough
- joint pain
- headache
- muscle spasms
- swelling of hands, ankles or feet
- neck pain
The above list includes the more common side effects of your medicine.
If any of the following happen, tell your doctor, nurse or pharmacist immediately or go to Accident and Emergency at your nearest hospital:
- swelling usually of the face, lips, mouth, tongue or throat which may cause difficult in swallowing or breathing
- rash, itching or hives on the skin
These are very serious side effects. If you have them, you may be having a serious allergic reaction. You may need urgent medical attention or hospitalisation.
Tell your doctor, nurse or pharmacist immediately if you notice any of the following:
- numbness or tingling in your fingers, toes or around the mouth
- muscle spasms, twitches or cramps
- chest pain, shortness of breath, headache
- numbness or weakness in face, arms or legs
- difficulty talking
- changes in vision
- loss of balance
These may be serious side effects. You may need urgent medical attention.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using Evenity
Storage
If you need to store your Evenity before use, keep it in the refrigerator between 2°C and 8°C. Do not freeze.
Keep your medicine in the original carton until use to protect it from light. If you remove the medicine from the carton it may not keep well.
You may leave Evenity in the original carton outside the refrigerator to reach room temperature (up to 25°C) before injection. This will make the injection more comfortable. Once Evenity has been left to reach room temperature (up to 25°C), it must be used within 30 days. After 30 days at room temperature Evenity should be discarded.
Do not shake. Protect from direct light and do not store above 25°C.
Do not store Evenity in extreme heat or cold.
Keep it where children cannot reach it.
Disposal
Return any unused or expired medicine to your pharmacist.
Product description
What it looks like
Evenity is a clear to opalescent, colourless to light yellow liquid.
Evenity is available as a pack that contains two single-use prefilled syringes or two single-use prefilled autoinjector/pens*.
*not currently available in Australia
Ingredients
The active ingredient is romosozumab.
Each 1.17 mL prefilled syringe or autoinjector/pen of Evenity contains 105 mg romosozumab (90 mg/mL).
Other ingredients:
- calcium
- acetate
- sucrose
- polysorbate 20
- Water for injection
- Sodium hydroxide
This medicine does not contain lactose, gluten, tartrazine or any other azo dyes.
Sponsor
Evenity is supplied in Australia by:
Amgen Australia Pty Ltd
ABN 31 051 057 428
Level 7, 123 Epping Road
North Ryde NSW 2113
Medical Information: 1800 803 638
Australian Registration Number:
105 mg prefilled syringe: AUST R 296831
105 mg prefilled autoinjector/pen: AUST R 296830
This leaflet was prepared in June 2019.
Published by MIMS April 2021



 Subgroup analyses of the primary endpoints showed that romosozumab for 12 months followed by alendronate for 12 months demonstrated a consistent treatment effect, as shown by odds ratios that favoured romosozumab/alendronate over alendronate/alendronate in all subgroups of baseline characteristics examined including age, presence or absence of severe vertebral fracture at baseline, number of prevalent vertebral fractures at baseline, race, geographic region, baseline lumbar spine BMD T-score, baseline total hip/femoral neck BMD T-score, baseline BMI, FRAX score, and history of nonvertebral fracture at or after age 55.
Subgroup analyses of the primary endpoints showed that romosozumab for 12 months followed by alendronate for 12 months demonstrated a consistent treatment effect, as shown by odds ratios that favoured romosozumab/alendronate over alendronate/alendronate in all subgroups of baseline characteristics examined including age, presence or absence of severe vertebral fracture at baseline, number of prevalent vertebral fractures at baseline, race, geographic region, baseline lumbar spine BMD T-score, baseline total hip/femoral neck BMD T-score, baseline BMI, FRAX score, and history of nonvertebral fracture at or after age 55. Evenity reduced the incidence of major nonvertebral fractures compared to alendronate as early as Month 12 and through Month 24.
Evenity reduced the incidence of major nonvertebral fractures compared to alendronate as early as Month 12 and through Month 24. Among women with BMD assessed at baseline and every 6 months, Evenity significantly increased BMD at the lumbar spine, total hip, and femoral neck compared to alendronate alone through month 24. Following the double-blind period, in patients who transitioned from Evenity to alendronate and in patients who continued on alendronate, BMD continued to increase through month 24. The differences in BMD achieved at month 12 between patients who initially received Evenity or alendronate were maintained at month 24 (Figure 3).
Among women with BMD assessed at baseline and every 6 months, Evenity significantly increased BMD at the lumbar spine, total hip, and femoral neck compared to alendronate alone through month 24. Following the double-blind period, in patients who transitioned from Evenity to alendronate and in patients who continued on alendronate, BMD continued to increase through month 24. The differences in BMD achieved at month 12 between patients who initially received Evenity or alendronate were maintained at month 24 (Figure 3).



 The secondary endpoint of nonvertebral fracture did not reach statistical significance at month 12 (p = 0.096) or month 24 (p = 0.057) with Evenity treatment.
The secondary endpoint of nonvertebral fracture did not reach statistical significance at month 12 (p = 0.096) or month 24 (p = 0.057) with Evenity treatment. Among women with BMD assessed at baseline and every 6 months, Evenity significantly increased BMD at the lumbar spine, total hip, and femoral neck relative to placebo at 6 and 12 months. Following the transition from Evenity to denosumab, BMD continued to increase through month 24. In patients who transitioned from placebo to denosumab, BMD also increased with denosumab use. The differences in BMD achieved at month 12 between Evenity and placebo patients were overall maintained at month 24, when comparing patients who transitioned from Evenity to denosumab versus patients who transitioned from placebo-to-denosumab (Figure 6). Subgroup analyses of the primary endpoints indicated that the efficacy of romosozumab was consistent regardless of baseline characteristics examined.
Among women with BMD assessed at baseline and every 6 months, Evenity significantly increased BMD at the lumbar spine, total hip, and femoral neck relative to placebo at 6 and 12 months. Following the transition from Evenity to denosumab, BMD continued to increase through month 24. In patients who transitioned from placebo to denosumab, BMD also increased with denosumab use. The differences in BMD achieved at month 12 between Evenity and placebo patients were overall maintained at month 24, when comparing patients who transitioned from Evenity to denosumab versus patients who transitioned from placebo-to-denosumab (Figure 6). Subgroup analyses of the primary endpoints indicated that the efficacy of romosozumab was consistent regardless of baseline characteristics examined.



