WHAT IS IN THIS LEAFLET
This leaflet answers some common questions about Fentanyl Sandoz.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking this medicine against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
WHAT FENTANYL SANDOZ IS USED FOR
Fentanyl Sandoz is used for the long-term management of pain that is severe enough to require daily around-the-clock pain relievers, when other treatment options are not able to effectively manage your pain or you cannot tolerate them.
Fentanyl Sandoz is only used in people who have previously been using other opioid-based pain relief. Fentanyl Sandoz is not used to treat pain that you only have once in a while.
It contains the active ingredient fentanyl. Fentanyl belongs to a group of medicines called opioid analgesics.
It works by blocking the nerves that recognise pain messages from the body.
Each patch is applied onto the skin every three days (72 hours). The patch releases a continuous amount of fentanyl that is absorbed through the skin in contact with the patch.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine may be addictive.
This medicine is available only with a doctor's prescription.
BEFORE YOU USE FENTANYL SANDOZ
Warning
Opioids can be abused and misused, and you are at risk for opioid addiction, even if you take your dose as prescribed. Opioid addiction, abuse and misuse can lead to overdose and death.
Fentanyl Sandoz may become habit-forming causing mental and physical dependence. If abused, it may become less able to reduce pain.
Fentanyl Sandoz patches may be retrieved and abused or misused illegally. Please ensure that used patches are concealed and disposed of carefully. Return unused patches to the pharmacy (see Disposal at the end of this leaflet).
Keep used and unused patches where children cannot reach them. A patch may be tempting to a child. Accidental exposure or ingestion of used or unused Fentanyl Sandoz patches, particularly in children, may result in breathing difficulties, with slow or shallow breathing, that could lead to death. Improper use including Fentanyl Sandoz patches sticking to another person can be life-threatening.
Fentanyl Sandoz can cause sleep apnoea (stopping breathing from time to time while sleeping) which can lead to low levels of oxygen in the blood. Tell your doctor if you have a history of sleep apnoea or if anyone notices you stop breathing from time to time whilst sleeping.
Tolerance
As with all opioid analgesics, Fentanyl Sandoz may lead to tolerance with continued use. Tolerance means that the effect of the medicine may decrease and more is needed to product the same effect. Therefore, it is possible your doctor will prescribe a higher dose of Fentanyl Sandoz after some time to product the same result.
Opioids can cause a high level of the hormone 'prolactin' and low levels of sex hormones in the blood. The effects of these hormone changes may include:
- In females – unexpected production of breast milk; loss of menstrual periods
- In males – enlarged breasts; difficulty getting and keeping an erection (impotence)
- In females or males – decreased sex drive; infertility
Tell your doctor if you notice any of these symptoms. You may need blood tests, and your doctor may tell you to stop using Fentanyl Sandoz.
Opioids can cause a decreased level of hormones in the blood caused by a problem with the adrenal glands. The effects of these hormone changes may include:
- Nausea, vomiting, loss of appetite, feeling very tired and weak, feeling dizzy, or low blood pressure.
Tell your doctor if you notice any of these symptoms. You may need blood tests, and your doctor may tell you to stop using Fentanyl Sandoz.
Dependence and withdrawal
As with other opioids, your body may become used to you taking Fentanyl Sandoz after several days to weeks of continued use resulting in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking Fentanyl Sandoz suddenly. Some examples of withdrawal symptoms include feeling uneasy and unwell, restless, agitated, anxious, increased pain, sweating, chills, weakness, stomach cramps, problems sleeping, nausea, vomiting, diarrhoea, increased blood pressure, fast breathing and fast heartbeat.
Fentanyl Sandoz must be stopped by decreasing the dose gradually. Your doctor will tell you how to do this, and will regularly monitor and support any increase in pain or withdrawal symptoms.
Increased sensitivity to pain
Rarely, increasing the dose of this medicine can make you more sensitive to pain. If this happens you need to speak to your doctor about your treatment.
When you must not use it
Do not use this medicine if you have an allergy to:
- fentanyl, the active ingredient, or to any of the other ingredients listed at the end of this leaflet under Product description.
- any other similar medicines.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not use this medicine if you have or have had any of the following medical conditions:
- acute pain or pain following surgery
- mild or intermittent pain
- any lung condition or breathing difficulties.
Do not use any Fentanyl Sandoz strength greater than the 25 micrograms/hour patch or if you have not used opioid analgesics in the past. This is because you may be more likely to experience some of the side effects.
Do not use this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start using this medicine, talk to your doctor.
Before you start to use it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- liver or kidney disease
- lung disease or breathing difficulties
- sleep apnoea or if anyone notices you stop breathing from time to time while sleeping
- brain lesions or head injuries
- heart disease
- problems with your stomach or intestine such as bowel blockage
- problems with your immune system or medical conditions which lower your resistance to diseases.
Tell your doctor if you are pregnant, plan to become pregnant, are breastfeeding or wish to breastfeed. If you have not told your doctor or pharmacist about any of the above, tell them before you start using or are given Fentanyl Sandoz.
Tell your doctor if you (or a family member) have ever abused or been dependent on alcohol, prescription medicines, illegal drugs or have a mental illness.
Children
Fentanyl Sandoz should not be used in children under 12 years of age or in adolescents under 18 years of age who weigh less than 50 kg.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Fentanyl Sandoz may interfere with each other. These include:
- medicines that slow down your central nervous system, for example:
- medicines that makes you sleepy, reduce anxiety or decrease awareness, such as sedatives, hypnotics, sleeping tablets, tranquillisers (benzodiazepine)
- other opioid medicines
- medicines used for surgery (anaesthetics) and muscle relaxants
- antihistamines or allergy medicine that makes you drowsy
- gabapentinoid medicines used to treat epilepsy or nerve pain such as gabapentin and pregabalin
- alcohol, cannabis or some illegal drugs
Combination of these medicines with FENTANYL may increase the sedative effect of these drugs or slow down your ability to react, have decreased awareness, breathing difficulties with slow or shallow breathing, coma and death. A change in dose by your doctor may be required if FENTANYL is used with these medicines. - antidepressant medicines belonging to the class monoamine oxidase inhibitors (MAOIs).
Fentanyl Sandoz should not be used together with MAOIs as this may cause severe serotonin syndrome which is a potentially life-threatening condition. Signs and symptoms can include confusion, restlessness, fever, heavy sweating, fast or irregular heart rate, diarrhoea, uncoordinated movement of limbs or eyes, uncontrollable jerking of muscles, seizures and unconsciousness. Do not use Fentanyl Sandoz if you have taken a MAOI in the last 14 days. - other antidepressant medicines belonging to the class selective serotonin re-uptake inhibitors (SSRIs), serotonin norepinephrine re-uptake inhibitors (SNRIs) or tricyclic antidepressants (TCA) and nefazodone.
Combination of these medicines with Fentanyl Sandoz may increase the risk of serotonin syndrome, a potentially life-threatening condition. - medicines used to treat mental illness or psychotic conditions
- medicines used to relieve severe nausea and vomiting, such as phenothiazines
- certain antibiotics used to treat infections such as erythromycin, clarithromycin and troleandomycin
- certain medicines used to treat fungal infections such as fluconazole, ketoconazole, itraconazole and voriconazole.
- ritonavir and nelfinavir, medicines used to treat HIV infections. Do not take ritonavir or nelfinavir while using Fentanyl Sandoz, unless you are closely monitored by your doctor.
- if taking rifampicin, carbamazepine, phenobarbital or phenytoin, careful monitoring by your doctor and dose adjustment may be required.
- Idelalisib (cancer treatment),
- certain medicines that act on the heart and blood vessels such as calcium-channel blockers like verapamil, diltiazem and nicardipine.
- certain medicines used to treat arrhythmias such as amiodarone.
These medicines may be affected by Fentanyl Sandoz or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while using this medicine.
Effect of alcohol
You must not drink alcohol while using Fentanyl Sandoz since their combined effect may cause severe drowsiness, decreased awareness, breathing problems, coma and death.
HOW TO USE FENTANYL SANDOZ
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions, ask your doctor or pharmacist for help.
How to use the patch
Ask your doctor or pharmacist if you are unsure of the correct dose for you. They will tell you exactly which patch or patches to use.
Follow the instructions they give you. If you use the wrong dose, Fentanyl Sandoz may not work as well in controlling your pain.
Adults
Fentanyl Sandoz is available in five different patches, each with a different size and strength. Your doctor will decide which patch, or combination of patches, is suitable to control your pain. The lowest effective strength should be used.
Each patch is applied onto the skin and lasts for three days (72 hours). After three days, remove the patch and apply a new patch to the skin at a different place.
You should not use more than one patch at a time, unless your doctor authorises otherwise (for example to obtain a dose that cannot be achieved with a single patch). The old patch should be removed before the new patch is applied.
Children
Fentanyl Sandoz should not be used in children under 12 years of age or in adolescents under 18 years of age who weigh less than 50 kg.
Using it for the first time
The first patch may take up to a day to take effect after it is applied onto the skin. This is because fentanyl is slowly absorbed through the skin into the blood. Your doctor may prescribe additional medicines to control your pain for the first day.
Applying the patch
- Find an intact and hairless spot of skin on the upper part of your body or on your upper arm. Do not place the patch onto skin that is red, burnt or damaged.
The skin should be healthy and undamaged.
- Trim any excess hair with scissors. Do not shave the hair off since this may affect the skin. If you need to wash the skin before applying the patch, use clean water only. Do not use soap, oils or lotions.
The skin should be completely dry before applying the patch.
- Open the pouch and remove the Fentanyl Sandoz patch. Do not apply the patch if it looks damaged in any way. Never cut or divide the patch.
- Remove the protective film.
Avoid touching the adhesive side of the patch.
- Apply the patch to the skin and press with the palm of the hand for about 30 seconds. Make sure all of the patch is in contact with skin and the corners are stuck tightly.
- Wash your hands after applying or removing the patch.
You can now leave the patch on the skin for three days (72 hours).
You may have a bath, shower or swim.
Always write the date and time you applied the patch on the pack.
It will help you to use Fentanyl Sandoz correctly and remember when the next patch is due. Changing the patch
- After three days (72 hours), remove the patch.
- Fold the used patch in half so that the adhesive side sticks to itself. Wrap the folded patch and carefully dispose of it in the garbage.
- Apply a new patch straight away to a different area of the skin, following the steps under 'Applying the patch'.
If you do not understand the instructions provided with this medicine, ask your doctor or pharmacist for help.
If your pain continues, see your doctor who may prescribe additional medicines to help control the pain or change the dose of Fentanyl Sandoz. Your doctor may advise you initially to change the patch every two days (48 hours) instead of every three days (72 hours) to achieve adequate pain relief.
How long to use Fentanyl Sandoz
Continue using your medicine for as long as your doctor tells you.
If you forget to use it
Apply a new patch as soon as you remember, and continue to use Fentanyl Sandoz as you would normally.
If it is almost time for your next dose, skip the dose you missed and apply your next dose when you are meant to.
Do not use a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
If you use too much (overdose)
The most important sign of overdose is difficulty in breathing. If a person using Fentanyl Sandoz has abnormally slow or weak breathing, remove the patch(es) and phone triple zero (000) for an ambulance. Keep the person awake by talking to them or gently shaking them every now and then.
Also, telephone your doctor or the Poisons Information Centre (telephone Australia 13 11 26 or New Zealand 0800 POISON or 0800 764766) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have used too much Fentanyl Sandoz. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
You should also follow the above steps if someone other than you have accidentally used Fentanyl Sandoz that was prescribed for you. If someone takes and overdose they may experience one or more of the following symptoms:
- Slow, unusual or difficult breathing
- Drowsiness, dizziness or unconsciousness
- Slow or weak heart beat
- Nausea or vomiting
- Convulsions or fits
Other signs of overdose can also include problems with the ‘nervous system’ caused by damage to the white matter of the brain (known as toxic leukoencephalopathy).
When seeking medical attention, take this leaflet and remaining medicine with you to show the doctor. Also tell them about any other medicines or alcohol which have been taken.
WHILE YOU ARE USING FENTANYL SANDOZ
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are using Fentanyl Sandoz.
Tell any other doctors, dentists, and pharmacists who treat you that you are using this medicine.
If you plan to become pregnant while using this medicine, tell your doctor immediately, who will then decide whether you may use Fentanyl Sandoz. Fentanyl Sandoz should not be used during childbirth as the medicine can slow the breathing of the newborn child. Prolonged used of Fentanyl Sandoz during pregnancy can cause withdrawal symptoms in your newborn baby that could be life-threatening if not recognised and treated. If you are breastfeeding, you should not use Fentanyl Sandoz since it may be present in your milk. See your doctor.
If your pain continues or returns, see your doctor. You may need additional medicines to control the pain or a change in the strength of the Fentanyl Sandoz patch.
Tell your doctor if you develop a fever. At high temperatures, the amount of fentanyl absorbed by the skin increases. Your doctor may need to adjust your Fentanyl Sandoz dose.
Things you must not do
Do not expose the patch to direct heat from electric blankets, heat pads, heated water beds, heat or tanning lamps, intensive sunbathing, hot water bottles, long hot baths, saunas or hot spa baths while you are using Fentanyl Sandoz. Direct exposure to such heat may cause an increase in the amount of fentanyl absorbed by the skin, resulting in possible overdose and death.
Do not use Fentanyl Sandoz to treat any other complaints unless your doctor tells you to.
Do not give the patches to anyone else, even if they have the same condition as you.
Do not stop using your medicine or lower the dosage without checking with your doctor.
Your doctor may want you to gradually reduce the amount you are using before stopping completely. If you have been using Fentanyl Sandoz for a long period of time but stop using it suddenly without your doctor's advice, you may experience withdrawal symptoms such as:
- trouble sleeping, nervousness, restlessness, agitation or anxiety
- body aches, weakness or stomach cramps
- nausea, loss of appetite, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- water eyes, runny nose, chills or yawning
- increased sweating.
Seek your doctor's advice if you experience these symptoms.
Do not cut, break, chew, crush, dissolve, snort or inject Fentanyl Sandoz. This can result in serious side effects and death.
Things to be careful of
Be careful driving or operating machinery until you know how Fentanyl Sandoz affects you. This medicine may affect your alertness and cause dizziness in some people. Do not drive, operate machinery or do anything else that could be dangerous until your doctor tells you that it is safe.
Avoid drinking alcohol while you are taking this medicine. If you drink alcohol, drowsiness may be worse.
If the patch accidentally adheres to another person (for example a family member sharing the same bed) or is accidentally swallowed (for example by a child), remove the patch and phone triple zero (000). Do this even if there are no signs of discomfort or drowsiness.
Different brands of fentanyl patches may vary in size, shape, colour or adhesiveness.
DO NOT switch brands of fentanyl patches unless your doctor and pharmacist authorise it.
SIDE EFFECTS
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Fentanyl Sandoz.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- nausea, vomiting, constipation, dry mouth, diarrhoea, uncomfortable feeling in stomach or belching after eating, indigestion, blockage in the bowel, problem with the movement of food or drink through the food pipe
- low blood pressure, headache, weakness or dizziness, high blood pressure, being less alert or aware, or loss of consciousness
- blurred vision
- sleepiness, confusion, hallucinations, euphoria, depression, loss of appetite, anxiety, trouble sleeping, agitation, loss of memory, pins and needles
- stopping breathing from time to time whilst sleeping (sleep apnoea)
- skin rash (local redness and itch at the site of the patch is usually mild and resolves when the patch is removed)
- thinning or redness where the patch has been on the skin; ulcer (sore) where the patch has been on the skin
- unusual tiredness or weakness, feeling of body temperature change; swelling of feet, ankles and hands.
Tell your doctor immediately if you notice any of the following asyou may need urgent medical care:
- slow heart beat
- fast heart beat.
REMOVE the Fentanyl Sandoz patch and phone triple zero (000) or go to the Emergency Department at your nearest hospital if the following happens:
- breathing slows or weakens
- temporarily stopped breathing
- difficulty in breathing
- numbness; seizures or fits
- involuntary muscle movements including muscle spasms
- sweating or trouble urinating
- sudden life-threatening allergic reaction
- sudden signs of allergy such as rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body, shortness of breath, wheezing or trouble breathing.
Make sure that you are with someone who can keep you awake by talking to you or gently shaking you every now and then. The risk of breathing problems is higher if you:
- have an existing lung condition
- have a small physique
- are an elderly patient
- have kidney or liver disease
- are given a high Fentanyl Sandoz dose
- you have not used opioid pain relief before.
Your doctor will carefully select the most appropriate dose for you.
Nausea, vomiting, diarrhoea, anxiety and shivering may occur initially when you are switched from other opioid analgesics to Fentanyl Sandoz or if therapy is stopped suddenly. Tell your doctor if you experience any of these effects.
Other side effects not listed above such as sexual dysfunction and withdrawal symptoms may also occur in some people. Tell your doctor if you notice any other effects.
AFTER USING FENTANYL SANDOZ
Storage
Keep your medicine in the original sealed pouch.
If you take it out of its original container it may not keep well.
Keep your medicine in a cool dry place where the temperature stays below 25°C.
Store this medicine securely, where other people cannot access it. It may harm people who may take this medicine by accident, or intentionally when it has not been prescribed for them.
Do not store Fentanyl Sandoz or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car.
Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
The contents of Fentanyl Sandoz patches may be retrieved and abused or misused illegally.
Fold used patches so that the adhesive side of the patch sticks to itself, wrap and dispose of carefully in the garbage.
If your doctor tells you to stop using this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
PRODUCT DESCRIPTION
What it looks like
Fentanyl Sandoz patches are transparent rounded oblong patches. They are individually packed in sachets and are available in packs containing 5 sachets.
Ingredients
Active ingredients:
- Fentanyl Sandoz 12 mcg/h - 12 microgram fentanyl released per hour
- Fentanyl Sandoz 25 mcg/h - 25 microgram fentanyl released per hour
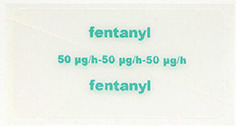
- Fentanyl Sandoz 50 mcg/h - 50 microgram fentanyl released per hour
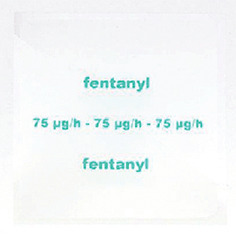
- Fentanyl Sandoz 75 mcg/h - 75 microgram fentanyl released per hour
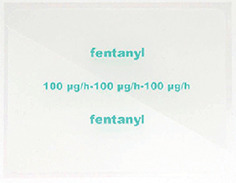
- Fentanyl Sandoz 100 mcg/h - 100 microgram fentanyl released per hour
Inactive ingredients:
- polyethylene terephthalate
- acrylic-vinylacetate copolymer
- siliconised polyethylene terephthalate.
This medicine does not contain lactose monohydrate, sucrose, gluten, tartrazine or any other azo dyes.
Supplier
Sandoz Pty Ltd
100 Pacific Highway
North Sydney, NSW 2060
Australia
Tel 1800 726 369
Sandoz New Zealand Limited
12 Madden Street
Auckland 1010
New Zealand
Tel 0800 726 369
This leaflet was revised in June 2024.
Australian Register Numbers
12 mcg/h transdermal patch: AUST R 152572
25 mcg/h transdermal patch: AUST R 152569
50 mcg/h transdermal patch: AUST R 152571
75 mcg/h transdermal patch: AUST R 152568
100 mcg/h transdermal patch: AUST R 152576
Published by MIMS August 2024





 The initial evaluation of the maximum analgesic effect of fentanyl should not be made before the patch has been worn for 24 hours. This is due to the gradual increase in serum fentanyl concentration in the 24 hours following initial application of the patch. Previous analgesic therapy should therefore be phased out gradually after the initial dose application until the analgesic efficacy with fentanyl is attained.
The initial evaluation of the maximum analgesic effect of fentanyl should not be made before the patch has been worn for 24 hours. This is due to the gradual increase in serum fentanyl concentration in the 24 hours following initial application of the patch. Previous analgesic therapy should therefore be phased out gradually after the initial dose application until the analgesic efficacy with fentanyl is attained. Adverse drug reactions not reported in Table 4 that were reported by ≥ 1% of fentanyl-treated subjects (N=1854) in 11 clinical trials of fentanyl used for the treatment of chronic malignant or nonmalignant pain (which includes trial FEN-EMA-1) are shown in Table 5. All subjects took at least one dose of fentanyl and provided safety data.
Adverse drug reactions not reported in Table 4 that were reported by ≥ 1% of fentanyl-treated subjects (N=1854) in 11 clinical trials of fentanyl used for the treatment of chronic malignant or nonmalignant pain (which includes trial FEN-EMA-1) are shown in Table 5. All subjects took at least one dose of fentanyl and provided safety data. Adverse drug reactions reported by < 1% of fentanyl treated subjects (N=1854) in the above clinical trial dataset are shown in Table 6.
Adverse drug reactions reported by < 1% of fentanyl treated subjects (N=1854) in the above clinical trial dataset are shown in Table 6.


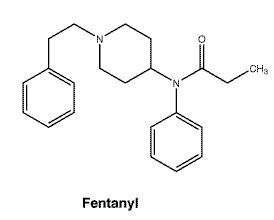 Chemical name: N-phenyl-N-[1- (2-phenylethyl)- 4-piperidinyl]propanamide.
Chemical name: N-phenyl-N-[1- (2-phenylethyl)- 4-piperidinyl]propanamide.