What is in this leaflet
This leaflet answers some common questions about FINASTERIDE ALPHAPHARM.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks. Your doctor has weighed the risks of you taking FINASTERIDE ALPHAPHARM against the benefits expected for you.
If you have any concerns about taking this medicine, talk to your doctor or pharmacist.
Keep this leaflet with your medicine.
You may need to read it again.
What FINASTERIDE ALPHAPHARM is used for
FINASTERIDE ALPHAPHARM is for use by men only.
FINASTERIDE ALPHAPHARM is used to treat a medical condition in men called benign prostatic hyperplasia or BPH. BPH is a condition where your prostate gland (which is near your bladder) has become bigger making it more difficult for you to pass urine. This can lead to symptoms such as:
- weak or interrupted stream of urine
- feeling that you cannot empty your bladder completely
- delay before you start to pass urine
- needing to pass urine often, especially at night
- feeling that you must pass urine right away
BPH occurs only in men and is common over the age of 50 years. In some men, BPH can lead to serious problems, including urinary tract infections and the sudden inability to pass urine at all. BPH can also lead to the need for surgery such as procedures to improve the flow of urine.
The prostate gland takes years to grow. Therefore, the symptoms of BPH take a long time to develop. FINASTERIDE ALPHAPHARM works by slowly reducing the size of your prostate gland. This may lead to gradual improvement in your urine flow and other symptoms over several months. FINASTERIDE ALPHAPHARM also helps reduce the risk of developing a sudden inability to pass urine (acute urinary retention) and the need for surgery. This may happen whether or not you notice any improvement or change in your symptoms.
Your doctor may have prescribed FINASTERIDE ALPHAPHARM for another reason.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Your doctor may have prescribed it for another reason.
This medicine is not addictive.
This medicine is available only with a doctor's prescription.
There is not enough information to recommend the use of this medicine for children.
Before you take FINASTERIDE ALPHAPHARM
When you must not take it
Do not take FINASTERIDE ALPHAPHARM if you have an allergy to:
- any medicine containing finasteride
- any of the ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering.
If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Women who are pregnant or may be pregnant must not take FINASTERIDE ALPHAPHARM or handle crushed or broken tablets.
If the active ingredient in FINASTERIDE ALPHAPHARM is absorbed after swallowing the tablet or through the skin by a woman who is pregnant with a male baby, it may cause the male baby to be born with abnormalities of the sex organs. FINASTERIDE ALPHAPHARM tablets are coated and will prevent contact with the active ingredient during normal handling, provided the tablets are not broken or crushed.
If a pregnant woman swallows FINASTERIDE ALPHAPHARM or handles crushed or broken tablets, her doctor must be consulted immediately.
Do not give this medicine to children or women.
The condition for which FINASTERIDE ALPHAPHARM is prescribed occurs only in men.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any medical conditions.
If you have not told your doctor about any of the above, tell them before you start taking FINASTERIDE ALPHAPHARM.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from a pharmacy, supermarket or health food shop.
Some medicines may affect the way other medicines work. However, FINASTERIDE ALPHAPHARM has not been shown to interfere with other medicines.
Driving and operating machinery
FINASTERIDE ALPHAPHARM should not affect your ability to drive or operate machinery.
How to take FINASTERIDE ALPHAPHARM
Follow all directions given to you by your doctor and pharmacist carefully.
They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box/bottle, ask your doctor or pharmacist for help.
How much to take
Take FINASTERIDE ALPHAPHARM only when prescribed by your doctor.
The usual dose in men is one tablet taken once each day.
How to take it
Swallow FINASTERIDE ALPHAPHARM with a full glass of water.
When to take it
Take FINASTERIDE ALPHAPHARM once a day at about the same time each day.
Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take this medicine before or after food.
How long to take it
FINASTERIDE ALPHAPHARM shrinks the prostate gland slowly. Therefore, you may need to take FINASTERIDE ALPHAPHARM for 6 months or longer to see whether it helps you. If it does help your symptoms, you may need to take FINASTERIDE ALPHAPHARM every day.
Continue taking your medicine for as long as your doctor tells you to.
If you stop taking the medicine the prostate gland is likely to grow again.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your tablet as you would normally.
Do not take a double dose to make up for the dose you missed.
This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much FINASTERIDE ALPHAPHARM. Do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention.
While you are taking FINASTERIDE ALPHAPHARM
Things you must do
Go to your doctor for regular checkups, including a physical check for prostate cancer once a year if you are over 50.
While BPH is not cancer and does not lead to cancer, the two conditions can exist at the same time. FINASTERIDE ALPHAPHARM is used for BPH not prostate cancer.
If you are having a blood test to measure your PSA (prostate-specific antigen) levels, tell your doctor you are taking FINASTERIDE ALPHAPHARM.
FINASTERIDE ALPHAPHARM can affect the results of this test.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking FINASTERIDE ALPHAPHARM.
Tell any other doctors, dentists and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine.
It may affect other medicines used during surgery.
Things you must not do
Do not take FINASTERIDE ALPHAPHARM to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or change the dosage without checking with your doctor.
If you stop taking it suddenly, your condition may worsen or you may have unwanted side effects.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking FINASTERIDE ALPHAPHARM.
FINASTERIDE ALPHAPHARM helps most men with BPH, but it may have unwanted side effects in a few people.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by the following list of side effects.
You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- impotence (inability to have an erection) that continues after stopping FINASTERIDE ALPHAPHARM.
- less desire for sex that continues after stopping FINASTERIDE ALPHAPHARM.
- changes or problems with ejaculation that continued after stopping the medication, such as decreased amount of semen released during sex (this decrease does not appear to interfere with normal sexual function).
- male infertility and/or poor quality of semen have been reported infrequently. Improvement in the quality of semen has been reported after stopping finasteride.
The above list includes the more common side effects of your medicine. They are usually mild and short-lived. In some cases, these side effects disappeared while the patient continued to take FINASTERIDE ALPHAPHARM. If symptoms persisted, they usually resolved on stopping the tablets.
Tell your doctor as soon as possible if you notice any of the following:
- breast swelling and/or tenderness In rare cases, male breast cancer has been reported
- breast lumps, pain or discharge from the nipples
- skin rash, itchiness
- hives or nettle rash (pinkish, itchy swellings on the skin)
- testicle pain
- depression (feelings of severe sadness and unworthiness).
The above list includes uncommon side effects that have been reported with FINASTERIDE ALPHAPHARM.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- swelling of the lips, tongue, throat or face
The above list includes very serious side effects that may cause difficulty in swallowing or breathing. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After taking FINASTERIDE ALPHAPHARM
Storage
Keep your tablets in the pack until it is time to take them.
If you take the tablets out of the pack they may not keep well.
Keep your tablets in a cool dry place where the temperature stays below 25°C.
Do not store FINASTERIDE ALPHAPHARM or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car.
Heat and dampness can destroy some medicines.
Keep it where children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
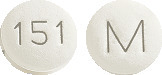
FINASTERIDE ALPHAPHARM 5 mg tablets is white film-coated, round, biconvex, beveled-edge tablet debossed with M on one side of the tablet and 151 on the other side.
Each blister pack contains 30 tablets.
Ingredients
FINASTERIDE ALPHAPHARM contains 5 mg of finasteride as the active ingredient.
It also contains the following inactive ingredients:
- povidone
- docusate sodium 50% in macrogol 400
- lactose anhydrous
- pregelatinised maize starch
- croscarmellose sodium
- microcrystalline cellulose
- magnesium stearate
- colloidal anhydrous silica
- Opadry II complete film-coating system Y-B22-7719 (ARTG no. 2578)
FINASTERIDE ALPHAPHARM contains lactose.
FINASTERIDE ALPHAPHARM does not contain gluten, sucrose, tartrazide or any other azo dyes.
Supplier
FINASTERIDE ALPHAPHARM is supplied in Australia by:
Alphapharm Pty Limited
(ABN 93 002 359 739)
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
Phone: (02) 9298 3999
www.alphapharm.com.au
Australian Registration Numbers:
FINASTERIDE ALPHAPHARM 5 mg blister pack: AUST R 186305
This leaflet was prepared on 5 December 2014.
FINASTERIDE ALPHAPHARM_cmi\Dec14/00