WHAT IS IN THIS LEAFLET
This leaflet answers some common questions about Flosix.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking this medicine against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor, nurse or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
WHAT FLOSIX IS USED FOR
Flosix is for use by men only.
This medicine is used for the relief of lower urinary tract symptoms (LUTS) associated with a condition called benign prostatic hyperplasia (BPH). BPH is NOT prostate cancer. BPH is a condition where your prostate gland (which is near your bladder) has become bigger making it more difficult for you to pass urine. This can lead to symptoms such as:
- weak or interrupted stream of urine
- delay before you start to pass urine, and you have to strain to do so.
- feeling that you cannot empty your bladder completely
- may dribble at the end of passing urine.
- needing to pass urine often, especially at night
- feeling that you must pass urine right away.
BPH occurs only in men and is common over the age of 50 years. In some men, BPH can lead to serious problems, including urinary tract infections and the sudden inability to pass urine at all.
This medicine contains the active ingredient tamsulosin hydrochloride. Tamsulosin hydrochloride belongs to a group of medicines called alpha-blockers.
Flosix works by helping relax the smooth muscles in the prostate, in that way it improves the flow of urine, thus relieving the pain when passing urine.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is available only with a doctor's prescription.
BEFORE YOU TAKE FLOSIX
When you must not take it
Do not take this medicine if you have an allergy to:
- tamsulosin hydrochloride, the active ingredient, or to any of the other ingredients listed at the end of this leaflet under Product Description
- any other alpha-blockers
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not take this medicine if you are a woman or a child. Flosix is for use in men only.
Do not take this medicine if you have or have had any of the following medical conditions:
- severe liver problems
- severe kidney problems
- feeling dizzy or light-headedness when you sit up or stand abruptly. This is a common symptom of a condition called orthostatic hypotension which is an excessive decrease in blood pressure that occurs when a person stands up, resulting in reduced blood flow to the brain and dizziness or fainting.
- are taking other medication which relaxes the smooth muscle of blood vessels (some of the tradenames are Minipress, Prasig and Hytrin.
Do not take this medicine if you are taking another alpha-blocker.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Use Flosix only if your doctor has prescribed it for you.
All medicines have benefits and risks. In deciding to prescribe Flosix for you, your doctor has weighed the risk of taking Flosix against the benefit it is expected to have for you.
Your doctor has prescribed Flosix for BPH. Flosix does not treat prostate cancer. BPH and prostate cancer may have similar symptoms. A man can have prostate cancer and BPH at the same time. You should be checked for prostate cancer before you start Flosix. It is recommended that men be checked for prostate cancer once a year, from 50 years of age onwards. These checks should continue while you are on Flosix.
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- dizziness and lightheadedness (low blood pressure)
- heart attack or chest pain, feeling of tightness, pressure or heaviness in the chest (angina pectoris) during the last six months
- are allergic to Flosix (or any of the ingredients (see 'Ingredients')
- have high, or low blood pressure, or your blood pressure is controlled by medication
- have had ejaculation problems
- are suffering from any other illness
- have had allergies to sulfa or any other medications
- have had or planning to have cataract or glaucoma surgery
- you previously have taken any other similar medicine in the same class as Flosix.
If you have not told your doctor about any of the above, tell him/her before you start taking Flosix.
Your doctor will discuss the risks and benefits of using Flosix.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Flosix may interfere with each other. These include:
- cimetidine, a medicine generally used to treat stomach ulcers or reflux
- diuretic medicines used to treat high blood pressure such as frusemide
- H2 antagonists or H2 blockers, a class of medicines used to treat stomach ulcer, reflux or heartburn such as cimetidine.
These medicines may be affected by Flosix or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
HOW TO TAKE FLOSIX
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions, ask your doctor or pharmacist for help.
How much to take
The standard dose for this medicine is one tablet daily.
Patients with severe liver problems should not take these tablets.
Patients with severe kidney problems should not take these tablets.
Ask your doctor or pharmacist if you are unsure of the correct dose for you. They will tell you exactly how much to take.
Follow the instructions they give you.
If you take the wrong dose, Flosix may not work as well and your problem may not improve.
How to take it
Swallow the tablet whole with a full glass of water.
Do not break, chew, crunch or dissolve the tablets. These tablets have a special coating to stop them dissolving until they have gone through the stomach and into the intestines, where they can start to work. If you chew them, the coating is destroyed.
When to take Flosix
Take your medicine at about the same time each day.
Flosix can be taken on an empty stomach, or before, with or after food.
How long to take Flosix
Continue taking your medicine for as long as your doctor tells you. This medicine helps to control your condition, but does not cure it.
If you forget to take it
Take your dose as soon as you remember, and continue to take it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
If you miss a whole day, continue to take your normal daily dose the next day.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
Do not take a double dose to make up for the dose that you missed.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone Australia 13 11 26 or New Zealand 0800 POISON or 088 764766) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much Flosix. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of an overdose may include vomiting, diarrhoea. Low blood pressure may also occur, leading to dizziness, lightheadedness and fainting. Medical attention should be sought immediately.
WHILE YOU ARE TAKING FLOSIX
Things you must do
Be sure to keep all of your doctor's appointments so that your progress can be checked.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Flosix.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
Tell your doctor if, for any reason, you have not taken your medicine exactly as prescribed.
Otherwise your doctor may think it is not working effectively and change your treatment unnecessarily.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you are going to have eye surgery for cataracts or glaucoma, please tell your surgeon that you are taking or have taken Flosix.
(See 'Side Effects')
Things you must not do
Do not take Flosix to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Things to be careful of
Be careful driving or operating machinery while taking Flosix. This medicine may cause dizziness in some people. If you feel dizzy do not drive, operate machinery or do anything else that could be dangerous.
If you feel dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
SIDE EFFECTS
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Flosix.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- changes or problems with ejaculation (retrograde ejaculation). Retrograde ejaculation is a condition described as abnormal ejaculation in which semen is ejaculated backward into the bladder rather than out through the penis. Retrograde ejaculation is painless.
- dizziness
- constipation, diarrhoea, nausea, vomiting, dry mouth
- insomnia
- headache
- fast heart beats
- unusual weakness
- blocked nose
- nose bleed
- skin rash, itchiness
- hives (pinkish, itchy swellings on the skin)
- fainting
- blurred vision or visual impairment
- inflammation and blistering of the skin and/or mucous membranes of the lips, eyes, mouth, nasal passages or genitals
Flosix can occasionally cause people to feel faint and dizzy. You should get up slowly from the sitting or lying position to reduce the risk of dizziness or lightheadedness. If you do feel faint on standing up, you should lie down for a short while. If the dizziness persists you should contact your doctor. You must not drive a car or operate machinery if you feel dizzy.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- persistent painful erection of the penis which occurs without sexual arousal
- skin rash, itchiness or swelling of the face, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing.
These are symptoms of an allergic reaction. You may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything else that is making you feel unwell. Other side effects not listed above may also occur in some people.
Tell your doctor if you notice any other effects or if the unwanted effects are particularly bothersome.
You should always tell your doctor about any problems you have whilst taking Flosix.
If you are having an operation on your eyes because of cataracts or glaucoma and are already taking or have taken Flosix, the pupil may dilate poorly and the iris (the coloured part of the eye) may become floppy during the procedure. This can be managed if your surgeon knows before carrying out the operation. If you are going to have eye surgery for cataracts or glaucoma, please tell your surgeon that you are taking or have taken Flosix.
AFTER TAKING FLOSIX
Storage
Keep your medicine in the original container.
If you take it out of its original container it may not keep well.
Keep your medicine in a cool dry place where the temperature stays below 25°C.
Do not store Flosix or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car.
Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Note the expiry date on the pack. Do not use after this expiry date.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
PRODUCT DESCRIPTION
What it looks like
Flosix 400 mcg - brown, round, biconvex modified release tablets with debossing '0.4' on one side and 'SZ' on the other side.
Available in blister packs of 10 and 30 tablets.
Ingredients
Active ingredients:
- Flosix 400 mcg - 400 mcg tamsulosin hydrochloride.
Inactive ingredients:
Excipients core:
- cellulose - microcrystalline
- hydroxypropyl cellulose
- polyethylene oxide
- butylated hydroxytoluene
- magnesium stearate
- silica - colloidal anhydrous.
Excipients coating:
- hypromellose
- hyprolose
- macrogol 400
- titanium dioxide
- purified talc
- quinoline yellow
- cochineal
- iron oxide black.
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Supplier
Sandoz Pty Ltd
ABN 60 075 449 553
54 Waterloo Road
Macquarie Park NSW 2113
Australia
Tel: 1800 726 369
This leaflet was revised in July 2020.
Australian Register Number:
Flosix 400 mcg modified release tablets: AUST R 336015
Published by MIMS June 2021

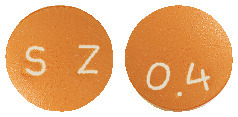
 The following treatment-related adverse events were reported from clinical trials, where Common is ≥ 1% and < 10%; Uncommon is ≥ 0.1% and < 1%; Rare is ≥ 0.01% and < 0.1%; and Very rare is < 0.01%.
The following treatment-related adverse events were reported from clinical trials, where Common is ≥ 1% and < 10%; Uncommon is ≥ 0.1% and < 1%; Rare is ≥ 0.01% and < 0.1%; and Very rare is < 0.01%.

 As a result of the prolonged release characteristic of tamsulosin, the trough concentrations - at steady state, of tamsulosin hydrochloride in plasma amount to approximately 40% of the peak plasma concentrations, under fasted and fed conditions.
As a result of the prolonged release characteristic of tamsulosin, the trough concentrations - at steady state, of tamsulosin hydrochloride in plasma amount to approximately 40% of the peak plasma concentrations, under fasted and fed conditions. Chemical formula: 5-[(2R)-2-[[2-(2-Ethoxyphenoxy) ethyl]amino]propyl]-2 methoxybenzenesulfonamide hydrochloride.
Chemical formula: 5-[(2R)-2-[[2-(2-Ethoxyphenoxy) ethyl]amino]propyl]-2 methoxybenzenesulfonamide hydrochloride.