What is in this leaflet
This leaflet answers some common questions about FLUTIFORM pressurised inhalation ("inhaler").
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using FLUTIFORM inhaler against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What FLUTIFORM inhaler is used for
FLUTIFORM inhaler is used to help with asthma in people who need regular treatment.
Asthma is a disease where the lining of the lungs become inflamed (red and swollen), making it difficult to breathe. This may be due to an allergy to house dust mites, smoke, air-borne pollution or other irritants. Symptoms of asthma include shortness of breath, wheezing, chest tightness and cough.
FLUTIFORM inhaler contains two different medicines called fluticasone propionate and formoterol (eformoterol) fumarate dihydrate.
Fluticasone propionate belongs to a group of medicines known as corticosteroids, frequently called 'steroids'. They are not 'anabolic steroids' which are sometimes misused by athletes. Corticosteroids help to reduce swelling and inflammation in the lungs.
Formoterol (Eformoterol) fumarate dihydrate belongs to a group of medicines known as long-acting beta-2-agonists. Beta-2-agonists help the airways in your lungs to stay open, making it easier for you to breathe.
When used together regularly, fluticasone propionate and formoterol (eformoterol) fumarate dihydrate help to control your asthma symptoms and prevent asthma attacks. FLUTIFORM inhaler is known as a 'preventer'. It does not work if you are already having an asthma attack, i.e. you are already wheezing and breathless. You will need to use a fast acting 'reliever' medicine if this happens.
Your doctor may have prescribed it for another reason.
Ask your doctor if you have any questions about why it has been prescribed for you.
FLUTIFORM inhaler is not addictive.
This medicine is only available with a doctor's prescription.
Before you use FLUTIFORM inhaler
When you must not use it
Do not use FLUTIFORM inhaler if you are allergic to fluticasone propionate, formoterol (eformoterol) fumarate dihydrate or any of the other ingredients listed at the end of this leaflet. Symptoms of an allergic reaction may include wheezing, swelling of the lips/mouth or other parts of the body, difficulty in breathing, lumpy rash ("hives") or fainting.
Do not give FLUTIFORM inhaler to a child under 12 years, unless directed to by the child's doctor. FLUTIFORM inhaler is not recommended for use in children under 12 years. The safety and efficacy of FLUTIFORM inhaler have not been assessed in children under 12 years of age.
Do not use FLUTIFORM inhaler after the expiry date (EXP) printed on the pack. If you use it after the expiry date has passed, it may not work very well.
Do not use it if the packaging is torn or shows signs of tampering.
If you are not sure whether you should be using FLUTIFORM inhaler, talk to your doctor or pharmacist.
Before you start to use it
Tell your doctor or pharmacist if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor or pharmacist if you have or have had any of the following medical conditions:
- heart problems or disease, an irregular heartbeat or if you have been told that your heart trace is abnormal
- an abnormal bulging of a blood vessel wall (aneurysm)
- liver problems
- diabetes
- high blood pressure
- glaucoma
- an overactive thyroid gland
- adrenal glands not working properly
- tumour of the adrenal gland
- low blood levels of potassium.
Tell your doctor or pharmacist if you currently have an infection. If you use FLUTIFORM inhaler while you have an infection, the medicine may hide some of the signs of an infection. This may make you think, mistakenly, that you are better or that it is not serious.
Tell your doctor or pharmacist if you have or have had tuberculosis (TB).
Tell your doctor or pharmacist if you are pregnant or intend to become pregnant. Your doctor or pharmacist will discuss the possible risks and benefits of using FLUTIFORM inhaler during pregnancy.
Tell your doctor or pharmacist if you are breastfeeding or plan to breastfeed. Your doctor or pharmacist will discuss the possible risks and benefits of using FLUTIFORM inhaler during breastfeeding.
Tell your doctor if you are going to have an operation, or are feeling stressed. You may need additional steroid medicine during these times.
If you have not told your doctor or pharmacist about any of the above, tell them before you use FLUTIFORM inhaler.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and FLUTIFORM inhaler may interfere with each other. These include:
- medicines to treat depression or mood/mental disorders (such as monoamine oxidase inhibitors, tricyclic antidepressants, phenothiazines), or you have taken any of these types of medicines in the last two weeks
- medicines known as beta-blockers, which may be used to treat high blood pressure or glaucoma including those that are eye drops
- medicines used to treat high blood pressure or fluid build-up
- medicines used to treat heart failure (such as digoxin)
- medicines used to treat abnormal heart rhythms (such as quinidine, disopyramide, procainamide)
- medicines used to treat fungal infections (such as ketoconazole, itraconazole)
- medicines used to treat viral infections, including HIV (such as ritonavir, atazanavir, indinavir, nelfinavir, saquinavir, cobicistat)
- medicines used to treat bacterial infections (antibiotics, such as clarithromycin, telithromycin, furazolidone)
- medicines used to treat allergies, hayfever, coughs, colds and runny nose
- other medicines containing steroids
- certain other medicines used to treat asthma or breathing conditions (such as theophylline or aminophylline)
- medicines used for weight reduction purposes
- medicine to treat Hodgkin's disease (procarbazine)
- medicine to treat Parkinson's disease (levodopa)
- medicine used to treat an underactive thyroid gland (levothyroxine)
- medicine to induce labour (oxytocin)
- alcohol.
If you are going to have an operation under a general anaesthetic, please tell the doctor at the hospital that you are using this inhaler.
These medicines may be affected by or may affect FLUTIFORM inhaler. If you are taking these medicines, talk to your doctor or pharmacist who will advise you on what to do. You may need to use different amounts of your medicines, or take different medicines.
FLUTIFORM inhaler contains a very small amount of sodium cromoglycate, however patients who are currently using cromoglycate (to treat conditions such as asthma, allergic rhinitis and allergic conjunctivitis) should continue their normal medication.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while using FLUTIFORM inhaler.
How to use FLUTIFORM inhaler
How much to use
Follow the instructions given to you by your doctor and pharmacist.
Your doctor or pharmacist will tell you exactly how much to use.
Adults and adolescents 12 years and older:
Your doctor will decide what dose and what strength of FLUTIFORM inhaler you should use, depending on the severity of your asthma.
The usual dose is two inhalations twice a day; two in the morning and two in the evening.
Adolescents should not use the highest strength inhaler (FLUTIFORM inhaler 250 micrograms/10 micrograms).
How to use it
The instructions for using FLUTIFORM inhaler are given in this leaflet. You should check with your doctor or pharmacist if you are not sure how to use FLUTIFORM inhaler.
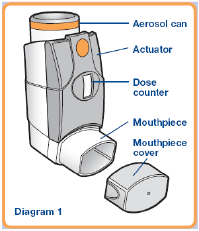
The medicine is contained in an aerosol can (canister) (see Diagram 1) which sits inside a plastic dispenser (also known as an actuator). The actuator has a colour-coded dose counter which displays the remaining actuations in increments of 10, down to 60 doses, and in increments of 5 thereafter. The numbers are separated by a dot. The indicator is initially green (120 to 45 puffs remaining), then yellow (45 to 25 puffs remaining), and then red (25 to 0 puffs remaining). When the indicator turns red, you should be contacting your doctor or returning to your pharmacy to have a replacement dispensed. Do not remove the canister from the actuator as this can interfere with the dose counter.
Before you use your inhaler for the first time, or if it has not been used for more than 3 days, it needs to be 'primed' to ensure it works properly and gives you the correct dose.
To 'prime' the inhaler:
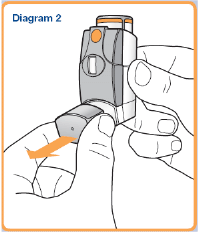
- Remove the mouthpiece cover and shake the inhaler well.
- Spray away from your face by pressing down on the canister. This step should be performed 4 times.
Also, in case of freezing conditions, the inhaler must be primed. Allow 30 minutes warm up to room temperature, and then spray away from your face 2 times before use.
Using your inhaler:
The medicine in FLUTIFORM inhaler must only be breathed into your lungs through the mouth.
- Remove the mouthpiece cover and check that your mouthpiece is clean and free from any dust. The inhaler should be shaken immediately before releasing each puff.
- Sit upright or stand. Breathe out slowly and as deeply as possible.
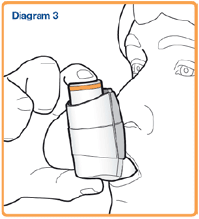
- Hold your inhaler upright (as shown in Diagram 3) and put the mouthpiece in your mouth with your lips around it. Do not bite the mouthpiece.
- Breathe in slowly and deeply through your mouth and, at the same time, press down on the canister to release one puff. (as shown in Diagram 4)
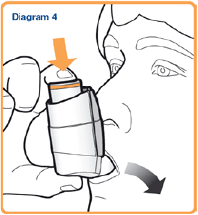
- Hold your breath for as long as possible. Finally, remove the inhaler from your mouth and breathe out slowly. Do not breathe out into the inhaler.
- Keep the inhaler in a vertical position for about half a minute and then shake the inhaler prior to slowly repeating Steps 2 to 5.
- Replace the mouthpiece cover.
Do not perform steps 2 to 5 too quickly.
If you see a 'mist' from the top of the inhaler or around your mouth when you use your inhaler, then you may not have inhaled your medicine properly. Have another dose by repeating from Step 2 above.
Always rinse your mouth out, gargle with water or brush your teeth after you have used your inhaler and spit out the residue. This may help prevent you developing a hoarse voice or sore mouth, throat or tongue.
If you have weak hands, it may be easier to hold the inhaler in both hands placing both index fingers on the canister and both thumbs on the base of the inhaler.
If you have difficulty using your inhaler, your doctor may also recommend a spacer, a device to help you use your medicine properly.
Your doctor or pharmacist will advise you how to use your spacer device with your inhaler. It should also come with instructions for use and care.
Caring for your inhaler:
It is important to clean your inhaler weekly.
To clean your inhaler:
- Remove the mouthpiece cover.
- Do not remove the canister from the actuator as this can interfere with the dose counter.
- Wipe the inside and outside of the mouthpiece and the outside of the actuator with a clean, dry cloth or tissue.
- Replace the mouthpiece cover.
DO NOT PUT THE METAL CANISTER INTO WATER.
If you have any difficulties or do not understand the instructions, talk to your doctor or pharmacist.
When to use it
FLUTIFORM inhaler is used to help people with asthma who need regular treatment.
It is very important that you use your FLUTIFORM inhaler every day, twice a day (morning and night). This will help prevent asthma symptoms throughout the day and night.
Do not use FLUTIFORM inhaler to relieve a sudden attack of breathlessness or wheezing. You will need a fast-acting 'reliever' medicine if this happens. You should carry your 'reliever' medicine with you at all times.
If you feel you are getting more breathless or wheezy more often than normal, or you are using your fast-acting 'reliever' medicine more than usual, you should talk to your doctor.
How long to use it
You must use your FLUTIFORM inhaler every day (twice a day). Do not stop using it, even if you feel better, unless your doctor tells you.
If you forget to use it
If you forget to have a dose, use it as soon as you remember. However, if it is almost time for your next dose, skip the missed dose, then use your next dose when you are meant to.
Do not use a double dose to make up for the dose you have missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to use your medicine, ask your pharmacist for hints. For example, use your medicine at the same time each morning and evening such as 8 a.m. and 8 p.m.
If you use too much (overdose)
Immediately telephone your doctor, or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at your nearest hospital, if you think you or anyone else may have used too much FLUTIFORM inhaler.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Keep telephone numbers for these places handy.
If you use too much of FLUTIFORM inhaler than you should, you may experience severe chest pain (angina), high or low blood pressure, a headache, muscle cramps, difficulty in sleeping, nervousness, a dry mouth, a loss of appetite, seizures, fits or convulsions. You may feel shaky, light headed, faint, tired, sick or generally unwell. You may also notice changes in the rate of your heart beat. There may be changes to potassium and sugar levels in your blood. You may also suffer from symptoms such as abdominal pain, being sick, weight loss or decreased level of consciousness (which could make you feel drowsy or confused).
If you have used more than the prescribed dose for a long period of time, you should talk to your doctor or pharmacist for advice.
When seeking medical attention, take this leaflet and your inhaler with you to show the doctor.
While you are using FLUTIFORM inhaler
Things you must do
Use FLUTIFORM inhaler exactly as your doctor has prescribed.
Before you start on a new medicine, remind your doctor and pharmacist that you are using FLUTIFORM inhaler.
Tell any other doctors, dentists and pharmacists who treat you that you are using this medicine.
If you have an Asthma Action Plan that you have agreed with your doctor, follow it closely at all times.
Tell your doctor if you are going to have an operation, have had an accident or have a severe infection.
If your body is stressed, you may need other medicines.
If you become pregnant while using this medicine, tell your doctor immediately.
Keep all of your doctor's appointments so that your progress can be checked.
Tell your doctor as soon as possible if:
- you are getting more breathless or wheezy
- you are using your 'reliever' medicine more than usual
- your 'reliever' medicine does not help your breathing as much as usual
- the effect of your 'reliever' medicine does not last as long as usual
- you need more doses of your 'reliever' medicine than usual to get relief.
Tell your doctor if, for any reason, you have not used your medicine exactly as prescribed. Otherwise your doctor may think that your medicine was not effective and change your treatment unnecessarily.
If you have used more than the prescribed dose for a long time, you should talk to your doctor for advice. This is because large doses may reduce the amount of steroid hormones produced by your body.
Make sure you have enough doses of FLUTIFORM inhaler with you to last over weekends and holidays.
Things you must not do
Do not take any other medicines for your breathing problems without checking with your doctor.
Do not use FLUTIFORM inhaler to treat any other complaint unless your doctor tells you to.
Do not give FLUTIFORM inhaler to anyone else, even if they have the same condition as you.
Do not stop using FLUTIFORM inhaler or change the dosage without checking with your doctor. If you need to stop using FLUTIFORM inhaler, your doctor will tell you how to do this. Usually your dose will be gradually reduced each day, before stopping the medicine completely. This is to reduce the risk of triggering an asthma attack.
Do not use FLUTIFORM inhaler to treat an acute asthma attack.
Things to be careful of
Be careful driving or operating machinery until you know how FLUTIFORM inhaler affects you. FLUTIFORM inhaler is unlikely to affect your ability to drive or operate machinery. However, as with many other medicines, some people may experience blurred vision, dizziness or spinning sensation, light-headedness or tiredness.
Tell your doctor or pharmacist if you notice that you have a sore mouth, throat or tongue, sore, creamy-yellow, raised patches in the mouth or a hoarse voice. These problems are less likely to happen if you use a spacer or rinse your mouth out each time after using FLUTIFORM inhaler.
Side effects
All medicines may have some unwanted side effects. Sometimes they are serious but most of the time they are not. You may need medical treatment if you get some of the side effects. Your doctor has weighed the risks of FLUTIFORM inhaler against the benefits they expect it will have for you.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using FLUTIFORM inhaler.
Ask your doctor or pharmacist to answer any questions you may have.
If you get any side effects, do not stop using FLUTIFORM inhaler without first talking to your doctor or pharmacist.
Tell your doctor if you notice any of the following and they worry you:
- headaches
- dizziness or spinning sensation
- shaking or tremors
- changes in taste
- hoarse voice
- dry mouth
- irritated throat
- raised patches in the mouth
- sore mouth, throat or tongue
- inflammation of the sinuses, runny or blocked nose
- diarrhoea
- nausea, vomiting
- indigestion
- muscle spasms or pain
- abnormal dreams or difficulty in sleeping
- agitation or anxiety
- tiredness or unusual weakness
- swelling of legs and ankles
- an increase in the amount of sugar in your blood
- blurred vision or other visual disturbances.
Tell your doctor or pharmacist immediately if you notice any of the following:
- irregular heart beat or palpitations
- chest pain
- bruising
- feeling nervous or restless
- mood changes
- worsening of your asthma (shortness of breath, coughing).
The above list includes serious side effects that may require medical attention.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- you have an allergic reaction: shortness of breath, wheezing, shallow or difficult breathing; swelling of the tongue, throat, face, lips or other parts of the body; rash, itching or hives on the skin.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using FLUTIFORM inhaler
Storage
Keep your inhaler in a cool, dry place, where the temperature stays below 25°C.
Do not keep your inhaler in the refrigerator or freezer.
Do not store it or any other medicine in the bathroom, near a sink or on a window sill. Do not leave it in the car on hot days. Heat and damp can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Do not use the inhaler if it has been removed from the foil pouch for over 3 months, or if the dose counter reads "0" (whichever comes first).
WARNING: The canister contains a pressurised liquid. Do not puncture, break or burn the can even when apparently empty.
Disposal
If your doctor tells you to stop using the inhaler, the inhaler has been removed from the foil pouch for over 3 months, the dose counter reads "0", or if the inhaler has passed their expiry date, ask your pharmacist how to dispose of medicines no longer required.
Product description
What it looks like
FLUTIFORM® inhalers are pressurised metered-dose inhalers. The inhalers are small canisters containing a white to off-white liquid suspension fitted with a metering valve. The canisters are inserted into plastic dispensers (actuators) fitted with a dust cap and a dose indicator which indicates the number of doses remaining. Each inhaler contains 120 actuations (puffs).
They are available in three strengths:
- 50 micrograms/5 micrograms
- 125 micrograms/5 micrograms
- 250 micrograms/10 micrograms.
FLUTIFORM® inhalers are packed in foil pouches within a box containing one inhaler.
Ingredients
Active ingredients:
Each dose contains:
- 50 micrograms of fluticasone propionate/5 micrograms of formoterol (eformoterol) fumarate dihydrate
- 125 micrograms of fluticasone propionate/5 micrograms of formoterol (eformoterol) fumarate dihydrate
- 250 micrograms of fluticasone propionate/ 10 micrograms of formoterol (eformoterol) fumarate dihydrate.
Inactive ingredients:
- sodium cromoglycate
- absolute ethanol
- apaflurane (HFA 227, propellant).
HFA 227 is a CFC-free propellant.
This medicine does not contain lactose, sucrose, gluten or tartrazine.
Supplier
FLUTIFORM® pressurised inhalation is supplied in Australia by:
Mundipharma Pty Limited
ABN 87 081 322 509
10 Carrington Street
Sydney, NSW, 2000
Phone: 1800 188 009
® FLUTIFORM is a registered trade mark of Jagotec AG used under licence by Mundipharma Pty Limited.
This leaflet was updated in September 2022
Australian Registration Numbers for FLUTIFORM® pressurised inhalation:
50 micrograms/5 micrograms: AUST R 177869
125 micrograms/5 micrograms: AUST R 177873
250 micrograms/10 micrograms: AUST R 177875
Published by MIMS November 2022


 As with other inhalation therapy, paradoxical bronchospasm may occur with an immediate increase in wheezing and shortness of breath after dosing. Paradoxical bronchospasm responds to a rapid acting inhaled bronchodilator and should be treated immediately. Inhalation therapy should be discontinued immediately, the patient assessed and alternative therapy instituted if necessary.
As with other inhalation therapy, paradoxical bronchospasm may occur with an immediate increase in wheezing and shortness of breath after dosing. Paradoxical bronchospasm responds to a rapid acting inhaled bronchodilator and should be treated immediately. Inhalation therapy should be discontinued immediately, the patient assessed and alternative therapy instituted if necessary.



