1 Name of Medicine
Inactivated trivalent influenza vaccine, split virion (influenza virus haemagglutinin).
2 Qualitative and Quantitative Composition
Fluzone for intramuscular injection is an inactivated influenza virus vaccine, prepared from influenza viruses propagated in embryonated chicken eggs. The virus-containing allantoic fluid is harvested and inactivated with formaldehyde. Influenza virus is concentrated and purified in a linear sucrose density gradient solution using a continuous flow centrifuge. The virus is then chemically disrupted using a non-ionic surfactant, octoxinol-9 (Triton X-100), producing a "split virus". The split virus is further purified and then suspended in sodium phosphate-buffered isotonic sodium chloride solution. Antigens from the three strains included in the vaccine are produced separately and then combined to make the trivalent formulation. See Table 1.
 Fluzone contains 45 microgram hemagglutinin (HA) per 0.5 mL dose, in the recommended ratio of 15 microgram HA per strain.
Fluzone contains 45 microgram hemagglutinin (HA) per 0.5 mL dose, in the recommended ratio of 15 microgram HA per strain.
The type and amount of viral antigens contained in Fluzone conform to the annual requirements of the Australian Influenza Vaccine Committee (AIVC) and the World Health Organization (WHO) recommendations for the season.
Neither antibiotics nor preservative are used during manufacture.
Fluzone is presented in prefilled syringes that are not made with natural rubber latex.
3 Pharmaceutical Form
Sterile aqueous suspension for injection.
Fluzone suspension for injection is clear and slightly opalescent in colour.
4.1 Therapeutic Indications
Fluzone is indicated for active immunisation of influenza disease caused by influenza A subtype viruses and type B viruses contained in the vaccine. Fluzone is indicated for use in adults and children 6 months and older.
Fluzone should be given in accordance with the national recommendation as per the current Immunisation Handbook.
4.2 Dose and Method of Administration
Inspect Fluzone visually for particulate matter and/or discolouration prior to administration. If any of these defects or conditions exist, the vaccine should not be administered.
Before administering a dose of vaccine, shake the prefilled syringe.
The syringe is for single use only and must not be reused. Discard any remaining unused contents.
For needle size and length, refer to the national recommendations as per the current Immunisation Handbook.
Fluzone should not be mixed with any other vaccine in the same syringe or vial.
Administration should be carried out by the intramuscular route. The dose and schedule are as follows (see Table 2):
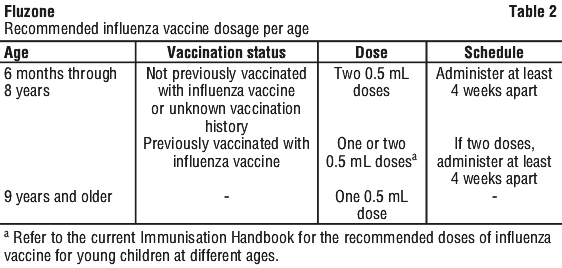 The preferred sites for intramuscular injection are the anterolateral aspect of the thigh in infants 6 months through 11 months of age, the anterolateral aspect of the thigh (or the deltoid muscle if muscle mass is adequate) in persons ≥ 12 months through 35 months of age, or the deltoid muscle in persons > 36 months of age.
The preferred sites for intramuscular injection are the anterolateral aspect of the thigh in infants 6 months through 11 months of age, the anterolateral aspect of the thigh (or the deltoid muscle if muscle mass is adequate) in persons ≥ 12 months through 35 months of age, or the deltoid muscle in persons > 36 months of age.
The vaccine should be administered into healthy well-developed muscle and should not be injected into the gluteal region where there may be a risk of local neural, vascular and tissue injury.
4.3 Contraindications
Fluzone should not be administered to anyone with a known systemic hypersensitivity reaction, such as anaphylaxis, after previous administration of any influenza vaccine or to any component of the vaccine (see Section 2 Qualitative and Quantitative Composition; Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
Do not administer intravenously.
Hypersensitivity.
Prior to any vaccine injection, all known precautions should be taken to prevent hypersensitivity reactions. This includes a review of the individual's prior vaccination history with respect to possible hypersensitivity to the vaccine or similar vaccines.
As each dose may contain traces of formaldehyde and octoxinol-9 which are used during vaccine production, caution should be exercised when the vaccine is administered to individuals with hypersensitivity to either one of these products.
As with all injectable vaccines, appropriate medical treatment and supervision should always be readily available in case of a rare anaphylactic event following administration of the vaccine especially for individuals who have a known allergy to egg protein.
Febrile or acute disease.
Vaccination should be postponed in case of moderate or severe acute or febrile disease with or without fever but a mild disease with low-grade fever is usually not a reason to postpone vaccination.
Neurological disorders.
Patients with a history of Guillain-Barré syndrome (GBS) with an onset related in time to influenza vaccination may be at increased risk of again developing GBS, but whether vaccination specifically might increase the risk for recurrence is unknown. Because patients with a history of GBS have an increased likelihood of again developing the syndrome, the chance of them coincidentally developing the syndrome following influenza vaccination may be higher than in individuals with no history of GBS. If GBS has occurred within 6 weeks following previous influenza vaccination, the decision to give Fluzone should be based on careful consideration of the potential benefits and risks. Refer to the current Immunisation Handbook for further details.
Immunosuppressive treatments or conditions.
The immunogenicity of Fluzone may be reduced by immunosuppressive treatment or in individuals with immune deficiency syndromes. Vaccination of individuals with chronic immunodeficiencies is recommended even though the antibody response may be limited.
Protection.
Influenza virus is remarkably unpredictable in that significant antigenic changes may occur from time to time. It is known that influenza vaccines, as now constituted, are not effective against all possible strains of influenza virus. Protection is limited to those strains of virus from which the vaccine is prepared or to closely related strains.
As with any vaccine, vaccination with Fluzone may not protect 100% of susceptible individuals.
Bleeding disorder.
Because any intramuscular injection can cause an injection-site haematoma in individuals with any bleeding disorder, such as haemophilia or thrombocytopenia, or in individuals on anticoagulant therapy, intramuscular injections with Fluzone should not be administered to such individuals unless the potential benefits outweigh the risk of administration. If the decision is made to administer any product by intramuscular injection to such individuals, it should be given with caution, with steps taken to avoid the risk of haematoma formation following injection.
Syncope.
Syncope (fainting) has been reported following vaccination with Fluzone. Procedures should be in place to prevent falling injury and manage syncopal reactions.
Use in the elderly.
Safety and immunogenicity of Fluzone were evaluated in adults 65 years of age and older (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Antibody responses to Fluzone are lower in individuals ≥ 65 years of age than in younger adults.
Adults aged ≥ 65 years are strongly recommended to receive either high-dose or adjuvanted influenza vaccine every year. Refer to the current Immunisation Handbook.
Paediatric use.
Safety and effectiveness of Fluzone in children below the age of 6 months have not been established. Children in Study GRC88 aged between 6 and < 12 months were required to be born at full term of pregnancy (≥ 37 weeks) and/or with a birth weight ≥ 2.5 kg.
Preterm infants (< 37 weeks gestation) are strongly recommended to receive influenza vaccine each year, starting at ≥ 6 months of age. Refer to the current Immunisation Handbook.
Effects on laboratory tests.
Interference of Fluzone with laboratory and/or diagnostic tests has not been studied.
Following influenza vaccination, false positive results in serology tests using the ELISA method to detect antibodies against HIV1, hepatitis C, and especially HTLV1 have been observed. An appropriate Western Blot test should be used to confirm or disprove the results of the ELISA test. The transient false-positive reactions could be due to a non-specific IgM response induced by the vaccine.4.5 Interactions with Other Medicines and Other Forms of Interactions
Fluzone should not be mixed with any other vaccine in the same syringe or vial.
Adult target groups for influenza and pneumococcal polysaccharide vaccination overlap considerably. Clinical studies show that influenza vaccine may be administered with pneumococcal vaccine using separate syringes at different sites.
No studies regarding the simultaneous administration of inactivated influenza vaccine and other childhood vaccines have been conducted. According to ACIP, inactivated vaccines usually do not interfere with the immune response to other inactivated or live vaccines and children at high risk for influenza-related complications, including those aged 6-23 months, can receive influenza vaccine at the same time they receive other routine vaccinations.
If Fluzone is to be given at the same time as another injectable vaccine(s), the vaccine(s) should always be administered at different injection sites. Refer to the current Immunisation Handbook for further details.
Although inhibition of hepatic clearance of phenytoin, theophylline and warfarin has been reported after influenza vaccination, subsequent studies have not shown any evidence of undesirable effects related to this phenomenon.
If the vaccine is used in individuals deficient in producing antibodies due to immunosuppressive therapy, the expected immune response may not be obtained.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Fluzone has not been evaluated for the possible effects on human fertility. A reproductive toxicity study in which female rabbits were administered a Fluzone QIV formulation, 24 and 10 days before insemination showed no effects on female fertility.
(Category A)
Animal reproductive studies have not been conducted with Fluzone. A developmental and reproductive toxicity study has been performed in female rabbits at a dose approximately 20 times the human dose (on a mg/kg basis) and has revealed no evidence of impaired female fertility or harm to the fetus due to Fluzone QIV formulation. There are, however, no adequate and well-controlled studies in pregnant women.
In the developmental and reproductive toxicity study, female rabbits were administered a Fluzone QIV formulation or control saline (each 0.5 mL/dose) by intramuscular injection 24 and 10 days before insemination, and on Days 6, 12, and 27 of gestation. The administration of a Fluzone QIV formulation did not result in systemic maternal toxicity (no adverse clinical signs and no change in body weight or food consumption). In addition, no adverse effects on pregnancy, parturition, lactation, or embryo-fetal or pre-weaning development were observed. There were no vaccine-related fetal malformations or other evidence of teratogenesis noted in this study.
It is also not known whether Fluzone can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity.
Data from studies involving large numbers of women (> 80,000) vaccinated during pregnancy with inactivated influenza vaccines do not indicate any adverse fetal and maternal outcomes attributable to the vaccine. Fluzone should be given to a pregnant woman following an assessment of the risks and benefits. Because of the known adverse consequences of influenza infection in pregnant women, health authorities recommend vaccination of pregnant women.
It is not known whether Fluzone is excreted in human milk hence, caution should be used when administering vaccine to breastfeeding women.4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive or use machines have been performed.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials experience.
Because clinical trials are conducted under widely varying conditions, adverse event rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trial of another vaccine and may not reflect the rates observed in practice.
Children 6 months through 8 years of age.
In a multi-centre study conducted in the US (FID07), children 6 months through 35 months of age received two 0.25 mL doses of Fluzone, and children 3 years through 8 years of age received two 0.5 mL doses of Fluzone, irrespective of previous influenza vaccination history. The two doses (year 2006-2007 formulation) were administered 26 to 30 days apart. The safety analysis set included 97 children 6 months through 35 months of age and 163 children 3 years through 8 years of age. Table 3 and Table 4 summarise solicited injection site reactions and systemic adverse events reported within 7 days post-vaccination via diary cards.
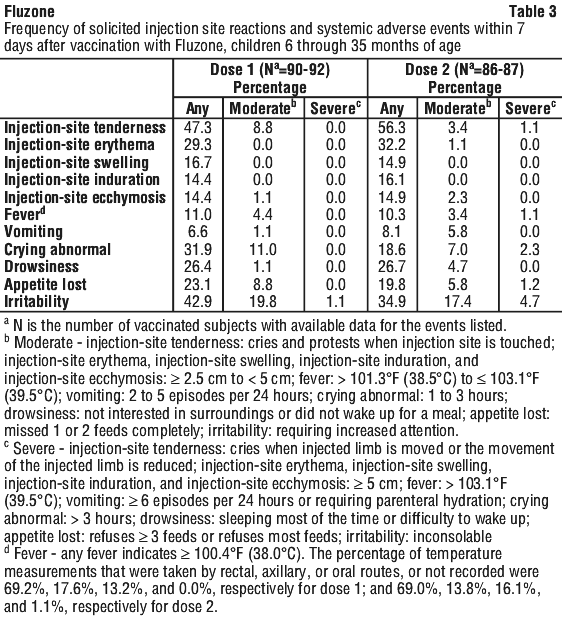
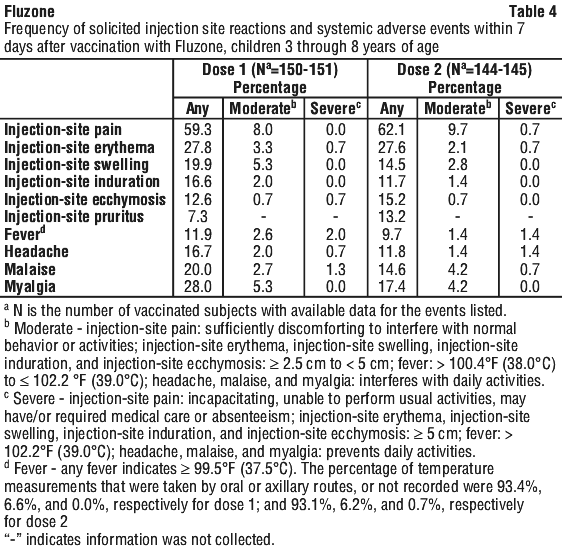 The most frequently reported solicited injection site reactions within 7 days following dose 1 and dose 2 vaccination were tenderness (47% and 56%) and redness (29% and 32%) in children 6 months through 35 months; or pain (59% and 62%) and erythema (27% and 27%) in children 3 years through 8 years. The most frequently reported solicited systemic adverse events within 7 days following dose 1 and dose 2 vaccination were irritability (43% and 35%), crying abnormal (32% and 19%), and drowsiness (26% and 27%) in children 6 months through 35 months; or myalgia (28% and 17%), malaise (20% and 15%), and headache (17% and 12%) in children 3 years through 8 years.
The most frequently reported solicited injection site reactions within 7 days following dose 1 and dose 2 vaccination were tenderness (47% and 56%) and redness (29% and 32%) in children 6 months through 35 months; or pain (59% and 62%) and erythema (27% and 27%) in children 3 years through 8 years. The most frequently reported solicited systemic adverse events within 7 days following dose 1 and dose 2 vaccination were irritability (43% and 35%), crying abnormal (32% and 19%), and drowsiness (26% and 27%) in children 6 months through 35 months; or myalgia (28% and 17%), malaise (20% and 15%), and headache (17% and 12%) in children 3 years through 8 years.
During the period from the first vaccination through 6 months following the second vaccination, there were no serious adverse events considered to be caused by vaccination and no deaths reported in this study.
Children 6 months through 35 months of age.
Safety data collected in the GRC88 study conducted using a Quadrivalent Influenza Vaccine (QIV) formulation can support the 0.5-mL dose of the TIV in children 6 to 35 months.
Removal of B/Yamagata strain from the Fluzone QIV is unlikely to impact the efficacy of A/H1N1, A/H3N2 and B/Victoria.
GRC88 was an observer-blind multi-centre study conducted in the US, including healthy children age between 6 and 35 months. Infants less than 12 months of age were of gestational age ≥ 37 weeks and/or birth weight ≥ 2.5 kg. Participants were randomly assigned to receive a Quadrivalent Influenza Vaccine dose of 0.25 mL (Group 1) or 0.5 mL (Group 2). For children requiring a second dose, the same dose was administered 4 weeks after the first.
Participants who received at least one dose of study vaccine were included in the safety analysis set: Group 1 n = 949; Group 2 n = 992.
Table 5 summarises solicited injection-site and systemic adverse reactions reported within 7 days post-vaccination via diary cards for the 0.25 mL and 0.5 mL volumes of Fluzone Quadrivalent in children 6 months through 35 months of age.
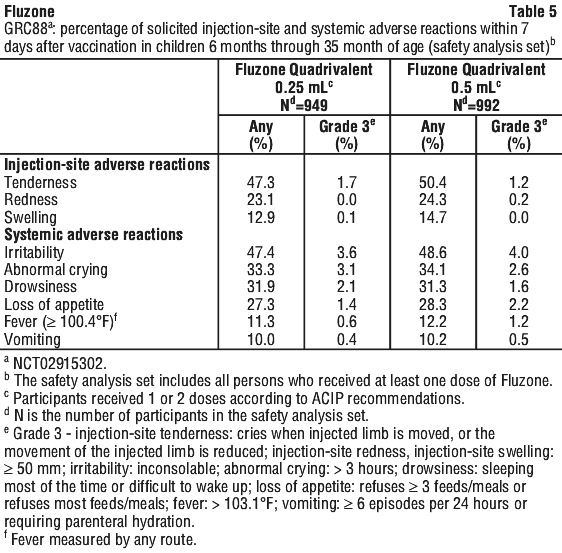 The difference in fever rate (Group 2 minus Group 1) was 0.84% (95% CI: -2.13%; 3.80%), meeting the prespecified non-inferiority criterion (upper limit of the 2-sided 95% CI of the difference in fever rates < 5%). Participants were monitored for unsolicited adverse events and SAEs during the 28 days following vaccination. Unsolicited non-serious adverse events were reported in 417 (44%) participants in Group 1 and 394 (40%) participants in Group 2. The most commonly reported unsolicited non-serious adverse events in both groups were cough and rhinorrhoea. Ten SAEs were reported during the 28-day follow-up period: 5 (0.5%) in Group 1 and 5 (0.5%) in Group 2.
The difference in fever rate (Group 2 minus Group 1) was 0.84% (95% CI: -2.13%; 3.80%), meeting the prespecified non-inferiority criterion (upper limit of the 2-sided 95% CI of the difference in fever rates < 5%). Participants were monitored for unsolicited adverse events and SAEs during the 28 days following vaccination. Unsolicited non-serious adverse events were reported in 417 (44%) participants in Group 1 and 394 (40%) participants in Group 2. The most commonly reported unsolicited non-serious adverse events in both groups were cough and rhinorrhoea. Ten SAEs were reported during the 28-day follow-up period: 5 (0.5%) in Group 1 and 5 (0.5%) in Group 2.
Adults.
Adults 18 through 64 years of age received Fluzone (year 2008-2009 formulation) in a multi-centre trial conducted in the US (FID31). The safety analysis set included 1421 Fluzone recipients. Table 6 summarises solicited injection-site reactions and systemic adverse events reported within 7 days post-vaccination via diary cards.
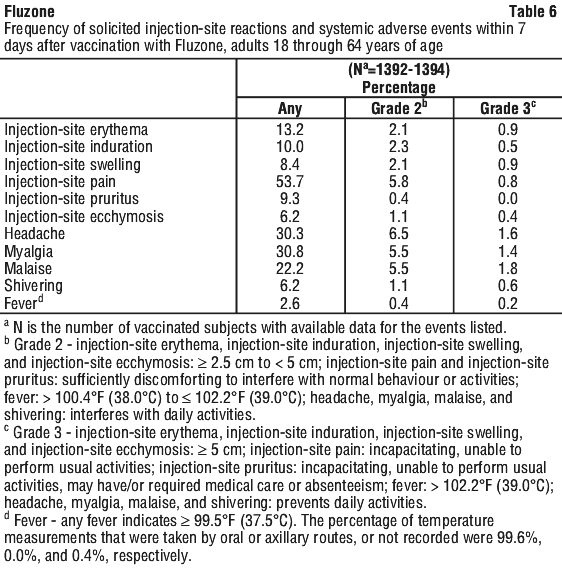 The most frequently reported solicited injection-site reaction was pain (54%); the most frequently reported solicited systemic adverse events were myalgia (31%), headache (30%), and malaise (22%).
The most frequently reported solicited injection-site reaction was pain (54%); the most frequently reported solicited systemic adverse events were myalgia (31%), headache (30%), and malaise (22%).
Within 28 days and six months post-vaccination, a serious adverse event was reported by 5 (0.4%) and 20 (1.4%) Fluzone recipients, respectively. No serious adverse event was considered to be caused by vaccination. No deaths were reported during the 6 months post-vaccination.
Geriatric adults.
Adults 65 years of age and older received Fluzone (year 2006-2007 formulation) in a multi-centre, double-blind trial conducted in the US (FIM05). The safety analysis set included 1260 Fluzone recipients.
Table 7 summarises solicited injection-site reactions and systemic adverse events reported within 7 days post-vaccination via diary cards. Onset was usually within the first 3 days after vaccination and a majority of the reactions resolved within 3 days.
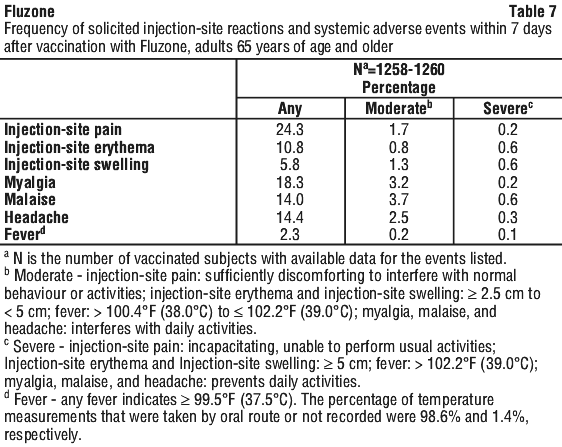 The most frequently reported solicited injection-site reaction was pain (24%); the most frequently reported solicited systemic adverse events were myalgia (18%), headache (14%), and malaise (14%).
The most frequently reported solicited injection-site reaction was pain (24%); the most frequently reported solicited systemic adverse events were myalgia (18%), headache (14%), and malaise (14%).
Within 6 months post-vaccination, 93 (7.4%) Fluzone recipients experienced a serious adverse event (N=1260). No deaths were reported within 28 days post-vaccination. A total of 7 deaths were reported during the period Day 29-180 post-vaccination: 7 (0.6%) among Fluzone recipients (N=1260). The majority of these participants had a medical history of cardiac, hepatic, neoplastic, renal, and/or respiratory diseases. No deaths were considered to be caused by vaccination.
Adverse reactions from post-marketing surveillance.
Based on spontaneous reporting, the following adverse events have been reported following commercial use of Fluzone. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to vaccine exposure. Adverse events were included based on one or more of the following factors: severity, frequency of reporting, or strength of evidence for a causal relationship to Fluzone.
Blood and lymphatic system disorders.
Thrombocytopenia, lymphadenopathy.
Immune system disorders.
Anaphylaxis, other allergic/hypersensitivity reactions (including urticaria, angioedema).
Eye disorders.
Ocular hyperemia.
Nervous system disorders.
Guillain-Barré syndrome (GBS), convulsions, febrile convulsions, myelitis (including encephalomyelitis and transverse myelitis), facial palsy (Bell's palsy), optic neuritis/neuropathy, brachial neuritis, syncope (shortly after vaccination), dizziness, paresthesia.
Vascular disorders.
Vasculitis, vasodilation/flushing.
Respiratory, thoracic and mediastinal disorders.
Dyspnea, pharyngitis, rhinitis, cough, wheezing, throat tightness.
Skin and subcutaneous tissue disorders.
Stevens-Johnson syndrome.
General disorders and administration site conditions.
Pruritus, asthenia/fatigue, pain in extremities, chest pain.
Gastrointestinal disorders.
Vomiting.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Influenza vaccine, ATC code: J07BB.
Mechanism of action.
Influenza illness and its complications follow infection with influenza viruses. Global surveillance of influenza identifies yearly antigenic variants. For example, since 1977, antigenic variants of influenza A (H1N1 and H3N2) viruses and influenza B viruses have been in global circulation. Specific levels of hemagglutination inhibition (HAI) antibody titre post-vaccination with inactivated influenza virus vaccines have not been correlated with protection from influenza virus infection. In some human studies, antibody titres ≥ 1:40 have been associated with protection from influenza illness in up to 50% of subjects.
Antibodies against one influenza virus type or subtype confer limited or no protection against another. Furthermore, antibodies to one antigenic variant of influenza virus might not protect against a new antigenic variant of the same type or subtype. Frequent development of antigenic variants through antigenic drift is the virological basis for seasonal epidemics and the reason for the usual change of one or more strains in each year's influenza vaccine. Therefore, influenza vaccines are standardised to contain the hemagglutinins of influenza virus strains representing the influenza viruses likely to be circulating in the upcoming season.
Clinical trials.
Immunogenicity of Fluzone in children 6 months to 8 years of age.
In a multi-centre study conducted in the US (FID07), 68 children 6 months through 35 months of age given two 0.25 mL doses of Fluzone and 120 children 3 years through 8 years of age given two 0.5 mL doses of Fluzone were included in the per-protocol analysis set. The two doses (year 2006-2007 formulation) were administered 26 to 30 days apart. Females accounted for 42.6% of the participants in the 6 months through 35 months age group and 53.3% of the participants in the 3 years through 8 years age group. Most participants in the 6 months through 35 months and 3 years through 8 years age groups, respectively, were Caucasian (70.6% and 79.2%), followed by Hispanic (19.1% and 13.3%), and Black (7.4% and 4.2%).
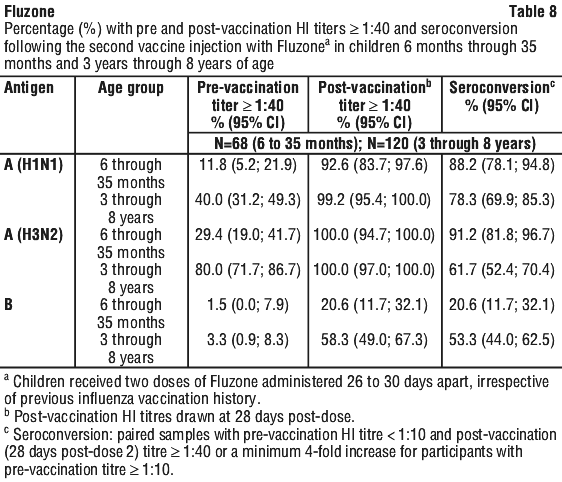
The percentage of participants who received influenza vaccination during the previous influenza season was 54.4% for the 6 months through 35 months age group and 27.5% for the 3 years through 8 years age group. Table 8 shows seroconversion rates and the percentage of participants with an HI titre ≥ 1:40 pre-vaccination and one month following the second dose of Fluzone.
Blood samples were obtained before dose 1 and 28 days post-dose 2. The proportion of children 6 months through 35 months of age with HI titres ≥ 1:40 increased from 12% pre-vaccination to 93% post-vaccination against A/H1N1, from 29% pre-vaccination to 100% post-vaccination against A/H3N2, and from 1.5% pre-vaccination to 21% post-vaccination against type B.
The proportion of children 3 years through 8 years of age with HI titres ≥ 1:40 increased from 40% pre-vaccination to 99% post-vaccination against A/H1N1, from 80% pre-vaccination to 100% post-vaccination against A/H3N2, and from 3.3% pre-vaccination to 58% post-vaccination against type B.
Seroconversion was defined for paired samples with a pre-vaccination HI titre < 1:10 and post-vaccination titre ≥ 1:40 or a minimum 4-fold increase for participants with a pre-vaccination titre ≥ 1:10. For children 6 months through 35 months of age, seroconversion rates against A/H1N1, A/H3N2, and B were 88%, 91%, and 21%, respectively. For children 3 years through 8 years of age, seroconversion rates were 78%, 62%, and 53%, respectively.
Immunogenicity of a 0.5 mL dose of quadrivalent influenza vaccine (QIV) in children 6 months to 35 months.
GRC88 (NCT02915302 see http://clinicaltrials.gov) was a randomised, observer-blind, multi-centre study including healthy children age between 6 and 35 months, randomly assigned to receive a Quadrivalent Influenza Vaccine dose of 0.25 mL (Group 1) or 0.5 mL (Group 2). For children requiring a second dose, the same dose was administered 4 weeks after the first. The per-protocol immunogenicity set included 715 participants in Group 1 and 745 in Group 2. Immunogenicity was assessed 28 - 35 days after the final vaccination. (Also see Section 4.8 Adverse Effects (Undesirable Effects)).
HI antibody GMTs following the 0.5 mL dose of Quadrivalent Influenza Vaccine were non-inferior to those following a 0.25 mL dose for all four strains, based on pre-specified criteria of lower limit of the 2-sided 95% CI of the ratio of GMTs (GMT 0.5 mL/GMT 0.25 mL) > 0.667.
A/H1N1 strain: GMT ratio = 1.42(95% CI: 1.16; 1.74).
A/H3N2 GMT ratio: = 1.48 (95% CI: 1.21; 1.82).
B Victoria lineage strain GMT ratio: = 1.33 (95% CI: 1.09; 1.62).
B Yamagata lineage strain GMT ratio: = 1.41 (95% CI: 1.17; 1.70).
Seroconversion rates 28 days following final vaccination with a 0.5 mL dose of Quadrivalent Influenza Vaccine were non-inferior to those following a 0.25 mL dose for all four strains, based on pre-specified criteria of lower limit of the 2-sided 95% CI of the difference in seroconversion rates > -10% (GMT 0.5 mL minus GMT 0.25 mL) > -10%).
A/H1N1 strain: difference in SC rates = 4.6% (95% CI: 0.4%; 9.6%).
A/H3N2 strain: difference in SC rates = 5.1% (95% CI: 0.4%; 9.8%).
B Victoria lineage strain: difference in SC rates = 1.3% (95% CI: -2.9%; 5.6%).
For the B Yamagata lineage strain: difference in SC rates = 2.6% (95% CI: -1.4%; 6.5%).
Immunogenicity of Fluzone in adults.
Adults 18 through 64 years of age received Fluzone (year 2008-2009 formulation) in a multi-centre trial conducted in the US (FID31). For immunogenicity analyses, there were 1287 participants who received Fluzone in the per-protocol analysis set. There were fewer males (35.8%) than females. The mean age was 42.6 years (ranged from 18.2 through 65.0 years). Most participants were Caucasian (80.0%), followed by Hispanic (11.0%), and Black (6.3%). Table 8 shows seroconversion rates at 28 days following vaccination and the percentage of participants with an HI titre ≥ 1:40 prior to vaccination and 28 days following vaccination.
Blood samples were obtained pre-vaccination and 28 days post-vaccination. The proportion participants with HI titres ≥ 1:40 increased from 39% pre-vaccination to 92% post-vaccination against A/H1N1, from 34% pre-vaccination to 91% post-vaccination against A/H3N2, and from 41% pre-vaccination to 89% post-vaccination against type B. Seroconversion rates (see Table 9) against A/H1N1, A/H3N2, and B were 61%, 75%, and 54%, respectively.

Immunogenicity of Fluzone in adults ≥ 65 years of age.
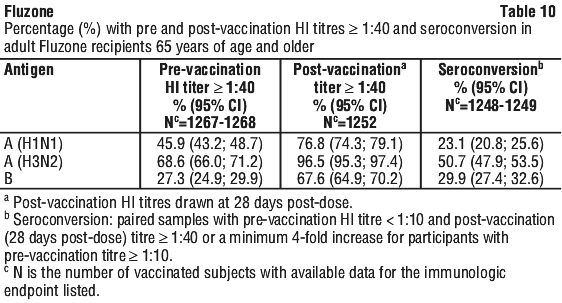
Adults 65 years of age and older received Fluzone (year 2006-2007 formulation) in a multi-centre trial conducted in the US (FIM05). For immunogenicity analyses, there were 1,275 participants who received Fluzone in the immunogenicity analysis set. Females accounted for 54.7% of participants. The mean age was 72.9 years (ranged from 65 through 94 years of age); 36% of participants were 75 years of age or older. Most participants were Caucasian (92.9%), followed by Hispanic (3.7%), and Black (2.7%). Table 10 shows seroconversion rates at 28 days following vaccination and the percentage of participants with an HI titre ≥ 1:40 prior to vaccination and 28 days following vaccination.
Blood samples were obtained pre-vaccination and 28 days post-vaccination. The proportion participants with HI titres ≥ 1:40 increased from 46% pre-vaccination to 77% post-vaccination against A/H1N1, from 69% pre-vaccination to 97% post-vaccination against A/H3N2, and from 27% pre-vaccination to 68% post-vaccination against type B. Seroconversion rates (see Table 10) against A/H1N1, A/H3N2, and B were 23%, 51%, and 30%, respectively.
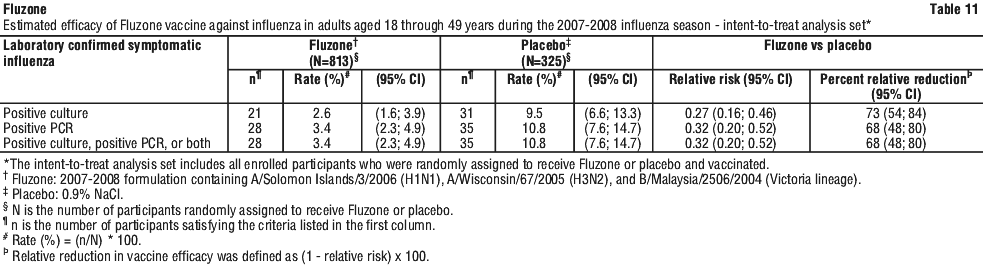
Efficacy of Fluzone in adults.
GRC70 was a randomised, double-blind, placebo-controlled study conducted in a single US center during the 2007-2008 influenza season. Participants received one dose of either Fluzone vaccine (N = 813), an active comparator (N = 814), or placebo (N = 325). The intent-to-treat analysis set included 1138 healthy adults who received Fluzone or placebo.
Participants were 18 through 49 years of age (mean age was 23.3 years); 63.3% were female, 83.1% were Caucasian, and 16.9% were of other racial/ethnic groups. Cases of influenza were identified through active and passive surveillance and confirmed by cell culture and/or real-time polymerase chain reaction (PCR). Influenza-like illness was defined as an illness with at least 1 respiratory symptom (cough or nasal congestion) and at least 1 constitutional symptom (fever or feverishness, chills, or body aches). Vaccine efficacy of Fluzone against all influenza viral types and subtypes is presented in Table 11.
5.2 Pharmacokinetic Properties
No pharmacokinetic studies have been performed.
5.3 Preclinical Safety Data
Fluzone has not been evaluated in non-clinical studies.
Genotoxicity.
Fluzone has not been evaluated for genotoxic potential.
Carcinogenicity.
Fluzone has not been evaluated for carcinogenic potential.6 Pharmaceutical Particulars
6.1 List of Excipients
Buffer solution: sodium chloride, dibasic sodium phosphate, monobasic sodium phosphate, water for injections.
Fluzone may also contain traces of octoxinol-9 (≤ 250 microgram), formaldehyde (≤ 100 microgram) and ovalbumin (≤ 1 microgram).
6.2 Incompatibilities
Fluzone should not be mixed with any other vaccine in the same syringe or vial.
6.3 Shelf Life
12 months when stored at 2°C to 8°C.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate, do not freeze). Discard if vaccine has been frozen.
6.5 Nature and Contents of Container
Prefilled syringe (clear syringe plunger rod), 0.5 mL with or without separate needle. Packs of 5 syringes.
Not all packs may be marketed.
The Fluzone presentation is not made with natural rubber latex.
6.6 Special Precautions for Disposal
After use, any remaining vaccine and container must be disposed of safely, according to locally acceptable procedures.
6.7 Physicochemical Properties
Not applicable for vaccines.
7 Medicine Schedule (Poisons Standard)
S4 Prescription Only Medicine.
Summary Table of Changes


 Fluzone contains 45 microgram hemagglutinin (HA) per 0.5 mL dose, in the recommended ratio of 15 microgram HA per strain.
Fluzone contains 45 microgram hemagglutinin (HA) per 0.5 mL dose, in the recommended ratio of 15 microgram HA per strain. The preferred sites for intramuscular injection are the anterolateral aspect of the thigh in infants 6 months through 11 months of age, the anterolateral aspect of the thigh (or the deltoid muscle if muscle mass is adequate) in persons ≥ 12 months through 35 months of age, or the deltoid muscle in persons > 36 months of age.
The preferred sites for intramuscular injection are the anterolateral aspect of the thigh in infants 6 months through 11 months of age, the anterolateral aspect of the thigh (or the deltoid muscle if muscle mass is adequate) in persons ≥ 12 months through 35 months of age, or the deltoid muscle in persons > 36 months of age.
 The most frequently reported solicited injection site reactions within 7 days following dose 1 and dose 2 vaccination were tenderness (47% and 56%) and redness (29% and 32%) in children 6 months through 35 months; or pain (59% and 62%) and erythema (27% and 27%) in children 3 years through 8 years. The most frequently reported solicited systemic adverse events within 7 days following dose 1 and dose 2 vaccination were irritability (43% and 35%), crying abnormal (32% and 19%), and drowsiness (26% and 27%) in children 6 months through 35 months; or myalgia (28% and 17%), malaise (20% and 15%), and headache (17% and 12%) in children 3 years through 8 years.
The most frequently reported solicited injection site reactions within 7 days following dose 1 and dose 2 vaccination were tenderness (47% and 56%) and redness (29% and 32%) in children 6 months through 35 months; or pain (59% and 62%) and erythema (27% and 27%) in children 3 years through 8 years. The most frequently reported solicited systemic adverse events within 7 days following dose 1 and dose 2 vaccination were irritability (43% and 35%), crying abnormal (32% and 19%), and drowsiness (26% and 27%) in children 6 months through 35 months; or myalgia (28% and 17%), malaise (20% and 15%), and headache (17% and 12%) in children 3 years through 8 years. The difference in fever rate (Group 2 minus Group 1) was 0.84% (95% CI: -2.13%; 3.80%), meeting the prespecified non-inferiority criterion (upper limit of the 2-sided 95% CI of the difference in fever rates < 5%). Participants were monitored for unsolicited adverse events and SAEs during the 28 days following vaccination. Unsolicited non-serious adverse events were reported in 417 (44%) participants in Group 1 and 394 (40%) participants in Group 2. The most commonly reported unsolicited non-serious adverse events in both groups were cough and rhinorrhoea. Ten SAEs were reported during the 28-day follow-up period: 5 (0.5%) in Group 1 and 5 (0.5%) in Group 2.
The difference in fever rate (Group 2 minus Group 1) was 0.84% (95% CI: -2.13%; 3.80%), meeting the prespecified non-inferiority criterion (upper limit of the 2-sided 95% CI of the difference in fever rates < 5%). Participants were monitored for unsolicited adverse events and SAEs during the 28 days following vaccination. Unsolicited non-serious adverse events were reported in 417 (44%) participants in Group 1 and 394 (40%) participants in Group 2. The most commonly reported unsolicited non-serious adverse events in both groups were cough and rhinorrhoea. Ten SAEs were reported during the 28-day follow-up period: 5 (0.5%) in Group 1 and 5 (0.5%) in Group 2. The most frequently reported solicited injection-site reaction was pain (54%); the most frequently reported solicited systemic adverse events were myalgia (31%), headache (30%), and malaise (22%).
The most frequently reported solicited injection-site reaction was pain (54%); the most frequently reported solicited systemic adverse events were myalgia (31%), headache (30%), and malaise (22%). The most frequently reported solicited injection-site reaction was pain (24%); the most frequently reported solicited systemic adverse events were myalgia (18%), headache (14%), and malaise (14%).
The most frequently reported solicited injection-site reaction was pain (24%); the most frequently reported solicited systemic adverse events were myalgia (18%), headache (14%), and malaise (14%).



