What is in this leaflet
Read all of this leaflet carefully before you are vaccinated.
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This vaccine has been prescribed for you. Do not pass it on to others.
- If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
In this leaflet:
- What Fluzone High-Dose is and what it is used for
- Before you are given Fluzone High-Dose
- How Fluzone High-Dose is given
- Possible side effects
- Storing Fluzone High-Dose
- Further information
What Fluzone High-Dose is and what it is used for
Fluzone High-Dose is a vaccine. Vaccines are used to help protect you against infectious diseases.
Fluzone High-Dose is recommended to help protect you against influenza. Fluzone High-Dose may be administered to persons aged 65 years and older. Influenza (flu) is a disease caused by different strains of the influenza virus. The influenza virus can spread rapidly, especially in homes or institutions, where it can be very easy for someone to catch flu and spread the disease.
Your doctor will be able to recommend the best time for you to be vaccinated. Flu is present every year, even when epidemics or outbreaks are not reported.
Fluzone High-Dose will only help to prevent flu if it is caused by one of the three strains of virus contained in the vaccine, or other strains closely related to them. Fluzone High-Dose will not prevent flu if you are incubating the disease before vaccination or if it is caused by another virus.
You should also note that there are many germs which can cause influenza-like illness. The vaccine will not protect you against these.
Previous injections of flu vaccine are unlikely to give you protection against the current strains of flu virus most common this year, so you will need to receive a vaccine against flu every year especially if you are at risk.
How Fluzone High-Dose works
Fluzone High-Dose works by helping the body to make antibodies. These antibodies help the body to recognise the flu virus and help prevent the infection. It takes a few weeks for the body to produce these antibodies. The vaccine will protect you only against the influenza strains contained in the vaccine. It will not protect you from viruses that are unrelated to those contained in the vaccine.
Before you are given Fluzone High-Dose
When you must not be given it
Do not have Fluzone High-Dose:
- If you have ever had a severe allergic reaction to:
- Eggs or egg products
- Any of the other ingredients of Fluzone High-Dose listed in the FURTHER INFORMATION section. - If you have ever had a severe allergic reaction after getting any flu vaccine.
- Before speaking to your doctor, if you have an acute illness with or without high temperature.
Take special care with Fluzone High-Dose
- Tell your doctor or pharmacist if you have or have had Guillain-Barré syndrome (severe muscle weakness) after getting a flu vaccine
- Tell your doctor or pharmacist if you have or have had an immune response problem because the immune response to the vaccine may be diminished
- Tell your doctor or pharmacist if you have bleeding problems or bruise easily
- Tell your doctor or pharmacist if you are pregnant or breast feeding.
Your doctor will discuss the possible risks and benefits of having Fluzone High-Dose during pregnancy or breastfeeding.
Your doctor should make sure the benefits of vaccination outweigh the risks when recommending Fluzone High-Dose.
Taking other medicines
- Tell your doctor or pharmacist if you are taking medicines that may reduce your immune response: such as corticosteroids (for example prednisone), medicines used to treat cancer (chemotherapy), radiotherapy or other medicines affecting the immune system.
Tell your doctor or pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without prescription.
Having other vaccines
- Your doctor will advise you if Fluzone High-Dose may be given with another vaccine.
How Fluzone High-Dose is given
Fluzone High-Dose is administered to you by a health care professional.
Dose and Schedule
Adults over 65 years: A single 0.5 mL injection
Method of Administration
Fluzone High-Dose is injected into the muscles of the upper arm (preferably).
The vaccines should not be injected directly into a blood vessel (intravascularly).
When is it given?
Fluzone High-Dose should be given annually
Possible side effects
Like all medicines, Fluzone High-Dose can cause side effects, although not everybody gets them.
The most common local side effects of Fluzone High-Dose include:
- pain, redness (erythema) and swelling, at the injection site
Systemic side effects include:
- muscle aches (myalgia)
- feeling unwell (malaise)
- headache
- fever
These side effects usually clear up within a few days. If events continue or become severe, please tell your doctor or pharmacist.
Other side effects not listed above have been reported occasionally with Fluzone High-Dose and other flu vaccines:
- inflammation of nerves leading to weakness, such as weakness of facial muscles (facial palsy), visual disturbances (optic neuritis/neuropathy) or shoulder and arm pain (brachial neuritis)
- fainting (syncope), dizziness, tingling or numbness of hands or feet (paraesthesia)
- Shortness of breath (dyspnoea), sore throat and discomfort when swallowing (pharyngitis), runny or blocked nose and sneezing (rhinitis)
- Vomiting
- temporary inflammation of nerves causing pain, paralysis and sensitivity disorders (Guillain-Barré syndrome [GBS])
- fits (convulsions) with or without fever
- unusual weakness (asthenia) or tiredness (fatigue), pain in extremities or chest pain
- Redness of the eyes (ocular hyperaemia)
- Skin disorders such as Stevens-Johnson syndrome
- severe allergic reaction (anaphylaxis)
- temporary reduction in the number of blood particles called platelets (thrombocytopenia), swollen glands in neck, armpit or groin (lymphadenopathy)
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please tell your doctor or pharmacist.
Storing Fluzone High-Dose
Fluzone High-Dose is usually stored in the doctor’s surgery or clinic, or at the pharmacy. However, if you need to store Fluzone High-Dose:
- Keep out of reach and sight of children.
- Keep Fluzone High-Dose in the original packaging until it is time for it to be given.
- Keep it in the refrigerator, store at 2C to 8C. Do not freeze Fluzone High-Dose.
Do not use Fluzone High-Dose after the expiry date which is stated on the carton after EXP.
Do not have Fluzone High-Dose if the packaging is torn or shows signs of tampering.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
Further Information
What Fluzone High-Dose contains
Ingredients
Active Ingredients:
Fluzone High-Dose contains 3 killed influenza virus strains recommended for the 2019 influenza season:
- A/Michigan/45/2015 (H1N1)pdm09-like virus (A/Michigan/45/2015 X-275)
- A/Switzerland/8060/2017 (H3N2)- like virus (A/Brisbane/1/2018 X-311)
- B/Phuket/3073/2013-like virus (B/Phuket/3073/2013)
The other ingredients include sodium chloride, dibasic sodium phosphate, monobasic sodium phosphate, octoxinol-9, water for injection, and traces of formaldehyde.
Fluzone High-Dose contains less than 1 microgram ovalbumin (egg protein) per dose.
Fluzone High-Dose does not contain any antibiotics or preservative.
Fluzone High-Dose syringes are not made with natural rubber latex.
What Fluzone High-Dose looks like and contents of the pack
Fluzone High Dose is supplied as:
- Single-dose, 0.5mL pre-filled syringe (gray plunger rod) without needles.
Name and Address of Australian Sponsor
sanofi-aventis australia pty ltd
Talavera Corporate Centre - Building D
12-24 Talavera Road
Macquarie Park NSW 2113
Australia
Tel: 1800 818 806
Name and Address of New Zealand Sponsor
sanofi-aventis new zealand limited
Level 856 Cawley St
Ellerslie
Auckland
New Zealand
Tel: 0800 283 684
AUST R number
AUST R 285932
Date of preparation
10 December 2018
fluzonehd-ccdsv1-cmiv3-10dec18
Published by MIMS April 2026


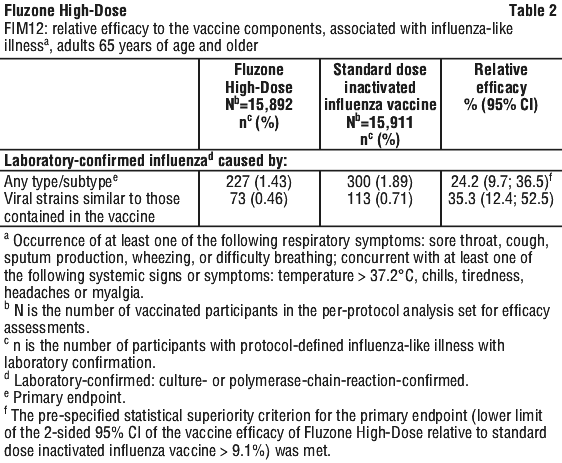 For the supplementary analysis, selected serious cardiorespiratory events reported in FIM12 were grouped into 7 pre-specified categories and represented the following endpoints: pneumonia events, asthma/chronic obstructive pulmonary disease (COPD)/bronchial events, influenza events (serious laboratory-confirmed influenza diagnosed outside study procedures by a subject's healthcare provider), other respiratory events, coronary artery events, congestive heart failure events and cerebrovascular events.
For the supplementary analysis, selected serious cardiorespiratory events reported in FIM12 were grouped into 7 pre-specified categories and represented the following endpoints: pneumonia events, asthma/chronic obstructive pulmonary disease (COPD)/bronchial events, influenza events (serious laboratory-confirmed influenza diagnosed outside study procedures by a subject's healthcare provider), other respiratory events, coronary artery events, congestive heart failure events and cerebrovascular events.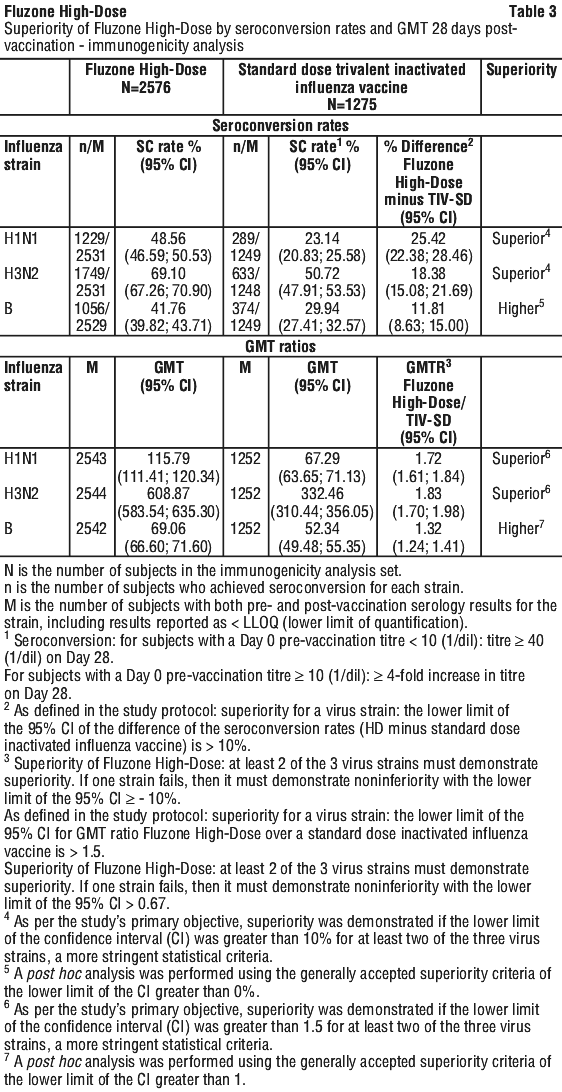 According to the criteria set in the protocol, Fluzone High-Dose elicited a superior immune response compared to a standard dose trivalent inactivated influenza vaccine for both seroconversion rates and GMTs.
According to the criteria set in the protocol, Fluzone High-Dose elicited a superior immune response compared to a standard dose trivalent inactivated influenza vaccine for both seroconversion rates and GMTs.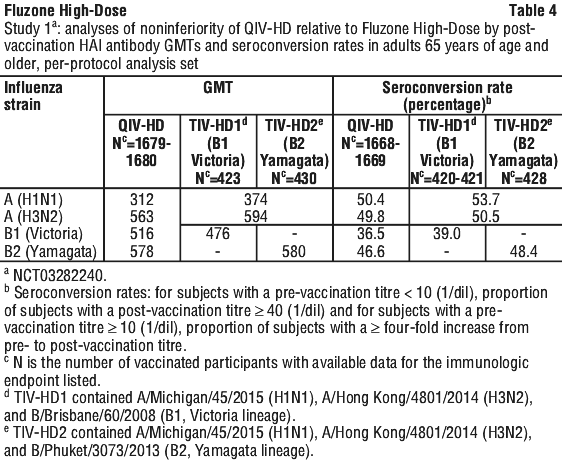 These data strongly support Fluzone High-Dose being as immunogenic as QIV-HD for all 3 strains in common, and thus allow inferring the immunogenicity, efficacy and effectiveness results of QIV-HD to Fluzone High-Dose.
These data strongly support Fluzone High-Dose being as immunogenic as QIV-HD for all 3 strains in common, and thus allow inferring the immunogenicity, efficacy and effectiveness results of QIV-HD to Fluzone High-Dose.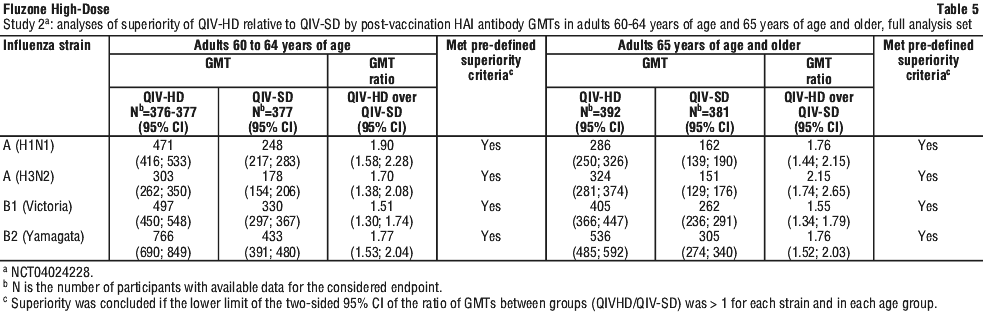 QIV-HD induced a superior immune response to QIV-SD for all 4 virus strains 28 days post-vaccination in adults 60 to 64 years of age, and this response was at least similar to the immune response in adults 65 years and above. The efficacy and effectiveness data from 65 years of age and above can thus be inferred to adults from 60 years of age and above.
QIV-HD induced a superior immune response to QIV-SD for all 4 virus strains 28 days post-vaccination in adults 60 to 64 years of age, and this response was at least similar to the immune response in adults 65 years and above. The efficacy and effectiveness data from 65 years of age and above can thus be inferred to adults from 60 years of age and above.