What is in this leaflet
This leaflet answers some common questions aboutGABARAN tablets (gabapentin).
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking GABARAN against the benefits it is expected to have for you.
If you have any concerns about using/taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with this medicine. You may need to read it again.
What GABARAN Tablets are used for
GABARAN contains gabapentin. Gabapentin belongs to a group of medicines used for treating epilepsy (antiepileptic medicines). These drugs are thought to work by controlling brain chemicals, which send signals to nerves so that convulsions do not happen.
Gabapentin also has analgesic effects. It may also be used for treating neuropathic pain (pain caused by damage to nerves).
Your doctor would have prescribed GABARAN tablets for one or more of the following:
- To treat certain types of epilepsy (partial onset seizures) in combination with other antiepileptic medicines, when the condition is not controlled with other anti-epileptic medicines or there are unacceptable side effects.
- To treat neuropathic pain (pain caused by damage to nerves). A variety of conditions can cause neuropathic pain, e.g., shingles, diabetes, trauma.
GABARAN 600 mg and 800 mg tablets will not be used to start your treatment. Your doctor will give you another brand of gabapentin when you begin your treatment. You will be givenGABARANtablets once your daily dose of gabapentin is 1800 mg (one 600 mg tablet three times a day) or more than 1800 mg.
Your doctor may have prescribed GABARAN tablets for another reason. Ask your doctor if you have any questions about why GABARAN tablets have been prescribed for you.
There is no evidence thatGABARAN tablets are addictive.
Gabapentin may make you sleepy or dizzy. This could affect your ability to drive a car or operate machinery.
Before you take GABARAN tablets
When you must not take it
Do not take GABARAN tablets if you are allergic to gabapentin or any of the inactive ingredients listed at the end of this leaflet. Symptoms of an allergic reaction may include skin rash, itchiness, shortness of breath, swelling of the tongue or face.
Do not take GABARAN tablets after the expiry date printed on the pack. If you take it after the expiry date has passed, it may not work as well. Do not take GABARAN if the tablets do not look quite right.
Do not take GABARAN tablets if the packaging is torn or shows signs of tampering.
Before you start to take it
Tell your doctor if you have any allergies to:
- any other substances, such as foods, preservatives or dyes
Tell your doctor if you are pregnant or intend to become pregnant. Like many other medicines, gabapentin may affect your developing baby if you take it during pregnancy. However, it is very important to control your fits while you are pregnant. If it is necessary for you to take gabapentin, your doctor can help you decide whether or not to take it during pregnancy.
Tell your doctor if you are breast-feeding or planning to breast-feed Your doctor will discuss the possible risks and benefits of using GABARAN during breast-feeding. If you do breast-feed, watch your baby carefully. If your baby develops a skin rash, becomes sleepy or has unusual symptoms, don't breast-feed again until you speak to your doctor.
Tell your doctor if you have or have had any medical conditions, especially the following:
- You have mixed seizure and/or absence seizures disorder
- You are pregnant, or planning to become pregnant, or are breast- feeding
- You have had mental illness
- You have problems with your kidneys
Your medicine also contains small amounts of an inactive ingredient called lactose monohydrate. If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor before taking this medicine.
Tell your doctor if you plan to have blood/urine tests.
If you need a laboratory investigation such as blood test or urine test, do inform your doctor about the medicine you are taking.
If you have not told your doctor about any of the above, tell them before you take GABARAN tablets.
Taking other medicines
Tell your doctor if you are taking any other medicines, including medicines you buy without a prescription from a pharmacy, supermarket or health food shop.
GABARAN tablets are not expected to interact with other anti-epileptic medicines or the oral contraceptive pill.
Some medicines may interfere with the absorption or action of GABARAN tablets.
These include:
- Antacids containing magnesium and aluminium (medicines for relieving symptoms of indigestion). These products can reduce the absorption of gabapentin.
- Cimetidine (a medicine for indigestion)
- Narcotic medicines (e.g. morphine)
These medicines may be affected byGABARAN tablets,or may affect how well it works. You may need to use different amounts of your medicine or you may need to take different medicines. Your doctor or pharmacist will be able to tell you what to do when taking/being given GABARAN tablets with other medicines.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while takingGABARAN tablets.
How GABARAN tablets are given
How much to take
Your doctor will tell you how many tablets you will need to take each day. This may depend on your age, your condition and whether or not you are taking any other medicines. Your doctor may recommend that you start with a low dose ofGABARAN and slowly increase the dose to the lowest amount needed to control your epilepsy/convulsions or neuropathic pain.
Take your medicine as instructed by your doctor. Do not take more than the doctor told you to. Check the label carefully for how much to take and how often to take. Your pharmacist or doctor can help if you are not sure.
How to take it
Note: GABARAN tablets are available as 600 mg and 800 mg tablets . GABARAN 600 mg and 800 mg tablets may be used for administering gabapentin in the dose range of 1800 to 3600 mg/day. Lower strengths of gabapentin are available in other dose forms (e.g. gabapentin 300 and 400 mg capsules). GABARAN 600 mg and800 mg tablets should not be broken into two halves to administer lower doses.
Swallow the tablet whole with a full glass of water. GABARAN tablets may be taken with or without food. If you are taking antacids containing magnesium and aluminium, it is recommended that you take GABARAN tablets at least 2 hours after taking your indigestion medicine.
Adults
Epilepsy (adults and children aged over 12 years)
Your doctor would usually start your treatment with 300 mg gabapentin taken once a day on the first day, followed by 300 mg gabapentin taken twice a day on the second day, and 300 mg gabapentin taken three times a day on the third day. Your doctor may also advise you to take 300 mg gabapentin three times daily on the first day. Your doctor will use another brand of gabapentin to start your treatment.
Thereafter, your doctor may increase your dose over the next few days to a maximum of 800 mg of gabapentin, taken three times a day. GABARAN 600 mg and 800 mg tablets may be used by your doctor to give you 600 to 800 mg of gabapentin, three times a day, as a maintenance dose.
Neuropathic pain (adults over the age of 18 years)
Your doctor would usually start your treatment with 300 mg gabapentin taken three times a day. Thereafter, your doctor may increase your dose over the next few days to a maximum of 1200 mg of gabapentin, taken three times a day. GABARAN 600 mg and 800 mg tablets may be used by your doctor to give you 600 to 1200 mg of gabapentin, three times a day, as a maintenance dose.
If you are elderly or have kidney problems, your doctor may prescribe a lower dose for you.
If gabapentin treatment is to be discontinued or another anti-epileptic medicine is to be added, this would be done gradually over a period of at least one week.
Children
Epilepsy (children aged between 3 to 12 years)
The dose that is right for your child would be decided by the doctor, based on your child’s weight.
Children below 3 years of age
The safety of GABARAN in the treatment of epilepsy has not been established in children below 3 years of age.
The safety of GABARAN in the treatment of neuropathic pain has not been established in children and adolescents below 18 years of age.
When to take it
Take your GABARAN tablets at about the same time. Take the first days dose at bedtime for best results. Taking your tablets at the same time will have the best effect. It will also help you to remember when to take the tablets.
How long to take it
Continue taking GABARAN tablets until your doctor tells you to stop. GABARAN helps control your condition, but does not cure it. Therefore you must take your medicine every day, even if you feel well.
Do not stop taking GABARAN, or lower the dosage, without checking with your doctor. Do not let yourself run out of medicine over the weekend or on holidays.
StoppingGABARANsuddenly may cause unwanted effects or make your condition worse. Your doctor will slowly reduce your dose before you can stop taking it completely.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, then go back to taking it as you would normally.
Do not double a dose to make up for the dose you have missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much (Overdose)
Immediately telephone your doctor or the Poisons Information Centre (Telephone No: 13 11 26), or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else has taken too much of GABARAN tablets. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention. If you take too many GABARAN tablets, you may feel drowsy, weak, unsteady when walking, have double vision, slurred speech or diarrhoea. Keep this telephone number handy.
While you are using GABARAN tablets
Things you must remember
Take GABARAN tablets exactly as your doctor tells you to.
Try not to miss any doses and take the medicine even if you feel well.
Visit your doctor regularly for check-ups.
Tell all doctors, dentists and pharmacists who are treating you that you are taking GABARAN tablets.
If you are about to be started on any new medicine, tell your doctor or pharmacist that you are taking GABARAN tablets.
Tell your doctor if you are to undergo any blood or urine test.
If you become pregnant while taking GABARAN tablets, tell your doctor immediately.
Tell your doctor immediately if you have any suicidal thoughts or mood changes
Persons taking antiepileptic medicines, including gabapentin, may be more likely to think about killing themselves or actually doing so.
If you or someone you know demonstrate any of the following warning signs of suicide-related behaviour while taking GABARAN, contact a healthcare provider immediately, or even go to the nearest hospital for treatment:
- thoughts or talk of death or suicide, or self-harm or harm to others
- any recent attempts of self-harm
- increase in aggressive behaviour, irritability or agitation
Things you must not do
Do not give this medicine to anyone else, even if they have the same condition as you.
Do not use GABARAN tablets to treat any other complaints unless your doctor tells you to.
Do not stop taking GABARAN tablets, or lower the dose, without first checking with your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how GABARAN affects you. Gabapentin may make you dizzy. This may affect your ability to drive or operate machinery. Make sure you know how you react to GABARAN tablets before you drive a car, operate machinery, or do anything else that could be dangerous if you are not alert.
Your doctor may suggest you avoid alcohol while you are being treated with GABARAN tablets.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking GABARAN tablets.
Ask your doctor or pharmacist any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- Uncontrolled shaking (tremors), memory loss, abnormal thinking, confusion, dizziness, sleep disturbances, anxiety, nervousness, depressed mood, emotional lability, hostile behaviour, hallucinations, difficulty in speech, uncontrolled jerking of facial muscles (twitching), reduced co-ordination or slowed reactions, gait disturbances, irregular involuntary contraction of a muscle
- Double vision, reduced vision, unusual jerky eye movements
- Hoarseness of voice
- Accidental injury, headache, unsteadiness, tiredness, decreased sensitivity to touch, weight gain, generalised weakness
- Feeling sick (nausea), being sick (vomiting), constipation, diarrhoea (loose stools), pain in the belly, indigestion, wind (flatulence), increased or decreased appetite, dryness of mouth, white patches inside the mouth or on the tongue (oral thrush)
- Chest infection, difficulty in breathing, sore throat
- Ringing in the ears
- Fast and irregular heart beat
- Dizziness and/or light-headedness
- Sweating
- Pins and needles sensation, numbness
- Joint pains, muscle aches, back ache
- Loss of hair
- Runny nose, cough
- Easy fracture and dislocation of bones
- Sexual inability in men (impotence)
- Inability to hold urine
- Fever, flu-like illness
- Distorted teeth
- Swelling of the hands or feet
- High blood pressure
- Excessive development of the breast in the male and female
These are the more common side effects of GABARAN tablets. These side effects are usually mild and occur at the start of treatment.
Tell your doctor immediately if you notice any of the following
- Unusual bleeding or increased tendency to bleed, persistent sore throat and frequent infections, and/or anaemia
- Sudden onset of severe abdominal pain associated with feeling sick (nausea) and being sick (vomiting) (manifestations of a condition called pancreatitis)
- Swelling of face, ankles or other parts of the body, with decrease in the amount of urine passed
- Yellowing of skin and whites of eyes with decreased appetite, abdominal pain
- More frequent or more severe seizures (fits)
- Myocardial Infarction (chest pain is more severe and prolonged than angina pain described above and may be associated with nausea (feeling sick), vomiting (being sick) and excessive sweating (occurs due to a complete blockade in one or more of the blood vessels, which supply the muscles of the heart, and the resulting failure to deliver oxygen for normal functioning of the heart)
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital if you notice any of the following:
- Thoughts of suicide or self-harm
- Symptoms of an allergic reaction may include rashes, hives, itching, chest constriction, shortness of breath or swelling of face, lips, tongue, hands/feet, fainting, fever.
- Severe skin reactions with blisters, sores or ulceration
- Rashes and hives with muscle and joint pains, and fever
These are very serious side effects. You may need urgent medical attention or hospitalisation. All these side effects are very rare.
Changes in laboratory tests
- Increased blood sugar in diabetic patients
- Abnormal liver function tests
Other side effects not listed above may also occur in some patients. Tell you doctor if you notice anything else that is making you feel unwell.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
After using it
Storage
Keep this medicine where young children cannot reach it.
Keep your tablets in the blister pack until it is time to take them.If you take the tablets out of the blister pack they may not keep well.
Keep your GABARAN in a cool, dry place where it stays below 25°C. Do not store it, or any other medicine, in the bathroom or near a sink.
Do not leave it in the car on hot days.
Disposal
If your doctor tells you to stop taking GABARAN tablets or you find that they have passed their expiry date, ask your pharmacist what to do with any tablets that are left over.
Product description
What it looks like
GABARAN 600 mg are white to off-white, oval-shaped, biconvex film-coated tablets imprinted with 'G 600' in black ink on one side and plain on the other side. Available in packs of 100 tablets.
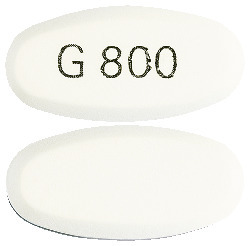
GABARAN 800 mg are white to off-white, oval-shaped, biconvex film-coated tablets imprinted with 'G 800' in black ink on one side and plain on the other side. Available in packs of 100 tablets.
Ingredients
Active ingredient
GABARAN 600 mg tablets - 600 mg of gabapentin
GABARAN 800 mg tablets - 800 mg of gabapentin
Inactive ingredients
Hydroxypropylcellulose, mannitol, crospovidone, purified talc, magnesium stearate, poloxamer, Opadry 20A59015, Opacode S-1-17823
Australian Registration Numbers
GABARAN 600 mg tablets blister pack – AUST R 121833
GABARAN 800 mg tablets blister pack – AUST R 121843
Sponsor
GABARAN Tablets are supplied in Australia by:
Ranbaxy Australia Pty Ltd.
Suite 4.02, Level 4, Building D
12 – 24 Talavera Rd
North Ryde, NSW 2113
GABARAN® is a registered trade mark owned by Ranbaxy Laboratories Limited.
This leaflet was prepared in May 2010.
Published by MIMS August 2010



 The relative risk for suicidal thoughts or behaviour was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
The relative risk for suicidal thoughts or behaviour was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
 Other events in more than 2% of children but equally or more frequent in the placebo group included: pharyngitis, upper respiratory infection, headache, rhinitis, convulsions, diarrhea, anorexia, coughing, and otitis media.
Other events in more than 2% of children but equally or more frequent in the placebo group included: pharyngitis, upper respiratory infection, headache, rhinitis, convulsions, diarrhea, anorexia, coughing, and otitis media.
 For patients undergoing hemodialysis who have never received gabapentin, a loading dose of 300 to 400 mg is recommended, then 200 to 300 mg of gabapentin following each 4 hours of hemodialysis.
For patients undergoing hemodialysis who have never received gabapentin, a loading dose of 300 to 400 mg is recommended, then 200 to 300 mg of gabapentin following each 4 hours of hemodialysis.