What is in this leaflet
This leaflet answers some common questions about Galvus.
It does not contain all the available information. It does not take the place of talking to your doctor, pharmacist or diabetes educator.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available.
You should ensure that you speak to your pharmacist or doctor to obtain the most up to date information on the medicine. You can also download the most up to date leaflet from www.novartis.com.au. Those updates may contain important information about the medicine and its use of which you should be aware.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking this medicine against the benefits they expect it will provide.
If you have any concerns about this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Galvus is used for
Galvus is used to lower blood sugar levels in patients with type 2 diabetes mellitus either alone (if you are unable to take metformin) or in combination with certain other medicines (metformin, or a sulfonylurea medicine, or pioglitazone, or metformin and a sulfonylurea, or insulin injections), when diet plus exercise plus the single or dual medicines do not provide adequate blood sugar level control.
Type 2 diabetes mellitus
Type 2 diabetes develops if the body does not produce enough insulin, or where the insulin that your body makes does not work as well as it should. It can also develop if the body produces too much glucagon.
Insulin is a substance which helps to lower the level of sugar in your blood, especially after meals. Glucagon is another substance which triggers the production of sugar by the liver, causing the blood sugar to rise. The pancreas makes both of these substances.
Galvus is a member of a class of medicines you take by mouth called DPP-4 inhibitors (dipeptidyl peptidase-4 inhibitors) that lowers blood sugar levels in patients with type 2 diabetes mellitus.
Your doctor will prescribe Galvus either alone or in combination with other antidiabetic medicines if that medicine or medicines do not sufficiently control your blood sugar level.
It is important that you continue to follow the diet and/or exercise recommended for you whilst you are on treatment with Galvus.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is only available with a doctor's prescription. It is not addictive.
There is not enough information to recommend this medicine for children.
Before you take Galvus
When you must not take it
Do not take Galvus if you have an allergy to:
- vildagliptin (the active ingredient) or to any of the other ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- shortness of breath;
- wheezing or difficulty breathing;
- swelling of the face, lips, tongue or other parts of the body;
- rash, itching or hives on the skin.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. In that case, return it to your pharmacist.
Before you start to take it
Tell your doctor if:
- you have allergies to any other medicines, foods, dyes or preservatives.
Your doctor will want to know if you are prone to allergies. - you are pregnant or intend to become pregnant.
Your doctor will discuss the possible risks and benefits involved. - you are breast-feeding or plan to breast-feed.
It is not known if the active ingredient of Galvus passes into breast milk and could affect your baby. - you have any of the following medical conditions:
- problems with your kidneys
- problems with your liver
- heart failure
- type 1 diabetes (formerly called 'juvenile onset' or 'insulin-dependent' diabetes mellitus or 'IDDM'), where the body does not produce any insulin
- diabetic ketoacidosis, a condition where chemicals called ketones build up in the body due to very low insulin levels.
Galvus is not a substitute for insulin. You should therefore not receive Galvus for the treatment of type 1 diabetes or diabetic ketoacidosis.
If you are not sure whether any of the above conditions apply to you, your doctor can advise you.
If you have not told your doctor about any of the above, tell him/her before you take any Galvus. Your doctor will do some blood and urine tests for sugar level regularly, and for liver functions at the start of treatment and regularly while you are on treatment. Your doctor will also test your kidney function before you start treatment with Galvus.
If your doctor has told you to stop your treatment with Galvus because of liver problems, you should never start taking Galvus again.
Tell your doctor if you suffer from lactose intolerance. This is because Galvus tablets contain lactose.
Alcohol, diet, exercise and your general health all strongly affect the control of your diabetes.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from a pharmacy, supermarket or health food shop. You may need to take different amounts of your medicines or to take different medicines while you are taking Galvus. Your doctor and pharmacist have more information.
If you have not told your doctor about any of these things, tell him/her before you start taking this medicine.
How to take Galvus
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the label, ask your doctor or pharmacist for help.
How much to take
The usual dose is 50mg or 100mg daily. The 50mg dose should be taken as 50mg once a day (morning). The 100mg dose is taken as 50mg twice a day (in the morning and evening).
Your doctor will monitor your blood glucose levels and may increase or decrease the dose of Galvus to maintain good control of your diabetes.
Your doctor will prescribe Galvus either alone or in combination with other antidiabetic medicines, if that medicine or medicines do not sufficiently control your blood sugar level.
How to take it
Swallow Galvus tablets whole with a glass of water.
If your doctor has prescribed 100mg a day, take one 50 mg tablet in the morning and one 50 mg tablet in the evening. Do not take two tablets together at any one time.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it. It does not matter if you take this medicine before or after food.
How long to take it
Continue taking your medicine for as long as your doctor tells you to. Your doctor will check your progress to make sure the medicine is working and will discuss with you how long your treatment should continue.
If your doctor has told you to stop your treatment with Galvus because of liver problems, you should never start taking Galvus again.
Do not stop taking Galvus unless your doctor tells you to.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone number: 13 11 26), or go to Accident and Emergency at your nearest hospital if you think that you or anyone else may have accidentally taken too much Galvus. Do this even if there are no signs of discomfort or poisoning. Keep the telephone numbers for these places handy. You may need urgent medical attention.
Symptoms of an overdose may include:
- Swelling in hands or feet
- Tingling or numbness in hands or feet
- Muscle pain
- Fever
While you are taking Galvus
Things you must do
If you become pregnant while taking this medicine, tell your doctor immediately. Galvus should not be taken if you are pregnant. Insulin is more suitable for controlling blood glucose during pregnancy.
Carefully follow your doctor's and/or dietician's advice on diet, drinking alcohol and exercise.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor will do regular checks to help prevent you from having side effects from the medicine or developing serious complications of diabetes.
Make sure you check your blood glucose levels regularly. This is the best way to tell if your diabetes is being controlled properly. Your doctor or diabetes educator will show you how and when to do this.
If you become ill or experience extra stress, injury, fever, infection or need surgery, tell your doctor. Your blood glucose may become difficult to control at these times.
Make sure you keep enough medicine to last over weekends and holidays. It is important to keep your blood glucose controlled at all times to prevent serious complications of diabetes from happening.
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Galvus.
Tell any other doctor, dentist or pharmacist who treats you that you are taking Galvus.
Things you must not do
Do not give this medicine to anyone else, even if their condition seems similar to yours.
Do not use it to treat any other complaints unless your doctor tells you to.
Things to be careful of
Be careful driving, operating machinery or doing jobs that require you to be alert until you know how this medicine affects you. If your blood glucose level becomes too low, you may feel dizzy, lightheaded, weak or tired and your reaction time may be slower than usual.
If you have any of these symptoms, do not drive or do anything else that could be dangerous.
Be careful when doing any of the following things, which increase the risk of your blood glucose becoming too low:
- drinking alcohol
- not eating enough
- doing unexpected or vigorous exercise
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Galvus even if you do not think it is connected with the medicine.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Do not be alarmed by these lists of possible side effects. You may not experience any of them.
Ask your doctor, pharmacist or diabetes educator to answer any questions you may have.
Tell your doctor if you notice any of the following side effects and they worry you:
- swelling of the hands, ankles or feet
- weakness
- dizziness
- headache
- trembling
- low blood glucose
- nausea
- chills
- constipation
- diarrhoea
- wind (flatulence)
- a burning sensation in the chest rising up to the throat ('heartburn')
- weight increase
- excessive decreased blood glucose
- itchy rash
- peeling of skin or blisters
- skin lesions
- joint pain
- excessive sweating
Tell your doctor immediately or go to Accident and Emergency if you notice any of the following:
- swelling of the face, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing
- sudden onset of rash or hives
- yellow skin and eyes, nausea, loss of appetite, dark urine (possible symptoms of liver problems)
- severe and persistent pain in the stomach area which might reach through your back, as well as nausea and vomiting (possible sign of inflamed pancreas or gallbladder)
Tell your doctor if you notice anything else that is making you feel unwell. Some people may have other side effects not yet known or mentioned in this leaflet. Some side effects (e.g. changes in liver function) can only be found by laboratory testing.
After using Galvus
Storage
- Keep your medicine in the original container until it is time to take it.
- Store it in a cool dry place where the temperature stays below 30°C.
- Do not store Galvus or any other medicine in the bathroom or near a sink.
- Do not leave it in the car or on window sills.
Keep the medicine where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any tablets you have left over.
Product description
What it looks like
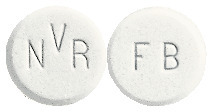
Galvus tablets are round, white to light yellowish, marked with "NVR" on one side and "FB" on the other.
Galvus tablets are available in boxes containing 14 or 60 tablets.
Ingredients
Each Galvus tablet contains 50 mg of vildagliptin as the active ingredient.
It also contains the following inactive ingredients:
- lactose anhydrous
- cellulose - microcrystalline
- sodium starch glycollate
- magnesium stearate
Galvus does not contain gluten, tartrazine or any other azo dyes.
Sponsor
Galvus is supplied in Australia by:
NOVARTIS Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone: 1 800 671 203
Web site: www.novartis.com.au
®= Registered Trademark
This leaflet was prepared in November 2023
Australian Registration Number:
50 mg tablet - AUST R 159561
Internal Document Code:
(gal151123c based on PI gal151123i)
Published by MIMS January 2024

 Long-term clinical trials of up to 2 years did not show any additional safety signals or unforeseen risks with vildagliptin monotherapy.
Long-term clinical trials of up to 2 years did not show any additional safety signals or unforeseen risks with vildagliptin monotherapy. Long-term clinical trials of up to more than 2 years did not show any additional safety signal or unforeseen risks when vildagliptin was added on to metformin.
Long-term clinical trials of up to more than 2 years did not show any additional safety signal or unforeseen risks when vildagliptin was added on to metformin.




 The adverse effect profiles for the vildagliptin and sitagliptin groups of the 24 week core study and 28 week extension study investigating patients with type 2 diabetes and severe renal insufficiency are shown in Table 9.
The adverse effect profiles for the vildagliptin and sitagliptin groups of the 24 week core study and 28 week extension study investigating patients with type 2 diabetes and severe renal insufficiency are shown in Table 9.

 In a double blind, active controlled 24 week trial (study 2354; n = 576), vildagliptin (100 mg/day; 50 mg in the morning and 50 mg in the evening) was compared to pioglitazone (30 mg once daily) in patients with type 2 diabetes inadequately controlled with metformin alone. Mean reductions from baseline HbA1c of 8.4% were -0.9% with vildagliptin added to metformin and -1.0% with pioglitazone added to metformin. The decrease in HbA1c from baseline > 9.0% was greater (-1.5%) in both treatment groups. Patients receiving pioglitazone in addition to metformin experienced an increase in weight of 1.9 kg while those receiving vildagliptin in addition to metformin experienced an increase in weight of 0.3 kg.
In a double blind, active controlled 24 week trial (study 2354; n = 576), vildagliptin (100 mg/day; 50 mg in the morning and 50 mg in the evening) was compared to pioglitazone (30 mg once daily) in patients with type 2 diabetes inadequately controlled with metformin alone. Mean reductions from baseline HbA1c of 8.4% were -0.9% with vildagliptin added to metformin and -1.0% with pioglitazone added to metformin. The decrease in HbA1c from baseline > 9.0% was greater (-1.5%) in both treatment groups. Patients receiving pioglitazone in addition to metformin experienced an increase in weight of 1.9 kg while those receiving vildagliptin in addition to metformin experienced an increase in weight of 0.3 kg. Molecular formula: C17H25N3O2.
Molecular formula: C17H25N3O2.