What is in this leaflet
This leaflet answers some common questions about Glatira20 mg/mL and Glatira 40 mg/mL.
It does not contain all the available information. It does not take the place of talking to your doctor and pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Glatira 20 mg/mL and Glatira 40 mg/mL is used for
Glatira 20 mg/mL and Glatira 40 mg/mL is used for the management of relapsing forms of Multiple Sclerosis (MS).
Glatira may also be used in patients who, for the first time, have experienced symptoms and have MRI changes that indicate a high risk for development of MS. Your doctor will rule out any other reasons which could explain these symptoms before you are treated.
The cause of MS is not yet known. MS affects the brain and spinal cord. In MS, the body's immune system reacts against its own myelin (the 'insulation' surrounding nerve fibres). In relapsing forms of MS, people can have 'exacerbations' from time to time (eg. blurred vision, weakness in the legs or arms, or loss of control of bowel or bladder function). These are followed by periods of recovery.
Although the exact mechanism of how Glatira works in MS is unknown, it is thought that Glatira works by changing the immune processes that are believed to be responsible for the disease.
Glatira has been shown to be effective in reducing the number of relapses in patients with relapsing remitting MS. Although it is not a cure, patients treated with Glatira generally find that they will experience fewer relapses.
Your doctor, however, may prescribe Glatira for another purpose.
Ask your doctor or pharmacist if you have any questions about why it has been prescribed for you.
This medicine is only available with a doctor's prescription.
Before you use it
When you must not use it
Do not use Glatira if you are allergic to it or any of the ingredients listed at the end of this leaflet. Some symptoms of an allergic reaction include skin rash, itching, shortness of breath or swelling of the face, lips or tongue, which may cause difficulty in swallowing or breathing.
Do not give Glatira to children. There is no experience with its use in children under 12 years of age.
Do not use it after the expiry date (EXP) printed on the pack. If you use it after the expiry date has passed, it may not work as well.
Do not use it if the packaging is damaged or shows signs of tampering.
Before you start to use it
Tell your doctor or pharmacist if you have allergies to:
- any of the ingredients listed at the end of this leaflet
- any other medicines
- any other substances, such as foods, preservatives or dyes.
Tell your doctor if you are pregnant or intend to become pregnant. Like most medicines of this kind, Glatira is not recommended to be used during pregnancy. You should discuss contraceptive options with your doctor. Your doctor will discuss the risks and benefits of using it if you are pregnant.
Tell your doctor if you are breast- feeding or plan to breast-feed. It is not known whether Glatira passes into breast milk. Your doctor will discuss the risks and benefits of using it if you are breast-feeding or planning to breast-feed.
Tell your doctor if you have or have ever had any medical conditions, especially the following:
- asthma
- a history of severe allergic reactions
Tell your doctor if you plan to have surgery.
If you have not told your doctor about any of the above, tell them before you use Glatira.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food store.
Some medicines may interfere with the absorption of Glatira.
These medicines may be affected by Glatira, or may affect how well it works. You may need to use different amounts of your medicine, or take different medicines. Your doctor or pharmacist will advise you.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while using Glatira 20 mg/mL or Glatira 40 mg/mL.
How to use it
Follow all directions given to you by your doctor, MS education nurse or pharmacist. They may differ from the information contained in this leaflet.
How much to use
The recommended dose of one Glatira 20mg/mL pre-filled syringe (1mL) is to be injected once a day.
The recommended dose of one Glatira 40 mg/mL pre-filled syringe (1mL) is to be injected three times a week and at least 48 hours apart. Glatira 40 mg/mL should be given, if possible, at the same time and on the same three days each week (for example, Monday, Wednesday and Friday) at least 48 hours apart.
Do not change the dose or dosing schedule or stop using Glatira without talking to your doctor.
How to use it
Glatira 20 mg/mL and Glatira 40 mg/mL is given by an injection into the fatty layer under the skin (subcutaneous injection).
Many people with MS learn to give themselves the injection or have it given by a carer.
Self-injection needs to be taught and practised. It is important that a qualified healthcare professional supervises your first injection.
Your doctor may teach you to self-inject or arrange for an MS education nurse to do so.
You may have a friend attend the injection training session as your assistant. Especially when you first start giving yourself injections, your assistant should be with you.
Talk to your doctor or the MS Society or telephone 1800 620 076 for more information.
Patient support kits that include a self-injection device are available by telephoning 1800 620 076.
After being taught to self-inject, you can refer to this leaflet for step-by-step instructions about how to prepare and inject Glatira 20 mg/mL and Glatira 40 mg/mL.
The following instructions explain how to inject Glatira 20mg/mL and Glatira 40 mg/mL yourself.
Do not attempt self-injection until you are confident that you understand how to inject yourself.
Please read these instructions carefully.
Before you Inject
Glatira 20 mg/mL and Glatira 40 mg/mL pre-filled injection is pre-mixed and ready for you to use.
Make sure that the blister with the syringe inside is taken out of the refrigerator for 20 minutes before you use it. It is important that the solution is at room temperature when you inject it. If you need to delay your injection, return the blister to its package in the refrigerator.
Always inspect Glatira solution in the pre-filled syringe before you use it. Do not inject the solution if it is cloudy or has particles in it.
Gathering the Materials
Gather the following items on a towel in a well-lit area:
- One blister with Glatira 20 mg/mL or Glatira 40 mg/mL pre-filled syringe (at room temperature - as described above)
- A dry cotton ball
- Disposal unit for used syringes
Wash and dry your hands. Do not touch your hair or skin afterwards. This will help prevent infection.
Deciding where to Inject
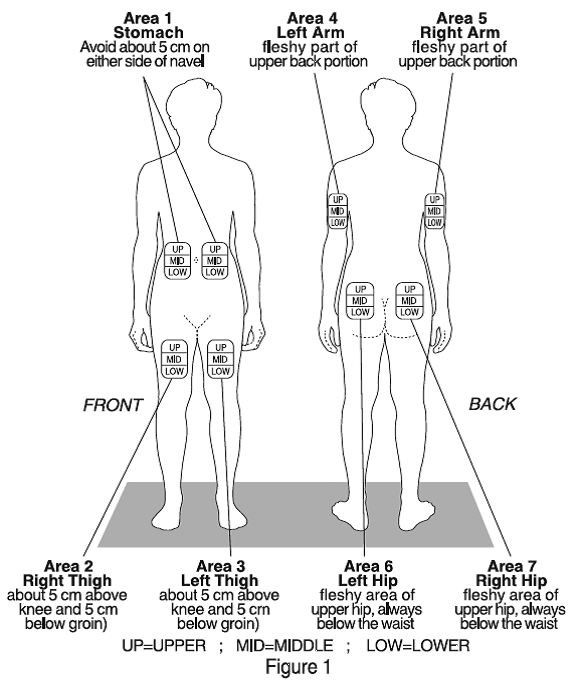
There are seven injection areas on your body; arms, thighs, back of the hips and stomach (belly). Within each injection area there are multiple injection sites (Figure 1).
Rotate the injection sites within an area.
You should not use any site more than once each week. Choose a different site for each injection. This will reduce the likelihood of irritation, pain or loss of fatty tissue (lipoatrophy) at the site of injection. Marking a calendar will help you keep track of the sites you have used each day.
Giving Yourself the Injection
- Remove the syringe from its protective blister by peeling back the paper label.
- Pick up the syringe as you would a pencil, using the hand you write with. Remove the plastic cover from the needle.
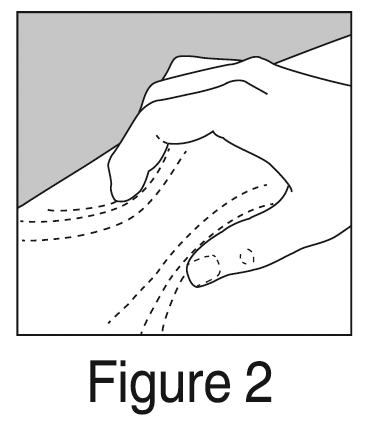
- Pinch about a 5 cm fold of skin between thumb and index finger.
When you inject in the upper back portions of your arms, it is not possible to pinch 5 cm of skin with one hand and inject yourself with the other hand. Ask your doctor / nurse for instruction on how to use these areas.
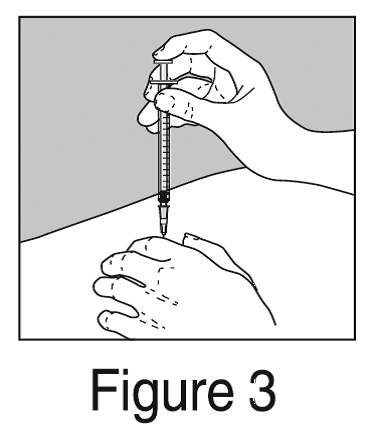
- Inject the medication by holding the syringe steady while pushing down on the plunger until the syringe is empty. The injection should take just a few seconds.
- Pull the needle straight out
- Press a dry cotton ball on the injection site for a few seconds, but do not massage it.
- Dispose of the pre-filled syringe in a safe hard-walled container, according to your doctor's instructions and the laws of your state (if any). Ask your doctor or pharmacist for advice if you are in doubt.
Some patients may experience a side-effect known as the Immediate Post Injection Reaction (see the information under the heading 'Side effects' below).
Proper use of needles and syringes
Do not reuse the pre-filled syringe. Each pre-filled syringe should be used only for one injection.
Ask your doctor or nurse about obtaining a "sharps container".
After use, place all used syringes and needles in a hard-walled plastic container, such as a liquid laundry detergent container. It is very important that you keep the cover of the container tightly shut and that you store it out of the reach of children. When the container is full, check with your doctor or nurse about proper disposal.
When to use it
Give yourself the Glatira 20 mg/mL injection at the same time each day.
Give yourself the injection at the same time on the same three days of the week (for example, Monday, Wednesday and Friday) at least 48 hours apart. Choose a time when you feel strongest.
How long to use it
Do not stop using Glatira without checking with your doctor first.
The positive effects of Glatira are not seen immediately. They occur with long-term treatment.
If you forget to use it
Glatira 20 mg/mL
If it is almost time for your next dose, skip the dose you missed and have your next injection when you are meant to.
If there is still a long time to go before your next dose, use it as soon as you remember, and then go back to using it as you would normally.
Glatira 40 mg/mL
If you miss a dose of Glatira 40 mg/mL, you should take your next dose as soon as you remember or are able to take it, then skip the following day. If possible, you should return to your regular administration schedule the following week.
Do not use a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side-effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to use your medicine, ask your pharmacist or MS education nurse for some hints.
It is important not to miss injections as this can affect the success of your treatment.
If you use too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone Australia 13 11 26), or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else may have used too much Glatira 20 mg/mL or Glatira 40 mg/mL.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using it
Things you must do
Tell all the doctors, dentists and pharmacists who are treating you that you are using Glatira.
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are using Glatira.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are using this medicine.
If you become pregnant while you are using this medicine, stop using it and tell your doctor immediately.
Things you must not do
Do not use more than the recommended dose unless your doctor tells you to.
Do not give this medicine to anyone else, even if they have the same condition as you.
Side effects
All medicines have some unwanted side effects. Sometimes they are serious, but most of the time they are not. Your doctor has weighed the risks of using this medicine against the benefits they expect it will have for you.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using Glatira.
It helps most people with MS, but it may have unwanted side effects in a few people.
Tell your doctor if you notice any of the following and they worry you:
- pain, redness, itching, hardening of the skin or an infection at the site where you have injected yourself
- nausea (feeling sick)
- pain in the joints
- sore muscles
- feeling of weakness
- dizziness
- constipation
- changes to your vision
- hay fever symptoms
- changes in your urination frequency
These side effects were reported by patients who used Glatira 20 mg/mL every day and by patients who use Glatira 40 mg/mL three times a week.
Some patients may experience a side-effect known as the Immediate Post Injection Reaction. This reaction consists of the following symptoms:
- reddening of the face and/or neck,
- chest pain or tightness,
- feeling your heartbeat quickly (heart palpitations),
- anxiety and difficulty in breathing.
These are mild side effects of this medicine and usually are short-lived.
Some patients may experience only one episode during the treatment period.
Stop administering Glatira and contact your doctor immediately if the above symptoms are severe.
If any of the following happen, stop using this medicine and tell your doctor immediately, or go to Accident and Emergency at your nearest hospital:
- swelling of the face, lips, mouth or throat, which may cause difficultly in swallowing or breathing
- hives
- chest pain
- trouble breathing
- severe pain, redness or swelling at the injection site that does not go away.
These are very serious side effects. If you have them, you may have had a serious allergic reaction to Glatira. You may need urgent medical attention or hospitalisation.
These side effects are very rare.
Tell your doctor or pharmacist if you notice anything else that is making you feel unwell. Other side effects not listed above may occur in some patients.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
After using it
If you have any queries about any aspect of your medicine, or any questions regarding the information in this leaflet, discuss them with your doctor or pharmacist.
Storage
Keep Glatira 20 mg/mL and Glatira 40 mg/mL in the pack until it is time to use it. Protect from direct light. The medicine will not keep as well if taken out of the packaging.
Keep Glatira pre-filled syringes in the refrigerator at 2°C to 8°C. In the event of refrigeration being unavailable, Glatira may be stored below 25°C for up to one month.
Glatira 20 mg/mL and Glatira 40 mg/mL must not be frozen. Do not place in the freezer or freezing compartment of a refrigerator.
Do not store it or any other medicine in the bathroom, near a sink or on a windowsill.
Do not leave it in the car. Heat and damp can destroy some medicines.
Keep it where children cannot reach it.
Disposal
Please read the package insert carefully for full details, including self-disposal of needles and syringes after use.
If your doctor tells you to stop using Glatira or the medicine has passed its expiry date, ask your pharmacist what to do with any that are left over.
Return any unused medicine to your pharmacist
Product description
What it looks like
Glatira 20 mg/mL and Glatira 40 mg/mL is a clear colourless solution supplied in 1mL glass single use pre-filled syringes fitted with a staked 1/2 inch needle.
Glatira 20 mg/mL is supplied in a box of 28 blister packed pre-filled syringes.
Glatira 40 mg/mL is supplied in a box of 12 blister packed pre-filled syringes
Ingredients
Active Ingredient:
Each Glatira pre-filled syringe contains 20 mg glatiramer acetate.
Each Glatira 40 mg/mL pre-filled syringe contains 40 mg glatiramer acetate.
Inactive Ingredients:
Each injection also contains:
- mannitol
- water for injections
Distributor
Glatira is distributed in Australia by:
Juno Pharmaceuticals Pty Ltd
42 Kelso Street,
Cremorne,
VIC 3121
Medical information please call 1800 620 076
Australian Register Number:
20 mg/1 mL: AUST R 303650
40 mg/1 mL: AUST R 303649
This leaflet was prepared in January 2022
Published by MIMS March 2022

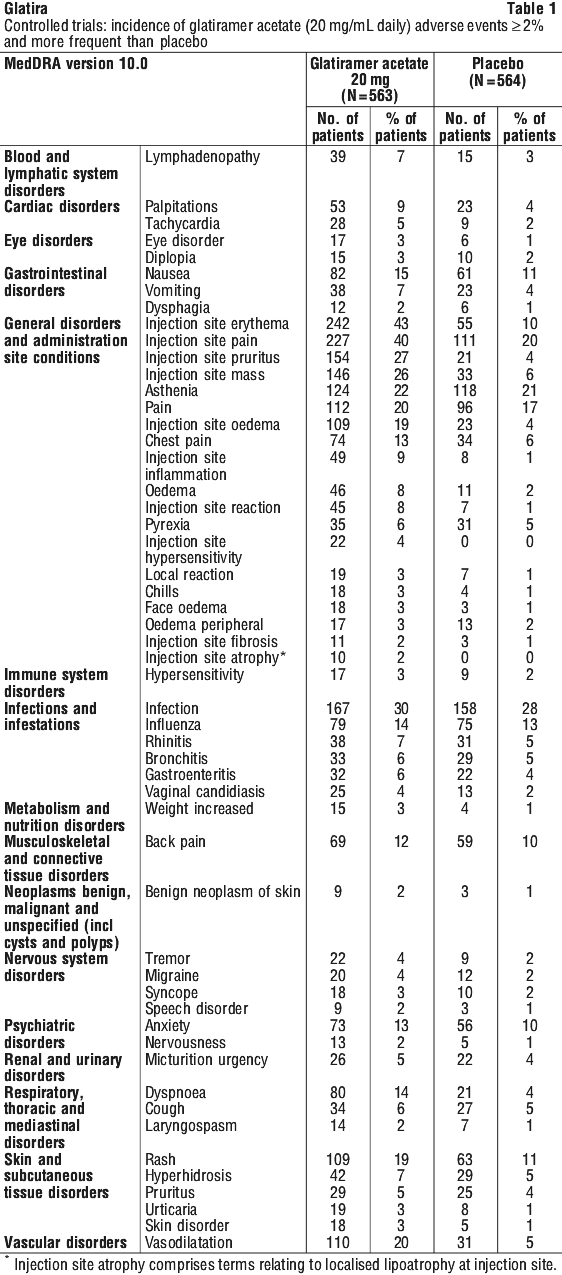 The overall frequency of adverse events and the frequencies of individual adverse events were higher in patients treated with glatiramer acetate for RRMS or SPMS, and lower in patients treated with glatiramer acetate for a first clinical episode of MS.
The overall frequency of adverse events and the frequencies of individual adverse events were higher in patients treated with glatiramer acetate for RRMS or SPMS, and lower in patients treated with glatiramer acetate for a first clinical episode of MS.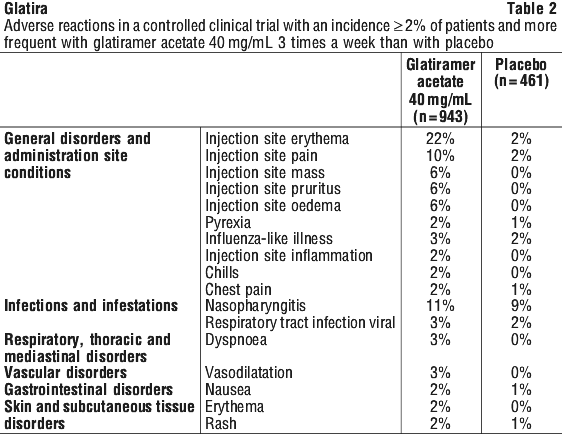
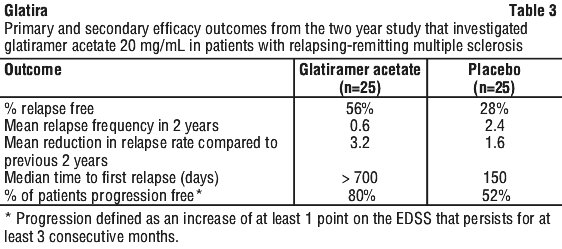 The second trial was of similar design to the first study. A total of 251 patients were enrolled. The primary outcome measure was the mean 2 year relapse rate. Table 4 presents the results of the analysis of this outcome for the intent-to-treat population, as well as other secondary measures.
The second trial was of similar design to the first study. A total of 251 patients were enrolled. The primary outcome measure was the mean 2 year relapse rate. Table 4 presents the results of the analysis of this outcome for the intent-to-treat population, as well as other secondary measures.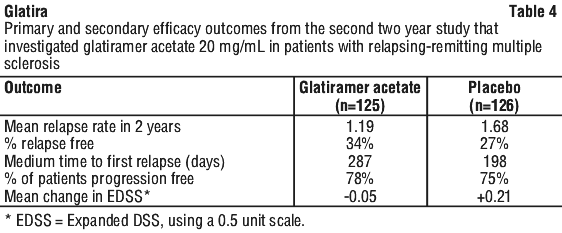 Results also showed that glatiramer acetate reactive antibodies were present in all treated patient's sera, with maximal levels attained after a duration of 3-4 months. Thereafter antibody levels slowly declined and stabilised at a level slightly higher than baseline.
Results also showed that glatiramer acetate reactive antibodies were present in all treated patient's sera, with maximal levels attained after a duration of 3-4 months. Thereafter antibody levels slowly declined and stabilised at a level slightly higher than baseline.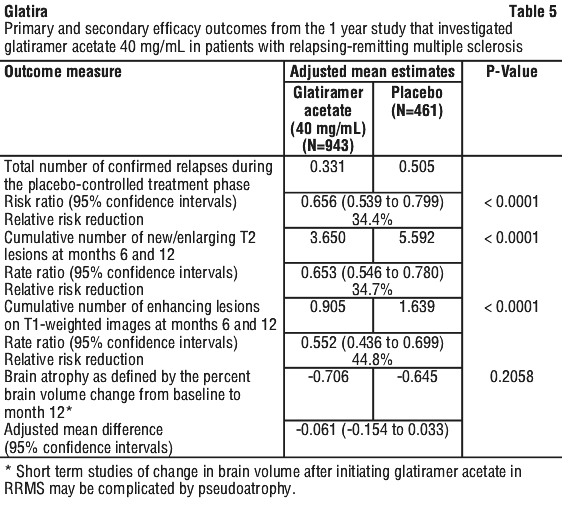
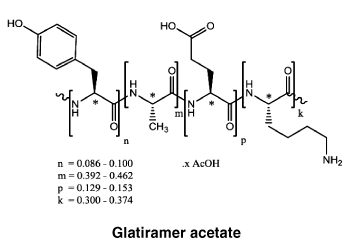 Structural Formula: Poly [ L-Glu13-15, L-Ala39-46, L-Tyr8.6-10, L-Lys30-37].n(CH3CO2H).
Structural Formula: Poly [ L-Glu13-15, L-Ala39-46, L-Tyr8.6-10, L-Lys30-37].n(CH3CO2H).