What is in this leaflet
- What GlucaGen® HypoKit is used for
- Before you use GlucaGen® HypoKit
- How to use GlucaGen® HypoKit
- While you are using GlucaGen® HypoKit
- Side Effects
- Storage
- Product Description
- Further Information
- Instructions For Use
This leaflet answers some common questions about GlucaGen® HypoKit. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using GlucaGen® HypoKit against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What GlucaGen® HypoKit is used for
GlucaGen® HypoKit contains glucagon (rys) hydrochloride. The abbreviation “rys” indicates the method of genetic engineering used to manufacture the glucagon.
Glucagon is a natural hormone, which has the opposite effect to insulin in the human body. It helps the body to convert glycogen into glucose (sugar) in the liver. Glucose is then released into the blood stream. This means that GlucaGen® HypoKit will only work to increase blood sugar if there are stores of glycogen in the liver.
Glucagon also reduces the movements of the smooth muscle in the digestive system and stimulates the production of insulin by the beta cells of the pancreas.
GlucaGen® HypoKit is used to treat severe hypoglycaemia (extremely low blood sugar levels, or “hypos”) in people using insulin or taking tablets to control their diabetes, who have become unconscious.
GlucaGen® HypoKit is also used during medical investigations of the gastrointestinal tract to relax and slow down the movements of the stomach and bowel. This makes it easier for your doctor to investigate your digestive tract.
GlucaGen® HypoKit is also used to stimulate the production of insulin by the beta cells. It can therefore be used to find out how well the beta cells in the pancreas are working.
Your doctor may have prescribed GlucaGen® HypoKit for another reason.
Ask your doctor if you have any questions about why GlucaGen® HypoKit has been prescribed for you.
Before you use GlucaGen® HypoKit
When you must not use it
You should not use or be given GlucaGen® HypoKit if you have the following medical conditions:
- phaeochromocytoma (a rare tumour of the adrenal gland that raises blood pressure)
- glucagonoma (a pancreatic tumour that causes increased levels of glucagon)
- insulinoma (a tumour that causes increased levels of insulin)
- allergies to glucagon or any of the ingredients listed in the “Ingredients” section of this leaflet.
GlucaGen® HypoKit contains latex. The cap of the syringe contains latex rubber. This may cause severe allergic reactions in people who are allergic to latex.
Before you start to use it
GlucaGen® will not work properly:
- If you have been fasting for a long time
- If you have low levels of adrenaline
- If you have chronic hypoglycaemia
- If you have hypoglycaemia caused by drinking too much alcohol
- If you have a tumour that releases glucagon or insulin.
If any of these apply, get medical advice.
Also seek medical advice if you are taking a sulfonylurea tablet to treat your diabetes.
Tell your doctor before you are given GlucaGen® HypoKit in connection with medical investigations of your gastrointestinal tract if you:
- have diabetes
- are elderly and have a heart condition.
GlucaGen® HypoKit should not be given via intravenous infusion.
You should not use or be given GlucaGen® HypoKit if:
- it does not look like a powder or powdery tablet before adding the water
- the powder does not dissolve properly after adding the water, or the solution looks like a gel
- the expiry date (“Expiry”) printed on the pack and on the vial has passed
- the packaging is damaged or shows signs of tampering, the tamper-proof plastic caps of the vials are loose or missing, or the cover on the syringe needle is missing.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
In particular, tell your doctor if you are taking:
- warfarin - used to prevent blood clots
- beta-blockers - used to treat high blood pressure or other heart conditions
- insulin - used to treat diabetes
- indomethacin - used to reduce the pain and inflammation of various conditions e.g. arthritis.
These medicines may be affected by GlucaGen® HypoKit or may affect how well GlucaGen® HypoKit works. Your doctor or pharmacist will be able to tell you what to do if you use GlucaGen® HypoKit while taking other medicines.
How to use GlucaGen® HypoKit
The freeze-dried GlucaGen® powder should be dissolved in the water provided and then injected under the skin, into a muscle or - by a medical person only - into a vein.
Read the ‘Instructions for use’ section of this leaflet carefully in order to correctly prepare GlucaGen® HypoKit for injection.
What to do in case of severe hypoglycaemia (a severe “hypo”)
The first symptoms of a hypo can come on suddenly. They may include: cold sweat, cool pale skin, fatigue, nervousness or tremor, drowsiness, anxious feeling, unusual tiredness and weakness, confusion, difficulty in concentration, excessive hunger, vision changes, headache, nausea or rapid heart beat.
If you experience any of the symptoms mentioned above you should immediately take sugary food or drink e.g., lollies, biscuits or fruit juice. So always carry one of these with you.
Tell your relatives, friends and close workmates that you have diabetes and how they can help you if you have a severe hypo. Make sure they know you should not be given anything to eat or drink if you are unconscious because you could choke.
If you are unconscious, you should be turned on your side and your relatives, friends or workmates should get medical help immediately.
An injection of GlucaGen® HypoKit may speed up recovery from unconsciousness. This can be given by someone who has been instructed in its use. Make sure that your family, friends, carer or co-workers know how to use GlucaGen® and where it is kept before an emergency arises.
Injection by a non-medical person:
Always use GlucaGen® HypoKit exactly as your doctor has told you to. You should check with your doctor or pharmacist if you are not sure.
Inject the dose into the fatty tissue just below the skin of the thigh, buttocks or upper arm.
Adults and children above 25kg: Inject full dose (1mL, marked on syringe as 1/1).
Children below 25kg: Inject half dose (1/2mL, marked on syringe as 1/2).
The patient will normally respond within 10 minutes to the injection of glucagon. Sugary food or drink should be given when the patient regains consciousness. This will help to build up liver glucose stores again and prevent relapse. Medical help must be sought for an unconscious person.
If GlucaGen® HypoKit has been used, tell your doctor or diabetes educator as you may require a change to your diabetes treatment.
Injection by a medical person:
Give 0.5-1mg by subcutaneous, intramuscular or intravenous injection. If the patient does not respond within 10 minutes intravenous glucose should be given. When the patient has responded to treatment, oral carbohydrate should be given to restore the liver glycogen and prevent secondary hypoglycaemia.
Using GlucaGen® HypoKit for diagnostic tests
Note that a syringe with a thinner needle and a finer graduation may be more suitable for use in diagnostic procedures.
Investigations of the digestive tract: Your doctor will determine how much glucagon you should be given. The dose will depend on your body weight, the test and the time it will take. Doses usually vary from 0.2-2mg depending on the diagnostic technique used and how the injection is given. The diagnostic dose for relaxation of the stomach, duodenal bulb, duodenum and small bowel is 0.2-0.5mg given intravenously or 1mg given intramuscularly; the usual dose to relax the colon is 0.5-0.75mg intravenously or 1-2mg intramuscularly.
The effect after an injection of 0.2-0.5mg into a vein starts within one minute and lasts for between 5 and 20 minutes depending on the organ under examination. The effect after an injection into the muscle of 1-2mg starts after 5-15 minutes and lasts about 10-40 minutes depending on the organ.
Stimulation of insulin production: 1mg of glucagon is injected into a vein in a fasting person. Plasma Cpeptide is measured before and 6 minutes after injection.
When the diagnostic test is completed: If you have been fasting, you should be given oral carbohydrate when the diagnostic test has been completed to restore the liver glycogen and prevent occurrence of secondary hypoglycaemia. In the case of severe hypoglycaemia, intravenous glucose may be required.
If you use too much (overdose)
Immediately telephone your doctor, or the Poisons Information Centre on 13 11 26 (Australia) or 0800 764 766 (New Zealand), if you think you or anyone else may have used or been given too much glucagon. Do this even if there are no signs of discomfort or poisoning.
If you have been given too much glucagon, it may cause nausea and vomiting. Specific treatment is not usually necessary. The levels of potassium in the blood may decrease. Patients on beta-blocker medicines may experience a short term increase in blood pressure.
While you are using GlucaGen® HypoKit
Things to be careful of
- Do not give drinks or food to an unconscious person.
- GlucaGen® has the opposite effect to insulin. You need to take extra care if you are using GlucaGen® HypoKit in connection with endoscopy or radiography, if you are a person with diabetes, or if you are elderly and have a heart condition.
- GlucaGen® HypoKit will only work if there are stores of glucose (as glycogen) in the liver; therefore, it will not treat hypoglycaemia if you have been fasting, or have low levels of adrenaline, chronic hypoglycaemia, or hypoglycaemia caused by drinking too much alcohol.
- Because GlucaGen® depletes glycogen stores, you must be given sugary food or drink by mouth as soon as you are able to take it after you have responded to the treatment or after the diagnostic test is over. This will prevent the occurrence of secondary hypoglycaemia.
If you are not sure whether you should use or be given GlucaGen® HypoKit, ask your doctor, pharmacist or diabetes educator.
When driving or operating machines
If you are still experiencing the effects after a severe hypo, do not drive or operate machinery.
Pregnancy and breast-feeding
GlucaGen® does not cross the human placenta and may be used in the treatment of a severe hypo during pregnancy.
While breast-feeding, very small amounts of glucagon may reach the mother’s milk if she is treated with glucagon for a severe hypo. However, any glucagon in the milk will be broken down in the baby’s digestive tract making it harmless. Breast-feeding following treatment of a severe hypo with GlucaGen® HypoKit does not put your baby at risk.
Side Effects
Tell your doctor or pharmacist as soon as possible if you do not feel well after using GlucaGen® HypoKit. This medicine helps most people who use it for the recommended purposes, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
When GlucaGen® HypoKit is used to treat a severe hypo
Tell your doctor if you notice any of the following and they worry you:
- allergic reaction - signs are wheezing, sweating, rapid heart beat, rash, swollen face, collapse (anaphylactic reaction). Get medical help immediately if any of these occur.
- abdominal pain, nausea or vomiting.
When GlucaGen® HypoKit is used in a diagnostic procedure
Tell your doctor if you notice any of the following and they worry you:
- allergic reaction - signs are wheezing, sweating, rapid heart beat, rash, swollen face, collapse (anaphylactic reaction). Get medical help immediately if any of these occur.
- abdominal pain, nausea or vomiting
- mild to moderate hypoglycaemia (which can occur after a patient responds to treatment with GlucaGen® HypoKit. It can be more pronounced in patients who have fasted prior to a diagnostic procedure)
- temporarily increased blood pressure
- decreased blood pressure
- increased heart rate
- decreased heart rate.
Very rarely it has been reported that patients treated with GlucaGen® HypoKit have developed hypoglycaemic coma or experienced heart attacks.
Other side effects not listed above may also occur in some people.
Ask for the advice of your doctor or pharmacist if you have any concerns about the effects of using this medicine.
Storage
GlucaGen® HypoKit should be stored at room temperature (i.e. less than 25°C).
Avoid freezing to prevent damage to the glass syringe.
The GlucaGen® HypoKit powder vial should be stored in the original package to protect from light.
The expiry date (“Expiry”) is printed on the pack. If your medicine has passed this date, do not use it. Check the expiry date from time to time to make sure that the glucagon in your GlucaGen® HypoKit has not expired, in case of an emergency.
Due to the instability of GlucaGen® in solution, the product should be used immediately after reconstitution. Use once only and discard any residue. Do not store it for later use.
Keep out of the reach of children.
Disposal
Ask your pharmacist how to dispose of medicines no longer required.
Product Description
What it looks like
GlucaGen® HypoKit is supplied as a single dose pack. It includes one vial containing a sterile white freeze-dried powder (GlucaGen®) and a disposable glass syringe pre-filled with water (water for injections). The powder may appear more like a powdery tablet upon settling.
When the glucagon powder is reconstituted with the water for injections, it forms a solution of 1mg (1 IU) glucagon in each mL.
Ingredients
GlucaGen® HypoKit contains glucagon (rys) 1mg (corresponding to 1 IU), as hydrochloride, as the active ingredient. The abbreviation “rys” indicates the method of genetic engineering used to manufacture the glucagon.
The powder vial also contains lactose monohydrate 107mg, sodium hydroxide and/or hydrochloric acid.
Sponsor
GlucaGen® HypoKit is supplied in Australia by:
Novo Nordisk Pharmaceuticals Pty Ltd
Level 3
21 Solent Circuit
Baulkham Hills NSW 2153
GlucaGen® HypoKit is supplied in New Zealand by:
Novo Nordisk Pharmaceuticals Ltd
58 Richard Pearse Drive
Airport Oaks
Mangere
New Zealand
This leaflet was prepared on 10 February 2015.
Australian Registration Number: AUST R 47105
GlucaGen® and NovoCare® are registered trademarks of Novo Nordisk A/S.
© 2015
Novo Nordisk A/S
Further Information
For further information call the NovoCare® Customer Care Centre on 1800 668 626 (Australia) or 0800 733 737 (New Zealand).
- www.novonordisk.com.au
- www.novonordisk.co.nz
You can also get more information about diabetes and insulin from Diabetes Australia and Diabetes New Zealand:
- freecall helpline 1300 136 588 (Australia)
- www.diabetesaustralia.com.au
- www.diabetes.org.nz
GlucaGen® HypoKit
Glucagon (rys) hydrochloride
Instructions For Use
Preparing GlucaGen® HypoKit for injection:
The glucagon solution should be prepared immediately before use. The freshly prepared glucagon solution should be clear. It should not be injected if it contains solid particles or looks like a gel. The glucagon solution should not be stored for later use.
- Remove the plastic cap from the vial. Pull the needle cover off the syringe. Insert the needle through the rubber stopper (within the marked circle) of the vial containing GlucaGen® and inject all the liquid from the syringe into the vial.
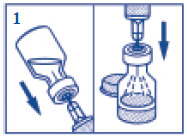
- Without taking the needle out of the vial, gently shake the vial until the GlucaGen® has completely dissolved and the solution is clear.

- Make sure the plunger is completely down. While keeping the needle in the liquid, slowly withdraw all the solution back into the syringe. Do not pull the plunger out of the syringe. It is important to remove any air bubbles from the syringe:
- With the needle pointing upwards, tap the syringe with your finger
- Push the plunger slightly to release any air that has collected at the top of the syringe. Continue to push the plunger until you have the correct dose for the injection. A small amount of liquid will be pushed out when you do this.

- Inject the dose under the skin or into a muscle. Please refer to the instructions on how to inject the solution.

Published by MIMS April 2015




