1 Name of Medicine
Glucose.
2 Qualitative and Quantitative Composition
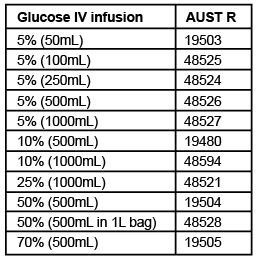
Baxter Glucose Intravenous (IV) Infusion preparations are sterile, non-pyrogenic parenteral solutions. They do not contain an antimicrobial agent or added buffer, and have a pH of 3.5-6.5 for 5% to 25% glucose concentrations, and a pH of 3.2 - 6.5 for 50% and 70% glucose concentrations. The concentrations of glucose dissolved in a litre of water for injections and their isotonicity are shown in Table 1 and 2.

 The glucose 5% intravenous infusion is an isotonic solution, whereas glucose ≥ 10% intravenous infusions are hypertonic.
The glucose 5% intravenous infusion is an isotonic solution, whereas glucose ≥ 10% intravenous infusions are hypertonic.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Clear slightly coloured solution for intravenous infusion.
4.1 Therapeutic Indications
Isotonic (glucose 5%) intravenous infusions are mainly indicated:
whenever nonelectrolyte fluid replacement is required;
as a vehicle for drug delivery, provided that the added components are compatible with glucose.
Hypertonic (glucose ≥ 10%) intravenous infusions are indicated:
as a source of energy incorporated with parenteral nutrition with minimal dilution effect;
for use with an appropriate protein (nitrogen) source in the prevention of nitrogen loss or in the treatment of negative nitrogen balance in patients where:
a) the alimentary tract cannot or should not be used;
b) gastrointestinal absorption of protein is impaired;
c) metabolic requirements for protein are substantially increased, as with extensive burns.
4.2 Dose and Method of Administration
To be used for intravenous administration as directed by the physician.
The infusion rate and volume of Baxter Glucose IV Infusion is dependent upon the age, weight, concomitant therapy, clinical and metabolic conditions of the patient as well as laboratory determinations. Electrolyte supplementation may be indicated according to the clinical needs of the patient. As indicated on an individual basis, vitamins and trace elements and other components (including amino acids and lipids) can be added to the parenteral regimen to meet nutrient needs and prevent deficiencies and complications from developing. For 25 to 70% glucose intravenous infusions, these are concentrated solutions that are components for use with amino acids only in parenteral nutrition (PN). Dilute hypertonic glucose intravenous solution before use to a concentration which will, when administered with an amino acid (nitrogen) source, result in an appropriate calorie to gram of nitrogen ratio and which has an osmolarity consistent with the route of administration.
When glucose is used as a diluent, the dosage administered will be principally dictated by the nature of the additive and the infusion rate will depend upon the dose regimen of the prescribed drug. Baxter Glucose 5% Intravenous Infusion may be administered intravenously to healthy individuals at a rate of 0.5 g/kg per hour without producing glycosuria; the maximum infusion rate should not exceed 0.8 g/kg per hour.
A hypertonic glucose intravenous infusion, such as Baxter Glucose 10% IV Infusion (556 mOsmol/L), if undiluted, is intended to be administered via intravenous catheter in a large central vein. A gradual increase of flow rate should be considered when starting administration of hypertonic glucose intravenous infusions. To reduce the risk of hypoglycaemia after discontinuation, a gradual decrease in flow rate before stopping the infusion should be considered. The usual dose is 20-50 mL of 50% glucose injection administered slowly, at a rate of 3 mL/minute. If a peripheral vein is used, a large arm vein should be selected and the infusion site should be changed daily.
Parenteral drug products should be inspected visually for particulate matter and discolouration prior to the administration (see Section 4.4 Special Warnings and Precautions for Use); only sterile and nonpyrogenic equipment must be used for intravenous administration. Do not administer unless the solution is clear and the seal is intact. Use of an in-line filter is recommended during administration of all parenteral solutions where possible.
Baxter Glucose IV Infusion is for single use only. Discard any unused portion. Do not reconnect partially used bags.
Additives may be introduced before infusion or during infusion through the injection site. Additives may be incompatible. Consult with pharmacist, if available. Check additive compatibility with both the solution and container prior to use. Complete information is not available. Those additives known to be incompatible should not be used. Before adding a substance or medication, verify that it is soluble and/or stable in water and that the pH range of Baxter Glucose IV Infusion is appropriate. The instructions for use of the medication to be added and other relevant literature must be consulted. When introducing additives to Baxter Glucose IV Infusion, aseptic technique must be used. After addition, check for a possible color change and/or the appearance of precipitates, insoluble complexes or crystals. Thorough and careful aseptic mixing of any additive is mandatory. Solutions containing additives should be used immediately and not stored.
The osmolarity of a final admixed infusion solution must be taken into account when peripheral administration is considered. Administration of hyperosmolar solutions may cause venous irritation and phlebitis.
Direction for use of Viaflex plastic container.
Do not remove unit from overwrap until ready for use. The inner bag maintains the sterility of the product.
Do not connect plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed. Pressurising intravenous solutions contained in flexible plastic containers to increase flow rate can also result in air embolism if the residual air in the container is not fully evacuated prior to administration. Use of a vented intravenous administration set with the vent in the open position could also result in air embolism. Vented intravenous administration sets with the vent in the open position should not be used with flexible plastic containers.
To open.
Tear overwrap down side at slit and remove solution container. Check solution for limpidity and absence of foreign matter. If solution is not clear or contains foreign matter, discard the solution. Some opacity of the plastic due to moisture absorption during the sterilisation process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually.
Check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution, as sterility may be impaired.
If supplemental medication is desired, follow directions below.
Preparation for administration.
Baxter Glucose IV Infusion is a sterile preparation. Thus, aseptic technique must be applied throughout the administration.
(1) Suspend container from eyelet support.
(2) Remove plastic protector from outlet port at the bottom of container.
(3) Attach administration set; use an aseptic method to set up the infusion.
To add medications.
Warning: Additives may be incompatible. Check the product information document(s) of the medication(s) and other relevant literature prior to their addition to Baxter Glucose IV Infusion.
To add medication before solution administration.
Prepare medication site. Using syringe with 19 to 22 gauge needle, puncture resealable medication port and inject. Mix solution and medication thoroughly. For high density medication such as potassium chloride, squeeze ports while ports are upright and mix thoroughly.
To add medication during solution administration.
Close clamp on the set. Prepare medication site. Using syringe with 19 to 22 gauge needle, puncture resealable medication port and inject. Remove container from IV pole and/or turn to upright position. Evaluate both ports by squeezing them while container is in the upright position. Mix solution and medication thoroughly. Return container to in use position, reopen the clamp and continue administration.4.3 Contraindications
The infusion of hypertonic glucose (≥ 10%) preparations are contraindicated in patients:
having intracranial or intraspinal haemorrhage;
with delirium tremens or those who are severely dehydrated;
who are anuric;
with diabetic coma.
Infusion of both isotonic and hypertonic glucose preparations are contraindicated in patients:
who have had head trauma within 24 hours, with blood glucose concentrations being closely monitored during intracranial hypertension;
with known hypersensitivity to the product;
with known allergy to corn or corn products, because cornstarch is used as raw material for glucose production;
with clinically significant hyperglycaemia.
Avoid use after an ischaemic stroke episode as under this condition, the induced lactic acidosis aggravates the recovery of the brain damage tissue.
4.4 Special Warnings and Precautions for Use
General.
The safety of the Viaflex plastic container used in Baxter Glucose IV Infusion preparations has been confirmed in tests with animals according to the USP biological tests for plastic container, as well as by tissue culture toxicity studies. Nevertheless, care should be exercised regarding a possible incompatibility outcome resulting either from the interaction between the plastic container or active ingredients and the added therapeutic substances (also see Section 4.2 Dose and Method of Administration).
The introduction of additives to any solution, regardless of type of container, requires special attention to assure that no incompatibilities results. While some incompatibilities are readily observed, one must be aware that subtle physical, chemical and pharmacological incompatibilities can occur. The medical literature, the package insert and other available sources of information should be reviewed for thorough understanding of possible incompatibility problems. In particular, the product information document of any added medication should be checked for any incompatibility with glucose infusions.
Do not administer Baxter Glucose IV Infusion unless the solution is clear and the seals intact.
Hypersensitivity reactions.
Hypersensitivity/ infusion reactions, including anaphylactic/ anaphylactoid reactions, have been reported with Baxter Glucose IV Infusion. The infusion must be stopped immediately if any signs or symptoms of a suspected hypersensitivity reaction develop. Appropriate therapeutic countermeasures must be instituted as clinically indicated.
Dilution and other effects on serum electrolytes.
The administration of Baxter Glucose IV Infusion can cause fluid and/or solute overloading resulting in dilution of the serum electrolyte concentrations, over hydration, congested states, or pulmonary oedema. The risk of dilution states is inversely proportional to the electrolyte concentrations of the injections. The risk of solute overload causing congested states with peripheral and pulmonary oedema is directly proportional to the electrolyte concentrations of the injections. Excessive administration of high concentration Baxter Glucose IV Infusion may result in significant hypokalaemia, requiring possible administration of concomitant potassium.
Depending on the volume and rate of infusion and depending on a patient's underlying clinical condition and capability to metabolise glucose, intravenous administration of glucose can cause:
hyperosmolality, osmotic diuresis and dehydration;
hypoosmolality;
electrolyte disturbances such as hypo- or hyperosmotic hyponatraemia (see below), hypokalaemia, hypophosphataemia, hypomagnesaemia, overhydration/ hypervolaemia and, for example, congested states, including pulmonary congestion and oedema.
The above effects do not only result from the administration of electrolyte-free fluid but also from glucose administration. In addition:
An increase in serum glucose concentration is associated with an increase in serum osmolality. Osmotic diuresis associated with hyperglycaemia can result in or contribute to the development of dehydration and in electrolyte losses.
Hyperglycaemia also causes a transcellular shift of water, leading to a decrease in extracellular sodium concentrations and hyponatraemia.
Since glucose is metabolised, infusion of glucose solution corresponds to increasing the body's load of free water, possibly leading to hypoosmotic hyponatraemia.
Monitoring of serum sodium is particularly important. High volume infusion must be used under specific monitoring in patients with cardiac or pulmonary failure, and in patients with non-osmotic vasopressin release (including SIADH), due to the risk of hospital-acquired hyponatraemia.
Acute hyponatraemia can lead to acute hyponatraemic encephalopathy (cerebral oedema) characterised by headache, nausea, seizures, lethargy and vomiting which can lead to coma, and death. Patients with cerebral oedema are at particular risk of severe, irreversible and lifethreatening brain injury. Acute symptomatic hyponatraemic encephalopathy is considered a medical emergency.
The risk for developing hypoosmotic hyponatraemia is increased, for example: in children; in elderly patients; in women; postoperatively; in persons with psychogenic polydipsia.
The risk for developing encephalopathy as a complication of hypoosmotic hyponatraemia is increased, for example:
in paediatric patients (≤ 16 years of age);
in women (in particular, premenopausal women);
in patients with hypoxemia;
in patients with underlying central nervous system disease.
Clinical evaluation and periodic laboratory determinations may be necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during prolonged parenteral therapy or whenever the condition of the patient or the rate of administration warrants such evaluation.
Particular caution is advised in patients at increased risk of and from water and electrolyte disturbances that could be aggravated by increased free water load. Hyperglycaemia or possibly required insulin administration (see Hyperglycaemia below).
Preventive and corrective measures must be instituted as clinically indicated.
Hyperglycaemia.
As with the intravenous administration of nutrients (e.g. glucose, amino acids and lipids) in general, metabolic complications may occur if the nutrient intake is not adapted to the patient's requirements, or the metabolic capacity of any given dietary component is not accurately assessed. Adverse metabolic effects may arise from administration of inadequate or excessive nutrients or from inappropriate composition of an admixture for a particular patient's needs.
Rapid administration of glucose solutions may produce substantial hyperglycaemia and a hyperosmolar syndrome. In order to avoid hyperglycaemia the infusion rate should not exceed the patient's ability to utilise glucose. To reduce the risk of hyperglycaemia-associated complications, the infusion rate must be adjusted and/or insulin administered if blood glucose levels exceed levels considered acceptable for the individual patient.
Intravenous glucose solution should be administered with caution in patients with, for example:
impaired glucose tolerance (such as in diabetes mellitus, renal impairment, or in the presence of sepsis, trauma, or shock);
severe malnutrition (risk of precipitating a refeeding syndrome);
water and electrolyte disturbances that could be aggravated by increased glucose and/or free water load.
Thiamine diphosphate, cocarboxylase, is an essential co-enzyme in the carbohydrate metabolism; therefore, patients having thiamine deficiency, e.g. in patients with chronic alcoholism (risk of severe lactic acidosis due to impaired oxidative metabolisation of pyruvate), should be treated cautiously with glucose intravenous infusions.
Baxter Glucose IV Infusions should be used with caution in patients with overt or subclinical diabetes mellitus (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Other groups of patients in whom glucose intravenous infusions should be used with caution include:
Patients with ischemic stroke. Hyperglycaemia has been implicated in increasing cerebral ischemic brain damage and impairing recovery after acute ischemic strokes (see Section 4.3 Contraindications).
Patients with severe traumatic brain injury. Early hyperglycaemia has been associated with poor outcomes in patients with severe traumatic brain injury (also see Section 4.3 Contraindications).
Newborns (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use).
Prolonged intravenous administration of glucose and associated hyperglycaemia may result in decreased rates of glucose-stimulated insulin secretion.
Refeeding syndrome.
Refeeding severely undernourished patients may result in the refeeding syndrome that is characterised by the shift of potassium, phosphorus, and magnesium intracellularly as the patient becomes anabolic. Thiamine deficiency and fluid retention may also develop. Careful monitoring and slowly increasing nutrient intakes while avoiding overfeeding can prevent these complications.
Liver disorders.
Hepatobiliary disorders including cholestasis, hepatic steatosis, fibrosis and cirrhosis, possibly leading to hepatic failure, as well as cholecystitis and cholelithiasis are known to develop in some patients on parenteral nutrition. The etiology of these disorders is thought to be multifactorial and may differ between patients. Patients developing abnormal laboratory parameters or other signs of hepatobiliary disorders should be assessed early by a clinician knowledgeable in liver diseases in order to identify possible causative and contributory factors, and possible therapeutic and prophylactic interventions.
Catheter infection and sepsis.
Infection and sepsis may occur as a result of the use of intravenous catheters to administer parenteral formulations, poor maintenance of catheters or contaminated solutions.
Immunosuppression and other factors such as hyperglycaemia, malnutrition and/or their underlying disease state may predispose patients to infectious complications.
Careful symptomatic and laboratory monitoring for fever/ chills, leukocytosis, technical complications with the access device, and hyperglycaemia can help recognise early infections. The occurrence of septic complications can be decreased with heightened emphasis on aseptic technique in catheter placement, maintenance, as well as aseptic technique in nutritional formula preparation.
Precipitates.
Pulmonary vascular precipitates have been reported in patients receiving parenteral nutrition. In some cases, fatal outcomes have occurred. Excessive addition of calcium and phosphate increases the risk of the formation of calcium phosphate precipitates. Precipitates have been reported even in the absence of phosphate salt in the solution. Precipitation distal to the in-line filter and suspected precipitate formation in the blood stream has also been reported.
In addition to inspection of the solution, the infusion set and catheter should also periodically be checked for precipitates.
If signs of pulmonary distress occur, the infusion should be stopped and medical evaluation initiated.
Others.
In a dilute condition, osmolarity/L is approximately the same as osmolality/kg. As shown in Table 2 (see Section 6.5 Nature and Contents of Container), high concentration glucose (≥ 10%) intravenous infusions are hypertonic, whereas the 5% preparations are isotonic (278 mOsmol/L, Table 1 (see Section 6.5 Nature and Contents of Container). The glucose 70% intravenous infusion is a concentrated preparation that should be diluted prior to infusion. However, the glucose 50% intravenous infusion should be administered without dilution in the management of severe hypoglycaemia by slow infusion via central vein, such as insulin induced hypoglycaemia. Undiluted glucose 50% should not be administered through peripheral veins.
Administration of substantially hypertonic solutions may lead to a wide variety of complications. These include crenation (shrinkage) of red blood cells and general cellular dehydration. Prolonged intravenous infusion of hypertonic glucose solutions may cause thrombophlebitis extending from the site of infusion. Thus, unless appropriately diluted, the infusion of hypertonic glucose injection solution into a peripheral vein may result in vein irritation, vein damage and thrombosis. Strongly hypertonic nutrient solutions should only be administered through an indwelling intravenous catheter with the tip located in a large central vein such as the superior vena cava. Care should be taken to avoid circulatory overload, particularly in patients with cardiac insufficiency.
Similarly, administration of hypertonic glucose intravenous infusions and amino acid solutions via central venous catheter may be associated with complications that can be prevented or minimised by careful attention to all aspects of procedure.
Glucose tolerance may be impaired in patients with renal failure. In patients with deficiency of sodium, administration of hypertonic Baxter Glucose IV Infusion without sodium may lead to peripheral collapse and oligouria.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance, electrolyte concentrations, and acid-base balance during prolonged parenteral therapy or whenever the condition of the patients warrants such evaluation.
Use in the elderly.
When selecting the type of infusion solution and the volume/ rate of infusion for a geriatric patient, consider that geriatric patients are generally more likely to have cardiac, renal, hepatic, and other diseases or concomitant drug therapy.
Paediatric use.
The infusion rate and volume depends on the age, weight, clinical and metabolic conditions of the child and concomitant therapy. Only consulting physicians experienced in paediatric intravenous fluid therapy should determine glucose intravenous infusion rate and volume.
Hypo-/ hyperglycaemia.
Neonates, especially those born premature and with low birth weight, are at increased risk of developing hypo- or hyperglycaemia. Close monitoring during treatment with intravenous glucose solutions is needed to ensure adequate glycaemic control in order to avoid potential long-term adverse effects. Hypoglycaemia in the neonate can cause prolonged seizures, coma and cerebral injury. Hyperglycaemia has been associated with cerebral injury (including intraventricular haemorrhage), late onset bacterial and fungal infection, retinopathy of prematurity, necrotizing enterocolitis, increased oxygen requirements, bronchopulmonary dysplasia, prolonged length of hospital stay, and death.
Hyponatraemia.
Children (including neonates and older children) are at increased risk of developing hypoosmotic hyponatraemia as well as for developing hyponatraemic encephalopathy. Acute hyponatraemia can lead to acute hyponatraemic encephalopathy (cerebral oedema) characterised by headache, nausea, seizures, lethargy, and vomiting which can lead to coma, and death. Patients with brain oedema are at particular risk of severe, irreversible and lifethreatening brain injury. Acute symptomatic hyponatraemic encephalopathy is considered a medical emergency. Plasma electrolyte concentrations should be closely monitored in the paediatric population. Rapid correction of hypoosmotic hyponatraemia is potentially dangerous (risk of serious neurologic complications). Dosage, rate, and duration of administration should be determined by a physician experienced in paediatric intravenous fluid therapy.
Effects on laboratory tests.
The effect of this medicine on laboratory tests has not been established.4.5 Interactions with Other Medicines and Other Forms of Interactions
Baxter Glucose IV Infusion preparations (an aqueous, i.e. electrolyte-free glucose solution) should not be administered simultaneously with blood preparations through the same administration set, because of the possibility of pseudoagglutination or haemolysis.
Both the glycaemic effects of Baxter Glucose IV Infusion and its effects on water and electrolyte balance should be taken into account when using Baxter Glucose IV Infusion in patients treated with other substances that affect glycaemic control, or fluid and/or electrolyte balance. Use of Baxter Glucose IV Infusion may necessitate review of a patient's oral hypoglycaemic or insulin requirements, so close monitoring of serum glucose levels is required.
Caution is advised when administering Baxter Glucose IV Infusion to patients treated with drugs leading to an increased vasopressin effect. The below listed drugs increase the vasopressin effect, leading to reduced renal electrolyte free water excretion and may increase the risk of hyponatraemia following treatment with IV fluids. (See Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
Drugs stimulating vasopressin release such as chlorpropamide, clofibrate, carbamazepine, vincristine, selective serotonin reuptake inhibitors (SSRIs), 3.4-methylenedioxy-N-methamphetamine, ifosfamide, antipsycotics, opioids.
Drugs potentiating vasopressin action such as chlorpropamide, non-steroidal antiinflammatories (NSAIDS), cyclophosphamide.
Vasopressin analogues such as desmopressin, oxytocin, vasopressin, terlipressin.
Caution is advised when administering Baxter Glucose IV Infusion to patients treated with drugs that may increase the risk of hyponatraemia, such as diuretics and antiepileptics (e.g. oxcarbazepine).
Caution must be taken with hypertonic Baxter Glucose IV Infusion in patients receiving corticosteroids or corticotropin.
See Section 6.2 Incompatibilities.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category C)
Animal reproduction studies have not been conducted with Baxter Glucose IV Infusion. It is also not known whether Baxter Glucose IV Infusion cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Intrapartum maternal intravenous glucose infusion may result in foetal insulin production, with an associated risk of foetal hyperglycaemia and metabolic acidosis as well as rebound hypoglycaemia in the neonate.
Physicians should carefully consider the potential risks and benefits for each specific patient before administering Baxter Glucose IV Infusion preparations.
Safety in lactation has not been established. Use of Baxter Glucose Intravenous Infusion in nursing woman only when it is clearly needed and the potential benefits outweigh the potential risks to the baby.4.7 Effects on Ability to Drive and Use Machines
There is no information on the effects of Baxter Glucose IV Infusion on the ability to operate an automobile or other heavy machinery.
4.8 Adverse Effects (Undesirable Effects)
Intravenous infusion of glucose can lead to the development of fluid and electrolytes disturbances including hypokalemia, hypomagnesaemia, and hypophosphatemia.
Hyperglycaemia and dehydration have resulted from inappropriate parenteral use. If administered to diabetic patients, insulin requirements may be modified (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Reactions that may occur because of the solution (e.g. from contamination), additive drugs or techniques of administration include fever response (due to possible introduction of pyrogens), infection at the site of injection, venous thrombosis or phlebitis extending from the site of injection, extravasation and hypervolemia. In case of such adverse reactions, the infusion should be stopped. If hypertonic glucose solutions are infused too rapidly, local pain and, rarely, vein irritation may occur.
It has been reported that a high concentration of glucose administered by intravenous route may induce a release of histamine leading to anaphylactoid manifestation, hypersensitivity, pyrexia and chills. This adverse reaction was reported in two patients with both asthma and diabetes mellitus. Rapid infusion of 25 to 50 g of glucose [equivalent to 50 mL to 100 mL of glucose (50%)] over 3 minutes may occasionally cause a generalised flush, which subsides within 10 minutes (see Section 4.4 Special Warnings and Precautions for Use).
Hyperglycaemia and glucosuria may occur if the rate of infusion is greater than 0.5 g/kg/h particularly with hypertonic Baxter Glucose IV Infusion; if undetected and untreated, this can lead to diuresis, dehydration, hyperosmolar coma and death. Continual clinical monitoring is recommended (see Section 4.4 Special Warnings and Precautions for Use). Vitamin B complex deficiency, thiamine and pantothenic acid in particular may occur in patients under prolonged parenteral nutrition.
If an adverse reaction does occur, discontinue the infusion, evaluate the patient, institute appropriate therapeutic countermeasures and save the remainder of the fluid for examination if deemed necessary. The nature of any additives should be considered in the event of other undesirable effects.
Post-marketing adverse reactions.
The following adverse reactions have been reported in the post-marketing experience, listed by MedDRA System Organ Class (SOC), then where feasible, by preferred term in order of severity.
Immune system disorders.
Hypersensitivity/ infusion reactions, including anaphylactic/ anaphylactoid reactions, including reactions with mild manifestations, e.g. pruritus, and reactions with severe manifestations, e.g. bronchospasm, cyanosis, angioedema and hypotension, pyrexia, chills
Metabolism and nutrition disorders.
Hyperglycaemia.
Vascular disorders.
Phlebitis.
Skin and subcutaneous tissue disorders.
Rash.
General disorders and administration side conditions.
Infusion site reactions, infusion site phlebitis, infusion site erythema.
Other adverse reactions (class reactions).
Other adverse reactions reported with other similar products include:
hyponatraemia (which may be symptomatic);
hyponatraemic encephalopathy;
infusion site thrombophlebitis (associated with hyperosmolar solutions);
adverse reactions reported with parenteral nutrition to which the glucose component may play a causal or contributory role include: hepatic failure, hepatic cirrhosis, hepatic fibrosis, cholestasis, hepatic steatosis, blood bilirubin increased, hepatic enzyme increased, cholecystitis, cholelithiasis, pulmonary vascular precipitates.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
Excessive administration of Baxter Glucose IV Infusion can cause hyperglycaemia, adverse effects on water and electrolyte balance, and corresponding complications (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)). For example, severe hyperglycaemia and severe dilutional hyponatraemia, and their complications, can be fatal. Clinically significant overdose of Baxter Glucose IV Infusion may, therefore, constitute a medical emergency.
Symptoms.
Prolonged administration or rapid infusion of large volumes of isotonic solutions may cause oedema or water intoxication. Typical overdosage is manifested by symptoms of hyperglycaemia and glycosuria. If these symptoms are not detected and treated, they can lead to dehydration, mental confusion, hyperosmolar coma and death.
Typical initial signs of hypertonic solution overdose are extracellular fluid excess, hyperglycaemia, lowering of haemoglobin and haematocrit, lowering of serum electrolyte concentrations, potassium transfer from the cell to the extracellular space and increased plasma osmolarity. In patients with intact renal function, overdosage causes osmotic diuresis to a greater or lesser extent, commensurate with the hyperosmolarity of the infused solution, accompanied by a concomitant loss of electrolytes, especially potassium. Through increased water binding of the infused hypertonic carbohydrate solution, overdosage may lead to dehydration to a greater or lesser extent during the course of osmotic diuresis. Dehydration is characterised by lowering of initially raised plasma osmolarity. Haemoglobin and haematocrit, which are lowered immediately after overdosage, may thus return more or less to normal during the course of diuresis.
If diuresis is slow to develop, metabolic disturbances associated with glucose overdose may occur, characterised in particular by increased lactic acid build up and lowering of pH. If diuresis does not occur, this may result in symptoms of circulatory overload, in particular oedema (including pulmonary oedema) and heavy intracellular potassium loss.
The signs and symptoms of over infusion will also be related to the nature of any additive drugs.
Treatment.
The infusion should be discontinued and the patient observed for appropriate signs and symptoms related to glucose and/or additive drugs administered, and appropriate symptomatic and supportive measures instituted as required, such as administration of insulin.
Fluid overload and biochemical imbalance resulting from overdosage with glucose should be treated with appropriate corrective therapy. If diuresis is adequate, administration of a slightly hypotonic electrolyte solution in a quantity calculated to replace the net quantity of fluid and specific electrolytes (particularly potassium) lost to osmodiuresis, whilst continuously monitoring serum electrolytes, fluid balance and acid-base status is recommended.
A suitable basic solution for replacing fluids and major electrolytes could be made up according to the following formulation per 1000 mL: Na+: approx. 120 mmol, K+: approx. 30 mmol, Cl-: approx. 150 mmol. Other electrolytes should also be replaced to make up for losses incurred.
In addition to replacement of net losses of fluids and electrolytes to diuresis, any acid-base imbalance should be corrected whilst continuing to monitor laboratory values.
In patients with oliguria or those with anuria, peritoneal dialysis or extracorporeal haemodialysis using carbohydrate free solutions can be considered as a last resort.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Glucose is readily metabolised into carbon dioxide and water, with a release of energy. As such, an administration of a glucose solution either by oral or parenteral route provides water for body hydration as well as calories. In addition, it may reduce catabolic loss of nitrogen from the body and aids in prevention of depletion of liver glycogen. That is, in the absence of glucose, amino acids undergo deamination. It is followed by oxidation, with a release of energy. Thus, Baxter Glucose IV Infusions have value as a source of water and energy.
Glucose is readily converted into fat in the body which can be used as a source of energy as required. Under a similar conversion into storage of energy, glucose is stored in the liver and muscles as glycogen. For a quick rise in plasma glucose, glycogen is readily converted into glucose.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
A glucose preparation administered by the oral route is rapidly absorbed from the gastrointestinal tract by an active mechanism. Following an oral administration in hypoglycaemic individuals, plasma glucose is built up within 10-20 minutes and peaks at about 40 minutes.
As Baxter Glucose IV Infusion preparations are directly administered to the systemic circulation by infusion, the bioavailability of the active components is complete (100%).
5.3 Preclinical Safety Data
Genotoxicity.
The active ingredient, glucose, in Baxter Glucose IV Infusion is not a mutagen. It is a basic nutrient in all living cells.
Carcinogenicity.
The active ingredient, glucose, in Baxter Glucose IV Infusion is not a carcinogen. It is a basic nutrient in all living cells.6 Pharmaceutical Particulars
6.1 List of Excipients
Water for injections.
6.2 Incompatibilities
Additives may be incompatible. Consult with pharmacist, if available. Check the product information document(s) of the medication(s) and other relevant literature prior to their addition to Baxter Glucose IV Infusion. Complete information is not available. Those additives known to be incompatible should not be used (see Section 4.2 Dose and Method of Administration).
Baxter Glucose IV Infusion preparations (an aqueous, i.e. electrolyte-free glucose solution) should not be administered simultaneously with blood preparations through the same administration set, because of the possibility of pseudo-agglutination or haemolysis.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
Exposure to the heat should be minimised. Avoid excessive heat.
6.5 Nature and Contents of Container
Baxter Glucose Intravenous Infusion preparations are sterile, non-pyrogenic solutions supplied in Viaflex plastic bags, either as a single unit or multiple units in a box. See Section 2 Qualitative and Quantitative Composition, for information on pack sizes.
6.6 Special Precautions for Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Glucose (D-(+)glucopyranose).
Molecular formula: C6H12O6. Molecular Weight: 180.2.
Appearance: a white crystal or granular powder.
Solubility: freely soluble in water, sparingly soluble in ethanol (96%).
Chemical structure.

CAS number.
CAS No.: 50-99-7.7 Medicine Schedule (Poisons Standard)
Unscheduled.
Summary Table of Changes



 The glucose 5% intravenous infusion is an isotonic solution, whereas glucose ≥ 10% intravenous infusions are hypertonic.
The glucose 5% intravenous infusion is an isotonic solution, whereas glucose ≥ 10% intravenous infusions are hypertonic.
