What is in this leaflet
This leaflet answers some of the common questions people ask about Go-Kit.
It does not contain all the information that is known about Go-Kit. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor will have weighed the risks of you taking Go-Kit against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Go-Kit is used for
Go-Kit is used for bowel cleansing in conjunction with barium meal, barium enema, air contrast examination and intravenous pyelography (IVP) of the kidneys.
The bowel needs to be clean before your doctor can examine it properly.
Go-Kit produces watery stools or bowel motions within 2 to 3 hours after the first administration and may cause frequent loose bowel motions during the night.
This medicine belongs to a group of medicines called Bowel Preparations.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Before you take Go-Kit
When you must not take it
Do not take Go-Kit if you have an allergy to:
- Any of the ingredients listed at the end of this leaflet
- Any similar medicines to Go-Kit
Some symptoms of an allergic reaction may include:
- Shortness of breath
- Wheezing or difficulty breathing
- Swelling of the face, lips, tongue or other parts of the body
- Skin rash, itching or hives
Do not take Go-Kit if you have, or have had any of the following medical conditions:
- Severe dehydration
- Appendicitis
- Gastrointestinal obstruction
- Gastric retention
- Bowel perforation
- Toxic megacolon
- A condition known as Paralytic Ileus where the small bowel does not work properly
- Toxic colitis
- A body weight less than 20kg
Caution should be exercised in “at risk” patients such as the elderly who are more at risk of dehydration as electrolyte depletion may occur. If you are elderly, ensure you receive adequate fluids during the administration of Go-Kit.
Go-Kit is not recommended in children as the safety and effectiveness in children has not been established.
Do not take Go-Kit after the expiry date printed on the pack, or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure when you should start taking Go-Kit, talk to your doctor or pharmacist.
Before you start to take it
Tell your doctor or pharmacist if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any medical conditions especially the following:
- Inflammatory bowel disease
- Kidney problems
- Heart problems
- Diabetes
- Dehydration
- Stoma
- Undiagnosed stomach pain
It may not be safe for you to take Go-Kit if you have these conditions.
Tell your doctor or pharmacist if you are pregnant, intend to become pregnant or are breast feeding. Go-Kit is not generally recommended for use in pregnant women unless the benefits outweigh the risk to the unborn baby. Your doctor will discuss the benefits and possible risks with you.
Tell your doctor if you are diabetic. The liquid diet recommended with this medication may affect your blood glucose levels and adjustment of your diabetic medication may be required.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Medications that are taken just before or during the course of Go-Kit may not be absorbed. This is due to the increased movement in the digestive tract and the watery diarrhoea that is caused by Go-Kit. These include:
- Oral contraceptives ‘the pill’
- Antibiotics
- Medicines for diabetes.
Your doctor or pharmacist will have more information on medicines to be careful with or avoid while taking Go-Kit.
How to take Go-Kit
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box or in this leaflet, ask your doctor or pharmacist for help.
Go-Kit will produce numerous bowel motions within a short period of time. Ensure you have bathroom facilities within easy access from the time you start taking Go-Kit.
How much to take
Go-Kit contains only enough medication for one treatment.
How to take it
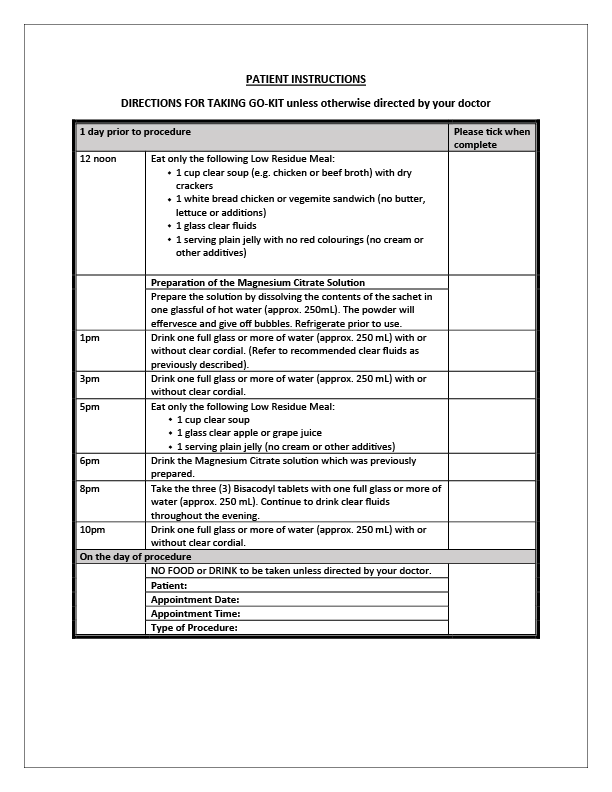
It should be prepared and taken according to the directions in this leaflet unless otherwise directed by your doctor.
Swallow the tablets whole.
Do not crush or chew the tablets.
Preparation for the procedure begins at 12 noon on the day before the procedure commencing with a low residue meal. (Refer to directions leaflet).
Recommended Clear Fluids include water, strained fruit juice without pulp (apple, white grape, pineapple, pear), clear broth, tea or coffee (without milk/cream), clear sugar-free cordials such as lemon or lime (no red or purple colourings), plain sugar-free jelly and clear ice blocks.
Do not drink carbonated beverages.
Do not drink alcoholic beverages.
REMEMBER you need to be close to toilet facilities whilst you are taking Go-Kit. Bowel movements may continue for several hours after the last dose of Go-Kit has been taken.
If you take too much (overdose)
Overdosage is unlikely as Go-Kit contains only enough medication for one treatment. However in the event of an overdose, dehydration is likely and immediate action should be taken to restore electrolyte balance with appropriate fluid replacement.
Contact the Poisons Information Centre for any further information.
Australia: 13 11 26
New Zealand: 0800 764 766
While you are taking Go-Kit
Things you must do
You must ensure that you drink the recommended amount of liquid to replace the large amounts of fluid that may be lost during bowel emptying.
Things you must not do
Do not take any additional bowel preparation or laxative products.
Do not give Go-Kit to anyone else, even if they have the same condition as you.
Do not stop taking Go-Kit or lower the dose without checking with your doctor.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Go- Kit.
Go-Kit helps most people that require bowel cleansing, but it may have unwanted side effects in a few people. All medicines can have some unwanted side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
The following is a list of possible side effects. Tell your doctor or pharmacist if you notice any of the following and they worry you:
- Nausea (feeling sick)
- Vomiting
- Stomach pain
- Stomach bloating
- Anal irritation
- Allergic reaction
Do not be alarmed by this list. You may not experience any of them.
If the effects are severe, you may need medical treatment.
However these side effects usually disappear when treatment with Go-Kit is finished.
If you get any side effects, do not stop taking Go-Kit without first talking to your doctor or pharmacist.
Other side effects not listed may also occur in some patients. Tell your doctor or pharmacist if you notice anything unusual that is making you feel unwell.
Further information
Storage
Keep Go-Kit in a cool dry place where the temperature stays below 25°C.
Do not store Go-Kit or any other medicine in the bathroom or near a sink.
Do not leave it on a window sill or in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one and a half metres above ground is a good place to store medicines.
Disposal
If your doctor or pharmacist tells you to stop taking Go-Kit or it has passed the expiry date, ask your pharmacist what to do with any medicine that is left over.
Product description
A composite pack containing a blister pack of three yellow Bisacodyl Tablets (5mg), a sachet containing 21.5g of dry white effervescent crystalline powder labelled Magnesium Citrate and a Consumer Medicine Information leaflet.
Ingredients
Each Go-Kit contains the following products:
Each Bisacodyl tablet contains 5 mg of bisacodyl as the active ingredient. It also contains: Lactose, sucrose, gelatin, starch, titanium dioxide (171), cellulose, macrogol and quinoline yellow (104).
Each sachet labelled Magnesium Citrate contains a white powder for solution, composed of magnesium carbonate 7.5g and citric acid 14g which together forms magnesium citrate in solution.
Go-Kit does not contain gluten, tartrazine or any other azo dyes.
Supplier
Fresenius Kabi Australia Pty Limited
Level 2, 2 Woodland Way
Mount Kuring-gai
NSW 2080
Tel: 1300 732 001
Fresenius Kabi New Zealand Limited
c/o Fresenius Medical Care Australia Pty Ltd
60 Pavilion Drive
Airport Oaks
Auckland
New Zealand
This leaflet was approved 2nd August 2012.
Australian Registration Number, AUST R 200381.
Published by MIMS July 2020







