What is in this leaflet
This leaflet answers some common questions about Hiprex tablets. It does not contain all the available information. It does not take the place of talking to your pharmacist or doctor.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet. You may need to read it again.
What Hiprex tablets are used for
Hexamine hippurate is used to suppress or eliminate urinary bacteria associated with chronic or recurrent infection.
Hiprex may be used for short or long term treatment.
Hexamine hippurate helps to acidify the urine and maintain a low urinary pH.
Hexamine hippurate has antibacterial activity.
Ask your pharmacist or doctor if you have any questions about this medicine. Your pharmacist or doctor may have given it for another reason.
Before you take Hiprex tablets
When you must not take it
Do not take this medicine if you have an allergy to:
- any medicine containing Hexamine (methenamine) hippurate.
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not take this medicine after the expiry date printed on the bottle or if the packaging is torn or shows signs of tampering.
If you are not sure whether you should start taking this medicine, talk to your pharmacist or doctor.
Before you start to take it
Tell your pharmacist if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your pharmacist if you have or have had any of the following medical conditions:
- kidney problems
- liver problems
Tell your pharmacist or doctor if you are pregnant or plan to become pregnant. Your pharmacist or doctor will discuss the benefits and possible risks of taking the medicine during pregnancy.
Ask your pharmacist or doctor about taking this medicine if you are breastfeeding. It is not known if hexamine hippurate passes into the breast milk. Your pharmacist or doctor will discuss the potential benefits and risks of taking the medicine if you are breastfeeding.
If you have not told your pharmacist or doctor about any of the above, tell him/her before you start taking Hiprex tablets.
Taking other medicines
Tell your pharmacist or doctor if you are taking any other medicines, including any that you get without a prescription or that are herbal from your pharmacy, supermarket or health food shop.
Some medicines and Hiprex may interfere with each other. These include:
- sulphur based drugs, this may include antibiotics, and medicines used to treat build up of fluids in the body, seizures or diabetes
- medicines taken to alkalise the urine
Your pharmacist or doctor will have more information on medicines to be careful with or avoid while taking Hiprex.
How to take Hiprex tablets
Follow all directions on the bottle or those given to you by your pharmacist or doctor carefully.
If you do not understand the instructions on the bottle, ask your pharmacist or doctor for help.
Hiprex can be taken with or without food.
How much to take
Adults & children over 12 years: - 1 g twice daily
Children 6-12 years - 500 mg to 1 g twice daily
Children under 6 years: - Do not use
When to take it
Take your medicine twice daily.
The maximum adult dose is 2 g per day.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre for advice, or go the Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too many Hiprex tablets. Do this even if there are no signs of discomfort or poisoning.
Poisons Information Australia:
Tel. 13 11 26
While you are using Hiprex tablets
Things you must do
Talk to your pharmacist or doctor if your symptoms do not improve. Your pharmacist or doctor will assess your condition and decide if you should continue to take the medicine.
Things you must not do
Do not take Hiprex tablets to treat any other complaints unless your pharmacist or doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not take more than the recommended dose unless your pharmacist or doctor tells you to.
Do not use this medicine in children under 6 years of age.
Things to be careful of
Some patients may have alkaline urine due to a vegetarian diet or an infection. As indicated by urinary pH or clinical response administration of ascorbic acid (1 g twice daily) is recommended.
Side effects
Tell your pharmacist or doctor as soon as possible if you do not feel well while you are taking Hiprex tablets.
This medicine helps most people, yet it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following list of possible side effects. You may not experience any of them.
Ask your pharmacist or doctor to answer any questions you may have.
Tell your pharmacist or doctor if you notice any of the following and they worry you:
- nausea
- upset stomach
- painful urination
- rash
- inflammation of the mouth
The above list includes the more common side effects of your medicine. They are usually mild.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare when used at the recommended dose for a short period of time.
Tell your pharmacist or doctor if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Before and after using Hiprex tablets
Storage
Keep your medicine in the original bottle until it is time to take it.
Keep your medicine in a cool dry place where the temperature stays below 30°C.
Do not store Hiprex or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep Hiprex out of reach of children. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
Ask your pharmacist what to do with any medicine that is left over, or if the expiry date has passed.
Product description
What it looks like
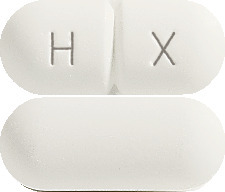
Hiprex tablets (1 g) are white capsule shaped, scored and marked HX on one side.
Hiprex is available in bottles of 20 and 100 tablets.
Ingredients
Active ingredient:
- hexamine hippurate
Other ingredients:
- magnesium stearate
- povidone
- silica - colloidal anhydrous
Hiprex is free from:
- sugar
- lactose
- gluten
- sodium
- artificial colours
Sponsor
iNova Pharmaceuticals (Australia) Pty Limited
ABN: 13 617 871 539
Level 10, 12 Help Street
Chatswood NSW 2067
Tel (Australia): 1800 630 056
™ = Trademark
This leaflet was prepared in November 2017.
Published by MIMS February 2018

 Occasionally superinfection with yeast may occur. At high dosage, chemical cystitis leading to dysuria may occur.
Occasionally superinfection with yeast may occur. At high dosage, chemical cystitis leading to dysuria may occur. Methenamine hippurate is a white to off-white powder. It is freely soluble in water.
Methenamine hippurate is a white to off-white powder. It is freely soluble in water.