What is in this leaflet
This leaflet answers some common questions about Hyrimoz.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you using this medicine against the benefits they expect it will have for you.
If you have any concerns about using this medicine, ask your doctor or pharmacist.
Read this leaflet carefully before you use Hyrimoz and keep it with the medicine. You may need to read it again.
What HYRIMOZ is used for
The active ingredient in this medicine is adalimumab, a fully human monoclonal antibody. Monoclonal antibodies are proteins made by a type of blood cell to fight a foreign protein in the body Adalimumab recognises and binds to a specific protein (tumour necrosis factor or TNF-alpha), which is present at increased levels in inflammatory diseases.
Hyrimoz is used for the treatment of:
- Rheumatoid arthritis
Hyrimoz is used to reduce the signs and symptoms of moderately to severely active rheumatoid arthritis, a painful disease of the joints, as well as to slow down and protect against damage to joints. Signs and symptoms of rheumatoid arthritis include joint pain, tenderness, swelling and stiffness. - Polyarticular Juvenile Idiopathic Arthritis
Hyrimoz is used for reducing the signs and symptoms of moderately to severely active polyarticular juvenile idiopathic arthritis, which is an inflammatory disease, involving multiple joints, in patients 2 years of age and older. - Enthesitis-related arthritis
Hyrimoz is used to treat enthesitis-related arthritis, an inflammatory disease of the joints in children. - Psoriatic arthritis
Hyrimoz is used to reduce the signs and symptoms, as well as inhibit the progression of joint damage of moderately to severely active psoriatic arthritis, a disease of the joints and skin, with some similarities to rheumatoid arthritis, as well as psoriasis and other factors. - Ankylosing spondylitis
Hyrimoz is used to reduce the signs and symptoms in patients with active ankylosing spondylitis, an inflammatory disease of the spine. Signs and symptoms of ankylosing spondylitis include back pain and morning stiffness. - Crohn's Disease
Hyrimoz is used for the treatment of moderate to severe Crohn's disease, an inflammatory disease of the digestive tract, in adults and children aged 6 years and above to reduce the signs and symptoms of the disease and to induce and maintain periods where the symptoms are no longer present. Hyrimoz can be given to patients who have not responded well enough to conventional therapies, or who have lost response to or are intolerant to these medicines. - Ulcerative Colitis
Hyrimoz is used for the treatment of moderate to severe ulcerative colitis, an inflammatory bowel disease, in patients who have not responded well enough to conventional therapy or who are intolerant to or have medical contraindications for such therapies. Patients should show a response within 8 weeks to continue treatment. - Psoriasis
Hyrimoz is used to treat the signs and symptoms of moderate to severe chronic plaque psoriasis, an inflammatory disease of the skin. Plaque psoriasis can also affect nails, causing them to crumble, thicken and lift away from the nail bed which can be painful.
Hyrimoz is used for moderate to severe forms of the disease in adults and severe forms in children and adolescents from 4 years of age who have not responded well enough to topical therapy and phototherapy, or who cannot be given those treatments. - Hidradenitis suppurativa
Hyrimoz is used for the treatment of adult and adolescents from 12 years of age with active moderate to severe hidradenitis suppurativa (acne inversa), a chronic and often painful inflammatory skin disease. Symptoms may include tender nodules (lumps) and abscesses (boils) that may leak pus. It most commonly affects specific areas of the skin, such as under the breasts, the armpits, inner thighs, groin and buttocks. Scarring may also occur in affected areas. Your doctor will schedule follow-up appointments to check on your progress to continue treatment. - Uveitis
Hyrimoz is used to treat noninfectious intermediate, posterior and pan-uveitis, an inflammatory disease of the uveal tract of the eye. Hyrimoz is used in adults who have not responded well to corticosteroids or whose disease flares when they taper off corticosteroids. Signs and symptoms include inflammation, vision impairment and pain.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
This medicine is only available with a doctor’s prescription.
The long term effects of Hyrimoz on the growth and development of children is not known.
Before you use HYRIMOZ
When you must not use it
Do not use Hyrimoz if you:
- have an allergy to any medicine containing adalimumab or any of the ingredients listed at the end of this leaflet. Symptoms of an allergic reaction may include:
- chest tightness
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- hives, itching or skin rash - have a severe infection including infection of the blood (sepsis), active tuberculosis and other infections that can be caused by viruses, fungi, parasites or bacteria.
Infections can occur when the body’s natural defences are lowered. - are already using anakinra (Kineret) – a medicine for rheumatoid arthritis.
- have moderate to severe heart failure.
Do not use this medicine after the expiry date printed on the label / blister / carton or if the packaging is torn or shows signs of tampering.
Return out of date or damaged medicines to your pharmacist for disposal.
Before you use it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- an infection, including a long-term or localised infection (for example, leg ulcer)
- a history of recurrent infections or other conditions that increase the risk of infections.
If you are over 65, you may be more likely to get an infection while taking Hyrimoz. It is important that you and your doctor pay special attention to signs of infection while you are being treated with Hyrimoz - a history of tuberculosis, or if you have been in close contact with someone who has had tuberculosis
If symptoms of tuberculosis (persistent cough, weight loss, listlessness, mild fever), or any other infection appear during or after therapy tell your doctor immediately.
As cases of tuberculosis have been reported in patients treated with Hyrimoz your doctor will check you for signs and symptoms of tuberculosis before starting this medicine. This will include a thorough medical history, a chest x- ray and tuberculin test. - the hepatitis B virus (HBV) if you are a carrier of, or you have active HBV or you think you might be at risk of contracting HBV.
Hyrimoz can cause reactivation of HBV in people who carry this virus. In some rare cases, especially if you are taking other medicines that suppress the immune system, reactivation of HBV can be life-threatening. - a fungal infection, or if you have lived or travelled in countries where some fungal infections are common. These infections may develop or become more severe if you take Hyrimoz.
- If you suffer from uveitis, your doctor may check for signs and symptoms of neurologic disease before starting this medicine.
- multiple sclerosis, a disease of the nervous system or other demyelinating diseases
- allergic reactions such as chest tightness, wheezing, dizziness, swelling or rash
- blood disorders
- low resistance to disease
- heart conditions including congestive heart failure, heart attack or worsening of existing heart conditions
- cancer or autoimmune disease
- a lung disease called chronic obstructive pulmonary disease (COPD)
- kidney or liver problems
Tell your doctor if you are scheduled for any vaccines. It is recommended that children, if possible, be brought up to date with all immunisations in agreement with current immunisation guidelines prior to initiating Hyrimoz therapy. Patients receiving Hyrimoz should not receive live vaccines.
Tell your doctor if you are a psoriasis sufferer who has undergone phototherapy.
Tell your doctor if you are pregnant or plan to become pregnant. Women of childbearing age are advised to use contraception to prevent pregnancy and continue its use for at least 5 months after the last Hyrimoz treatment.
Hyrimoz should only be used during pregnancy if clearly needed.
A pregnancy study found that there was no higher risk of birth defects when the mother had used adalimumb during pregnancy, compared with mothers with the same disease who did not use adalimumab.
If you use Hyrimoz during pregnancy, your baby may have a higher risk of getting an infection.
It is important that you tell your baby's doctors and other healthcare professionals about your Hyrimoz use during your pregnancy before the baby receives any vaccine.
Tell your doctor if you are breastfeeding or plan to breastfeed. It is not known whether Hyrimoz passes into breast milk. If you are breastfeeding, your doctor may advise you to stop breastfeeding while you are using this medicine.
If you have not told your doctor or pharmacist about any of the above, tell them before you start using Hyrimoz.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket naturopath or health food shop.
Some medicines and Hyrimoz may interfere with each other. Your doctor and pharmacist have more information on medicines to be careful with or avoid while using this medicine.
Tell your doctor or pharmacist if you are taking anakinra (Kineret) or abatacept (Orencia), other medicines used to treat some forms of arthritis. Taking the two medicines together may increase the risk of infection.
Tell your doctor if you are taking azathioprine or 6-mercaptopurine with Hyrimoz.
Hyrimoz can be taken together with medicines such as: methotrexate, and other disease-modifying anti-rheumatic agents (for example, sulfasalazine, hydroxychloroquine, leflunomide and injectable gold preparations), steroids or pain medications including non-steroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen.
Tell your doctor if you are taking any other medicines to treat your condition.
How to use HYRIMOZ
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the label or in this leaflet, ask your doctor or pharmacist for help.
Always use Hyrimoz exactly as your doctor has instructed you.
How much to use
Adults
The usual dose for adults with rheumatoid arthritis, psoriatic arthritis and ankylosing spondylitis is one 40 mg injection fortnightly.
For adults with rheumatoid arthritis, if you are receiving Hyrimoz without methotrexate, your doctor may change your Hyrimoz dose to 40 mg every week or 80 mg every fortnight, depending on your response.
The usual dose for adults with Crohn's disease or ulcerative colitis is an initial dose of 160 mg (given as four injections on one day or two injections a day over two days), followed by 80 mg two weeks later (given as two injections on one day) then 40 mg starting two weeks later and continuing every two weeks.
Your doctor may change this ongoing (maintenance) dose to 40 mg every week or 80 mg every fortnight, depending on your response.
The usual dose for adults with psoriasis or uveitis is an initial dose of 80 mg, followed by 40 mg given fortnightly starting one week after the initial dose.
For adults with psoriasis, your doctor may change the dosage to 40 mg every week or 80 mg every fortnight, depending on your response.
The usual dose for adults with hidradenitis suppurativa is an initial dose of 160 mg (given as 4 injections in one day or 2 injections per day for two consecutive days), followed by an 80 mg dose (as 2 injections on the same day) two weeks later. After a further two weeks, continue with a dose of 40mg every week or 80 mg every fortnight.
Your doctor may prescribe other medicines for your condition to take with this medicine.
Children
There is no available dosage form of Hyrimoz that allows dosage of less than 40 mg.
The usual dose for children with polyarticular juvenile idiopathic arthritis, or enthesitis-related arthritis depends on body weight:
- with a body weight of 30 kg or above, the usual dose is 40 mg given fortnightly.
The usual dose for children with Crohn's disease depends on body weight.
For a body weight of 40 kg or above:
- an initial dose of 160 mg (day 1) (given as four injections on one day OR two injections a day over two days)
- 80 mg two weeks later (day 15) (given as two injections on one day)
- 40 mg every fortnight starting at day 29, which then continues (maintenance dose).
Your doctor may change this ongoing (maintenance) dose to 40mg every week or 80mg every fortnight (given as two injections), depending on your child’s response.
Treatment of Crohn's disease in children should be supported by good nutrition to allow appropriate growth.
The usual dose for children with psoriasis depends on the body weight:
- with a body weight of 40 kg or above, the usual dose is 40 mg given once weekly for the first two weeks, then fortnightly.
The usual dose for adolescents (from 12 years, weighing at least 30 kg) with hidradenitis suppurativa is as follows:
- initial dose of 80 mg (day 1) (given as two 40 mg injections in one day)
- 40 mg starting one week later (day 8)
- 40 mg every fortnight starting at day 22, which then continues (maintenance dose).
Your doctor may change this ongoing (maintenance) dose to 40 mg every week or 80 mg every fortnight (given as two injections on one day), depending on your response.
It is recommended you use an antiseptic body wash daily on the affected areas.
How to use it
Hyrimoz is injected under the skin. The injection can be self-administered or given by another person, for example a family member or friend after proper training in injection technique, or your doctor or his / her assistant.
Comprehensive instructions are given in the “Instructions for use for Hyrimoz pre-filled syringe / Hyrimoz SensoReady® pen”.
How long to use it
Keep using Hyrimoz for as long as your doctor tells you.
Hyrimoz will not cure your condition but should help control your symptoms.
Ask your doctor if you are not sure how long to take this medicine for.
If you forget to use it
If you forget to give yourself an injection, you should inject the next dose of Hyrimoz as soon as you remember. Then inject your next dose as you would have on your originally scheduled day, had you not forgotten a dose. Do not try to make up for missed doses by taking more than one dose at a time.
If you take too much (overdose)
If you accidentally inject Hyrimoz more frequently than told to by your doctor, immediately telephone your doctor or the Poisons Information Centre (Australia Telephone 13 11 26), or go to Accident and Emergency at your nearest hospital. Do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention. Always take the outer carton of the medicine with you.
While you are using HYRIMOZ
Things you must do
Check with your doctor before you receive any vaccines.
It is recommended that children, if possible, be brought up to date with all immunisations in agreement with current immunisation guidelines prior to initiating Hyrimoz therapy.
Some vaccines, such as oral polio vaccine, should not be given while receiving Hyrimoz.
If you become pregnant while using Hyrimoz, tell your doctor immediately.
If you are about to be started on any new medicine, tell your doctor you are using Hyrimoz.
Tell all doctors, dentists, and pharmacists who are treating you that you are using Hyrimoz.
If you are going to have surgery, tell the surgeon or anaesthetist that you are using Hyrimoz. Your doctor may recommend temporary discontinuation of Hyrimoz.
Keep all of your doctor's appointments so that your progress can be checked.
Things you must not do
Do not give Hyrimoz to anyone else, even if they have the same condition as you.
Do not use Hyrimoz to treat any other complaints unless your doctor tells you to.
Do not stop taking Hyrimoz, without checking with your doctor.
Do not take Hyrimoz and anakinra (Kineret) together.
Do not take Hyrimoz and abatacept (Orencia) together. Anakinra and abatacept are other medicines used to treat certain forms of arthritis.
Things to be careful of
It is important to tell your doctor if you get symptoms such as fever, wounds, feeling tired or dental problems.
You might get infections more easily while you are receiving Hyrimoz-treatment. These infections may be serious and include tuberculosis, infections caused by viruses, fungi or bacteria, or other opportunistic infections and sepsis that may, in rare cases, be life- threatening. Your doctor may recommend temporary discontinuation of Hyrimoz.
Be careful driving or operating machinery until you know how Hyrimoz affects you. The effects on your ability to drive and use machines whilst taking this medicine are not known.
Side effects
Tell your doctor as soon as possible if you have any problems while using Hyrimoz, even if you do not think the problems are connected with the medicine or are not listed in this leaflet.
All medicines have some unwanted side effects. Sometimes they are serious, but most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist any questions you may have.
Tell your doctor immediately or go to Accident and Emergency at your nearest hospital, if you experience any of the following:
- signs of an allergic reaction such as:
- chest tightness
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- hives, itching or skin rash - signs and symptoms suggestive of heart failure such as shortness of breath with exertion or upon lying down or swelling of the feet
- signs and symptoms suggestive of blood disorders such as persistent fever, bruising, bleeding, paleness
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are uncommon.
Tell your doctor as soon as possible if you notice any of the following:
- signs of tuberculosis such as persistent cough, weight loss, listlessness, fever
- signs of infection such as fever, lack of energy, malaise, skin sores, wounds, dental problems, burning on urination
- You might get infections more easily while you are receiving Hyrimoz treatment.
- signs of nervous system disorders such as numbness or tingling throughout your body, arm or leg weakness, double vision
- signs of soft tissue infection, such as a bump or open sore that doesn't heal
The above list includes serious side effects. You may need urgent medical attention. Serious side effects are rare.
Tell your doctor if you notice any of the following and they worry you:
- injection site reactions (including pain, swelling, redness or itching)
- upper respiratory tract infections (including cold, runny nose, sinus infection, sore throat, cough, congestion on the chest, asthma or worsening of asthma symptoms)
- lower respiratory tract infections (such as bronchitis, pneumonia)
- ear infections
- eye inflammation, inflammation of the eye lid
- burning or pain when passing urine, or blood in the urine
- skin bumps or sores that don’t heal
- headache, dizziness, vertigo, sensation disorders
- muscle weakness or numbness, difficulty balancing
- fever, flushing, increased sweating
- abdominal symptoms such as nausea, vomiting, abdominal pain
- reflux or heartburn
- rash, itching redness or scaly patches
- problems with your finger or toe nails
- hair loss
- fatigue, tiredness, lack of energy
- mouth inflammation and ulcers
- muscle, joint or bone pain
- bleeding or bruising more easily than usual
- feeling overwhelmed or sad, or depression, anxiety
- increased heart rate
- viral infections (including the flu, cold sore blisters, chicken pox and shingles)
- bacterial infections (including urinary tract infection)
- fungal infections
The above list includes the more common side effects of Hyrimoz. They are usually mild and short-lived.
There have been cases of certain kinds of cancer in patients taking Hyrimoz or other TNF blockers. People with more serious rheumatoid arthritis that have had the disease for a long time may have a higher chance of getting a kind of cancer that affects the lymph system, called lymphoma, or that affects the blood, called leukaemia. If you take Hyrimoz your risk may increase.
Tell your doctor if new skin lesions (skin spots or sores) appear, or if existing lesions change appearance, during or after Hyrimoz treatment. Very rare cases of skin cancer have been observed in patients taking Hyrimoz.
If you have COPD, or are a heavy smoker, you should discuss with your doctor whether treatment with a TNF blocker is appropriate for you. There have been cases of cancers other than lymphoma in patients with COPD treated with another TNF blocker.
Tell your doctor or pharmacist if you notice anything else that is making you feel unwell.
Laboratory results
Some side effects observed with Hyrimoz may not have symptoms and may only be discovered through blood tests. These include increased lipids in the blood, elevated liver enzymes, and increased uric acid in the blood.
Other side effects not listed above may occur in some people.
After using HYRIMOZ
Storage
Keep your pre-filled syringe / prefilled pen in the pack until it is time to use it.
Keep Hyrimoz in a refrigerator (2 °C–8 °C). Do not freeze. Do not shake.
Keep Hyrimoz in the refrigerator in a way children cannot get to it.
If needed (for example when you are travelling), Hyrimoz may be stored up to a maximum of 25 °C for a period of up to 21 days – be sure to protect it from light. When you remove your pre-filled syringe / pre-filled pen from the refrigerator and it has warmed to room temperature, your pre-filled syringe / pre-filled pen must be used within 21 days or discarded, even if it is later returned to the refrigerator.
Write down the date you first removed the syringe / pen from the refrigerator on the label, so you can check how long it has been.
For additional information about Hyrimoz, contact Sandoz Medical Information on 1800 726 369.
Disposal
After injecting Hyrimoz, throw away the used pre-filled syringe / pre-filled pen immediately in a special “sharps” container as instructed by your doctor, nurse or pharmacist.
If your doctor tells you to stop using Hyrimoz or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
Hyrimoz is a colourless to slightly yellowish as well as clear to slightly opalescent solution of 40 mg adalimumab in 0.8 mL water in a syringe available in a pre-filled syringe with needle guard / pre-filled pen for patient use in packs containing 1, 2 or 6 pre-filled syringe(s) / pre-filled pen(s)*.
* Not all pack sizes or presentations may be marketed
Ingredients
Hyrimoz contains 40 mg of adalimumab as the active ingredient.
It also contains other inactive ingredients:
- adipic acid
- Citric acid monohydrate
- Sodium chloride
- mannitol
- polysorbate 80
- hydrochloric acid
- sodium hydroxide
- water for injections
Sponsor
Hyrimoz is supplied in Australia by:
Sandoz Pty Ltd
ABN 60 075 449 553
54 Waterloo Road
Macquarie Park NSW 2113
Telephone: 1800 726 369
This leaflet was prepared in March 2021.
Australian Registration Numbers:
Pre-filled syringe AUST R 291937
Pre-filled pen AUST R 291938
Published by MIMS May 2021

 Available data suggest that a clinical response is usually achieved within 12 weeks of treatment. Continued therapy should be carefully reconsidered in a patient not responding within this time period.
Available data suggest that a clinical response is usually achieved within 12 weeks of treatment. Continued therapy should be carefully reconsidered in a patient not responding within this time period. Some patients who experience a decrease in their response may benefit from an increase in dosage of Hyrimoz to 40 mg every week or 80 mg fortnightly.
Some patients who experience a decrease in their response may benefit from an increase in dosage of Hyrimoz to 40 mg every week or 80 mg fortnightly.
 Some patients may benefit from increasing the dosage if a disease flare or an inadequate response is experienced during maintenance dosing: < 40 kg: 20 mg every week; ≥ 40 kg: 40 mg every week or 80 mg fortnightly.
Some patients may benefit from increasing the dosage if a disease flare or an inadequate response is experienced during maintenance dosing: < 40 kg: 20 mg every week; ≥ 40 kg: 40 mg every week or 80 mg fortnightly. During maintenance treatment, corticosteroids may be tapered in accordance with clinical practice guidelines.
During maintenance treatment, corticosteroids may be tapered in accordance with clinical practice guidelines. If re-treatment with Hyrimoz is indicated, the above guidance on dose and treatment duration should be followed.
If re-treatment with Hyrimoz is indicated, the above guidance on dose and treatment duration should be followed. Table 7 contains adverse drug reactions (ADRs), which in some cases represent groups of related Preferred Terms to represent a medical concept. The ADRs presented in the table were included based on criteria including statistical significance, doubling in rate in adalimumab treated patients compared to placebo treated patients, a rate greater than 1% for adalimumab treated patients and medical importance assessment.
Table 7 contains adverse drug reactions (ADRs), which in some cases represent groups of related Preferred Terms to represent a medical concept. The ADRs presented in the table were included based on criteria including statistical significance, doubling in rate in adalimumab treated patients compared to placebo treated patients, a rate greater than 1% for adalimumab treated patients and medical importance assessment.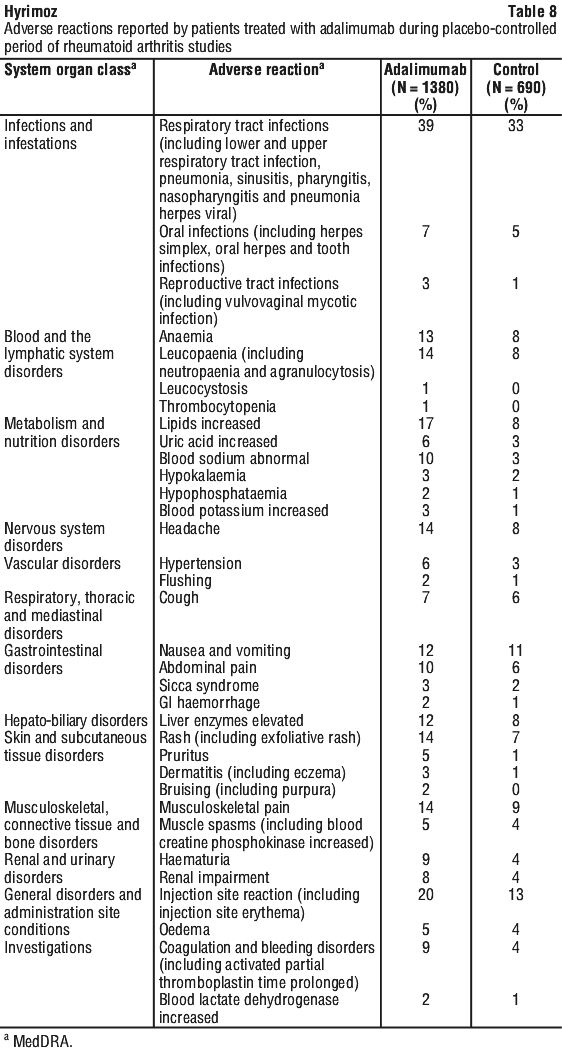
 The proportions of patients in the continued groups (stayed on one treatment for the entire study) who experienced AEs suspected of being related to study drug was similar between treatment groups during entire study duration. Safety profiles were similar. See Table 11.
The proportions of patients in the continued groups (stayed on one treatment for the entire study) who experienced AEs suspected of being related to study drug was similar between treatment groups during entire study duration. Safety profiles were similar. See Table 11. The proportions of patients in the four individual groups (reference medicine to Hyrimoz, continued reference medicine, Hyrimoz to reference medicine, and continued Hyrimoz) who experienced AEs suspected related to study drug during Week 17 to Week 51 were similar among treatment groups. See Table 12.
The proportions of patients in the four individual groups (reference medicine to Hyrimoz, continued reference medicine, Hyrimoz to reference medicine, and continued Hyrimoz) who experienced AEs suspected related to study drug during Week 17 to Week 51 were similar among treatment groups. See Table 12.


 EC50 estimates ranging from 0.8 to 1.4 microgram/mL were obtained through pharmacokinetic/ pharmacodynamic modelling of swollen joint count, tender joint count and ACR20 response from patients participating in Phase II and III trials.
EC50 estimates ranging from 0.8 to 1.4 microgram/mL were obtained through pharmacokinetic/ pharmacodynamic modelling of swollen joint count, tender joint count and ACR20 response from patients participating in Phase II and III trials. Patients receiving adalimumab 40 mg every week in RA Study II also achieved statistically significant ACR20, 50 and 70 response rates of 53.4%, 35.0% and 18.4%, respectively, at six months.
Patients receiving adalimumab 40 mg every week in RA Study II also achieved statistically significant ACR20, 50 and 70 response rates of 53.4%, 35.0% and 18.4%, respectively, at six months. The results of the components of the ACR response criteria for RA Study III are shown in Table 16. ACR response rates and improvement in all ACR response criteria were maintained to Week 104. Over the 2 years in RA Study III, 20% of adalimumab patients achieved a major clinical response, defined as maintenance of an ACR70 response over a > 6 month period.
The results of the components of the ACR response criteria for RA Study III are shown in Table 16. ACR response rates and improvement in all ACR response criteria were maintained to Week 104. Over the 2 years in RA Study III, 20% of adalimumab patients achieved a major clinical response, defined as maintenance of an ACR70 response over a > 6 month period. In RA Study III, 84.7% of patients with ACR20 responses at Week 24 maintained the response at 52 weeks. Clinical responses were maintained for up to 5 years in the open-label portion of RA Study III. ACR responses observed at Week 52 were maintained or increased through 5 years of continuous treatment with 22% (115/534) of patients achieving major clinical response. A total of 372 (67.8%) subjects had no change in their methotrexate dose during the study, 141 (25.7%) subjects had a dose reduction and 36 (6.6%) subjects required a dose increase. A total of 149 (55.6%) subjects had no change in their corticosteroid dose during the study, 80 (29.9%) subjects had a dose reduction and 39 (14.6%) subjects required a dose increase. Figures 2 and 3 illustrate the durability of ACR20 responses to adalimumab in RA Studies III and II.
In RA Study III, 84.7% of patients with ACR20 responses at Week 24 maintained the response at 52 weeks. Clinical responses were maintained for up to 5 years in the open-label portion of RA Study III. ACR responses observed at Week 52 were maintained or increased through 5 years of continuous treatment with 22% (115/534) of patients achieving major clinical response. A total of 372 (67.8%) subjects had no change in their methotrexate dose during the study, 141 (25.7%) subjects had a dose reduction and 36 (6.6%) subjects required a dose increase. A total of 149 (55.6%) subjects had no change in their corticosteroid dose during the study, 80 (29.9%) subjects had a dose reduction and 39 (14.6%) subjects required a dose increase. Figures 2 and 3 illustrate the durability of ACR20 responses to adalimumab in RA Studies III and II.
 In the open-label extension for RA study V, ACR responses were maintained when followed for up to 10 years. However, no statistical hypothesis was tested in the OLE period. Of 542 patients who were randomised to adalimumab 40 mg fortnightly, 170 patients continued on adalimumab 40 mg fortnightly for 10 years. Among those, 154 patients (90.6%) had ACR20 responses; 127 patients (74.7%) had ACR50 responses and 102 patients (60.0%) had ACR70 responses.
In the open-label extension for RA study V, ACR responses were maintained when followed for up to 10 years. However, no statistical hypothesis was tested in the OLE period. Of 542 patients who were randomised to adalimumab 40 mg fortnightly, 170 patients continued on adalimumab 40 mg fortnightly for 10 years. Among those, 154 patients (90.6%) had ACR20 responses; 127 patients (74.7%) had ACR50 responses and 102 patients (60.0%) had ACR70 responses.
 In RA Study V, adalimumab-treated patients had a mean duration of rheumatoid arthritis of less than 9 months and had not previously received methotrexate. Structural joint damage was assessed radiographically and expressed as change in modified Total Sharp Score. The Week 52 results are shown in Table 20. A statistically significant difference for change in modified Total Sharp Score and the erosion score was observed at Week 52 and maintained at Week 104.
In RA Study V, adalimumab-treated patients had a mean duration of rheumatoid arthritis of less than 9 months and had not previously received methotrexate. Structural joint damage was assessed radiographically and expressed as change in modified Total Sharp Score. The Week 52 results are shown in Table 20. A statistically significant difference for change in modified Total Sharp Score and the erosion score was observed at Week 52 and maintained at Week 104.
 At Year 2, 94/207 (45.4%) of patients who originally entered the study achieved a -0.5 reduction in HAQ. 79.5% (115/195) of the patients who achieved a reduction in HAQ of -0.5 at the end of one year of adalimumab treatment maintained this response over 5 years of active treatment.
At Year 2, 94/207 (45.4%) of patients who originally entered the study achieved a -0.5 reduction in HAQ. 79.5% (115/195) of the patients who achieved a reduction in HAQ of -0.5 at the end of one year of adalimumab treatment maintained this response over 5 years of active treatment.

 In subjects treated with adalimumab with no radiographic progression from baseline to Week 48 (n = 102), 84% continued to show no radiographic progression through 144 weeks of treatment.
In subjects treated with adalimumab with no radiographic progression from baseline to Week 48 (n = 102), 84% continued to show no radiographic progression through 144 weeks of treatment.
 A low level of disease activity (defined as a value < 20 [on a scale of 0-100 mm] in each of the four ASAS response parameters) was achieved at 24 weeks in 22% of adalimumab-treated patients vs. 6% in placebo-treated patients (p < 0.001) (see Table 26).
A low level of disease activity (defined as a value < 20 [on a scale of 0-100 mm] in each of the four ASAS response parameters) was achieved at 24 weeks in 22% of adalimumab-treated patients vs. 6% in placebo-treated patients (p < 0.001) (see Table 26). Results of this study were similar to those seen in the second randomised trial (AS Study II or M03-606), a multicenter, double-blind, placebo-controlled study of 82 patients with ankylosing spondylitis. Patient Reported Outcomes were assessed in both ankylosing spondylitis studies using the generic health status questionnaire SF-36 and the disease specific Ankylosing Spondylitis Quality of Life Questionnaire (ASQoL). The adalimumab-treated patients had significantly greater improvement in SF-36 Physical Component Score (mean change: 6.93) compared to placebo-treated patients (mean change: 1.55; p < 0.001) at Week 12, which was maintained through Week 24.
Results of this study were similar to those seen in the second randomised trial (AS Study II or M03-606), a multicenter, double-blind, placebo-controlled study of 82 patients with ankylosing spondylitis. Patient Reported Outcomes were assessed in both ankylosing spondylitis studies using the generic health status questionnaire SF-36 and the disease specific Ankylosing Spondylitis Quality of Life Questionnaire (ASQoL). The adalimumab-treated patients had significantly greater improvement in SF-36 Physical Component Score (mean change: 6.93) compared to placebo-treated patients (mean change: 1.55; p < 0.001) at Week 12, which was maintained through Week 24.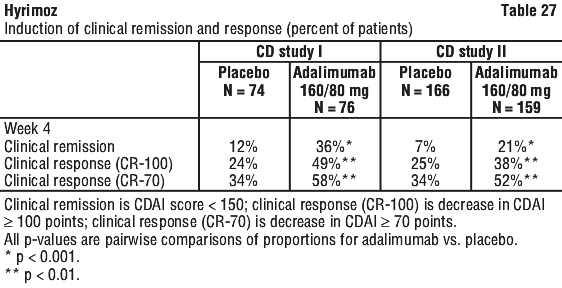
 Clinical remission results presented in Table 29 remained relatively constant irrespective of previous TNF antagonist exposure.
Clinical remission results presented in Table 29 remained relatively constant irrespective of previous TNF antagonist exposure. Of those in response at Week 4 who attained remission during the study, patients in adalimumab maintenance groups maintained remission for a significantly longer time than patients in the placebo maintenance group (see Figure 5). Among patients who were not in response by Week 12, therapy continued beyond 12 weeks did not result in significantly more responses. The group that received adalimumab every week did not show significantly higher remission rates than the group that received adalimumab fortnightly.
Of those in response at Week 4 who attained remission during the study, patients in adalimumab maintenance groups maintained remission for a significantly longer time than patients in the placebo maintenance group (see Figure 5). Among patients who were not in response by Week 12, therapy continued beyond 12 weeks did not result in significantly more responses. The group that received adalimumab every week did not show significantly higher remission rates than the group that received adalimumab fortnightly.


 Statistically significant increases (improvement) from Baseline to Week 26 and 52 in Body Mass Index and height velocity were observed for both treatment groups. Statistically and clinically significant improvements from Baseline were also observed in both treatment groups for quality of life parameters (including IMPACT III).
Statistically significant increases (improvement) from Baseline to Week 26 and 52 in Body Mass Index and height velocity were observed for both treatment groups. Statistically and clinically significant improvements from Baseline were also observed in both treatment groups for quality of life parameters (including IMPACT III).
 Adalimumab should be discontinued in patients who do not achieve a clinical response during the first 8 weeks of therapy because very few patients will achieve clinical remission with continuing treatment. In UC-1 and UC-2, of patients given adalimumab 160/80 mg at baseline who did not achieve a clinical response at Week 8, 5.2%, and 17.0% went on to be in remission and response, respectively at Week 52. See Table 35.
Adalimumab should be discontinued in patients who do not achieve a clinical response during the first 8 weeks of therapy because very few patients will achieve clinical remission with continuing treatment. In UC-1 and UC-2, of patients given adalimumab 160/80 mg at baseline who did not achieve a clinical response at Week 8, 5.2%, and 17.0% went on to be in remission and response, respectively at Week 52. See Table 35. Statistically significant reductions of both all-cause and UC-related rates of hospitalisation were observed in a pooled analysis of Studies UC I and II.
Statistically significant reductions of both all-cause and UC-related rates of hospitalisation were observed in a pooled analysis of Studies UC I and II.
 Two of the continuous treatment populations entering trial M03-658 were those from Period C of Study I and those from Study II.
Two of the continuous treatment populations entering trial M03-658 were those from Period C of Study I and those from Study II. Of those who continued to receive reference medicine treatment until Week 52, 65.0% achieved mNAPSI 75 response and 61.3% achieved PGA-F response.
Of those who continued to receive reference medicine treatment until Week 52, 65.0% achieved mNAPSI 75 response and 61.3% achieved PGA-F response. Patients who achieved PASI 75 and PGA clear or minimal were withdrawn from treatment for up to 36 weeks and monitored for loss of disease control (loss of PGA response). Patients were then re-treated with adalimumab 0.8 mg/kg fortnightly for an additional 16 weeks. Among patients who were responders to the initial 16 weeks of treatment but who relapsed upon withdrawal and were retreated, PASI 75 response of 78.9% (15 of 19 subjects) and PGA clear or minimal of 52.6% (10 of 19 subjects) was observed.
Patients who achieved PASI 75 and PGA clear or minimal were withdrawn from treatment for up to 36 weeks and monitored for loss of disease control (loss of PGA response). Patients were then re-treated with adalimumab 0.8 mg/kg fortnightly for an additional 16 weeks. Among patients who were responders to the initial 16 weeks of treatment but who relapsed upon withdrawal and were retreated, PASI 75 response of 78.9% (15 of 19 subjects) and PGA clear or minimal of 52.6% (10 of 19 subjects) was observed. There is a statistically significantly higher HiSCR rate at Week 36 in patients who continued to receive weekly adalimumab compared to those who stopped adalimumab at Week 12.
There is a statistically significantly higher HiSCR rate at Week 36 in patients who continued to receive weekly adalimumab compared to those who stopped adalimumab at Week 12.

 In both studies, all components of the primary endpoint contributed cumulatively to the overall difference between adalimumab and placebo groups (Table 42).
In both studies, all components of the primary endpoint contributed cumulatively to the overall difference between adalimumab and placebo groups (Table 42). Additionally, in Study UV I, statistically significant differences in favour of adalimumab versus placebo were observed for the secondary endpoints changes in AC cell grade, vitreous haze grade, and log MAR BCVA (mean change from best state prior to Week 6 to the final visit; P Values: 0.011, < 0.001 and 0.003, respectively).
Additionally, in Study UV I, statistically significant differences in favour of adalimumab versus placebo were observed for the secondary endpoints changes in AC cell grade, vitreous haze grade, and log MAR BCVA (mean change from best state prior to Week 6 to the final visit; P Values: 0.011, < 0.001 and 0.003, respectively). Key efficacy results for the pooled continued and pooled switched treatment groups are shown below. PASI response rates (PASI50, PASI75, PASI90 and PASI100) as well as the mean absolute PASI scores and percent changes from baseline in the TP2+EP were similar across pooled groups. The results indicate that treatment effect in patients continuing their original treatment (Hyrimoz or reference medicine) or patients who experienced multiple switches are similar. See Figure 8.
Key efficacy results for the pooled continued and pooled switched treatment groups are shown below. PASI response rates (PASI50, PASI75, PASI90 and PASI100) as well as the mean absolute PASI scores and percent changes from baseline in the TP2+EP were similar across pooled groups. The results indicate that treatment effect in patients continuing their original treatment (Hyrimoz or reference medicine) or patients who experienced multiple switches are similar. See Figure 8.
 Following the administration of 24 mg/m2 (up to a maximum of 40 mg) subcutaneously fortnightly to patients with enthesitis-related arthritis, the mean trough steady-state (values measured at Week 24) serum adalimumab concentrations were 8.8 ± 6.6 microgram/mL for adalimumab without concomitant methotrexate and 11.8 ± 4.3 microgram/mL with concomitant methotrexate. Based on a population pharmacokinetic (PK) modelling approach, simulated steady-state adalimumab serum trough concentrations for a weight-based dosing regimen (20 mg adalimumab fortnightly for body weight < 30 kg and 40 mg adalimumab fortnightly for body weight ≥ 30 kg) were comparable to the simulated trough concentrations for the body surface area-based regimen.
Following the administration of 24 mg/m2 (up to a maximum of 40 mg) subcutaneously fortnightly to patients with enthesitis-related arthritis, the mean trough steady-state (values measured at Week 24) serum adalimumab concentrations were 8.8 ± 6.6 microgram/mL for adalimumab without concomitant methotrexate and 11.8 ± 4.3 microgram/mL with concomitant methotrexate. Based on a population pharmacokinetic (PK) modelling approach, simulated steady-state adalimumab serum trough concentrations for a weight-based dosing regimen (20 mg adalimumab fortnightly for body weight < 30 kg and 40 mg adalimumab fortnightly for body weight ≥ 30 kg) were comparable to the simulated trough concentrations for the body surface area-based regimen.
