1 Name of Medicine
Idelvion (recombinant fusion protein linking coagulation factor IX with albumin (rIX-FP); albutrepenonacog alfa).
2 Qualitative and Quantitative Composition
Idelvion is a preservative-free, sterile, non-pyrogenic, lyophilised powder to be reconstituted with water for injections (WFI) for intravenous injection.
No human or animal-derived proteins are added during any stage of manufacturing or formulation of Idelvion.
The potency in international units (IU) is determined using an in vitro activated partial thromboplastin time (aPTT)-based one-stage clotting assay calibrated against the World Health Organisation (WHO) international standard for FIX concentrate.
Idelvion is available in five different presentations as detailed in Table 1.
 Idelvion contains approximately 75 mmol/L (1.7243 g/L) sodium. Idelvion is essentially sodium-free, containing less than 1 mmol sodium (23 mg) per vial. For the full list of excipients, see Section 6.1 List of Excipients.
Idelvion contains approximately 75 mmol/L (1.7243 g/L) sodium. Idelvion is essentially sodium-free, containing less than 1 mmol sodium (23 mg) per vial. For the full list of excipients, see Section 6.1 List of Excipients.
Not all registered presentations may be supplied.
3 Pharmaceutical Form
Powder and diluent for solution for injection.
Powder: pale yellow to white.
Diluent (WFI): clear, colourless.
4.1 Therapeutic Indications
Idelvion is indicated in all patients with haemophilia B for:
Routine prophylaxis to prevent or reduce the frequency of bleeding episodes;
Control and prevention of bleeding episodes;
Control and prevention of bleeding in the perioperative setting.
4.2 Dose and Method of Administration
Initiate treatment of Idelvion under the supervision of a physician experienced in the treatment of haemophilia B.
It is recommended that prescribed doses of Idelvion be expressed using 'international units' written in full.
The decision for an individual patient on the use of home treatment of bleeding and prophylaxis of bleeding in patients with haemophilia B should be made by the treating physician. The physician should ensure that appropriate training is provided and the use is reviewed at intervals.
Dosage.
The dose and duration of the substitution therapy depend on the severity of the FIX deficiency, the location and extent of the bleeding and the patient's clinical condition and response.
The number of units of FIX administered is expressed in international units (IU), which are related to the current WHO standard for FIX products. One IU of FIX activity is equivalent to that quantity of FIX in 1 mL of normal human plasma. FIX activity in plasma is expressed either as a percentage (relative to normal human plasma) or in IU (relative to an international standard for factor IX in plasma).
Prophylaxis. Adults and adolescents (≥ 12 years of age).
For routine prophylaxis to prevent bleeding in patients ≥ 12 years of age with haemophilia B, the recommended dose regimen is:
25-40 IU/kg once weekly (every 7 days).
Adult and adolescent patients who are well controlled on a 7-day regimen may be switched to:
50-75 IU/kg every 14 days.
Adjust dosing regimen based on individual patient's clinical condition and response.
Paediatrics (< 12 years of age).
For routine prophylaxis to prevent bleeding in paediatric patients with haemophilia B, the recommended dose regimen is:
35-50 IU/kg once weekly (every 7 days).
Based on the individual patient's clinical condition and response, it may be appropriate for the treating physician to increase dose and extend dosing interval during routine clinical management.
In some cases, especially in younger patients, and depending on individual patient pharmacokinetics, age, bleeding phenotype and physical activity, shorter dosage intervals or higher doses may be necessary.
See Section 5.1 Pharmacodynamic Properties, Clinical trials; Section 5.2 Pharmacokinetic Properties for relevant data.
On demand treatment. The calculation of the required dose of FIX is based on the empirical finding that 1 IU FIX per kg body weight is expected to increase the circulating level of FIX by an average of 1.3 IU/dL (1.3% of normal) in patients ≥ 12 years of age and by 1.0 IU/dL (1.0% of normal) in patients < 12 years of age. The required dose is determined using the following formulae:
Required dose (IU) = body weight (kg) x desired FIX increase (IU/dL or % of normal) x {reciprocal of observed recovery (IU/kg per IU/dL)};
Or
Expected FIX increase (IU/dL or % of normal) = dose (IU) x recovery (IU/dL per IU/kg)/body weight (kg).
The amount to be administered and the frequency of administration should always be oriented to the clinical effectiveness in the individual case.
For determination of adequate maintenance dose, take into consideration the extended half-life of the product.
Adults and adolescents (≥ 12 years of age).
For an incremental recovery of 1.3 IU/dL per 1 IU/kg, the dose is calculated as follows:
Dose (IU) = body weight (kg) x desired FIX increase (IU/dL) x 0.77 dL/kg.
Example:
1. A peak level of 50% of normal is required in an 80 kg patient with severe haemophilia B. The appropriate dose would be 80 kg x 50 IU/dL x 0.77 dL/kg = 3080 IU.
2. A dose of 2000 IU of Idelvion, administered to an 80 kg patient, should result in a peak post-injection FIX increase of 2000 IU x 1.3 (IU/dL per IU/kg) /80 kg = 32.5 IU/dL (32.5% of normal).
Paediatrics (< 12 years of age).
For an incremental recovery of 1 IU/dL per 1 IU/kg, the dose is calculated as follows:
Dose (IU) = body weight (kg) x desired FIX increase (IU/dL) x 1 dL/kg.
Example:
3. A peak level of 50% of normal is required in a 20 kg patient with severe haemophilia B. The appropriate dose would be 20 kg x 50 IU/dL x 1 dL/kg = 1000 IU.
4. A dose of 1000 IU of Idelvion, administered to a 25 kg patient, should result in a peak post-injection FIX increase of 1000 IU/25 kg x 1.0 (IU/dL per IU/kg) = 40 IU/dL (40% of normal).
Control and prevention of bleeding episodes and in the perioperative setting. A guide for dosing Idelvion in the control and prevention of bleeding episodes and in the perioperative setting is provided in Table 2. Ensure the FIX activity level is achieved and maintained in the corresponding period. The recommended circulating FIX level requirement for paediatric patients is the same as for adults.
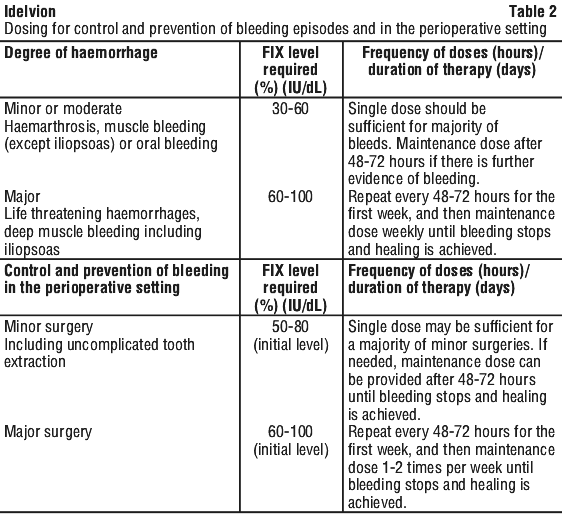
Monitoring advice.
Patients should be monitored to confirm adequate FIX levels have been achieved and maintained, and for the development of FIX inhibitors. See Section 4.4 Special Warnings and Precautions for Use.
General instructions.
For intravenous use only after reconstitution.
The solution should be clear or slightly opalescent, yellow to colourless. After filtering/withdrawal (see Reconstitution), the reconstituted product should be inspected visually for particulate matter and discolouration prior to administration.
Do not use solutions that are cloudy or have deposits.
Reconstitution and withdrawal must be carried out under aseptic conditions.
Reconstitution.
1. Bring the WFI to room temperature. Ensure Idelvion and WFI vial flip caps are removed and the stoppers are treated with an antiseptic solution and allowed to dry prior to opening the Mix2Vial package.
Open the Mix2Vial package by peeling off the lid. Do not remove the Mix2Vial from the blister package!
2. Place the WFI vial on an even, clean surface and hold the vial tight. Take the Mix2Vial together with the blister package and push the spike of the blue adaptor end straight down through the WFI vial stopper.
3. Carefully remove the blister package from the Mix2Vial set by holding at the rim, and pulling vertically upwards. Make sure that you only pull away the blister package and not the Mix2Vial set. Do not touch the exposed end of the Mix2Vial set.
4. Place the Idelvion vial on an even and firm surface. Invert the WFI vial with the Mix2Vial set attached and push the spike of the transparent adaptor end straight down through the Idelvion vial stopper. The WFI will automatically flow into the Idelvion vial.
5. With one hand grasp the Idelvion side of the Mix2Vial set and with the other hand grasp the WFI side and unscrew the set carefully anti-clockwise into two pieces.
Discard the WFI vial with the blue Mix2Vial adaptor attached.
6. Gently swirl the Idelvion vial with the transparent adaptor attached until the substance is fully dissolved. Do not shake.
7. Draw air into an empty, sterile syringe. While the Idelvion vial is upright, connect the syringe to the Mix2Vial's luer lock fitting by screwing clockwise. Inject air into the Idelvion vial.
Withdrawal and application.
8. While keeping the syringe plunger pressed, turn the system upside down and draw the solution into the syringe by pulling the plunger back slowly.
9. Now that the solution has been transferred into the syringe, firmly hold on to the barrel of the syringe (keeping the syringe plunger facing down) and disconnect the transparent Mix2Vial adaptor from the syringe by unscrewing anti-clockwise.
For injection of Idelvion, the provided administration sets are recommended to be used because treatment failure can occur as a consequence of FIX adsorption to the internal surface of some injection equipment.
Care should be taken that no blood enters the syringe filled with Idelvion, as there is a risk that the blood could coagulate in the syringe and fibrin clots could therefore be administered to the patient. If blood enters the syringe, discard and prepare a new vial of Idelvion.
The Idelvion solution must not be diluted.
Administration.
Intravenous use.
For instructions on reconstitution of the medicinal product before administration, see General instructions. The reconstituted preparation should be injected slowly intravenously at a rate comfortable for the patient.
The patient should be observed for any immediate reaction. If any reaction takes place that might be related to the administration of Idelvion, the rate of injection should be decreased or the application should be stopped, as required by the clinical condition of the patient. See Section 4.4 Special Warnings and Precautions for Use.
Use in one patient on one occasion only.
It is strongly recommended that every time that Idelvion is administered to a patient, the name and batch number of the product are recorded in order to maintain a link between the patient and the batch of the medicinal product.4.3 Contraindications
Idelvion is contraindicated in patients who have a known hypersensitivity to Idelvion, any of its components, excipients, or hamster protein (see Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
Monitoring laboratory tests.
To confirm adequate FIX levels have been achieved and maintained, monitor plasma FIX activity by performing the one-stage clotting assay.
When using an in vitro thromboplastin time (aPTT)-based one-stage clotting assay for determining FIX activity in patients' blood samples, plasma FIX activity results can be significantly affected by both the type of aPTT reagent and the reference standard used in the assay. Measurement with a one-stage clotting assay using a kaolin-based aPTT reagent or Actin FS aPTT reagent will likely result in approximately 50% lower than expected recovery based on labelled potency. This is of importance particularly when changing the laboratory and/or reagents used in the assay.
Hypersensitivity.
Allergic type hypersensitivity reactions are possible. The product contains traces of hamster proteins. If symptoms of hypersensitivity occur, discontinue use of the medicinal product immediately and initiate appropriate treatment. Patients should be informed of the early signs of hypersensitivity reactions including hives, generalised urticaria, tightness of the chest, wheezing, hypotension and anaphylaxis. Advise patients to discontinue use of Idelvion and contact their physician. All FIX products have potential of allergic reactions. It is recommended that the initial administration of FIX should be performed under medical observation where proper medical care for allergic reactions could be provided.
Thromboembolism.
Because of the potential risk of thrombotic complications with the use of FIX-containing products, clinical surveillance for early signs of thrombotic and consumptive coagulopathy should be initiated with appropriate biological testing when administering this product to patients with liver disease, to patients post-operatively, to new-born infants, or to patients at risk of thrombotic phenomena or disseminated intravascular coagulation (DIC). In each of these situations, the benefit of treatment with Idelvion should be weighed against the risk of these complications.
Inhibitors.
Formation of neutralising antibodies (inhibitors) to FIX has been reported during factor replacement therapy with Idelvion in the treatment of haemophilia B (see Section 4.8 Adverse Effects (Undesirable Effects)).
Patients should be monitored for the development of inhibitors by appropriate clinical observations and laboratory tests. If expected plasma FIX activity levels are not attained, or if the bleeding is not controlled after Idelvion administration, the presence of an inhibitor should be suspected. A specialised haemophilia treatment centre should be contacted if a patient develops an inhibitor.
Perform a Bethesda inhibitor assay if expected FIX plasma levels are not attained or if bleeding is not controlled with the expected dose of Idelvion. Use Bethesda units (BU) to report inhibitor levels.
There have been reports in the literature showing a correlation between the occurrence of a FIX inhibitor and allergic reactions. Therefore, patients experiencing allergic reactions should be evaluated for the presence of an inhibitor. It should be noted that patients with FIX inhibitors may be at an increased risk of anaphylaxis with subsequent challenge with FIX.
The safety and efficacy of using Idelvion for immune tolerance induction has not been established.
Continuous infusion.
The safety and efficacy of Idelvion administration by continuous infusion have not been established.
Use in the elderly.
Clinical studies specific to the use of Idelvion in subjects aged 65 and over have not been performed.
Paediatric use.
The listed precautions apply to both adults and children.
Effects on laboratory tests.
See Monitoring laboratory tests.4.5 Interactions with Other Medicines and Other Forms of Interactions
No interactions of Idelvion with other medicinal products have been reported.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
Fertility studies in animals have not been conducted with Idelvion.
(Category B2)
Animal reproduction studies have not been conducted with Idelvion. Based on the rare occurrence of haemophilia B in women, experience regarding the use of Idelvion during pregnancy is not available. Therefore, Idelvion should be used during pregnancy only if clearly indicated.
Lactation studies in animals have not been conducted with Idelvion. Based on the rare occurrence of haemophilia B in women, experience regarding the use of Idelvion during lactation is not available. Therefore, Idelvion should be used during lactation only if clearly indicated.4.7 Effects on Ability to Drive and Use Machines
No effects on ability to drive and use machines have been observed.
4.8 Adverse Effects (Undesirable Effects)
During open label clinical trials with Idelvion conducted in 114 previously treated patients (PTPs), there were 1,078 treatment-emergent adverse events reported in 103/114 (90.4%) subjects who received a total of 16,326 injections. Of these 1,078 events, 18 were reported as related to Idelvion in 11/114 (9.6%) subjects.
A total of 16,326 injections were administered to the overall safety population during a median of 1,543.5 days (min, max: 25 to 2,565 days), with a median 3,835.29 IU per injection (min, max: 849.8 to 8,852.3 IU). The median total amount of Idelvion administered was 445,128 IU (min, max: 1,900 to 1,607,781.3).
In the completed study with previously untreated patients (PUPs) there were 135 treatment-emergent adverse events reported in 11 of 12 PUPs, of which most were mild or moderate. Two PUPs had 5 events that were considered related to Idelvion.
The most frequent adverse reactions in clinical trials are headache, dizziness and injection site reactions. The frequency of these events is in the common category. The adverse reactions presented in Table 3 are from the clinical trials and considered related by the investigator. The frequency is estimated on a per-patient basis, by system organ class (SOC, and preferred term level). The frequencies have been evaluated according to the following convention:
Very common: ≥ 1/10;
Common: ≥ 1/100 and < 1/10;
Uncommon: ≥ 1/1000 and < 1/100;
Rare: ≥ 1/10,000 and < 1/1000;
Very rare: < 1/10,000;
Not known (cannot be estimated from the available data).
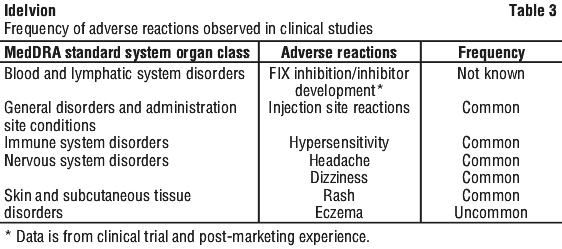 No neutralising antibodies (inhibitors) (see Section 4.4 Special Warnings and Precautions for Use), or antibodies to hamster proteins have been detected in PTPs in the clinical studies with Idelvion. Inhibitor development has been observed in the post-marketing experience.
No neutralising antibodies (inhibitors) (see Section 4.4 Special Warnings and Precautions for Use), or antibodies to hamster proteins have been detected in PTPs in the clinical studies with Idelvion. Inhibitor development has been observed in the post-marketing experience.
One case of inhibitor development to FIX was reported in the clinical trial which evaluated PUPs. The 11 year old PUP developed a low titre FIX inhibitor after 8 exposure days and a mild hypersensitivity reaction to Idelvion. Thereafter, a high titre inhibitor was observed and two further mild hypersensitivity reactions were experienced after receiving Idelvion. Following this, the subject was withdrawn from the study. There are insufficient data to provide information on inhibitor incidence in PUPs.
No thrombotic events were reported in the clinical studies with Idelvion.
With the use of FIX products, hypersensitivity or allergic reactions (which may include angioedema, burning and stinging at the injection site, chills, flushing, generalised urticaria, headache, hives, hypotension, lethargy, nausea, restlessness, tachycardia, tightness of the chest, tingling, vomiting, wheezing) have been observed. In rare cases, these reactions have progressed to anaphylaxis, and they have occurred in close temporal association with development of FIX inhibitors. No anaphylactic reactions have been observed in the clinical studies with Idelvion.
Paediatric population.
Frequency, type and severity of adverse reactions in children are similar to adults.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
No symptoms of overdose with Idelvion have been reported.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Haemophilia B is a sex-linked hereditary disorder of blood coagulation due to decreased levels of FIX and results in profuse bleeding into joints, muscles or internal organs, either spontaneously or as a result of accidental or surgical trauma. By replacement therapy, the plasma levels of FIX are increased, thereby enabling a temporary correction of the factor deficiency and correction of the bleeding tendencies.
Idelvion (albutrepenonacog alfa) is a recombinant fusion protein linking recombinant coagulation FIX with recombinant albumin that effectively replaces the missing coagulation FIX needed for haemostasis and provides for longer dose regimens. The prolongation of the half-life of FIX and the enhanced systemic exposure are achieved by fusion with recombinant albumin. Albumin is a natural, inert carrier protein in plasma with a long half-life of approximately 20 days that is not involved in immune defence or immune response. Genetic fusion of recombinant coagulation FIX with albumin extends the half-life of FIX (see Section 5.2 Pharmacokinetic Properties).
Idelvion remains intact in the circulation until FIX is activated, whereupon albumin is cleaved, releasing activated FIX (FIXa) when it is needed for coagulation.
FIX is activated by factor VII/tissue factor complex in the extrinsic pathway as well as factor XIa in the intrinsic coagulation pathway. Activated FIX, in combination with activated factor VIII, activates factor X. This results ultimately in the conversion of prothrombin to thrombin. Thrombin then converts fibrinogen into fibrin and a clot can be formed. FIX activity is absent or greatly reduced in patients with haemophilia B and substitution therapy may be required.
Clinical trials.
The safety, efficacy and pharmacokinetics (PK) of Idelvion were evaluated in prospective, open-label and multi-centre clinical studies. The relationship of the PK profile to the clinical response, namely the prevention of bleeding episodes with once weekly routine prophylaxis, was explored in a phase I/II study (Study 2004). The efficacy of Idelvion that compared episodic (on demand) treatment to weekly routine prophylaxis; compared weekly routine prophylaxis to every 10- or 14-day routine prophylaxis; haemostatic efficacy in the treatment of bleeding episodes and in the perioperative setting was evaluated in a pivotal phase II/III study (Study 3001). A study in the paediatric population (Study 3002) also characterised the safety, PK, and efficacy of Idelvion when used as routine prophylaxis (7-day dosing regimen) for the prevention of bleeding episodes and in the treatment of bleeding episodes.
The subjects enrolled had previously been treated with FIX replacement products (i.e. rFIX or pdFIX), had no confirmed history of FIX inhibitor formation, and were immunocompetent. All subjects in studies 2004 and 3001, were between the ages of 12 and 61 years and were required to have > 150 exposure days (EDs) of previous FIX treatment. In Study 3002 all subjects were between 1 and 10 years (those aged < 6 years were required to have > 50 EDs).
The long-term safety and efficacy of Idelvion was further evaluated in a prospective phase III, open-label, uncontrolled, multi-centre extension study (Study 3003) in subjects who had participated in Study 3001 and Study 3002.
Routine prophylaxis. In studies 2004, 3001 and 3002, the occurrence of bleeding episodes was recorded over the prophylaxis treatment period and the annualised spontaneous bleeding rate (AsBR) was determined.
Adult and adolescent subjects (≥ 12 to 65 years of age).
In Study 3001, a total of 63 male, previously treated patients (PTPs) with haemophilia B (≤ 2% endogenous FIX activity), between 12 and 61 years of age received Idelvion for up to 27 months. Forty subjects in the prophylaxis arm received weekly routine prophylaxis at an initial dose of 35-50 IU/kg, with median dose of 40 IU/kg of Idelvion at the end of the weekly prophylaxis period.
Following ≥ 26 weeks of a 7-day dosing interval prophylaxis regimen, 26 out of 40 subjects (26/40) switched to a longer prophylaxis dosing interval of either 10 or 14 days at a dose of 50-75 IU/kg based on meeting all of the following criteria: no dose adjustment in the previous month; currently on weekly prophylaxis of ≤ 50 IU/kg; did not experience a spontaneous bleeding episode in the previous month and willing to switch to a longer treatment interval. The extended interval was 10 days if the current weekly dose was > 40 to ≤ 50 IU/kg, 14 days if the current weekly dose was ≤ 40 IU/kg. The median dose was 74.2 IU/kg for 14-day prophylaxis.
The median AsBR of 0.00 for subjects on a 7-day regimen was the same as that for subjects on the extended treatment intervals: 10-day regimen, 0.00; 14-day regimen, 0.00 (see Table 4).
 In addition, based on a matched pairs design, every 14-day prophylaxis with Idelvion was demonstrated to be as effective as every 7-day prophylaxis with Idelvion. The AsBRs for 7-day and 14-day prophylaxis are summarised in Table 5.
In addition, based on a matched pairs design, every 14-day prophylaxis with Idelvion was demonstrated to be as effective as every 7-day prophylaxis with Idelvion. The AsBRs for 7-day and 14-day prophylaxis are summarised in Table 5.
 In Study 3001, 23 subjects in the on demand arm received Idelvion as needed for the treatment of bleeding episodes. Nineteen subjects subsequently crossed-over to weekly prophylaxis after approximately 26 weeks of episodic treatment. These subjects showed a statistically significant reduction in the rate of spontaneous and total bleeding episodes after switching from on demand treatment to prophylaxis. During on demand treatment, the subjects had a median AsBR of 15.4 bleeding episodes per year per subject. After switching to weekly prophylaxis treatment, the median AsBR was 0.00 bleeding episodes per year per subject. The corresponding median and mean percent reductions in AsBR were 100% and 96.0% (P < 0.0001), respectively. Similar results were observed in the efficacy population. A comparison of the AsBRs and ABRs in the subjects evaluable for efficacy is summarised in Table 6.
In Study 3001, 23 subjects in the on demand arm received Idelvion as needed for the treatment of bleeding episodes. Nineteen subjects subsequently crossed-over to weekly prophylaxis after approximately 26 weeks of episodic treatment. These subjects showed a statistically significant reduction in the rate of spontaneous and total bleeding episodes after switching from on demand treatment to prophylaxis. During on demand treatment, the subjects had a median AsBR of 15.4 bleeding episodes per year per subject. After switching to weekly prophylaxis treatment, the median AsBR was 0.00 bleeding episodes per year per subject. The corresponding median and mean percent reductions in AsBR were 100% and 96.0% (P < 0.0001), respectively. Similar results were observed in the efficacy population. A comparison of the AsBRs and ABRs in the subjects evaluable for efficacy is summarised in Table 6.
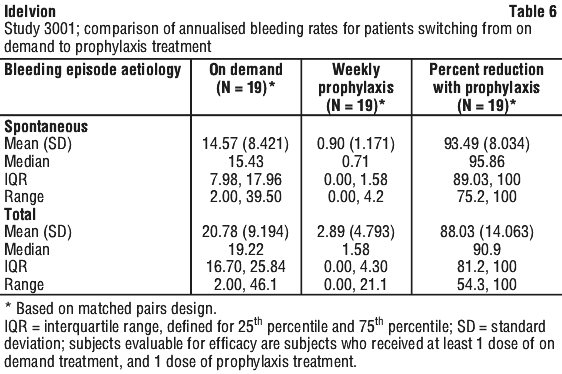 In Study 2004, subjects receiving weekly prophylaxis treatment had a lower AsBR (median: 1.13; N = 13) than subjects receiving on demand treatment only (median: 22.2; N = 4).
In Study 2004, subjects receiving weekly prophylaxis treatment had a lower AsBR (median: 1.13; N = 13) than subjects receiving on demand treatment only (median: 22.2; N = 4).
Consumption of Idelvion was measured as a secondary endpoint in Study 3001. Adult and adolescent subjects on routine prophylaxis regimens, exhibited mean monthly consumption of Idelvion that was approximately 50% lower than their previous FIX prophylaxis treatment; the mean consumption was reduced from 321 IU/kg of the previous FIX products to 203 IU/kg per month of Idelvion on once weekly regimen and 157 IU/kg on once every 14-day regimen.
Paediatric subjects (0 to < 12 years of age).
In Study 3002, all 27 male subjects were PTPs on a 7-day prophylaxis regimen. The overall median AsBR was 0.00 bleeds per year per subject. No differences between subjects < 6 years of age and subjects 6 to < 12 years of age were observed with regard to the efficacy of a 7-day routine prophylaxis regimen. These results are consistent with the AsBR derived for the adult population receiving routine prophylaxis in Study 3001.
Consumption of Idelvion was measured as a secondary endpoint in Study 3002. In paediatric subjects on a 7-day Idelvion routine prophylaxis regimen, consumption was similar to that of adults on the same prophylaxis regimen. In addition, the monthly prophylaxis consumption of Idelvion administered per subject was lower than for their previous FIX; the mean (SD) monthly consumption of Idelvion was 200 (40) IU/kg (N = 27) compared with 390 (209) IU/kg FIX (N = 24). There was no difference in Idelvion consumption between the 2 paediatric age groups (subjects < 6 years of age and subjects 6 to < 12 years of age).
The long-term safety and efficacy of Idelvion routine prophylaxis treatment in PTPs was confirmed in an open-label extension study (3003) for up to 5 years. The extension study enrolled 83 PTPs including 59 adults and adolescents (≥ 12 years of age) and 24 paediatrics (< 12 years of age). These patients received Idelvion for either prophylaxis and/or for the treatment of bleeding episodes on an on-demand basis.
Control and prevention of bleeding episodes. Adult and adolescent subjects (≥ 12 to 65 years of age).
In studies 2004 and 3001, bleeding episodes were treated with Idelvion when they occurred (i.e. on demand). Successful treatment was defined as achieving haemostasis with no more than 2 injections. In addition, investigators evaluated the haemostatic efficacy via a 4-point scale (excellent, good, moderate, poor / no response).
Across studies 2004 and 3001, 65 subjects experienced a total of 443 bleeding episodes that were treated with Idelvion (432 were assessed for efficacy). Of these 443 bleeding episodes, 412 (93.0%) were controlled with a single Idelvion injection and another 26 (5.9%) were controlled with 2 injections. Five bleeding episodes (1.1%) required more than 2 injections.
For 94.6% of bleeding episodes the haemostatic efficacy rating was either excellent or good.
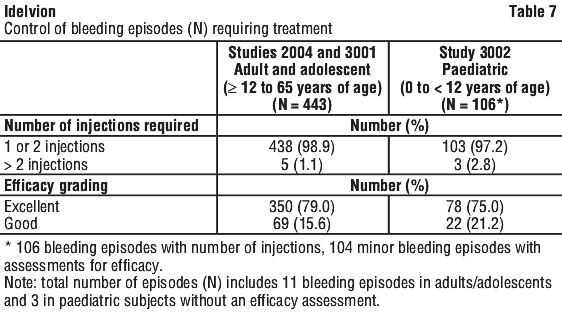
Paediatric subjects (0 to < 12 years of age).
In Study 3002, 23/27 subjects experienced a total of 106 bleeding episodes that were treated with Idelvion; the majority of bleeding episodes (103/106; 97.2%) were successfully treated with 1 or 2 injections of Idelvion. For most bleeding episodes requiring treatment, the investigator's assessment of haemostatic efficacy of Idelvion was either excellent (78/104 bleeding episodes; 75.0%) or good (22/104 bleeding episodes; 21.2%). These results were consistent for both age groups (see Table 7).
 Control and prevention of bleeding episodes in the perioperative setting. In the 2 pivotal studies (Study 3001 and Study 3002) and the extension study (Study 3003), a total of 21 subjects (5-59 years of age) received Idelvion for perioperative management of 30 surgical procedures, including 4 surgeries in children < 12 years of age. Dose was individualised based on the subject's PK and clinical response to treatment. These included 15 orthopaedic surgeries, a double mastectomy liposuction (n=1), haemorrhoidectomy (n=2), rhinoplasty, submucosal resection and inferior turbinectomy (n=1), circumcision (n=2), umbilical hernia and circumcision (n=1), teeth extractions (n=5), embolism of scrotal variceal (n=1), excision of pigmented nevus (n=1) and endoscopic mucosal resection (n=1).
Control and prevention of bleeding episodes in the perioperative setting. In the 2 pivotal studies (Study 3001 and Study 3002) and the extension study (Study 3003), a total of 21 subjects (5-59 years of age) received Idelvion for perioperative management of 30 surgical procedures, including 4 surgeries in children < 12 years of age. Dose was individualised based on the subject's PK and clinical response to treatment. These included 15 orthopaedic surgeries, a double mastectomy liposuction (n=1), haemorrhoidectomy (n=2), rhinoplasty, submucosal resection and inferior turbinectomy (n=1), circumcision (n=2), umbilical hernia and circumcision (n=1), teeth extractions (n=5), embolism of scrotal variceal (n=1), excision of pigmented nevus (n=1) and endoscopic mucosal resection (n=1).
A single preoperative bolus was used in 96.7% (n=29) of surgeries. Haemostatic efficacy was rated as excellent or good in ≥ 95.5% of the surgeries. During the 14-day postoperative period, patients received between 0 and 11 infusions and the Idelvion consumption was between 0 and 444.1 IU/kg.
PUP population.
The safety and efficacy of Idelvion was also evaluated in a multi-centre open-label clinical study (Study 3003) with 12 previously untreated paediatric patients (PUPs) with haemophilia B (≤ 2% endogenous FIX activity) who received at least one dose of Idelvion as part of routine prophylaxis once every 7 days, on-demand treatment and control of bleeding episodes. Eleven PUPs were below 6 years of age with a median (range) age of 0.0 (0 to 1) years and one PUP was 11 years old. The overall median (range) time on Idelvion was 22.65 (4.5 to 33.0) months, and the mean (SD) EDs to Idelvion in PUPs was 68.3 (37.94) EDs.
Routine prophylaxis in PUPs. All 12 PUPs received routine prophylaxis: 11 PUPs were on the 7-day regimen, and 1 PUP was on the 10-day regimen. The overall median time on prophylaxis was 11.50 (range: 3.1 to 32.3) months. In the 11 PUPs on the 7-day prophylaxis regimen, total annualised bleeding rate (ABR) ranged from 0 to 3.89. Five of the 11 PUPs had an ABR of 0 and 8 PUPs had an annualised spontaneous bleeding rate (AsBR) of 0. In the PUP study the mean (SD) monthly Idelvion dose was 193.66 (17.066) IU/kg for the 7-day prophylaxis regimen (N = 11) and 132.10 IU/kg for the 10-day prophylaxis regimen (N =1).
Control of bleeding episodes in PUPs. There were a total of 44 bleeding events in 12 PUPs across all study periods. Of those, 37 bleeding events observed in 10 PUPs were treated with Idelvion. Of the treated bleeding events, 16 were spontaneous, 17 traumatic, and 4 of unknown cause; 93.8% of spontaneous bleeding events were successfully controlled with 1 or 2 Idelvion infusions.
5.2 Pharmacokinetic Properties
Adult population (≥ 18 years to < 65 years).
The PK of Idelvion were evaluated following an intravenous injection of a single dose of 25, 50 and 75 IU/kg. The PK parameters (see Table 8) were based on plasma FIX activity measured by the one-stage clotting assay. Blood samples for PK analysis were collected prior to dosing and up to 336 hours (14 days) after dosing.
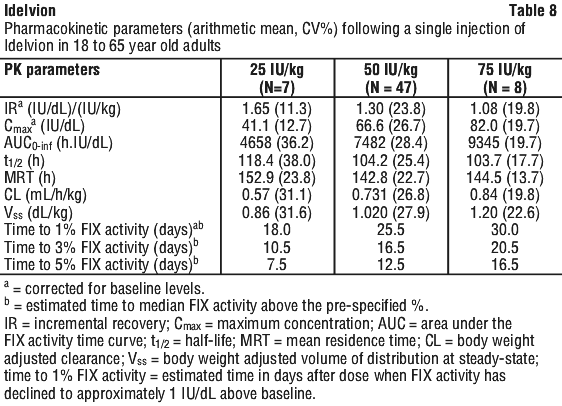 The PK data demonstrate that Idelvion has an improved PK profile with a prolonged circulating half-life, increased area under the FIX activity time curve, lower clearance and an increased incremental recovery compared with short-acting FIX replacement products. In the pivotal study, the mean (CV%) incremental recovery of Idelvion was 1.30 (23.8%) which is higher than that achieved 1.00 (25.7%) with the previous FIX product (plasma-derived FIX [pdFIX] or recombinant FIX [rFIX]). Therefore, 1 IU/kg Idelvion provides a mean increase of 1.30 IU/dL in the circulating level of FIX.
The PK data demonstrate that Idelvion has an improved PK profile with a prolonged circulating half-life, increased area under the FIX activity time curve, lower clearance and an increased incremental recovery compared with short-acting FIX replacement products. In the pivotal study, the mean (CV%) incremental recovery of Idelvion was 1.30 (23.8%) which is higher than that achieved 1.00 (25.7%) with the previous FIX product (plasma-derived FIX [pdFIX] or recombinant FIX [rFIX]). Therefore, 1 IU/kg Idelvion provides a mean increase of 1.30 IU/dL in the circulating level of FIX.
Repeat PK assessment for up to 30 weeks demonstrated a stable PK profile and incremental recovery was consistent over time.
The mean FIX activity at day 14 following a single dose of 75 IU/kg Idelvion was 6.65%. The mean FIX activity at days 7, 10, and 14 following a single dose of 50 IU/kg Idelvion was 13.76%, 9.59%, and 6.1%, respectively. The mean FIX activity at days 7, 10, and 14 following a single dose of 25 IU/kg Idelvion was 8.62%, 5.02%, and 2.96%, respectively.
Paediatric population.
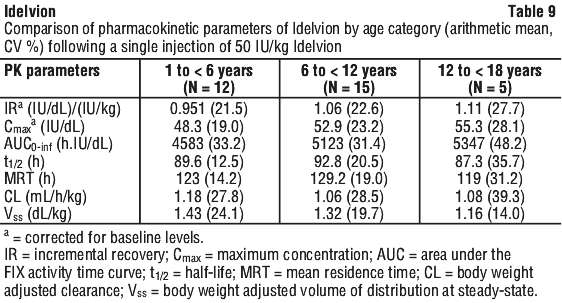
The PK parameters of Idelvion were evaluated in 5 adolescents (12 to < 18 years of age) and 27 children (1 to < 12 years of age) in open-label, multi-centre studies following an intravenous injection of a single dose of 50 IU/kg. The PK samples were collected prior to dosing and at multiple time points up to 336 hours (14 days) after dosing.
PK parameters were estimated based on the plasma FIX activity over time profile. Table 9 summarises the PK parameters calculated from the paediatric data of 32 subjects, 1 to < 18 years of age. Compared with adults, incremental recovery appeared to be slightly lower and body weight adjusted clearance appeared to be higher in children.
Population PK modelling.
Based on population PK modelling simulations (median data), the estimated time to reach 1% and 5% plasma FIX activity following a single dose of 25 IU/kg, 50 IU/kg and 75 IU/kg Idelvion are shown in Table 10.

5.3 Preclinical Safety Data
Genotoxicity.
Idelvion did not show evidence of genotoxicity in a reverse mutation assay in bacteria or a chromosomal aberration assay in human lymphocytes.
Carcinogenicity.
Carcinogenicity studies have not been conducted with Idelvion.6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium citrate dihydrate; polysorbate 80; mannitol; sucrose; hydrochloric acid (in small amounts for pH adjustment).
6.2 Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products or diluents.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the carton packaging.
Reconstituted product.
After reconstitution, the chemical and physical in-use stability has been demonstrated for 8 hours at room temperature (at or below 25°C). Idelvion contains no antimicrobial preservatives. If the product is not used immediately after reconstitution, store at room temperature (at or below 25°C) and use within 4 hours.
6.4 Special Precautions for Storage
Store at 2°C to 25°C. Do not freeze. Keep vials in the outer carton in order to protect from light. Do not use after the expiry date.
6.5 Nature and Contents of Container
Idelvion is available in the following presentations:
Powder (250/500/1000 IU) in a glass vial, with a rubber stopper, a plastic disc and an aluminium cap;
2.5 mL of water for injections in a glass vial, with a rubber stopper, a plastic disc and an aluminium cap.
Powder (2000/3500 IU) in a glass vial, with a rubber stopper, a plastic disc and an aluminium cap;
5 mL of water for injections in a glass vial, with a rubber stopper, a plastic disc and an aluminium cap.
Idelvion is packaged in latex free materials.
Idelvion is supplied as:
One pack with 250, 500 or 1000 IU containing: 1 vial with powder; 1 vial with 2.5 mL water for injections; 1 filter transfer set 20/20 (Mix2Vial); one administration pack containing: 1 disposable 5 mL syringe; 1 venipuncture set; 2 alcohol swabs.
One pack with 2000 IU or 3500 IU containing: 1 vial with powder; 1 vial with 5 mL water for injections; 1 filter transfer set 20/20 (Mix2Vial); one administration pack containing: 1 disposable 10 mL syringe; 1 venipuncture set; 2 alcohol swabs.
Not all registered presentations may be supplied.
6.6 Special Precautions for Disposal
Any unused medicinal product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
Idelvion contains albutrepenonacog alfa, a purified protein produced by recombinant DNA technology, generated by the genetic fusion of recombinant albumin to recombinant coagulation factor IX (FIX). The genetic fusion of the cDNA of human albumin to the cDNA of human coagulation FIX enables the protein to be produced as a single recombinant protein and assures product homogeneity by avoiding chemical conjugation. The recombinant FIX portion is identical to the Thr148 allelic form of plasma-derived FIX. The cleavable linker between the recombinant FIX and albumin molecules is derived from the endogenous activation peptide in native FIX. Idelvion remains intact in the circulation until FIX is activated, whereupon albumin is cleaved, releasing activated FIX (FIXa) when it is needed for coagulation.
Full length rIX-FP is expressed in recombinant Chinese hamster ovary cells as a single chain glycopeptide of 1018 amino acids with a molecular weight of ~125 kD.
CAS number.
1357448-54-4.7 Medicine Schedule (Poisons Standard)
Unscheduled.
Summary Table of Changes


 Idelvion contains approximately 75 mmol/L (1.7243 g/L) sodium. Idelvion is essentially sodium-free, containing less than 1 mmol sodium (23 mg) per vial. For the full list of excipients, see Section 6.1 List of Excipients.
Idelvion contains approximately 75 mmol/L (1.7243 g/L) sodium. Idelvion is essentially sodium-free, containing less than 1 mmol sodium (23 mg) per vial. For the full list of excipients, see Section 6.1 List of Excipients.
 No neutralising antibodies (inhibitors) (see Section 4.4 Special Warnings and Precautions for Use), or antibodies to hamster proteins have been detected in PTPs in the clinical studies with Idelvion. Inhibitor development has been observed in the post-marketing experience.
No neutralising antibodies (inhibitors) (see Section 4.4 Special Warnings and Precautions for Use), or antibodies to hamster proteins have been detected in PTPs in the clinical studies with Idelvion. Inhibitor development has been observed in the post-marketing experience. In addition, based on a matched pairs design, every 14-day prophylaxis with Idelvion was demonstrated to be as effective as every 7-day prophylaxis with Idelvion. The AsBRs for 7-day and 14-day prophylaxis are summarised in Table 5.
In addition, based on a matched pairs design, every 14-day prophylaxis with Idelvion was demonstrated to be as effective as every 7-day prophylaxis with Idelvion. The AsBRs for 7-day and 14-day prophylaxis are summarised in Table 5. In Study 3001, 23 subjects in the on demand arm received Idelvion as needed for the treatment of bleeding episodes. Nineteen subjects subsequently crossed-over to weekly prophylaxis after approximately 26 weeks of episodic treatment. These subjects showed a statistically significant reduction in the rate of spontaneous and total bleeding episodes after switching from on demand treatment to prophylaxis. During on demand treatment, the subjects had a median AsBR of 15.4 bleeding episodes per year per subject. After switching to weekly prophylaxis treatment, the median AsBR was 0.00 bleeding episodes per year per subject. The corresponding median and mean percent reductions in AsBR were 100% and 96.0% (P < 0.0001), respectively. Similar results were observed in the efficacy population. A comparison of the AsBRs and ABRs in the subjects evaluable for efficacy is summarised in Table 6.
In Study 3001, 23 subjects in the on demand arm received Idelvion as needed for the treatment of bleeding episodes. Nineteen subjects subsequently crossed-over to weekly prophylaxis after approximately 26 weeks of episodic treatment. These subjects showed a statistically significant reduction in the rate of spontaneous and total bleeding episodes after switching from on demand treatment to prophylaxis. During on demand treatment, the subjects had a median AsBR of 15.4 bleeding episodes per year per subject. After switching to weekly prophylaxis treatment, the median AsBR was 0.00 bleeding episodes per year per subject. The corresponding median and mean percent reductions in AsBR were 100% and 96.0% (P < 0.0001), respectively. Similar results were observed in the efficacy population. A comparison of the AsBRs and ABRs in the subjects evaluable for efficacy is summarised in Table 6. In Study 2004, subjects receiving weekly prophylaxis treatment had a lower AsBR (median: 1.13; N = 13) than subjects receiving on demand treatment only (median: 22.2; N = 4).
In Study 2004, subjects receiving weekly prophylaxis treatment had a lower AsBR (median: 1.13; N = 13) than subjects receiving on demand treatment only (median: 22.2; N = 4).
 The PK data demonstrate that Idelvion has an improved PK profile with a prolonged circulating half-life, increased area under the FIX activity time curve, lower clearance and an increased incremental recovery compared with short-acting FIX replacement products. In the pivotal study, the mean (CV%) incremental recovery of Idelvion was 1.30 (23.8%) which is higher than that achieved 1.00 (25.7%) with the previous FIX product (plasma-derived FIX [pdFIX] or recombinant FIX [rFIX]). Therefore, 1 IU/kg Idelvion provides a mean increase of 1.30 IU/dL in the circulating level of FIX.
The PK data demonstrate that Idelvion has an improved PK profile with a prolonged circulating half-life, increased area under the FIX activity time curve, lower clearance and an increased incremental recovery compared with short-acting FIX replacement products. In the pivotal study, the mean (CV%) incremental recovery of Idelvion was 1.30 (23.8%) which is higher than that achieved 1.00 (25.7%) with the previous FIX product (plasma-derived FIX [pdFIX] or recombinant FIX [rFIX]). Therefore, 1 IU/kg Idelvion provides a mean increase of 1.30 IU/dL in the circulating level of FIX.

