1 Name of Medicine
Illuccix 25 microgram, kit for Ga-68 Glu-urea-Lys(ahx)-hbed-CC radiopharmaceutical preparation.
2 Qualitative and Quantitative Composition
Illuccix is supplied as a sterile, multi-dose kit for the preparation of Ga-68 Glu-urea-Lys(ahx)-hbed-CC for intravenous use. There are 2 different kit configurations, each containing 3 vials.
Illuccix Configuration "A" is recommended for. but not limited to use with Ga-68 produced from a cyclotron and purified via GE FASTlab or Eckert and Ziegler GalliaPharm Ge 68/Ga-68 generator and includes:
Vial 1 (Glu-urea-Lys(ahx)-hbed-CC Vial): contains 25 microgram Glu-urea-Lys(ahx)-hbed-CC, 10 microgram mannose and water for injections as a lyophilized powder in a sterile 10 mL vial with a blue flip-off cap.
Vial 2 (Buffer Vial, Configuration A): contains 150 mg sodium acetate, 0.077 mL hydrochloric acid and water for injections (2.5 mL volume) in a sterile 10 mL vial with a red flip off cap.
Vial 3 (Sterile Vacuumed Vial): an evacuated sterile vial with white flip off cap used to collect Ga-68 chloride from generators or cyclotron.
Illuccix Configuration "B" is recommended for, but not limited to use with Ga-68 produced from an IRE Galli Eo Ge 68/Ga-68 generator and includes:
Vial 1 (Glu-urea-Lys(ahx)-hbed-CC Vial): contains 25 microgram Glu-urea-Lys(ahx)-hbed-CC, 10 microgram mannose and water for injections as a lyophilized powder in a sterile 10 mL vial with a blue flip-off cap.
Vial 2 (Buffer Vial, Configuration B): contains 150 mg sodium acetate, 0.15 mL hydrochloric acid and water for injections (6.4 mL volume) in a sterile 10 mL vial with a green flip off cap.
Vial 3 (Sterile Vacuumed Vial): an evacuated sterile vial with white flip off cap used to collect Ga-68 chloride from generator.
The radionuclide is not part of the kit. Before reconstitution and radiolabeling with Ga-68, the contents of this kit are not radioactive.
The Illuccix Cold Kit is an ARTG-registered product (please refer to https://www.tga.gov.au/resources/auspmd/illuccix) and undergoes evaluation for Quality, Safety, and Efficacy, in accordance with the TGA regulations for registration.
Reconstitution of radiopharmaceuticals is exempted from registration in Australia, in accordance with Item 6 of Schedule 5 of the Therapeutic Goods Regulation 1990. Therefore, the TGA does not impose control over the choice of Ge68/Ga68 generator to be used in the production of Illuccix Ga68-PSMA.
The prepared Ga-68 Glu-urea-Lys(ahx)-hbed-CC injection for intravenous use is a sterile, pyrogen free, clear, colourless, buffered solution with a pH between 4.0-5.0.
Physical characteristics.
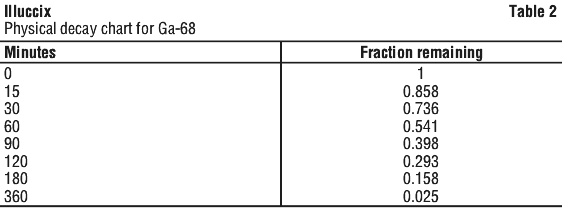
Gallium 68 (Ga-68) decays with a half-life of 68 minutes to stable Zinc-68. Principal radiation emission data and physical decay chart for Ga-68 listed in Table 1 and Table 2.

External radiation.
Radiation attenuation by lead shielding listed in Table 3.

3 Pharmaceutical Form
Powder for injection.
Glu-urea-Lys(ahx)-hbed-CC is white to off-white traces, practically free of particles. Buffer is clear and colourless solution, practically free of visible particles.
4.1 Therapeutic Indications
Illuccix, after radiolabeling with Ga-68, is a radioactive diagnostic agent indicated for use with positron emission tomography (PET) imaging combined with computerised tomography (CT) in patients with prostate cancer:
who are at risk of metastasis and who are suitable for initial definitive therapy;
who have suspected recurrence based on elevated serum prostate specific antigen (PSA) level;
for the selection of patients with metastatic prostate cancer in whom lutetium Lu 177 vipivotide tetraxetan PSMA-directed therapy is indicated.
4.2 Dose and Method of Administration
Radiation safety - drug handling.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC is a radioactive drug and should be handled with appropriate safety measures to minimize radiation exposure (see Section 4.4 Special Warnings and Precautions for Use). Use waterproof gloves, effective radiation shielding, and appropriate safety measures when preparing and handling Ga-68 Glu-urea-Lys(ahx)-hbed-CC.
Radiopharmaceuticals should be used by or under the control of physicians who are qualified by specific training and experience in the safe use and handling of radionuclides, and whose experience and training have been approved by the appropriate governmental agency authorized to license the use of radionuclides.
Patient preparation.
Advise patients to drink adequate amounts of water to ensure that they are sufficiently hydrated prior to administration of Ga-68 Glu-urea-Lys(ahx)-hbed-CC. During the first hour following administration, patients should drink and void frequently to reduce radiation exposure.
Recommended dosage and administration instructions.
The recommended amount of activity to be administered intravenously is 185 MBq (5 mCi) with a range of 166.5 to 203.5 MBq (4.5 to 5.5 mCi) and a maximum dose of 25 microgram of Glu-urea-Lys(ahx)-hbed-CC.
After reconstitution with Ga-68 eluate from an appropriate source [see Drug preparation], Ga-68 Glu-urea-Lys(ahx)-hbed-CC is administered by intravenous injection (bolus). The speed of administration depends on the venous tolerance to low pH solution, which is mainly dependent of the blood flow of the vein used for the injection. Tolerance can be increased by diluting Gallium (68Ga) Glu-urea-Lys(ahx)-hbed-CC Injection in isotonic saline solution.
Before administration to the patient, measure the radioactivity of the vial containing Ga-68 Glu-urea-Lys(ahx)-hbed-CC using a dose calibrator and calculate the necessary volume to administer based on calibration date and time. Ensure that the radioactivity to be injected is within ± 10% of the recommended dose.
Drug preparation.
Illuccix is supplied as 3 vials which allows for direct preparation of Illuccix with eluate from any appropriate Ga-68 source. The below instructions can be used with Telix's suggested Ga-68 sources:
Eckert and Ziegler GalliaPharm Germanium-68/Gallium-68 (Ge-68/Ga-68) generator (Configuration A).
Cyclotron produced via GE FASTlab (Configuration A).
IRE ELIT Galli Eo Ge-68/Ga-68 generator (Configuration B).
The Ge-68/Ga-68 generators and cyclotron are not supplied with Illuccix.
Components of Illuccix include:
Vial 1 (Glu-urea-Lys(ahx)-hbed-CC Vial) contains 25 microgram of Glu-urea-Lys(ahx)-hbed-CC and 10 microgram of D-mannose.
Vial 2 (Acetate Buffer Vial) contains 150 mg sodium acetate in HCl buffer.
Vial 3 (Vacuumed Vial) is a sterile, evacuated vial that serves as the collection vial for Ga-68 chloride.
Prepare Ga-68 Glu-urea-Lys(ahx)-hbed-CC for intravenous injection according to the following aseptic procedure:
a. Use suitable shielding to reduce radiation exposure.
b. Wear waterproof gloves.
c. If Ga-68 is generator produced, test the Ga-68 chloride eluate for Ge-68 breakthrough by a suitable method according to manufacturer recommendations. Ge-68 breakthrough and other gamma emitting radionuclides should meet the specifications provided by the manufacturer.
d. Place a "radioactive" label on Vial 3 (Vacuumed Vial).
e. Remove the flip-off disc top from the Vial 1, Vial 2 and Vial 3.
f. Swab the top of each vial with alcohol to disinfect the surface and allow the top of each vial to dry.
g. Note that to minimize any potential metallic contamination, the shortest possible needle should be used for the transfer of the gallium solution from the generator. The needle should be clean and dilute acid resistant.
h. Note that all syringes used in the steps below should be plastic. Syringes with rubber plungers should not be used.
i. Note that prior to use of any vial, confirm the correct vial is being used by a visual check of the vial label.
j. Follow the specific reconstitution procedure below, dependent on Ga-68 source. Then continue with the dilution and radiosynthesis procedure.
Reconstitution with Eckert and Ziegler GalliaPharm generator.
1. Pierce Vial 3 (Vacuumed Vial) with a sterile needle connected to a 0.22 micron sterile vented filter (not supplied) to maintain atmospheric pressure within the vial during the reconstitution process.
2. Connect the male luer of the outlet line of the GalliaPharm generator to a sterile needle.
3. Connect Vial 3 (Vacuumed Vial) directly to the outlet line of the GalliaPharm generator by pushing the needle through the rubber septum and place the vial in a radiation shielded container.
4. Prepare a syringe containing 5 mL of sterile ultrapure 0.1 M HCl provided with the GalliaPharm generator for elution.
5. Elute the generator directly into Vial 3 (Vacuumed Vial) according to the instructions for use of the GalliaPharm generator that are supplied by Eckert and Ziegler. Perform the elution manually or by means of a pump. Collect eluate.
6. At the end of the elution, disconnect the generator from Vial 3 (Vacuumed Vial) by removing the needle from the rubber septum.
Reconstitution with IRE Galli Eo generator.
1. Connect the male luer of the outlet line of the Galli Eo generator to a sterile needle.
2. Elute the generator directly into Vial 3 (Vacuumed Vial) according to the instructions for use of the Galli Eo generator that are supplied by IRE. Collect eluate.
3. At the end of the elution, disconnect the generator from Vial 3 (Vacuumed Vial) by removing the needle from the rubber septum.
Reconstitution with cyclotron produced Ga-68 via GE FASTlab.
1. After purification by the FASTlab, the [68Ga]GaCl3 solution is passed through a sterile filter and into the cassette product vial automatically by the FASTlab.
2. Pierce Vial 3 (Vacuumed Vial) with a sterile needle connected to a 0.22 micron sterile vented filter (not supplied) to maintain atmospheric pressure within the vial during the reconstitution process.
3. Aseptically transfer [68Ga]GaCl3 solution into Vial 3 (Vacuumed Vial).
Dilution.
1. Insert a sterile 10 mL syringe into Vial 2 (Acetate Buffer Vial) and draw up the contents of the vial (target 2.5 mL or target 6.4 mL depending on the configuration).
2. Inject the contents of the 10 mL syringe into Vial 1 (Glu-urea-Lys(ahx)-hbed-CC Vial).
3. Gently swirl Vial 1 to ensure the product is thoroughly dissolved.
Radiosynthesis procedure.
1. Insert a sterile 10 mL syringe into Vial 1 (Sterile Glu-urea-Lys(ahx)-hbed-CC Vial) and draw up the entire contents of the vial.
2. Transfer the contents of the 10 mL syringe to Vial 3 containing the Ga-68 chloride.
3. Wait for 5 minutes for radiolabeling to take place.
4. Assay the whole vial containing the Ga-68 Glu-urea-Lys(ahx)-hbed-CC Injection for total radioactivity concentration using a dose calibrator and record the result.
5. Perform the quality control of the reconstituted solution according to the recommended methods to check the compliance with the specifications (see Specifications and quality control).
6. Prior to use, visually inspect the solution behind a shielded screen for radioprotection purposes. Only use solutions that are clear without visible particles.
7. Keep the vial containing the Ga-68 Glu-urea-Lys(ahx)-hbed-CC Injection upright in a radioprotective shield container at ambient room temperature until use.
8. After transfer of the contents of Vial 1 (Glu-urea-Lys(ahx)-hbed-CC in acetate buffer) into Vial 3 containing the Ga-68, use Ga-68 Glu-urea-Lys(ahx)-hbed-CC Injection within 4 hours. The final volume of the reconstituted product is 7.5 mL.
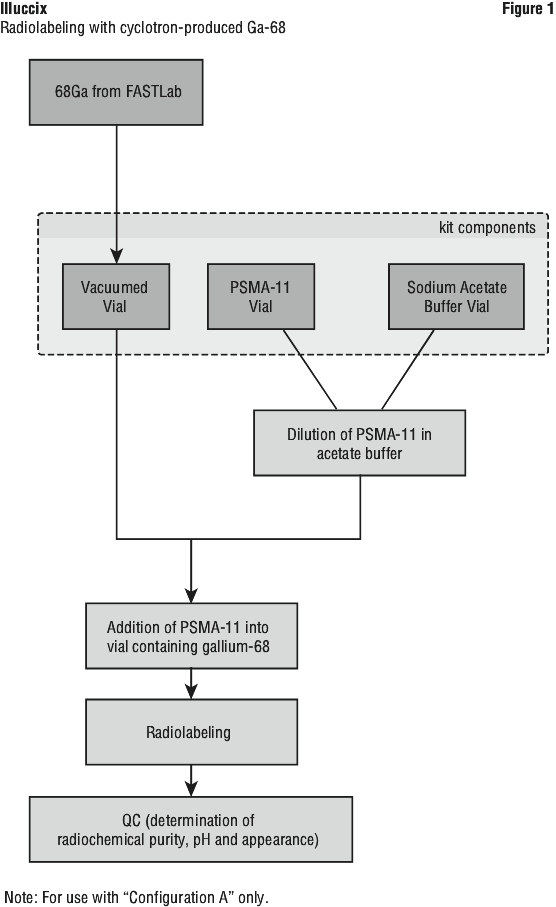
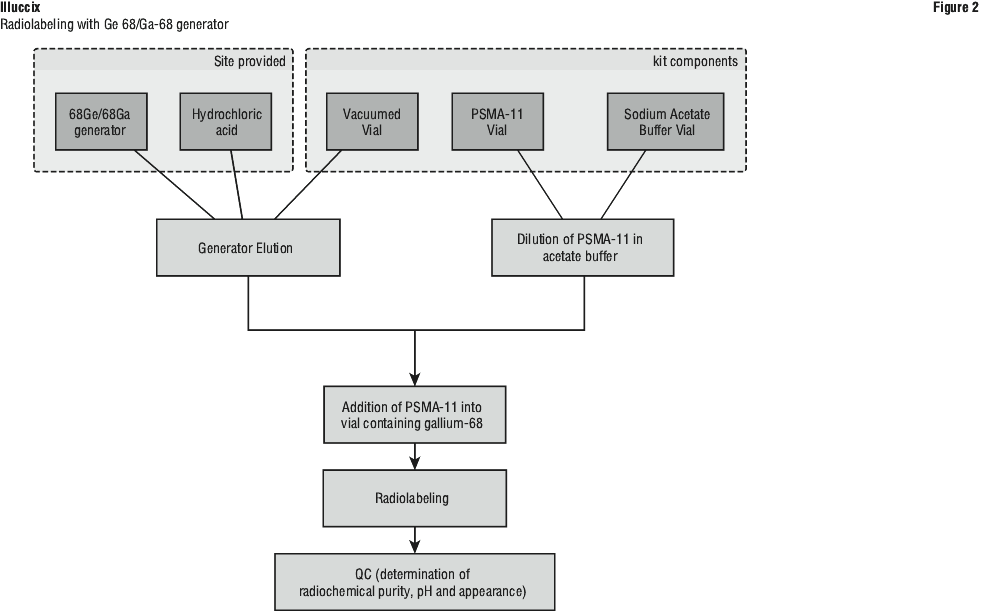
Flow diagrams are provided for the radiosynthetic process to be followed at the radiopharmacy site for:
Illuccix Configuration "A" when using Ga-68 prepared by cyclotron (Figure 1) or;
Illuccix Configurations "A" or "B" when using an appropriate Ge 68/Ga-68 generator (Figure 2).
Specifications and quality control.
Perform the quality controls in Table 4 behind a lead glass shield for radioprotection purposes.
 Perform the following:
Perform the following:
1. Pour ammonium acetate 1M/methanol (1/1 V/V) solution to a depth of 3 to 4 mm in the developing chamber, cover the chamber and allow it to equilibrate.
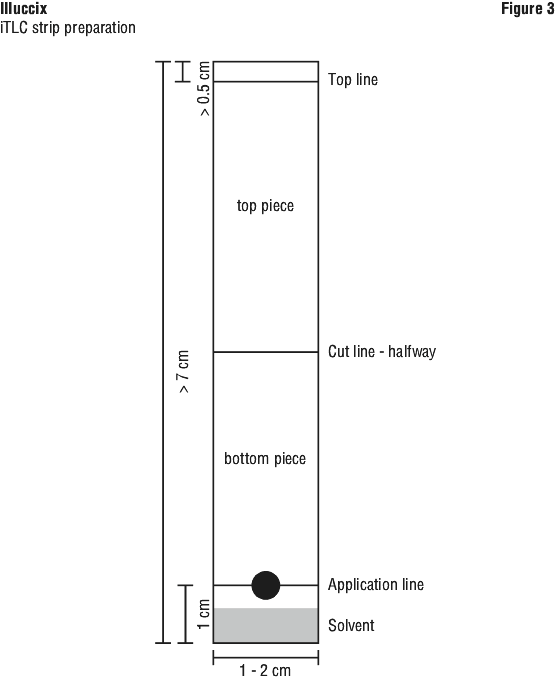
2. Prepare the Chromatography strip that is at least 7 cm long and 1- 2 cm wide.
3. Using a pencil, mark a line on the iTLC strip, 1 cm from the bottom, and at least 0.5 cm from the top (see Figure 3).
4. Using a pencil, mark the iTLC strip halfway between two markings from step 3 (1 cm from the bottom and 0.5 cm from the top) (see Figure 3).
5. Apply one to two drops of Ga-68 Glu-urea-Lys(ahx)-hbed-CC on the pencil line at the 1 cm mark from the bottom of the iTLC strip. Apply the drop on the appropriate side of the iTLC strip, as directed by the manufacturer's instructions.
6. Place the iTLC strip in the developing chamber. The lower side of the strip containing the 1 cm mark and the drop(s) of Ga-68 Glu-urea-Lys(ahx)-hbed-CC should be placed inside the chamber. Ensuring that the bottom of the iTLC strip sits in the liquid in the chamber, but that the liquid does not touch the drop of Ga-68 Glu-urea-Lys(ahx)-hbed-CC placed at the 1 cm marker.
7. Allow the iTLC strip to develop in the covered chamber until it reaches the mark at the top end of the iTLC strip.
8. Cut horizontally at the line marked halfway along the iTLC strip.
9. Using a dose calibrator, measure and record the radioactivity in the top half of the iTLC strip.
10. Using a dose calibrator, measure and record the radioactivity in the bottom half of the iTLC strip.
11. Calculate the quantity (in percent) of Ga-68 Glu-urea-Lys(ahx)-hbed-CC in the solution using the formula, accounting for any background radiation (see Equation 1):
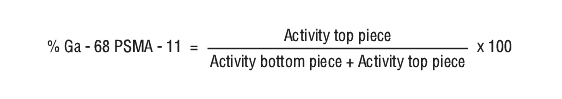 12. Scan the iTLC with a radiometric iTLC scanner.
12. Scan the iTLC with a radiometric iTLC scanner.
i. Calculate the quantity (in percent) of Ga-68 Glu-urea-Lys(ahx)-hbed-CC in the solution by integration of the peaks on the chromatogram.
ii. The retention factor (Rf) specifications are:
Free and colloidal Ga-68 species, Rf = 0 to 0.1,
Ga-68 Glu-urea-Lys(ahx)-hbed-CC, Rf = 0.6 to 1.
Administration.
The vial containing the Ga-68 Glu-urea-Lys(ahx)-hbed-CC solution is intended for multi-use. Prior to use, visually inspect the prepared Ga-68 Glu-urea-Lys(ahx)-hbed-CC behind a shielded screen for radioprotection purposes. Only use solutions that are clear, colourless or at most slightly yellow, and without visible particles. Use a single-dose syringe fitted with a sterile needle and protective shielding for each dose. Aseptically withdraw the prepared Ga-68 Glu-urea-Lys(ahx)-hbed-CC prior to administration. Measure the total radioactivity in the syringe by a dose calibrator immediately prior to administration. The dose calibrator must be calibrated with the National Institute of Standards and Technology (NIST) traceable standards.
If repeated withdrawals are made, the replacement of the contents of the vial with air should be minimized, and separate syringes must be used for each patient.
Handle and dispose of any unused drug in accordance with applicable regulations.
Image acquisition.
Position the patient supine with arms above the head. Begin PET scanning approximately 60 minutes after the intravenous administration of Ga-68 Glu-urea-Lys(ahx)-hbed-CC. It is recommended that image acquisition should include a whole-body acquisition from the top of the skull through midthigh, with the acquisition beginning at mid-thigh. Adapt imaging acquisition delay and duration according to the equipment used, and the patient and tumor characteristics, to obtain the best image quality possible.
Image interpretation.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC binds to the Prostate Specific Membrane Antigen (PSMA). Ga-68 Glu-urea-Lys(ahx)-hbed-CC images should be interpreted by appropriately trained personnel. PET/CT images with Ga-68 Glu-urea-Lys(ahx)-hbed-CC should be interpreted visually. Suspicion of cancer in sites typical for prostate cancer recurrence is based on Ga-68 Glu-urea-Lys(ahx)-hbed-CC uptake in comparison with tissue background. Usually attention should be paid to prostate gland/bed, seminal vesicles, regional ad distant lymph nodes, bones, lungs, and liver, as the regions that may relate to any symptoms given by the patient.
Normal and variable PSMA ligand update can be found in the lacrimal gland, salivary glands, liver, spleen, celiac lymph nodes, small intestine, kidney and the ureters. Variably high activity can be observed in the urinary bladder. Minor but visible uptake can be observed in the pharyngeal and laryngeal area and the cavum, the thyroid gland and mediastinal lymph nodes. Usually, tumour lesions inside and outside the prostate gland show a strong tumour-background ratio compared to surrounding tissue.
The impact of quantitative/semiquantitative measurement of Ga-68 Glu-urea-Lys(ahx)-hbed-CC uptake as an aid to image interpretation has not been assessed. Image interpretation errors can occur with Gallium (68Ga) Glu-urea-Lys(ahx)-hbed-CC Injection PET/CT (see Section 4.4 Special Warnings and Precautions for Use, Risk for image misinterpretation).
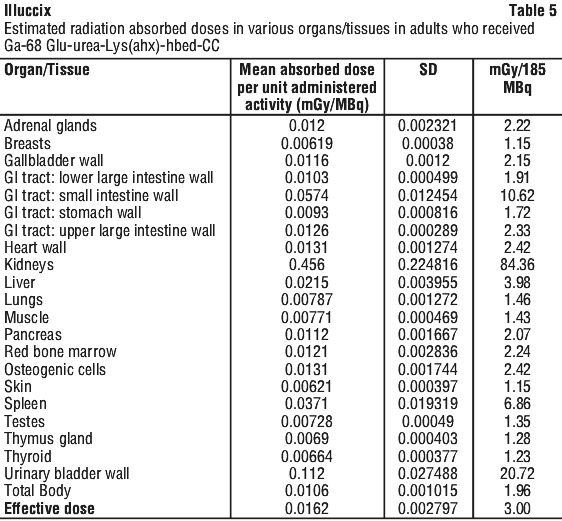
Radiation dosimetry.
The radiation absorbed doses estimated for adult patients following intravenous injection of Ga-68 Glu-urea-Lys(ahx)-hbed-CC are shown in Table 5, based on a dosimetry study using Illuccix (Configuration B) in healthy volunteers. Values were calculated from human biodistribution data using OLINDA/EXM (Organ Level Internal Dose Assessment/Exponential Modeling) software.
The (radiation absorbed) effective dose resulting from the administration of the recommended activity of 185 MBq of Ga-68 Glu-urea-Lys(ahx)-hbed-CC is 3.00 mSv. For an administered activity of 185 MBq (5 mCi), the highest-magnitude radiation doses are delivered to the kidney, bladder wall, small intestine wall, and spleen, with absorbed doses of 84.4 mGy, 20.7 mGy, 10.6 mGy, and 6.9 mGy, respectively. If a CT scan is simultaneously performed as part of the PET procedure, exposure to ionizing radiation will increase in an amount dependent on the settings used in the CT acquisition.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in Section 2.
4.4 Special Warnings and Precautions for Use
Extravasation risk.
Due to acidic pH of Gallium (68Ga) Glu-urea-Lys(ahx)-hbed-CC Injection, local vein irritation can occur. Accidental extravasation may also cause local irritation.
In case of local vein irritation, injection must be adapted accordingly by the health care professional. In case of extravasation, the injection must be stopped, the site of injection must be changed and the affected area should be irrigated with sodium chloride solution. The impact of extravasation on the actually injected dose and on image quality has to be considered when interpreting the imaging results.
Risk for image misinterpretation.
Image interpretation errors can occur with Ga 68 Glu-urea-Lys(ahx)-hbed-CC PET. A negative image does not rule out the presence of prostate cancer and a positive image does not confirm the presence of prostate cancer. It is recommended that appropriately trained and experienced personnel perform and interpret 68Ga Glu-urea-Lys(ahx)-hbed-CC PET.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC uptake is not specific for prostate cancer. An important pitfall is uptake in coeliac ganglia of the autonomic nervous system which may be misinterpreted as retroperitoneal lymph node metastases. Ga-68 Glu-urea-Lys(ahx)-hbed-CC uptake may occur with other types of cancer and with benign conditions such as prostatitis, benign prostatic hyperplasia, Paget's disease, fibrous dysplasia, and osteophytosis. False-positive cases have been also described in association with an inflammatory response after cryotherapy and radiation artefacts in patients previously treated with radiotherapy.
Clinical correlation, which may include histopathological evaluation of the suspected recurrence site, should be considered where appropriate.
Radiation risks.
Like other radiopharmaceuticals, Ga-68 Glu-urea-Lys(ahx)-hbed-CC contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Safe handling should be ensured to minimize radiation exposure to the patient and health care workers. Radiopharmaceutical agents should be used only by qualified personnel with the appropriate government authorisation for use and manipulation of radionuclides. [See Section 4.2 Dose and Method of Administration, Radiation safety - drug handling].
After the procedure.
Close contact with infants and pregnant women should be restricted during the initial 2 hours following the injection.
The patient should be encouraged to drink sufficient amounts and void as often as possible during the first hours after the scan in order to reduce radiation exposure of the bladder.
Use in renal impairment.
Gallium (68Ga) Glu-urea-Lys(ahx)-hbed-CC Injection prepared by Illuccix has not been studied in patients with renal impairment.
Considering the renal excretion of Illuccix, careful consideration of the activity to be administered is required since an increased radiation exposure is possible in these patients.
Use in the elderly.
Of the patients included in the clinical studies of Ga-68 Glu-urea-Lys(ahx)-hbed-CC, the mean age was 69 years with a range of 44.7 to 94.0 years. No overall difference in efficacy was observed between older subjects and younger subjects. Subjects aged 65 years or more reported more adverse events compared to subjects aged less than 65 years. However, the number of patients in the trials was not large enough to allow definitive comparisons.
Paediatric use.
Safety and effectiveness have not been established in paediatric patients.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
There are no known interactions to date. Given the microdose administered, and given that significant hepatic metabolism is unlikely, the risk of clinically significant pharmacokinetic drug interactions is very low. The reduction in signal from the urinary bladder seen with the concomitant administration of furosemide, suggested that the use of furosemide and possibly other diuretics, could reduce the scatter severity in Ga-68 Glu-urea-Lys(ahx)-hbed-CC PET/CT.
Androgen deprivation therapy (ADT) and other therapies targeting the androgen pathway, such as androgen receptor antagonists, may result in changes in uptake of Ga-68 Glu-urea-Lys(ahx)-hbed-CC in prostate cancer. The effect of these therapies on performance of Ga-68 Glu-urea-Lys(ahx)-hbed-CC PET has not been established.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
The effects of Ga-68 Glu-urea-Lys(ahx)-hbed-CC on fertility have not been evaluated in animal studies. Patients with partner of childbearing potential should be informed of potential impact of PET/CT on spermatogenesis as the binding and internalization of [68Ga] Glu-urea-Lys(ahx)-hbed-CC at the level of the prostate could however theoretically lead to DNA damage during spermatogenesis.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC is not indicated for use in females. There are no studies with Ga-68 Glu-urea-Lys(ahx)-hbed-CC in pregnant women to inform any drug-associated risks; however, all radiopharmaceuticals, including Ga-68 Glu-urea-Lys(ahx)-hbed-CC have the potential to cause fetal harm. Animal reproduction studies have not been conducted with Ga-68 Glu-urea-Lys(ahx)-hbed-CC.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC is not indicated for use in females. There is no information on the presence of Ga-68 Glu-urea-Lys(ahx)-hbed-CC in human milk, the effect on the breastfed infant, or the effect on milk production.4.7 Effects on Ability to Drive and Use Machines
Gallium (68Ga) Glu-urea-Lys(ahx)-hbed-CC Injection has no known influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials experience.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The safety of Ga-68 Glu-urea-Lys(ahx)-hbed-CC was evaluated in one retrospective efficacy and safety study, one prospective safety study (n=206), one retrospective observational study (n=194), in a survey of the scientific literature in patients with prostate cancer who received a single dose of Ga-68 Glu-urea-Lys(ahx)-hbed-CC (n=1882) and in the VISION study (n=1003). See Section 5.1 Pharmacodynamic Properties, Clinical trials.
Tabulated list of adverse reactions.
The following list of adverse reactions is based on experience from clinical trials and in available literature. Adverse reactions are displayed by system organ class and frequency in Table 6 and defined as very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000) and not known (cannot be estimated from the available data).
Adverse reactions were reported rarely (< 1/1,000) during clinical trials and in available literature.

Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
In the event of a radiation overdose, the absorbed dose to the patient should be reduced where possible by increasing the elimination of the radionuclide from the body by reinforced hydration and frequent bladder voiding. A diuretic might also be considered. If possible, an estimate of the radioactive dose given to the patient should be performed.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC binds to the prostate specific membrane antigen (PSMA) on cells that express PSMA including malignant prostate cancer cells, which overexpress PSMA. Ga-68 is a β+ emitting radionuclide with an emission yield that allows positron emission tomography (PET) imaging.
Clinical trials.
The efficacy of Ga-68 Glu-urea-Lys(ahx)-hbed-CC was established in a retrospective, open-label single center study.
Findings from the retrospective study are further supported by relevant evidence systematically collected from clinical studies in peer reviewed literature.
Additional results have been gained in the phase 3 VISION study investigating the radioligand lutetium Lu 177 vipivotide tetraxetan in a specific population of previously treated metastatic castrate-resistant prostate cancer patients using Ga-68 Glu-urea-Lys(ahx)-hbed-CC for screening of patients with a PSMA-positive gallium Ga-68 Glu-urea-Lys(ahx)-hbed-CC PET/CT scan and hence eligible for this radioligand therapy.
The relationship between Ga-68 PSMA 11 plasma concentrations and successful imaging was not explored in clinical trials.
Retrospective study.
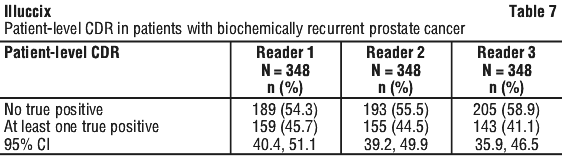
A total of 422 adult patients (mean age 68.4) with biochemical recurrence were evaluated with Ga-68 Glu-urea-Lys(ahx)-hbed-CC PET. All patients had biopsy proven prostate cancer that had been treated with curative intent and who presented with rising PSA. The mean age was 68 years, 93% were white, 80% had had prior prostatectomy, and mean baseline PSA was 11 nanogram/mL. Each participant received a single 68Ga Glu-urea-Lys(ahx)-hbed-CC PET from mid-thigh to skull base. The mean activity dose administered was 244 MBq and scans were performed on average 70 minutes after administration. There were 295 participants who received 68Ga Glu-urea-Lys(ahx)-hbed-CC as prepared using the Illuccix kit and 127 who received 68GA Glu-urea-Lys(ahx)-hbed-CC prepared by an alternate method. The images were read by three independent readers blinded to clinical and conventional imaging information. Diagnostic performance of Ga-68 Glu-urea-Lys(ahx)-hbed-CC in localizing tumor sites was assessed in 348 patients for whom a composite truth standard using biopsy/histopathology and/or conventional imaging was available. The co-primary efficacy endpoints were patient-level correct detection rate (CDR) and region-level verified localization rate (VLR). Patient-level CDR was defined as the percentage of patients who have at least one true positive lesion exactly localized between PET imaging and the reference standard, regardless of any coexistent false positive findings, out of all patients scanned and for whom a reference standard was available. Region-level VLR was defined as the percentage of regions containing at least one TP lesion (exactly localized correspondence between PET imaging and the reference standard), regardless of any co-existent FP findings within the same region, out of all regions containing at least one PET-positive finding. The regions evaluated include prostate bed, pelvic lymph nodes, skeleton, and other distant sites (extra-pelvic lymph nodes and viscera). Table 7 and Table 8 show the CDR and VLR of Ga-68 Glu-urea-Lys(ahx)-hbed-CC. The CDR results for the 3 independent readers ranged from 41.1% to 45.7%, all of which were significantly greater than the prespecified 20% threshold. The VLR results for the 3 independent readers ranged from 36.2% to 50%, all of which were significantly greater than the prespecified threshold of 10%. The results of the independent reads were generally consistent with one another.
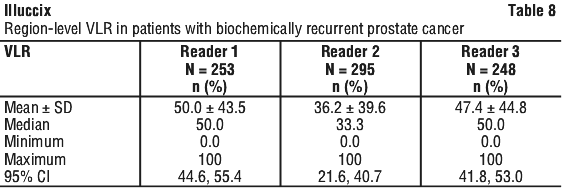 VLR was also evaluated separately by region (prostate bed, pelvic lymph nodes, skeleton, and other distant sites [extra-pelvic lymph nodes and viscera]). Ga-68 Glu-urea-Lys(ahx)-hbed-CC localized prostate cancer lesions in all regions and showed the highest localization rates in bone (VLR range 50 to 68%) and the lowest localisation rate in the prostate bed (VLR range 31 to 44%).
VLR was also evaluated separately by region (prostate bed, pelvic lymph nodes, skeleton, and other distant sites [extra-pelvic lymph nodes and viscera]). Ga-68 Glu-urea-Lys(ahx)-hbed-CC localized prostate cancer lesions in all regions and showed the highest localization rates in bone (VLR range 50 to 68%) and the lowest localisation rate in the prostate bed (VLR range 31 to 44%).
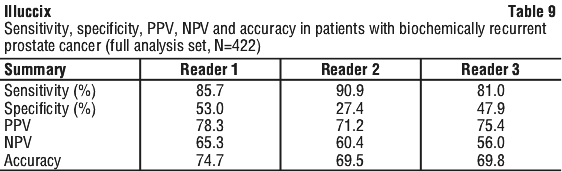
Ga-68 Glu-urea-Lys(ahx)-hbed-CC sensitivity, specificity, PPV, NPV, and accuracy are presented by reader in Table 9.
 The percentage of patients who were scanned and had at least one false positive finding, regardless of any coexistent TP lesions, ranged from 54.9% to 73.6% in the 348 participants.
The percentage of patients who were scanned and had at least one false positive finding, regardless of any coexistent TP lesions, ranged from 54.9% to 73.6% in the 348 participants.
An analysis of CDR and VLR according to 68Ga Glu-urea-Lys(ahx)-hbed-CC preparation method found similar results to the main analysis.
Literature review.
The diagnostic efficacy of Ga-68 Glu-urea-Lys(ahx)-hbed-CC is also based on a review of the worldwide literature: A systematic search identified 56 articles in patients with newly diagnosed prostate cancer published through 31 March 2020 and 27 articles in patients with biochemical recurrence published through 20 May 2020. Only studies that included biopsy/histology as a standard of truth were included. In patients newly diagnosed with PC, the sensitivity and specificity of Ga-68 Glu-urea-Lys(ahx)-hbed-CC was good and ranged from 24%-100% and 45%-100%, respectively; accuracy ranged from 60%-100%. A subset of studies in patients with newly diagnosed prostate cancer systematically evaluated change in management (n=11) associated with Ga-68 Glu-urea-Lys(ahx)-hbed-CC scan results. In these studies, 13%-42.5% of patients had their treatment plan modified based on Ga-68 Glu-urea-Lys(ahx)-hbed-CC scan results. In patients with biochemical recurrence, the sensitivity of Ga-68 Glu-urea-Lys(ahx)-hbed-CC ranged from 63.3%-100%; specificity ranged from 63%-100%; accuracy ranged from 71%-100%. In the subset of studies that evaluated change in management (n=7), 30%-75% of patients with biochemical recurrence had their treatment plan modified based on the Ga-68 Glu-urea-Lys(ahx)-hbed-CC scan results.
VISION.
The efficacy of gallium Ga-68 Glu-urea-Lys(ahx)-hbed-CC for selecting patients for radioligand therapy was evaluated in the VISION study (NCT03511664), a randomized (2:1), multicenter, open-label trial evaluating lutetium Lu 177 vipivotide tetraxetan radioligand therapy (7.4 GBq every 6 weeks for four to six cycles, n=551) plus standard care or standard care alone (control group, n=280) in men with progressive metastatic castration resistant prostate cancer. Ga-68 Glu-urea-Lys(ahx)-hbed-CC was used to identify PSMA-positive patients on PET imaging. Only PSMA-positive patients were eligible for randomization and receipt of lutetium Lu 177 vipivotide tetraxetan radioligand therapy.
A total of 1003 adult male patients received gallium Ga-68 Glu-urea-Lys(ahx)-hbed-CC (median decay-corrected dose of 166.60 MBq (range: 92.8-287.5), or 1.92 MBq/kg (range: 0.9-3.7) when corrected for body weight) by intravenous administration and underwent PET imaging approximately 60 minutes (range 50 to 100 minutes) after injection (i.e. at baseline, 1-6 weeks before the start of radioligand treatment). Ga-68 Glu-urea-Lys(ahx)-hbed-CC PET imaging was interpreted in conjunction with post-contrast CT and/or MRI of the chest, abdomen, and pelvis for all patients, and in addition to brain MRI and/or CT in patients with history of CNS metastasis.
Patients were of median age 70 years (range: 40 to 94), white (87%), black or African American (7%) and Asian (2.4%) and had median baseline serum PSA levels of 74 nanogram/mL (range: 0 to 8995 nanogram/mL) and a Gleason score of 8-10 at diagnosis in 58.8% and 60.7% in the lutetium Lu 177 vipivotide tetraxetan plus standard care group and the standard care alone group, respectively.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC PET and anatomical imaging was interpreted by one central reader who was blinded to clinical information and other PET and bone imaging. PSMA-positive (eligible) patients were defined as those having at least one tumor lesion with Ga-68 Glu-urea-Lys(ahx)-hbed-CC uptake greater than normal liver and all tumor lesions larger than size criteria with Ga-68 Glu-urea-Lys(ahx)-hbed-CC uptake greater than liver [short axis size criteria: organs ≥ 1 cm, lymph nodes ≥ 2.5 cm, bones (soft tissue component) ≥ 1 cm]. Of the patients evaluated by the central reader, 869 (87%) were found to be PSMA-positive (eligible) and 126 (13%) were found to be PSMA-negative (ineligible) for lutetium Lu 177 vipivotide tetraxetan therapy.
A reader sub-study evaluated imaging interpretation agreement. Among the 869 patients found eligible and 126 patients found ineligible for therapy, 75 and 50, respectively, were selected at random (total of 125). Ga-68 Glu-urea-Lys(ahx)-hbed-CC PET and anatomical imaging from these 125 patients was interpreted by three additional central readers (rereaders) who were blinded to clinical information and other PET and bone imaging. Imaging from 20 of the 125 patients was also randomly selected to assess intra-reader reproducibility. The estimated inter-reader Fleiss κ was 0.60 (95% CI: 0.50, 0.70) across the three re-readers, while the estimated intra-reader Cohen κ was 0.78 (95% CI: 0.49, 0.99), 0.76 (95% CI: 0.46, 0.99) and 0.89 (95% CI: 0.67, 0.99) for each re-reader.
Given the observed levels of inter-reader agreement, patient's PSMA interpretations were further evaluated by eligibility and eligibility sub-criteria. Of the 125 patients with four readers (reader and re-readers), at least one reader found the patient to be PSMA-negative for lutetium Lu 177 vipivotide tetraxetan therapy in 59 patients. All four readers agreed in 20 of the 59 patients (34% reader unanimity). Based on the sub-criteria involving evaluation of any one tumor lesion and evaluation of all lesions larger than size criteria, at least one reader found the patient to be PSMA-negative in 21 patients (43% reader unanimity) and 45 patients (16% reader unanimity), respectively.
5.2 Pharmacokinetic Properties
Distribution.
Intravenously injected Ga-68 Glu-urea-Lys(ahx)-hbed-CC is rapidly cleared from the blood (biological half-life 6.5 minutes for 91% of activity; 4.4 hours for the remaining 9%). Ga-68 Glu-urea-Lys(ahx)-hbed-CC distributes to all PSMA-expressing organs such as spleen, adrenals, kidney, prostate, liver, and salivary glands; and accumulates primarily in the liver (15%), kidneys (7%), spleen (2%), and salivary glands (0.5%). There is no uptake in the cerebral cortex or in the heart, and usually thymus and lung uptakes are low.
Excretion.
A total of 14% of the injected dose is excreted in urine in the first two hours post-injection.
5.3 Preclinical Safety Data
Genotoxicity.
No animal studies on genotoxic potential have been conducted with Ga-68 Glu-urea-Lys(ahx)-hbed-CC.
Carcinogenicity.
No animal studies on carcinogenic potential have been conducted with Ga-68 Glu-urea-Lys(ahx)-hbed-CC.6 Pharmaceutical Particulars
6.1 List of Excipients
Illuccix configuration "A".
Vial 1 (Glu-urea-Lys(ahx)-hbed-CC Vial): contains 25 microgram Glu-urea-Lys(ahx)-hbed-CC, 10 microgram mannose and water for injections as a lyophilized powder in a sterile 10 mL vial with a blue flip-off cap.
Vial 2 (Buffer Vial, Configuration A): contains 150 mg sodium acetate, 0.077 mL hydrochloric acid and water for injections (2.5 mL volume) in a sterile 10 mL vial with a red flip off cap.
Vial 3 (Sterile Vacuumed Vial): an evacuated sterile vial with white flip off cap used to collect Ga-68 chloride from generators or cyclotron.
Illuccix configuration "B".
Vial 1 (Glu-urea-Lys(ahx)-hbed-CC Vial): contains 25 microgram Glu-urea-Lys(ahx)-hbed-CC, 10 microgram mannose and water for injections as a lyophilized powder in a sterile 10 mL vial with a blue flip-off cap.
Vial 2 (Buffer Vial, Configuration B): contains 150 mg sodium acetate, 0.15 mL hydrochloric acid and water for injections (6.4 mL volume) in a sterile 10 mL vial with a green flip off cap.
Vial 3 (Sterile Vacuumed Vial): an evacuated sterile vial with white flip off cap used to collect Ga-68 chloride from generator.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store Illuccix upright in the original packaging at 2-8°C. Do not freeze. After reconstitution and radiolabeling, keep Ga-68 Glu-urea-Lys(ahx)-hbed-CC upright with appropriate shielding to protect from radiation at room temperature. Use Ga-68 Glu-urea-Lys(ahx)-hbed-CC within 4 hours of preparation.
6.5 Nature and Contents of Container
Glu-urea-Lys(ahx)-hbed-CC vial.
The container closure system for the Glu-urea-Lys(ahx)-hbed-CC Sterile Vial is a clear, 10 mL Type I borosilicate glass vial, coated with 100% Silicon Dioxide, with a gray chlorobutyl rubber stopper, and aluminum seal with a flip off cap.
Vacuum vial.
The container closure system for the Sterile Vacuum Vial is a clear, 10 mL Type I borosilicate glass vial, coated with 100% Silicon Dioxide, with a gray chlorobutyl rubber stopper, and aluminum seal with a flip off cap.
Acetate buffer vials.
The container closure system for the sterile acetate buffer vials is a clear, 10 mL Type I borosilicate glass vial, coated with 100% Silicon Dioxide, with a gray rubber serum stopper, and flip off cap.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
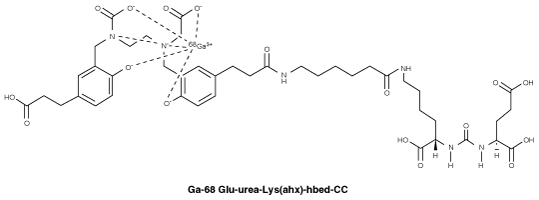
Chemical structure of Ga-68 Glu-urea-Lys(ahx)-hbed-CC.
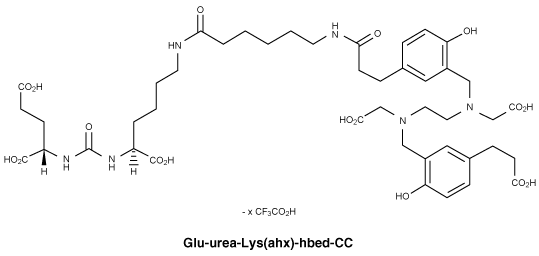
 Chemical structure of Glu-urea-Lys(ahx)-hbed-CC.
Chemical structure of Glu-urea-Lys(ahx)-hbed-CC.
CAS number.
Ga-68 Glu-urea-Lys(ahx)-hbed-CC - None.
Glu-urea-Lys(ahx)-hbed-CC - 1366302-52-4.7 Medicine Schedule (Poisons Standard)
Not scheduled.
Summary Table of Changes







 Perform the following:
Perform the following: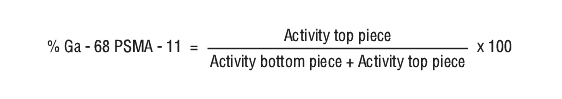 12. Scan the iTLC with a radiometric iTLC scanner.
12. Scan the iTLC with a radiometric iTLC scanner.



 VLR was also evaluated separately by region (prostate bed, pelvic lymph nodes, skeleton, and other distant sites [extra-pelvic lymph nodes and viscera]). Ga-68 Glu-urea-Lys(ahx)-hbed-CC localized prostate cancer lesions in all regions and showed the highest localization rates in bone (VLR range 50 to 68%) and the lowest localisation rate in the prostate bed (VLR range 31 to 44%).
VLR was also evaluated separately by region (prostate bed, pelvic lymph nodes, skeleton, and other distant sites [extra-pelvic lymph nodes and viscera]). Ga-68 Glu-urea-Lys(ahx)-hbed-CC localized prostate cancer lesions in all regions and showed the highest localization rates in bone (VLR range 50 to 68%) and the lowest localisation rate in the prostate bed (VLR range 31 to 44%). The percentage of patients who were scanned and had at least one false positive finding, regardless of any coexistent TP lesions, ranged from 54.9% to 73.6% in the 348 participants.
The percentage of patients who were scanned and had at least one false positive finding, regardless of any coexistent TP lesions, ranged from 54.9% to 73.6% in the 348 participants. Chemical structure of Glu-urea-Lys(ahx)-hbed-CC.
Chemical structure of Glu-urea-Lys(ahx)-hbed-CC.
