What is in this leaflet
This leaflet answers some common questions about INCRELEX. It does not contain all the available information. It does not take the place of talking to your child's doctor or pharmacist.
All medicines have risks and benefits. Your child's doctor has weighed the risks of them being given INCRELEX against the benefits they expect it will have.
If you have any concerns about your child being given this medicine, ask your child's doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What INCRELEX is used for
INCRELEX is a solution for injection that contains mecasermin which is a synthetic insulin-like growth factor-1 (IGF 1), which is similar to the IGF 1 made by your body.
It is used to treat children and adolescents from 2 to 18 years old who are very short for their age because their bodies do not make enough IGF 1. This condition is called primary IGF 1 deficiency.
Ask your doctor if you have any questions about why this medicine has been prescribed for your child. Your child's doctor may have prescribed it for another reason.
This medicine is available only with a doctor's prescription.
There is not enough information to recommend the use of this medicine for children under the age of 2 years.
Before giving INCRELEX
When your child must not be given it
Do not give INCRELEX if your child:
- currently has any tumour or growth, either cancerous or non-cancerous
- have had cancer in the past
- has any conditions which may increase the risk of cancer.
There is an increased risk of tumours and growths (both cancerous and non-cancerous) in children and adolescents treated with INCRELEX. If any new growth, skin lesion or symptom of cancer occurs during treatment or after treatment tell your child's doctor immediately.
Do not give INCRELEX to premature babies or newborns because it contains benzyl alcohol. Benzyl alcohol can cause toxic or allergic reactions in premature babies or newborns.
Do not give INCRELEX if your child has an allergy to:
- The active ingredient, mecasermin
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not give INCRELEX to your child if they have finished growing (the bone growth plates are closed). In this case INCRELEX cannot help them grow and should not be used. Your child's doctor will advise you of this.
Do not use this medicine if your child is pregnant. It may affect the developing baby if used during pregnancy.
Do not breast-feed while being given this medicine. It is not known if the active ingredient in INCRELEX (mecasermin) will pass into breast milk and it is not known if it will affect the baby.
Do not give this medicine to a child under the age of 2 years. Safety and effectiveness in children younger than 2 years have not been established.
Do not give this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether your child should start taking this medicine, talk to your child's doctor.
Before giving it
Tell your doctor if your child has allergies to any other medicines, foods, preservatives or dyes.
Tell your child's doctor if they have or have had any of the following medical conditions:
- if they have a curved spine (scoliosis).
- if they develop a limp or hip or knee pain
- if they have enlarged tonsils (tonsillar hypertrophy).
- if they have symptoms of increased pressure in the brain (intracranial hypertension), such as visual changes, headache, nausea and/or vomiting.
- if they have a localised reaction at the injection site or generalised allergic reaction with INCRELEX from a previous injection.
Tell your child's doctor if they are pregnant or plan to become pregnant or are breast-feeding. If they are pregnant or breast-feeding or think they may be pregnant or are planning to have a baby, ask your child's doctor for advice before using this medicine.
A negative pregnancy test is recommended for all women of child bearing potential prior to treatment with INCRELEX. It is also recommended that all women of childbearing potential use adequate contraception during treatment.
Mecasermin therapy should be discontinued if pregnancy occurs.
Mecasermin should not be administered to a breast-feeding mother.
If you have not told your child's doctor about any of the above, tell them before giving them INCRELEX.
Before starting treatment with INCRELEX, your child's doctor will check the function of your child's heart via ultrasound (echocardiogram).
Taking other medicines
Tell your child's doctor or pharmacist if they are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and INCRELEX may interfere with each other. These include:
- insulin or other anti-diabetes medicines. A dose adjustment may be needed for these medicines.
These medicines may be affected by INCRELEX or may affect how well it works. Your child may need different amounts of their medicines, or they may need to take different medicines.
Your child's doctor and pharmacist have more information on medicines to be careful with or avoid while being given this medicine.
How to give INCRELEX
Follow all directions given to you by your child's doctor or pharmacist carefully. Always give this medicine exactly as they have told you. Their instructions may differ from the information contained in this leaflet.
If you do not understand the instructions, ask your child's doctor or pharmacist for help.
How much to give
Your child's doctor or pharmacist will tell you how much to give.
The typical dose is 0.08 to 0.12 mg/kg of patient weight administered twice a day. See the 'Instructions for Use' at the end of this leaflet.
How to give it
Inject INCRELEX just under your child's skin.
Inject INCRELEX just below the skin in your child's upper arm, upper leg (thigh), stomach area (abdomen), or buttocks. Never inject it into a vein or muscle. Change the injection site for each injection.
Only give INCRELEX that is clear to slightly cloudy and colourless to slightly yellow.
Treatment with mecasermin is a long-term therapy. For further information ask your child's doctor.
INCRELEX is for use in one patient only.
When to give it
Give INCRELEX at about the same time each day. Giving it at the same time each day will have the best effect. It will also help you to remember when to give it.
Give INCRELEX shortly before or after a meal or snack (within 20 minutes) because it may cause low blood sugar levels.
If an INCRELEX dose cannot be given shortly before or just after a meal, that dose should not be given. The following dose should be the usual one and not doubled to make up for the missed dose
Do not inject the dose of INCRELEX if your child cannot eat for any reason.
Until your child’s doctor advises otherwise, vigorous physical activity should be avoided for 2-3 hours after the injection. If physical activity takes place after breakfast, a snack should be taken.
How long to give it
Continue giving the medicine for as long as your child's doctor tells you.
Treatment with mecasermin is a long-term therapy. For further information ask your child's doctor.
If you forget to give it
Do not make up the missed dose by giving two doses the next time. The next dose should be given as usual, with a meal or snack.
If you are not sure what to do, ask your child's doctor or pharmacist.
If you have trouble remembering to give the medicine, ask your pharmacist for some hints.
If you give too much (overdose)
Immediately telephone your child's doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or take your child to Accident and Emergency at the nearest hospital, if you think that your child or anyone else may have been given too much INCRELEX. Do this even if there are no signs of discomfort or poisoning. Your child may need urgent medical attention.
Symptoms of an overdose may include low blood sugar levels.
While being given INCRELEX
Things you must do
If you are about to be give your child any new medicine, remind your child's doctor and pharmacist that you are using INCRELEX.
Tell any other doctors, dentists, and pharmacists who treat your child that they are being given this medicine.
If they are going to have surgery, tell the surgeon or anaesthetist that your child is being given this medicine. It may affect other medicines used during surgery.
Hypoglycaemia
Hypoglycaemia is the medical name for a low blood sugar level. INCRELEX is related to insulin, a naturally occurring hormone in the body that decreases blood sugar levels so, INCRELEX can affect blood sugar levels. As a result, hypoglycaemia is a possible side effect of INCRELEX therapy.
Hypoglycaemia is one of the most common side effects and can happen when the correct dose is given.
If hypoglycaemia does occur, it is usually in the first month of treatment and the problem lessens as treatment goes on.
It is recommended that the blood glucose levels of your child should be regularly monitored. The blood glucose monitoring of your child is recommended at the start of treatment, when their dose is increased or decreased, when they are not taking enough food or if they are unwell and suffering from any other disease. Discuss the need for blood glucose monitoring for your child with their doctor.
It is important that you read all of this leaflet and any other information provided by your child's doctor about hypoglycaemia to minimise the risks of it occurring.
Make sure that you, your child, friends, family, your child's teachers and other carers can recognise the symptoms of hypoglycaemia and know how to treat them. Hypoglycaemia can occur suddenly.
Two of the most important risk factors for hypoglycaemia are:
- Not eating enough e.g. delaying or missing meals
- Physical exercise without eating sufficiently
Symptoms of hypoglycaemia may include:
- Dizziness
- Tiredness
- Restlessness
- Irritability
- Hunger
- Trouble concentrating
- Sweating
- Nausea
- Fast or irregular heartbeats
In younger children, the most important symptoms to look out for are your child becoming pale, stopping playing and remaining seated, or becoming sleepy.
If the hypoglycaemia is severe, your child may have reduced consciousness or have convulsions.
Symptoms of hypoglycaemia that occur during the night can be: restlessness, anxiety or excitability, nightmares, bed wetting, or tiredness in the morning
If your child experiences any of the symptoms of hypoglycaemia, you need to raise their blood glucose levels.
You can do this by giving your child one of the following:
- 5 to 7 jelly beans
- 3 teaspoons of sugar or honey
- half a can of non-diet soft drink
- 2 to 3 concentrated glucose tablets or glucose gel.
Unless your child is within 20 minutes of their next meal or snack, follow up with extra carbohydrates such as plain biscuits, fruit or milk.
It is important to keep snacks available, especially for younger children, and to teach them to eat as soon as they feel hungry. If hypoglycaemia occurs despite adequate food intake you should tell your child’s doctor, who may reduce the dose of INCRELEX.
In very rare cases, severe hypoglycaemia can occur. Should it occur, your child may not be conscious enough to swallow. In such cases, you should not try to give your child anything to eat or drink, but instead, you should give an injection of glucagon. Glucagon raises blood sugar levels when injected. Your doctor may show you how to use glucagon, in case you need to give it to your child
Tell your child's doctor about all of the occasions when hypoglycaemia may have occurred since your last visit. Keeping a record may help with this.
Intracranial Hypertension (Increased Pressure in the Brain)
High pressure in fluid around the brain (intracranial hypertension) can occur in some patients receiving INCRELEX treatment. Increased pressure in the brain may be caused by one of several factors other than treatment with INCRELEX. Therefore, if your child experiences the symptoms of increased pressure in the brain, which include severe headache, pain behind the eyes or visual changes such as blurred vision with nausea and vomiting, it is important to determine the reason for these symptoms.
It is important to tell your doctor if your child has an unexplained, severe, persistent headache or visual disturbance. By examining your child’s eyes, your doctor can confirm whether or not your child has increased pressure in their brain. Your doctor may then perform further tests to determine the cause of these symptoms and may adjust the dose of INCRELEX or stop treatment if necessary. It may be possible to restart treatment after the symptoms disappear.
Lipohypertrophy (increase in fat under the skin)
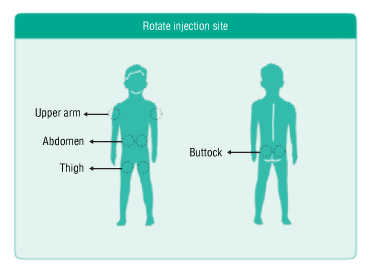
INCRELEX must be given using a different site at every injection, usually the stomach, thigh, buttocks or upper arm, to avoid an increase in fat tissue, also known as lipohypertrophy, around the area you inject. It is very important to be careful to rotate the injection sites, as lipohypertrophy will stop INCRELEX being absorbed into the body, and it will therefore not be effective. See the instructions for administration at the end of this leaflet for more information.
If your child becomes pregnant while taking this medicine, tell their doctor immediately.
If your child is about to have any blood tests, tell their doctor that they are being given this medicine. It may interfere with the results of some tests.
Keep all of your child's doctor's appointments so that their progress can be checked. Your child's doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
If your child has or develops scoliosis (sideways curvature of the spine caused by rapid growth), your child's doctor should monitor its progression.
Your child should have periodic examinations by their doctor for enlarged tonsils.
Your child's doctor will also continue to monitor their heart function (echocardiogram) during treatment and if treatment is stopped.
It is recommended that your child's doctor perform a skin check at before starting treatment and regularly during treatment to check for lesions growths or coloured patches on the skin such as birthmarks or moles.
Things you must not do
Do not give INCRELEX to treat any other complaints unless your child's doctor tells you to.
Do not give your child's medicine to anyone else, even if they have the same condition.
Do not stop giving their medicine or lower the dosage without checking with your child's doctor. A disruption or early ending of treatment with mecasermin may impair the success of the growth therapy. Please ask your child's doctor for advice before stopping the treatment.
Things to be careful of
Your child should be careful driving or operating machinery until you know how INCRELEX affects them. Mecasermin may cause low blood sugar levels (hypoglycaemia) that may affect their ability to drive and use machines because their ability to concentrate or react may be reduced. Other side effects of INCRELEX include dizziness and convulsions which could also affect their ability to drive or use machines.
They should avoid engaging in any high-risk activities (such as driving or operating machinery) within 2-3 hours after dosing, particularly at the start of INCRELEX treatment, until a dose has been found which does not cause side effects that make these activities risky.
Children should be careful when riding bicycles or climbing trees.
Side effects
Tell your child's doctor or pharmacist as soon as possible if they do not feel well while they are being given INCRELEX.
This medicine helps most people with primary IGF 1 deficiency, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. Your child may need medical attention if they get some of the side effects.
Do not be alarmed by the following lists of side effects. Your child may not experience any of them.
Ask your child's doctor or pharmacist to answer any questions you may have.
Tell your child's doctor or pharmacist if you notice any of the following and they worry you:
- signs of low blood sugar (hypoglycaemia) such as: dizziness, tiredness, restlessness, hunger, irritability, trouble concentrating, sweating, nausea, fast or irregular heartbeats
- injection site reactions such as: pain, irritation, bleeding, bruising, redness, hardening, swelling
- pain in the upper belly
- joint pain, pain in limbs
- headache
- dizziness, tremors
- breast enlargement
- skin thickening, moles, abnormal hair texture
- weight increase
The above list includes the more common side effects of the medicine. They are usually mild and short-lived.
Tell your child's doctor or pharmacist as soon as possible if you notice any of the following:
- increased blood sugar levels
- infection (especially in the middle ear)
- hearing loss, ear pain
- enlarged tonsils/adenoids, snoring, difficulty breathing or swallowing, sleep apnoea (a condition where breathing stops briefly during sleep), infections of the ear, excessive daytime sleepiness
If any of the following happen, tell your child's doctor immediately or take them to Accident and Emergency at your nearest hospital:
- seizures/fits, becoming unconscious. These are signs of severe low blood sugar levels (hypoglycaemia), if they are not responsive and cannot drink sugar-containing fluids, you should give an injection of glucagon. The doctor or nurse will instruct you how to give the injection. Glucagon raises the blood sugar when it is injected.
- visual changes, headache, nausea, vomiting
- a localised rash or other signs of a generalised allergic reaction (hives, trouble breathing, faintness or collapse and feeling generally unwell).
The above list includes very serious side effects. Your child may need urgent medical attention or hospitalisation.
Cancerous and non-cancerous tumours:
An increase in both cancerous and non-cancerous tumours has been reported in patients treated with INCRELEX. The risk of such tumours may be higher if INCRELEX is used for a condition other than what is listed above (see 'What INCRELEX is used for') or used at a higher than recommended dose. The proportion of patients affected is not known and cannot currently be estimated based on the available data.
Tell your child's doctor or pharmacist if you notice anything that is making them feel unwell.
Other side effects not listed above may also occur in some children.
Some of these side effects (for example, an enlarged thymus (a specialized organ of the immune system)), swelling at the back of the eye (due to increased pressure within the brain) or worsening of scoliosis (sideways curvature of the spine - caused by rapid growth)) can only be found when your child's doctor does tests from time to time to check your progress.
Talk to your doctor or pharmacist:
- if your child feels down, depressed, or if they feel they have no interest or take any pleasure in doing normal activities, whilst using this medicine. Depression is an uncommon side effect of the medicine.
After giving INCRELEX
Storage
Keep the vial in the outer carton until it is time to use it. This will protect from it light
Keep the medicine in the refrigerator where the temperature stays between 2°C - 8°C). Do not freeze. Protect from light.
After first use, the vial may be stored for up to 30 days at 2°C to 8°C.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your child's doctor tells you to stop giving this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over. Do not throw away any medicines via wastewater or household waste.
Product description
What it looks like
INCRELEX is a clear to slightly cloudy and colourless to slightly yellow solution for injection (injection) supplied in a clear glass vial closed with grey stopper and an aluminium seal with a plastic flip-off button. The vial contains 4 mL of solution.
Pack size of 1 vial.
Ingredients
INCRELEX contains 10 mg of mecasermin as the active ingredient. It also contains:
- benzyl alcohol,
- sodium chloride
- polysorbate 20
- glacial acetic acid
- sodium acetate trihydrate
- water for injections
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Sponsor
INCRELEX is sponsored in Australia by:
Ipsen Pty Ltd
Level 5
627 Chapel Street
South Yarra Victoria 3141
Australian Registration Number:
AUST R 308494
This leaflet was prepared in March 2025.
Full Product Information intended for healthcare professionals can be found at the following link:
www.ebs.tga.gov.au/ebs/picmi/picmirepository.nsf/pdf?OpenAgent&id=CP-2019-PI-02289-1&d=201911291016933
INCRELEX Injection Instructions for administration of the product
The following instructions explain how to inject INCRELEX.
PLEASE READ ALL OF THE INSTRUCTIONS CAREFULLY BEFORE STARTING THE INJECTION.
Your child's doctor or other healthcare professional will show you how to inject INCRELEX before first use.
INCRELEX should be administered using sterile disposable syringes and needles.
Preparing the dose:
- Wash your hands
Wash your hands before you get INCRELEX ready for the injection.
- Use a new needle and syringe
Use a new needle and syringe every time you give a dose.
- Preparing the syringe
Check the liquid to make sure it is clear to slightly cloudy and colourless to slightly yellow – do not use it if it is cloudy or if you see particles, and do not use it after the expiry date.
If you are using a new vial, remove the protective cap but do not remove the rubber stopper, which you should wipe with an alcohol swab to prevent contamination.
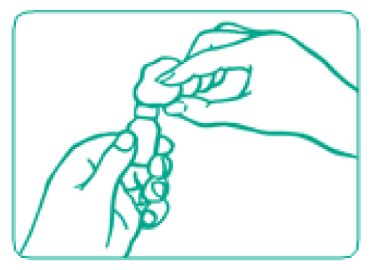
- Inject air into the vial
Before putting the needle into the vial, withdraw the plunger to draw an amount of air into the syringe that is equal to the Increlex dose.
Push the needle through the rubber top of the vial and then push the plunger to inject air into the vial.
- Withdrawing the dose
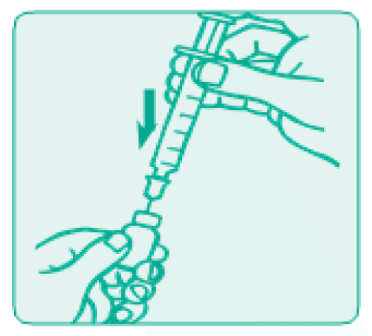
Leave the syringe in the vial and turn both upside down. Hold the syringe and vial firmly. Make sure that the tip of the needle is in the liquid and then pull the plunger to withdraw the correct dose into the syringe.
- Remove air bubbles and fill syringe
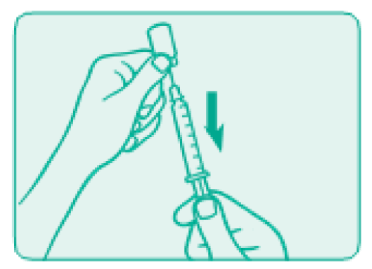
Before you take the needle out of the vial, check the syringe for air bubbles. If air bubbles are in the syringe, hold the vial and syringe with the needle straight up and tap the side of the syringe until the bubbles float to the top. Push the bubbles out by using the plunger, and then draw liquid back until you have the correct dose.
- Ready to inject
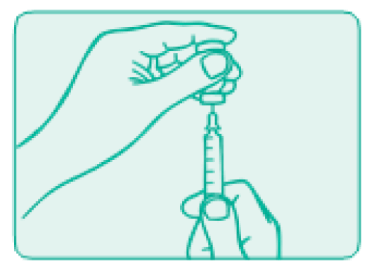
Remove the needle from the vial.
Do not let the needle touch anything. You are now ready to inject.
Injecting the dose:
Inject INCRELEX as instructed by your child’s doctor.
Do not give the INCRELEX injection if your child is unable to eat within 20 minutes before or after the injection.
- Decide on a site of injection
Decide on an injection area – upper arm, thigh, buttock, or abdomen. The injection site should be changed for each injection.
- To inject the dose
Use alcohol, or soap and water, to clean the skin where you are going to inject your child. The injection site should be dry before you inject.
Lightly pinch the skin. Insert the needle in the way that your child’s doctor has showed you. Release the skin.
Slowly push in the plunger of the syringe all the way, making sure that you have injected all the liquid.
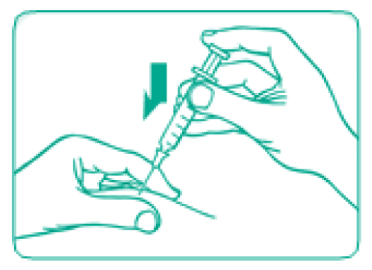
- Pull the needle straight out and gently press (do not rub) with gauze or cotton.
Pull the needle straight out and gently press on the spot where you injected your child with gauze or a cotton ball for a few seconds.
Do not rub the area.
Do not recap the syringe. The used needle and syringe should be placed in a sharps container which should be sealed and disposed of properly.
Published by MIMS May 2025