What is in this leaflet
This leaflet answers some common questions about Jardiance.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Jardiance against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
This leaflet was last updated on the date at the end of this leaflet. More recent information may be available. The latest Consumer Medicine Information is available from your pharmacist, doctor, or from www.medicines.org.au and may contain important information about the medicine and its use of which you should be aware.
Keep this leaflet with the medicine. You may need to read it again.
What Jardiance is used for
Jardiance is used to lower blood sugar levels in patients with type 2 diabetes mellitus.
It may be used when diet plus exercise do not provide adequate blood sugar level control either:
- alone as a single medicine, or
- in combination with certain other anti-diabetic medicines such as:
- metformin, or
- sulfonylurea medicines such as glimepiride and glibenclamide, or
- pioglitazone, or
- insulin, or
- dipeptidyl peptidase-4 inhibitor medicines such as linagliptin or sitagliptin.
If you have type 2 diabetes mellitus and cardiovascular disease, Jardiance can be used to reduce your risk of dying from your cardiovascular disease.
Type 2 diabetes mellitus is also called non-insulin-dependent diabetes mellitus or NIDDM. Type 2 diabetes develops if the body does not make enough insulin, or if the insulin that your body makes does not work as well as it should.
Insulin is a substance which helps to lower the level of sugar in your blood, especially after meals.
When the level of sugar builds up in your blood, this can cause damage to the body's cells and lead to serious problems with your heart, eyes, circulation or kidneys.
How Jardiance works
Jardiance contains the active ingredient, empagliflozin, which belongs to a group of medicines called SGLT2 (sodium-glucose co-transporter 2) inhibitors.
Jardiance works by blocking a protein in the kidneys called SGLT2. Jardiance removes blood sugar, which is too high because of your type 2 diabetes mellitus, via the urine. As a result, the levels of glucose in your blood are reduced.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
Jardiance is available only with a doctor's prescription.
Before you take Jardiance
When you must not take it
Do not take Jardiance if you have an allergy to:
- any medicine containing empagliflozin (the active ingredient in Jardiance)
- any of the other ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not take Jardiance if you have severe problems with your kidneys.
Do not take this medicine if you are pregnant. It may affect your developing baby if you take it during pregnancy.
Do not breastfeed if you are taking this medicine. It is not known if the active ingredient in Jardiance passes into human breast milk and there is a possibility that your baby may be affected.
Do not give this medicine to a child under the age of 18 years. Safety and effectiveness in children younger than 18 years has not been established.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- type 1 diabetes, a condition where your body does not produce insulin
- diabetic ketoacidosis, a condition in which substances called 'ketone bodies' accumulate in the blood and which can lead to diabetic pre-coma. Symptoms include: rapid weight loss, feeling sick or being sick, stomach pain, excessive thirst, fast and deep breathing, unusual sleepiness or tiredness, a sweet smell to your breath, a sweet or metallic taste in your mouth, or a different odour to your urine or sweat
- kidney problems
- galactosaemia, a rare hereditary condition of galactose intolerance
- frequent genital or urinary tract infections (infections of the bladder, kidney, or tubes that carry urine)
- illnesses that will make you dehydrated (e.g. diarrhoea or a severe infection)
- increased urine loss which may affect the fluid balance in your body.
Tell your doctor if you have heart problems, a history of low blood pressure or are 75 years of age or older.
Increased passing of urine due to the medicine may affect fluid balance in your body and increase your risk of dehydration.
Tell your doctor if you are 85 years of age or older. You should not start taking Jardiance if you are over 85 years of age.
If you have not told your doctor about any of the above, tell him/her before you start taking Jardiance.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Jardiance may interfere with each other. These include medicines used to treat high blood pressure known as water pills (diuretics).
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take Jardiance
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
The recommended dose is one 10 mg tablet once a day.
Your doctor may increase your dose to 25 mg once a day if your blood glucose levels remain high after starting treatment on 10 mg.
Your doctor will prescribe Jardiance alone, or in combination with another anti-diabetic medicine if that medicine alone is not sufficient to control your blood sugar level.
How to take it
Swallow the tablet whole with a full glass of water.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take this medicine before or after food.
How long to take it
Continue taking your medicine for as long as your doctor tells you.
This medicine helps to control your condition, but will not cure it. It is important to keep taking your medicine even if you feel well.
If you forget to take it
If it is less than 12 hours before your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone 13 11 26) for advice, or go to Emergency if you think that you or anyone else may have taken too much Jardiance. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking Jardiance
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Jardiance.
Tell any other doctors and pharmacists who treat you that you are taking this medicine.
If you become pregnant while taking this medicine, tell your doctor immediately.
If you are about to have any blood or urine tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Follow your doctor's and/or dietician's advice on diet, drinking alcohol and exercise. Diet and exercise can help your body use its blood sugar better. It is important to stay on the diet and exercise program recommended by your doctor while taking Jardiance.
Make sure you check your blood glucose regularly. This is the best way to tell if your diabetes is being controlled properly. Your doctor or diabetes educator will show you how and when to do this.
Check your feet regularly and see your doctor if you notice any problems. Follow any other advice regarding foot care given by your doctor.
Talk to your doctor about when to stop taking Jardiance and when to start taking it again if you are about to have surgery.
Things you must not do
Do not take Jardiance to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how Jardiance affects you.
When taken with other anti-diabetic medicines, such as sulfonylurea or insulin, your risk of getting low blood sugar is higher.
This may cause dizziness, light-headedness, tiredness, drowsiness in some people. Low blood glucose levels may also slow your reaction time and affect your ability to drive or operate machinery.
If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Be careful when doing any of the following things, which may increase the risk of your blood glucose becoming too low:
- drinking alcohol
- not eating enough
- doing unexpected or vigorous exercise.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Jardiance.
This medicine helps most people with type 2 diabetes mellitus, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- genital burning, redness, pain and discharge which may be signs of a genital yeast infection
- passing more urine than normal
- itching
- thirst.
The above list includes the more common side effects of your medicine. They are usually mild and short-lived.
Tell your doctor as soon as possible if you notice any of the symptoms of low blood sugar such as:
- sweating
- weakness
- hunger
- dizziness
- trembling
- headache
- flushing or paleness
- numbness
- a fast pounding heartbeat.
Low blood sugar may occur in patients who already take another medication to treat diabetes, such as a sulfonylurea or insulin. The dose of your sulfonylurea or insulin medicine may need to be reduced while taking Jardiance.
Tell your doctor as soon as possible if you notice any of the following:
- burning sensation when passing urine
- urine that appears cloudy
- pain in the pelvis, or mid-back pain
- straining or pain when passing urine
- unusual thirst
- light-headedness, or dizziness upon standing
- fainting or loss of consciousness.
The above list includes serious side effects that may require medical attention. Serious side effects are rare.
Tell your doctor immediately if you experience pain or tenderness, itching, swelling in the genital or back passage area, fever or are generally feeling unwell. These may be symptoms of a serious and life-threatening infection called Fournier's gangrene. Your doctor may tell you to stop taking Jardiance.
Tell your doctor immediately if you experience swelling of the penis that makes it difficult to pull back the skin around the tip of the penis (uncircumcised men).
Tell your doctor immediately or go to Emergency if you notice any of the following:
- swelling of the face, lips, mouth, tongue or throat which may cause difficulty in swallowing or breathing
- severe and sudden onset of itchy or raised skin rash, hives or nettle rash.
Tell your doctor immediately or go to Emergency if you notice any of the symptoms of diabetic ketoacidosis such as:
- rapid weight loss
- feeling sick or being sick
- stomach pain
- excessive thirst
- fast and deep breathing
- confusion
- unusual sleepiness or tiredness
- a sweet smell to your breath, a sweet or metallic taste in your mouth or a different odour to your urine or sweat.
In rare cases, empagliflozin, the active substance in Jardiance, can cause a serious side effect called diabetic ketoacidosis.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are very rare.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Some of these side effects can only be found when your doctor does tests from time to time to check your progress.
After using Jardiance
Storage
Keep your tablets in the pack until it is time to take them. If you take the tablets out of the pack they may not keep well.
Keep your tablets in a cool dry place where the temperature stays below 30°C.
Do not store Jardiance or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
Jardiance is the brand name of your medicine.
Jardiance is available in two strengths:
- Jardiance 10 mg tablets are pale yellow, round, biconvex and bevel-edged, marked with the BI company logo on one side and 'S10' on the other side.
- Jardiance 25 mg tablets are pale yellow, oval and biconvex, marked with the BI company logo on one side and 'S25' on the other side.
Jardiance tablets are available in PVC/Aluminium blister packs of 10 (sample) or 30 tablets.
Ingredients
Active ingredient:
- Jardiance 10 mg - 10 mg of empagliflozin.
- Jardiance 25 mg - 25 mg of empagliflozin.
Inactive ingredients:
- lactose monohydrate
- microcrystalline cellulose
- hyprolose
- croscarmellose sodium
- colloidal anhydrous silica
- magnesium stearate.
Tablet coating:
- hypromellose
- titanium dioxide
- purified talc
- macrogol 400
- iron oxide yellow.
This medicine does not contain sucrose, gluten, tartrazine or any other azo dyes.
Supplier
Jardiance tablets are supplied in Australia by:
Boehringer Ingelheim Pty Limited
ABN 52 000 452 308
Sydney NSW
www.boehringer-ingelheim.com.au
This Consumer Medicine Information was updated in January 2020.
® Jardiance is a registered trade mark of Boehringer Ingelheim.
© Boehringer Ingelheim Pty Limited 2020.
Australian Registration Numbers:
Jardiance 10 mg (AUST R 208829)
Jardiance 25 mg (AUST R 208827)
Published by MIMS March 2020





 During the placebo-controlled phase, the most frequent adverse event was hypoglycaemia (empagliflozin 10 mg and 25 mg, pooled: 23.1%, placebo: 9.4%).
During the placebo-controlled phase, the most frequent adverse event was hypoglycaemia (empagliflozin 10 mg and 25 mg, pooled: 23.1%, placebo: 9.4%).








 In a prespecified subgroup of patients with baseline HbA1c greater or equal than 8.5% the reduction from baseline in HbA1c with empagliflozin 25 mg/linagliptin 5 mg was -1.3% at 24 weeks (p < 0.0001 versus placebo [background linagliptin 5 mg]) and with empagliflozin 10 mg/linagliptin 5 mg -1.3% at 24 weeks (p < 0.0001 versus placebo [background linagliptin 5 mg]).
In a prespecified subgroup of patients with baseline HbA1c greater or equal than 8.5% the reduction from baseline in HbA1c with empagliflozin 25 mg/linagliptin 5 mg was -1.3% at 24 weeks (p < 0.0001 versus placebo [background linagliptin 5 mg]) and with empagliflozin 10 mg/linagliptin 5 mg -1.3% at 24 weeks (p < 0.0001 versus placebo [background linagliptin 5 mg]).
 In a prespecified pooled analysis of 4 placebo controlled studies, treatment with empagliflozin resulted in a reduction in SBP (-3.9 mmHg for empagliflozin 10 mg and -4.3 mmHg for empagliflozin 25 mg) compared with placebo (-0.5 mmHg), and in DBP (-1.8 mmHg for empagliflozin 10 mg and -2.0 mmHg for empagliflozin 25 mg) compared with placebo (-0.5 mmHg), at week 24, that were maintained up to week 52.
In a prespecified pooled analysis of 4 placebo controlled studies, treatment with empagliflozin resulted in a reduction in SBP (-3.9 mmHg for empagliflozin 10 mg and -4.3 mmHg for empagliflozin 25 mg) compared with placebo (-0.5 mmHg), and in DBP (-1.8 mmHg for empagliflozin 10 mg and -2.0 mmHg for empagliflozin 25 mg) compared with placebo (-0.5 mmHg), at week 24, that were maintained up to week 52.


 The cardiovascular benefits (CV death and hospitalisation for heart failure or CV death) of Jardiance observed were consistent across major demographic and disease subgroups.
The cardiovascular benefits (CV death and hospitalisation for heart failure or CV death) of Jardiance observed were consistent across major demographic and disease subgroups.
 Treatment with empagliflozin preserved eGFR and eGFR increased during the post-treatment 4 week follow-up. However, the placebo group showed a gradual decline in GFR during the course of the study with no further change during 4 week follow-up (see Figure 4).
Treatment with empagliflozin preserved eGFR and eGFR increased during the post-treatment 4 week follow-up. However, the placebo group showed a gradual decline in GFR during the course of the study with no further change during 4 week follow-up (see Figure 4).
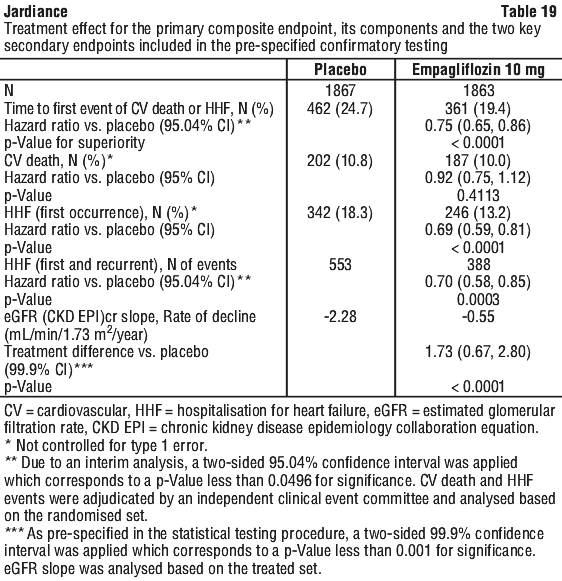

 The results of the primary composite endpoint were generally consistent with a hazard ratio (HR) below 1 across the pre-specified subgroups, including heart failure patients with and without type 2 diabetes mellitus (see Figure 7).
The results of the primary composite endpoint were generally consistent with a hazard ratio (HR) below 1 across the pre-specified subgroups, including heart failure patients with and without type 2 diabetes mellitus (see Figure 7).



 The results of the primary composite endpoint were consistent across each of the pre-specified subgroups categorised by e.g. LVEF, diabetes status or renal function down to an eGFR of 20 mL/min/1.73 m2 (see Figure 11).
The results of the primary composite endpoint were consistent across each of the pre-specified subgroups categorised by e.g. LVEF, diabetes status or renal function down to an eGFR of 20 mL/min/1.73 m2 (see Figure 11).



 The results of the primary composite endpoint were generally consistent across the pre-specified subgroups, including eGFR categories, underlying cause of renal disease, diabetes status, or background use of RAS inhibitors. Treatment benefits were more clearly evident in patients with higher levels of albuminuria.
The results of the primary composite endpoint were generally consistent across the pre-specified subgroups, including eGFR categories, underlying cause of renal disease, diabetes status, or background use of RAS inhibitors. Treatment benefits were more clearly evident in patients with higher levels of albuminuria.

