1 Name of Medicine
Leniolisib 70 mg film-coated tablets.
2 Qualitative and Quantitative Composition
Each film-coated tablet contains leniolisib phosphate equivalent to 70 mg leniolisib.
Excipient(s) with known effect.
Each tablet contains 241.16 mg of lactose monohydrate.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Film-coated tablet.
Yellow, oval shaped, biconvex, bevelled edge film coated tablet debossed with "70" on one side and "LNB" on the other side.
4.1 Therapeutic Indications
Joenja is indicated for the treatment of activated phosphoinositide 3-kinase delta syndrome (APDS) in adults and adolescents 12 years of age and older.
4.2 Dose and Method of Administration
Testing prior to treatment with Joenja.
Treatment with Joenja should only be commenced in patients in whom a pathogenic or likely pathogenic variant in an APDS-associated gene has been identified through a NATA accredited laboratory.
Verify pregnancy status in females of reproductive potential prior to initiating treatment with Joenja (see Section 4.4; Section 4.6).
Dosage.
The recommended dose in adult and paediatric patients who weigh more than 45 kg is 70 mg Joenja twice daily approximately 12 hours apart.
Method of administration.
Oral use.
The film coated tablet can be taken with or without food (see Section 5.2 Pharmacokinetic Properties).
Dose adjustment.
Combination with CYP3A4 inhibitors.
If use of strong CYP3A4 inhibitors is required, it is recommended that Joenja be discontinued 2 days before administration of the CYP3A4 inhibitor. Joenja may be restarted 7 days after CYP3A inhibitor discontinuation (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 5.2 Pharmacokinetic Properties).
Combination with CYP1A2 metabolised medicinal products with a narrow therapeutic index (NTI).
Interrupt Joenja dosing for at least 2 days prior to initiating therapy with an NTI CYP1A2 substrate. Restart Joenja after 7 days of an adequate washout of the NTI CYP1A2 substrate (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions; Section 5.2 Pharmacokinetic Properties).
Missed dose.
If a dose is missed by more than 6 hours, the patient should not take the missed dose but resume dosing at the next scheduled time.
Vomiting.
If vomiting occurs within 1 hour after taking Joenja, take another Joenja tablet as soon as possible. If vomiting occurs more than 1 hour after dosing, do not take an additional dose.4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in Section 6.1.
4.4 Special Warnings and Precautions for Use
Embryo-fetal toxicity.
Based on findings in animals, Joenja may cause fetal harm when administered to a pregnant woman. Administration of leniolisib to rats and rabbits during the period of organogenesis caused embryo-fetal toxicity including malformations at exposures that were 2 to 6 times higher than the recommended human dose of 70 mg twice a day based on AUC comparisons. Verify the pregnancy status of patients of reproductive potential prior to starting treatment. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use highly effective methods of contraception during treatment and for 1 week after the last dose (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy).
Immune-related adverse events.
Serious, sometimes fatal, immune-related adverse events such as severe infections, severe cutaneous adverse reactions (SCARs), pneumonitis, severe diarrhoea/colitis potentially resulting in intestinal perforation, neutropenia, and hepatotoxicity have occurred in patients receiving other PI3K delta inhibitors for the treatment of haematological or solid cancers. Joenja is not approved for treatment of haematological or solid cancers. While these serious events have not been associated with the use of Joenja in APDS patients, the safety data for Joenja is limited.
Vaccinations.
Live, attenuated vaccinations may be less effective if administered during Joenja treatment (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in hepatic impairment.
Joenja has not been studied in patients with hepatic impairment. Use of Joenja in patients with moderate to severe hepatic impairment is not recommended (see Section 5.2 Pharmacokinetic Properties).
Use in renal impairment.
Joenja has not been studied in patients with renal impairment.
Use in the elderly.
The safety and efficacy of leniolisib in patients over the age of 65 years have not been established.
Paediatric use.
The safety and efficacy of Joenja in children aged less than 12 years have not yet been established. No data are available.
Effects on laboratory tests.
Joenja has been associated with decreases in neutrophil counts. See Section 4.8 Adverse Effects (Undesirable Effects).
Use in combination with CYP3A4 inhibitors.
Concomitant use of Joenja with strong CYP3A inhibitors is expected to increase leniolisib exposure. (see Section 4.2 Dose and Method of Administration).
Use in combination with CYP1A2 metabolised medicinal products with a narrow therapeutic index (NTI).
Concomitant use with strong and moderate CYP3A4 inducers may result in reduced leniolisib exposure and thus reduced Joenja efficacy (see Section 4.2 Dose and Method of Administration).
Lactose content.
This medicinal product contains lactose monohydrate. Patients with rare hereditary problems of galactose intolerance, total lactase deficiency, or glucose-galactose malabsorption must not take this medicine.
Sodium content.
This medicinal product contains less than 1 mmol sodium (23 mg) per film-coated tablet.4.5 Interactions with Other Medicines and Other Forms of Interactions
Medicinal products affecting the pharmacokinetics of Joenja.
CYP3A4 inhibitors.
Leniolisib is cleared primarily through oxidative metabolism (primarily hydroxylation and dealkylation) by cytochrome P450 isoenzymes (predominantly CYP3A4, 95.4%). In a study of healthy adults, co-administration of leniolisib and itraconazole, a strong CYP3A inhibitor, resulted in a 2-fold increase in leniolisib exposure. Concomitant use of Joenja with strong CYP3A4 inhibitors (e.g. cobicistat, danoprevir, dasabuvir, elvitegravir, grapefruit juice, indinavir, itraconazole, ketoconazole, lopinavir, ombitasvir, paritaprevir, posaconazole, ritonavir, saquinavir, telithromycin, tipranavir, troleandomycin, voriconazole) should be avoided (see Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties).
CYP3A4 inducers.
No interaction studies have been conducted with Joenja and strong and moderate CYP3A4 inducers. Concomitant use may result in reduced leniolisib exposure and thus reduced Joenja efficacy. Therefore, concomitant use of Joenja with strong and moderate CYP3A4 inducers (e.g. avasimibe, carbamazepine, mitotane, phenobarbital, phenytoin, rifabutin, rifampin, St. John's Wort, bosentan, efavirenz, etravirine, genistein, modafinil, nafcillin, ritonavir, thioridazine, tipranavir) should be avoided.
Gastric acid reducing agents.
Leniolisib exhibits pH-dependent solubility, with lower solubility at higher pH-values. Avoid coadministration of Joenja with locally acting antacids. Antacids should be taken either 2 hours before or 2 hours after Joenja administration.
Medicinal products that have their exposure altered by Joenja.
OATP1B1, OATP1B3, and breast cancer resistance protein (BCRP) substrates.
In vitro, leniolisib is an inhibitor of the hepatic efflux transporters OATP1B1, OATP1B3, and BCRP. The effect of leniolisib as an inhibitor on OATP1B1, OATP1B3, and BCRP substrates has not been studied clinically. Although the likelihood of an in vivo inhibition is low when co-administered with Joenja, an increase in systemic exposure of medicinal products that are solely a substrate of these transporters (e.g. rosuvastatin, pitavastatin, letermovir) cannot be excluded. Co-administration with these medicinal products should be avoided.
OCT2, MATE1, and MATE2-K substrates.
In vitro, leniolisib is an inhibitor of the renal transporters OCT2, MATE1, and MATE2-K. Joenja may increase systemic exposure of medicines that are substrates of these transporters.
UGT1A1 substrates.
In vitro concentration-dependent inhibition of UGT1A1-mediated ethinylestradiol glucuronidation by leniolisib was observed. Joenja could increase plasma concentrations of drugs predominantly metabolised by UGT1A1.
CYP1A2 metabolized medicinal products with an NTI.
Time-dependent (irreversible) inhibition of CYP1A2 was observed in the presence of leniolisib in vitro. Clinically relevant interactions at the therapeutic dose of leniolisib (70 mg twice daily) cannot be excluded for inhibition of CYP1A2. Co-administration with CYP1A2 substrates (e.g. alosetron, caffeine, duloxetine, melatonin, ramelteon, tasimelteon, tizanidine) should be avoided (see Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties).
Hormonal contraceptives.
Administration of leniolisib with a single dose oral contraceptive containing ethinylestradiol and levonorgestrel increased ethinylestradiol exposure by approximately 30% with no effect on levonorgestrel exposure. The increase in ethinylestradiol exposure is unlikely to reduce the effectiveness of a combined oral contraceptive composed of ethinylestradiol and levonorgestrel.
Vaccines.
No vaccine interaction studies have been performed using Joenja. Vaccinations may be less effective if administered during Joenja treatment.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No human data on the effect of Joenja on fertility are available.
In repeat-dose toxicity studies in adult and juvenile rats, lower testes and epididymis weights and lower sperm counts were linked to decreases in the germinal epithelium and round spermatids and loss of spermatocytes at doses of ≥ 40 mg/kg/day (~2 times the exposure in humans at the recommended dose of 70 mg/day BID, based on AUC). There were no effects on mating or fertility in rats dosed with up to 90 mg/kg/day leniolisib from post-natal day 7 for 10 weeks. In the 26-week study, these effects were observed in the high dose group with similar incidence and severity with no progression by increasing the treatment duration. The described histological changes were limited to studies in rats and were not observed in studies in monkeys with up to 39 weeks of exposure. The clinical relevance of these findings is unknown.
(Category D)
There are no data from the use of Joenja in pregnant women.
Embryonic and fetal development studies in rats and rabbits have demonstrated that oral administration of leniolisib during organogenesis induced fetal-toxicity and teratogenicity. Decreased fetal bodyweight and an increased incidence of fetal malformations (eye bulge, microphthalmia, anophthalmia, and small orbital socket) and variations (dilated ureter and multiple skeletal variations related to ossification and alignment) were observed in rats at 120 mg/kg/day (6 times the exposure in humans at the recommended dose of 70 mg/day BID, based on AUC). In rabbits, agnathia, microstomia, aglossia, microphthalmia, and multiple skeletal malformations (small orbital socket and split palatine) and variations were observed at 100 mg/kg/day (2 times the exposure in humans at the recommended dose of 70 mg/day BID, based on AUC).
Joenja is not recommended during pregnancy and in women of childbearing potential not using highly effective methods of contraception. Women of childbearing potential should use highly effective methods of contraception during treatment with Joenja and for 1 week after the last dose. Verify pregnancy status in females of reproductive potential prior to initiating treatment with Joenja.
It is unknown whether leniolisib and its metabolites are excreted in human milk. Available pharmacokinetic/toxicological data in animals have shown excretion of leniolisib in milk. A risk to breastfed newborns/infants cannot be excluded. Breast-feeding should be discontinued during treatment with Joenja.
In a pre- and post-natal development study in rats, decreased lactation index and decreased pup bodyweight gain was observed during pre-weaning and low pup weights persisted into the postweaning period at 90 mg/kg/day (3 times the exposure in humans at the recommended dose of 70 mg/day BID, based on AUC).
In lactating rats, leniolisib was detected in all lactation study samples, with leniolisib concentrations increasing in a dose-dependent manner.4.7 Effects on Ability to Drive and Use Machines
Joenja has no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
The safety of Joenja reflects exposure based on 38 adult and paediatric patients 12 years of age and older with activated phosphoinositide 3-kinase delta (PI3Kδ) syndrome (APDS) from the placebo-controlled portion of Study 2201 (Part 2) and additional open-label clinical safety data (Study 2201E1).
In Study 2201 Part 2, the most common adverse events (> 10%) were headache, sinusitis, and atopic dermatitis. Adverse events occurring in ≥ 2 Joenja-treated patients and occurring more frequently than placebo are shown in Table 1.
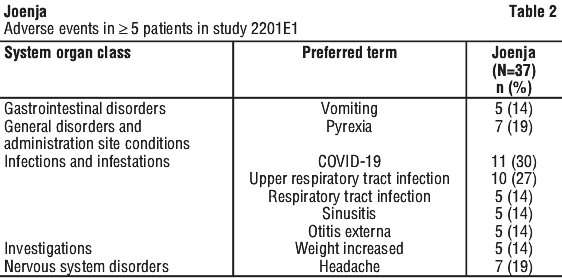
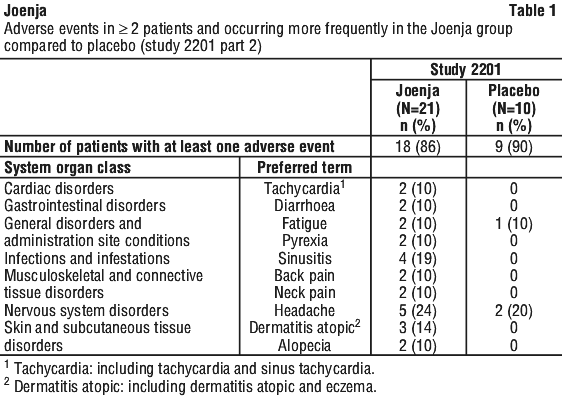 Table 2 summarizes safety data from Study 2201E1, an open-label, long term extension study. Thirty-seven patients received Joenja 70 mg orally twice daily for at least 60 weeks and 81% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
Table 2 summarizes safety data from Study 2201E1, an open-label, long term extension study. Thirty-seven patients received Joenja 70 mg orally twice daily for at least 60 weeks and 81% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
Laboratory abnormalities.
Neutrophil count decreased.
Seven (33%) patients receiving Joenja developed a transient absolute neutrophil count (ANC) between 500 and 1500 cells/microL. No patients developed an ANC < 500 cells/microL and there were no reports of infection associated with neutropenia.
Post-marketing reports.
Adverse events have been reported during post-approval use of leniolisib. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to leniolisib exposure.
Immune system disorders.
Hypersensitivity (anaphylaxis).
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
If overdose occurs, the patient must be monitored for evidence of toxicity. Treatment of overdose with Joenja consists of general supportive measures including monitoring of vital signs as well as observation of the clinical status of the patient.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: other immunostimulants, ATC code: L03AX22.
Mechanism of action.
Leniolisib selectively inhibits phosphoinositide 3-kinase delta (PI3Kδ) by blocking the active binding site of PI3Kδ. In cell-free isolated enzyme assays, leniolisib was selective for PI3K-delta over PI3Kalpha (28-fold), PI3K-beta (43-fold), and PI3K-gamma (257-fold), as well as the broader kinome. In cell-based assays, leniolisib reduced pAkt pathway activity and inhibited proliferation and activation of B and T cell subsets. Gain-of-function variants in the gene encoding p110δ catalytic subunit (resulting in APDS1) or loss-of-function variants in the p85α regulatory subunit (resulting in APDS2) both lead to hyperactive PI3Kδ signalling leading to increased production of phosphatidylinositol 3,4,5 trisphosphate (PIP3) and downstream phosphorylated protein kinase B (pAkt). Through inhibiting PI3Kδ thus decreasing production of PIP3, leniolisib reduces hyperactivity of the downstream Akt/mammalian target of rapamycin (mTOR) pathway, subsequently modifying dysregulation of B and T cell populations and disease progression.
Pharmacodynamics.
Ex vivo pharmacodynamics of leniolisib (proportion pAkt-positive B cells) were assessed intra-individually at 10, 30, and 70 mg twice daily for 4 weeks at each dose level in patients with APDS. Within the explored dose range, higher leniolisib plasma concentrations were generally associated with higher reduction of pAkt-positive B cells and higher doses were associated with a slightly higher peak reduction as well as more sustained reduction. Treatment with Joenja 70 mg twice a day at steady state is estimated to produce time-averaged reduction of pAkt-positive B cells by approximately 80%.
Clinical trials.
The efficacy of Joenja was assessed in Study 2201, a 12-week randomised, blinded, placebo-controlled study in 31 patients with confirmed APDS-associated pathogenic variant in either PIK3CD or PIK3R1. Patients were randomized 2:1 to receive either Joenja 70 mg or placebo twice a day. Patient demographics at baseline are presented in Table 3.
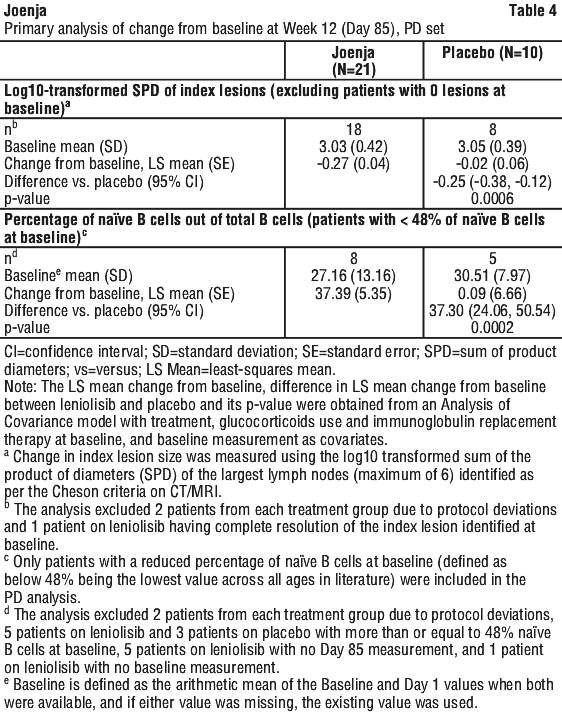
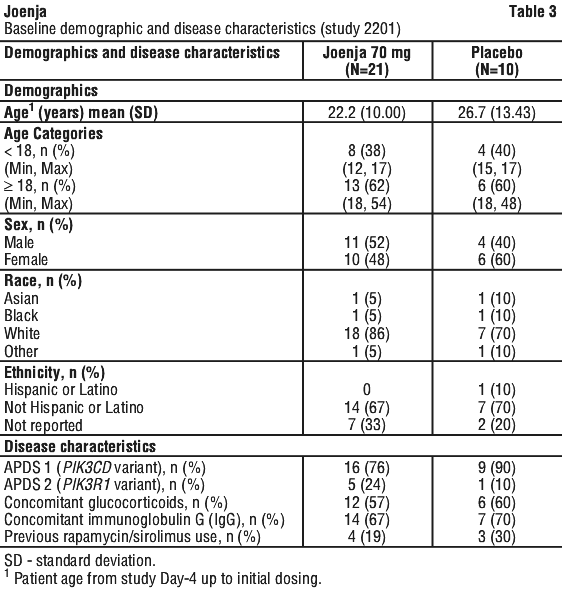 The co-primary efficacy endpoints were improvement in lymphoproliferation as measured by a change from baseline in lymphadenopathy measured by the log10-transformed sum of product diameters (SPD) of index lesions, and the normalisation of immunophenotype as measured by the percentage of naïve B cells out of total B cells. Table 4 presents the results for the co-primary endpoints.
The co-primary efficacy endpoints were improvement in lymphoproliferation as measured by a change from baseline in lymphadenopathy measured by the log10-transformed sum of product diameters (SPD) of index lesions, and the normalisation of immunophenotype as measured by the percentage of naïve B cells out of total B cells. Table 4 presents the results for the co-primary endpoints.
In a supportive analysis of naïve B cells in the pharmacodynamic analysis (PD) set (including patients with naïve B cells > 48% at baseline), the adjusted mean difference between the two treatment groups (95% CI) on change from baseline at Day 85 was 27.94 (15.02, 40.85); the 2-sided p-value observed for Joenja (N=13) vs placebo (N=8) was p=0.0003.
 Secondary endpoints were supportive of improvement in lymphoproliferation, with significant reduction in spleen size (see Table 5).
Secondary endpoints were supportive of improvement in lymphoproliferation, with significant reduction in spleen size (see Table 5).
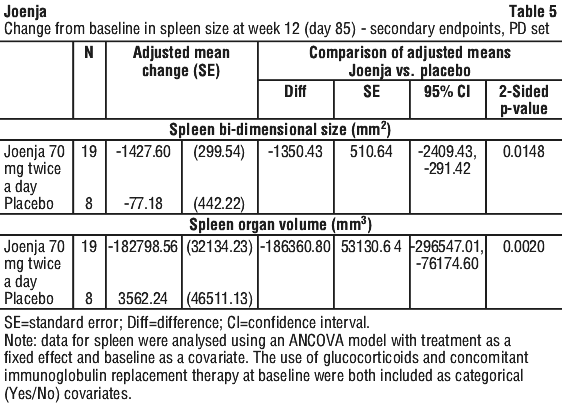 In the open-label long-term extension study, 37 patients continued treatment with Joenja 70 mg twice daily for 60 weeks or longer and 68% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
In the open-label long-term extension study, 37 patients continued treatment with Joenja 70 mg twice daily for 60 weeks or longer and 68% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
There was a continued reduction of lymphoproliferation within the first year, as measured by mean reductions from baseline of 52% for lymph node SPD, 38% for spleen volume, and 19% for spleen bidimensional size. Immunophenotype continued to improve within the first year, with an increase from baseline of 61% in naïve to total B cells, a reduction of 61% in transitional B cells, a reduction of 81% in senescent CD57+ CD4+ T cells, and a reduction of 38% in exhausted PD-1+ CD4+ T cells. Additionally, there were clinically meaningful reductions in infection rates (-0.262 infections per year of Joenja, p=0.0030), accompanied by post hoc observation of reduction in immunoglobulin replacement therapy (IRT), with 37% of patients reducing or completely stopping IRT after about 2 years.
5.2 Pharmacokinetic Properties
The pharmacokinetics of leniolisib have been studied in healthy subjects and adult and adolescent patients with APDS. Dose proportionality analysis of systemic drug exposure (area under the curve [AUC] and maximum plasma concentration [Cmax]) indicates that the pharmacokinetics of leniolisib are linear with respect to both dose (20 to 140 mg twice a day dosing and single doses of 10 to 400 mg/day) and time. During twice daily dosing approximately 12 hours apart, leniolisib accumulates approximately 1.4-fold in achieving steady state (range of 1.0 to 2.2), consistent with an effective half-life (t1/2) of approximately 7 hours. Steady state drug concentrations can be expected to be reached after approximately 2 to 3 days of Joenja treatment. The pharmacokinetics of leniolisib are similar between healthy participants and APDS patients.
Absorption.
In a placebo-controlled, ascending single and multiple dose study in healthy participants, leniolisib was rapidly absorbed in the fasted state, with median time to maximum plasma concentration (Tmax) at about 1 hour post dose. Tmax appeared independent of dose and was not altered after multiple oral doses.
Food effect.
Co-administration of a single 70 mg dose of Joenja with a high fat meal delayed the rate of absorption (Tmax) by 3 hours (0.64 h [fasting] to 3.51 h [fed]) and decreased Cmax on average by 41% but not the extent of absorption (AUC). The impact of the food effect on pAkt inhibition is expected to be minor and not leading to lower average or trough inhibition. Patients may therefore take Joenja irrespective of food.
Distribution.
The systemic decay in leniolisib plasma concentration over time is bi-exponential, indicating a distribution delay towards peripheral tissues. The apparent terminal elimination t1/2 is approximately 10 hours (estimate from steady-state drug washout). The median oral volume of distribution during the terminal phase ranged from 33 L to 57 L, indicating that leniolisib has a moderate-to-low volume of distribution. In human erythrocytes, the in vitro blood/plasma ratio is 0.643.
Leniolisib is highly bound (94.5%) to plasma proteins.
Metabolism.
Leniolisib was 60% metabolized by the liver, with CYP3A4 as the most predominant enzyme involved (95.4%) in the primary oxidative metabolism of leniolisib, with minor contribution from other enzymes (3.5% CYP3A5, 0.7% CYP1A2 and 0.4% CYP2D6). The strong activity of recombinant CYP1A1 suggests a possible involvement of this enzyme in the biotransformation of leniolisib in extra-hepatic tissues. Intestinal secretion by P-gp and extrahepatic CYP1A1 cannot be excluded as elimination routes.
Excretion.
The mass balance of an oral dose of 70 mg 14C-leniolisib was 92.5% (standard deviation: 2.3%) 168 hours post dose (morning of Day 8).
14C-leniolisib was excreted predominately via faeces (67.0%), while excretion via urine was approximately 25.5%. Approximately 70% of the 14C-leniolisib was recovered within 48 hours.
5.3 Preclinical Safety Data
Genotoxicity.
Leniolisib was not mutagenic in an in vitro bacterial reverse mutation (Ames) assay, clastogenic, or aneugenic in human cell micronucleus and chromosome aberration tests in vitro. Leniolisib was not genotoxic in an in vivo rat micronucleus test.
Carcinogenicity.
Long term carcinogenicity studies have not been conducted with leniolisib. In a 26-week carcinogenicity study in CByB6F1-Tg(HRAS)2Jic mice, there was no evidence of carcinogenicity at leniolisib doses up to 80 mg/kg/day.6 Pharmaceutical Particulars
6.1 List of Excipients
Tablet core.
Lactose monohydrate, microcrystalline cellulose, hypromellose, sodium starch glycollate type A, magnesium stearate, silicon dioxide.
Tablet film-coating.
Hypromellose, titanium dioxide, iron oxide yellow, iron oxide red, purified talc, macrogol 4000.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Do not refrigerate.
6.5 Nature and Contents of Container
High density polyethylene bottles with aluminum induction seal and child-resistant polypropylene screw cap.
Each pack contains 1 bottle with 60 tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
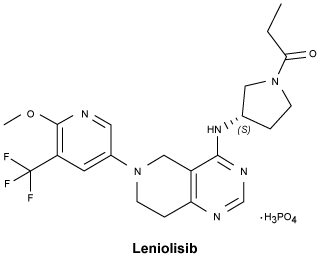
CAS number.
1354691-97-6.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription only medicine.
Summary Table of Changes


 Table 2 summarizes safety data from Study 2201E1, an open-label, long term extension study. Thirty-seven patients received Joenja 70 mg orally twice daily for at least 60 weeks and 81% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
Table 2 summarizes safety data from Study 2201E1, an open-label, long term extension study. Thirty-seven patients received Joenja 70 mg orally twice daily for at least 60 weeks and 81% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
 The co-primary efficacy endpoints were improvement in lymphoproliferation as measured by a change from baseline in lymphadenopathy measured by the log10-transformed sum of product diameters (SPD) of index lesions, and the normalisation of immunophenotype as measured by the percentage of naïve B cells out of total B cells. Table 4 presents the results for the co-primary endpoints.
The co-primary efficacy endpoints were improvement in lymphoproliferation as measured by a change from baseline in lymphadenopathy measured by the log10-transformed sum of product diameters (SPD) of index lesions, and the normalisation of immunophenotype as measured by the percentage of naïve B cells out of total B cells. Table 4 presents the results for the co-primary endpoints. Secondary endpoints were supportive of improvement in lymphoproliferation, with significant reduction in spleen size (see Table 5).
Secondary endpoints were supportive of improvement in lymphoproliferation, with significant reduction in spleen size (see Table 5). In the open-label long-term extension study, 37 patients continued treatment with Joenja 70 mg twice daily for 60 weeks or longer and 68% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
In the open-label long-term extension study, 37 patients continued treatment with Joenja 70 mg twice daily for 60 weeks or longer and 68% were exposed for 96 weeks or longer. Median duration of Joenja treatment was approximately 3 years, and 5 patients had more than 5 years of Joenja exposure.
