What is in this leaflet
This leaflet answers some common questions about JORVEZA tablets. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risk of you taking this medicine against the benefits it is expected to have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What JORVEZA is used for
JORVEZA tablets contain the active ingredient, budesonide. Budesonide belongs to a group of medications called corticosteroids. JORVEZA tablets are used to treat eosinophilic oesophagitis (EoE) which is an inflammatory condition of the gullet (food pipe) that causes problems with swallowing food.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
This medicine is not expected to affect your ability to drive a car or operate machinery.
JORVEZA is only available on a doctor’s prescription.
There is not enough information to recommend the use of this medicine for children or adolescents.
Before you take JORVEZA
When you must not take it
Do not take this medicine if you have an allergy to:
- any medicine containing budesonide
- any ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not take JORVEZA if you have uncontrolled infections or active tuberculosis.
Before you start to take it
Tell your doctor if you have any allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you are pregnant or plan to become pregnant or are breastfeeding or wish to breastfeed. Your doctor will discuss the risks and benefits of taking JORVEZA if you are pregnant or breastfeeding.
Tell your doctor if you have or have had any medical conditions, especially the following:
- liver disease or kidney disease
- lung disease (e.g. tuberculosis)
- high blood pressure
- diabetes, when the level of sugar in the blood is too high
- disease which causes bones to become less dense, gradually making them weaker, more brittle and likely to break (osteoporosis)
- ulcer in stomach or duodenum
- glaucoma (high pressure in the eye)
- cataracts (cloudy area in your eye)
- family history of diabetes or glaucoma
- any infection
- recent or planned surgery
- other stresses
- any other disease where use of corticosteroids may have unwanted effects.
If you have not told your doctor about any of the above, tell him/her before you start taking JORVEZA tablets.
Tell your doctor if you are on other corticosteroid treatments.
Keep away from people who have chicken pox, measles or herpes zoster (shingles), if you have never had them.
Contact your doctor if you have been exposed to chicken pox, measles and shingles infections. These illnesses may become more severe when you take JORVEZA. JORVEZA treatment may need to be discontinued if you have one of these infections.
If you develop swelling of your face, particularly around your mouth (lips, tongue or throat) and/or difficulties to breathe or swallow, stop taking Jorveza and seek urgent medical attention. These may be signs of an allergic reaction, which may also include rash and itching.
Tell your doctor if you have not yet had measles.
If you know that you need to be vaccinated please speak to your doctor first.
Tell your doctor if you get fungal infections in the mouth, throat (symptoms can be white spots) or in the gullet or if you think you have any infection during treatment with JORVEZA. The symptoms of some infections can be unusual or less pronounced when taking JORVEZA.
If you know that you are due to have an operation please tell your doctor that you are taking JORVEZA.
If you have been treated with a stronger corticosteroid preparation before starting treatment with JORVEZA, your symptoms may reappear when the medicine is changed.
If this happens, contact your doctor.
Contact your doctor if you experience blurred vision or other visual disturbances.
JORVEZA 0.5 mg and 1 mg contains 26 mg of sodium(the main component of cooking/table salt) in each tablet. Therefore, the maximum daily dose of sodium from either strength when used as advised (2 x 1 mg or 2 x 0.5 mg tablets) is 52 mg per day. This is equivalent to approximately 2.6% of the recommended maximum daily dietary intake of sodium for an adult.
JORVEZA could affect the results of adrenal function tests (ACTH stimulation test) ordered by your doctor or in hospital. Tell your doctors that you are taking JORVEZA before you have any tests.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and JORVEZA may interfere with each other. These include:
- cardiac glycosides such as digoxin (medicines used to treat heart conditions)
- diuretics (medicines used to treat excess fluid in your body)
- ketoconazole and itraconazole (medicines used to treat fungal infections)
- antibiotics such as clarithromycin and erythromycin (medicines used to treat infections)
- ritonavir and cobicistat (medicines used to treat HIV infections)
- oestrogens (contraceptive pill)
These medicines may be affected by JORVEZA or may affect how well it works. You may need to take different amounts of your other medicine(s), or you may need to take different medicines.
Avoid drinking grapefruit juice while you are taking JORVEZA as this can alter its effects.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking JORVEZA.
How to take JORVEZA
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
How much to take
Adults: The recommended dose for treatment of acute episodes is two 1 mg tablets per day. Take one 1 mg tablet in the morning and one in the evening.
The recommended dose for prevention of further episodes is two 0.5 mg tablets (1 mg budesonide) per day or two 1 mg tablets (2 mg budesonide) per day, depending on your response to the treatment. Take one tablet in the morning and one tablet in the evening. Your doctor will decide which dose is best for you.
Speak to your doctor if you want to interrupt or end your JORVEZA treatment early. It is important that you do not stop taking JORVEZA without talking to your doctor.
Keep taking JORVEZA until your doctor tells you to stop, even if you feel better.
How to take it
Take the JORVEZA tablet immediately once removed from the blister package.
Take JORVEZA after a meal.
Place the tablet on the tip of your tongue and close your mouth. Press it gently against the roof of your mouth with your tongue until it has disintegrated completely (this takes between 2 and 5 minutes on average but may take up to 10 minutes or longer in some patients). Saliva will be produced while the JORVEZA tablet is disintegrating. Swallow the disintegrated material slowly, as the tablet breaks up.
Do NOT take any liquid or food with the tablet.
Do not chew or swallow the tablet if not disintegrated.
Do not eat, drink, brush your teeth or rinse your mouth for at least 30 minutes after you have taken the tablet. Do not use any oral solutions, sprays or chewable tablets for at least 30 minutes before or after administration of the tablet. This will ensure that your medicine works properly.
When to take JORVEZA
Take your medicine twice a day (once in the morning and once at night) after a meal.
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
How long to take JORVEZA
Initially, your treatment should last about 6 to 12 weeks.
After this initial treatment, your doctor will decide for how long and which dose (1 mg/day or 2 mg/day) you should continue to take, depending on your condition and your response to the treatment.
Continue taking JORVEZA for as long as your doctor tells you.
If you forget to take it
Take your dose as soon as you remember, and continue to take it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed. This may increase the chance of getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice or go to Accident and Emergency at your nearest hospital, if you think you or anyone else has taken too much JORVEZA. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking JORVEZA
Things you must do
If you are about to start on any new medicine, remind your doctor and pharmacist that you are taking JORVEZA.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking JORVEZA.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking JORVEZA. It may affect other medicines used during surgery.
If you become pregnant while taking JORVEZA, tell your doctor immediately.
Things you must not do
Do not stop taking JORVEZA or change the dosage without checking with your doctor.
Do not take JORVEZA to treat any other complaint unless your doctor tells you to.
Do not give your JORVEZA to anyone else, even if they have the same condition as you.
Things to be careful of
Avoid drinking grapefruit juice. Grapefruit juice contains some components that influence the metabolism (break-down) of JORVEZA. This can worsen its side effects.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking JORVEZA.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following that are troublesome or ongoing:
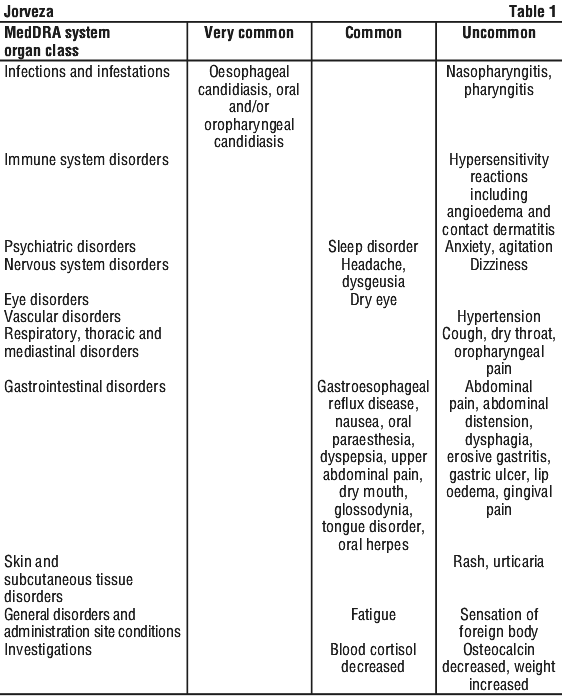
Very common side effects include:
- fungal infections in the gullet (which can cause pain or discomfort when swallowing)
- fungal infections in the mouth and throat (symptoms can be white spots)
Common side effects include:
- headache
- feeling sick (nausea)
- heartburn
- indigestion
- tingling or numbness in your mouth, dry mouth
- taste disorder, burning tongue
- upper abdominal (belly) pain
- tiredness
- decreased amount of the hormone cortisol in your blood.
- dry eyes
- difficulty in sleeping
- problems with tongue
- cold sore (oral herpes).
Uncommon side effects include:
- high blood pressure
- anxiety, agitation
- dizziness
- cough, dry throat, sore throat, common cold
- abdominal (belly) pain, abdominal distension (bloating)
- difficulty swallowing
- inflammation of the stomach, ulcers in the stomach
- swelling of the lips
- rash, itching rash
- sensation of foreign body
- pain in the mouth or throat
- painful gums
- decreased level of osteocalcin, weight gain.
When JORVEZA is taken, side effects may occur which are typical of the more active corticosteroid preparations (Cushing-like properties). The side effects listed below depend on the dose, the period of JORVEZA treatment, whether there has been prior or accompanying treatment with other corticosteroid preparations and on the individual sensitivity.
- Cushing Syndrome: roundness of the face, weight gain, acne, increased risk of high blood sugar, fluid retention (e.g. swollen legs)
- slowed growth in children
- increased risk of infection
- irregular periods in women, male hair growth patterns in women
- impotence
- mood changes such as depression, irritation or euphoria
- restlessness with increased physical activity and aggression
- blurred vision (e.g. glaucoma and cataract)
- increased risk of blood clotting, disease of the blood vessels (associated with stopping corticosteroid use after long term therapy)
- ulcers in the small intestine, pancreatitis and constipation
- muscle and joint pain, muscle weakness, muscle twitching
- bone weakness (osteoporosis), bone damage due to poor circulation of blood (osteonecrosis)
- rash from hypersensitivity reactions (allergic exanthema), formation of red stripes (striations) and bleeding in the skin, delayed wound healing, bruising
- general feeling of being ill
- increased brain pressure with possible additional swelling of the optic disk in adolescents.
If any of the following happen, stop taking JORVEZA and tell your doctor immediately, or go to Accident and Emergency at your nearest hospital:
- swelling of the face, particularly of the eyelids, lips, tongue or throat (angioedema)
- shortness of breath
- itchy rash or hives.
These are the symptoms of an allergic reaction.
These are serious side effects. You may need urgent medical attention or hospitalisation. Serious side effects are rare.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After taking JORVEZA
Storage
Keep JORVEZA in the original container.
JORVEZA tablets should not be removed from the individual blisters until immediately before dosing.
If you take it out of its container it may not keep well.
Keep JORVEZA in a cool dry place where the temperature stays below 25°C. Protect from light and moisture.
Do not store JORVEZA or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines, including JORVEZA.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking JORVEZA or the expiry date has passed, ask your pharmacist or doctor what to do with any medicine that is left over.
Product description
What it looks like
JORVEZA 1 mg orally disintegrating tablets: white or almost white, round tablets with a smooth surface and facet.
They are available in blisters of 20, 30, 60 and 90 tablets*.
* Not all pack sizes may be marketed
JORVEZA 0.5 mg orally disintegrating tablets: white, round tablets debossed with “0.5” on one side and smooth on the other side.
They are available in blisters of 20, 60, 100 and 200 tablets*.
* Not all pack sizes may be marketed
Ingredients
Active ingredients:
JORVEZA 1 mg orally disintegrating tablets – 1 mg budesonide
JORVEZA 0.5 mg orally disintegrating tablets – 0.5 mg budesonide
Inactive ingredients:
- magnesium stearate
- sodium acid citrate
- sodium dihydrogen citrate
- docusate sodium
- sucralose
- sodium bicarbonate
- mannitol
- macrogol 6000
- povidone
Supplier
JORVEZA® is supplied in Australia by:
Dr Falk Pharma Australia Pty Ltd
815 Pacific Highway,
Chatswood, NSW 2067
Phone: 1800 DRFALK (373 255)
JORVEZA® is supplied in New Zealand by:
Dr Falk Pharma New Zealand Ltd
29 Northcroft Street,
Takapuna, Auckland 0622
New Zealand
Phone: 0800 44 88 69
This leaflet was prepared in April 2024.
Australian Register Number
JORVEZA 1 mg orally disintegrating tablets:
AUST R 322645
JORVEZA 0.5 mg orally disintegrating tablets
AUST R 350996
Published by MIMS June 2024



 The following known adverse reactions of the therapeutic class (glucocorticoids), especially for those which are systemically acting, however, may also occur with the topically acting Jorveza (frequency = not known). See Table 2.
The following known adverse reactions of the therapeutic class (glucocorticoids), especially for those which are systemically acting, however, may also occur with the topically acting Jorveza (frequency = not known). See Table 2.
 Chemical name: 16α,17α-butylidene dioxy-11β,21-dihydroxy-1,4-pregnadiene-3,20-dione.
Chemical name: 16α,17α-butylidene dioxy-11β,21-dihydroxy-1,4-pregnadiene-3,20-dione.