1 Name of Medicine
Rabies immunoglobulin.
2 Qualitative and Quantitative Composition
KamRAB is supplied in single-use vials containing 2 mL or 10 mL. One mL of solution contains 150 IU rabies immunoglobulin (from human plasma), corresponding with 300 IU in 2 mL or 1,500 IU in 10 mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection (sterile).
KamRAB solution is a clear to slightly opalescent, colourless to pale yellow. The solution may contain some protein particles.
4.1 Therapeutic Indications
KamRAB is rabies immunoglobulin indicated for passive, transient post-exposure prophylaxis (PEP) of rabies infection, when given immediately after contact with a rabid or possibly rabid animal. KamRAB should be administered concurrently with a full course of rabies vaccine.
Do not administer additional (repeat) doses of KamRAB once vaccine treatment has been initiated, since this may interfere with the immune response to the rabies vaccine.
Do not administer KamRAB to patients with a history of a complete pre-exposure or post-exposure vaccination regimen and confirmed adequate rabies antibody titre.
4.2 Dose and Method of Administration
For wound infiltration and intramuscular use.
Product is for single use in one patient only. Discard any residue.
Local treatment of wounds prior to KamRAB administration.
The World Health Organisation (WHO) and the Australian Communicable Diseases Network Australia (CDNA) have outlined recommendations for passive and active immunisation after exposure to an animal suspected of having rabies. Immediate and thorough cleansing of all bite wounds and scratches with soap and water is an important component of post-exposure prophylaxis (PEP). A viricidal antiseptic solution (e.g. povidone-iodine solution or alcohol) should be used to irrigate the wounds. Tetanus prophylaxis and measures to control bacterial infection should be given if medically indicated.
Dosage.
Post-exposure prophylaxis consists of a single dose of KamRAB and a full course of rabies vaccine. The recommended dose of KamRAB is 20 IU/kg body weight, given at the time of the first vaccine dose. KamRAB and the first dose of rabies vaccine should be given as soon as possible after exposure, and regardless of the time interval between exposure and initiation of post-exposure prophylaxis, as delays are potentially lethal. However, should a delay occur, KamRAB should be administered at any time up to and including seven days after the first dose of vaccine. The rabies vaccine should be given according to the manufacturer's instructions.
No more than the recommended dose of KamRAB should be given because KamRAB partially suppresses active antibody production following vaccination. For the same reason, additional doses of KamRAB should not be given, even if the antibody response to vaccination is delayed.
Method of administration.
When the bite site is known and infiltration at the bite site is feasible, infiltrate as much of the dose as possible into and around any detectable bite wounds. Administer the remaining KamRAB intramuscularly into anatomical site(s) distant from the site of the rabies vaccine.
When the bite site is unknown or indeterminate (undetectable) or if infiltration is difficult at the bite site (e.g. lips, fingers, knee), administer the full KamRAB dose by the intramuscular route at a site distant from the site of rabies vaccination.
If a large intramuscular volume is required (> 2 mL for children or > 5 mL for adults), administer the total volume in divided doses at different sites.
If intramuscular administration is contraindicated (e.g. in patients with uncorrectable bleeding disorders), administer KamRAB subcutaneously. However, note that there are no clinical efficacy data to support administration of KamRAB by the subcutaneous route.
Do not mix with the rabies vaccine or administer in the same syringe with the rabies vaccine. Do not administer into the same anatomical site(s) as rabies vaccine.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. If either of these conditions exists, do not use KamRAB; discard the vial.4.3 Contraindications
Because of the life-threatening risk due to rabies, there are no contraindications to the administration of rabies immunoglobulin.
4.4 Special Warnings and Precautions for Use
Transmissible infectious agents.
Standard measures to prevent infections resulting from the use of medicinal products prepared from human blood or plasma include selection of donors, screening of individual donations and plasma pools for specific markers of infection and the inclusion of effective manufacturing steps for the inactivation/ removal of viruses. Despite this, when medicinal products prepared from human blood or plasma are administered, the possibility of transmitting infective agents cannot be totally excluded. This also applies to unknown or emerging viruses and other pathogens.
The measures taken are considered effective for enveloped viruses such as human immunodeficiency virus (HIV), hepatitis B virus (HBV) and hepatitis C virus (HCV), and for the nonenveloped hepatitis A and parvovirus B19 viruses.
There is reassuring clinical experience regarding the lack of hepatitis A or parvovirus B19 transmission with immunoglobulins and it is also assumed that the antibody content makes an important contribution to the viral safety.
It is strongly recommended that every time that KamRAB is administered to a patient, the name and batch number of the product are recorded in order to maintain a link between the patient and the batch of the product.
Previous rabies vaccination.
Patients who can document previous complete rabies pre-exposure prophylaxis or complete postexposure prophylaxis should only receive a booster rabies vaccine without KamRAB, because KamRAB may interfere with the anamnestic response to the vaccine.
Live attenuated virus vaccines.
KamRAB administration may interfere with the development of an immune response to live attenuated virus vaccines.
Avoid immunization with measles vaccine within 4 months after KamRAB administration.
Avoid immunization with other live attenuated virus vaccines within 3 months after KamRAB administration.
Anaphylactic shock.
KamRAB should not be injected into a blood vessel because of the risk of severe allergic or hypersensitivity reactions, including anaphylactic shock. KamRAB can induce a fall in blood pressure associated with an anaphylactic reaction, even in patients who tolerated previous treatment with human immunoglobulin.
Discontinue KamRAB injection immediately if there is an allergic or anaphylactic type reaction. In case of shock, implement standard medical treatment. Epinephrine should be available for treatment of acute anaphylactic symptoms.
Hypersensitivity.
Patients with a history of prior systemic allergic reactions following administration of human immunoglobulin preparations should be monitored for hypersensitivity.
KamRAB contains a small quantity of IgA. Individuals who are deficient in IgA have potential for developing IgA antibodies and may have anaphylactic reactions after administration of blood components containing IgA. The healthcare provider should assess the risks of this reaction against the benefits of administering KamRAB.
Use in the elderly.
The safety and effectiveness of KamRAB in the elderly population have not been established.
Clinical studies of KamRAB did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. Clinical experience with human rabies immune globulin products has not identified differences in effectiveness between elderly and younger patients.
Paediatric use.
Safety and effectiveness have been established in children. In a paediatric study of 30 patients ranging in age from 0.5 to 14.9 years, KamRAB presented no serious adverse reactions through day 84. Of the 30 patients, 28 (93.3%) achieved a Day 14 rabies virus neutralising activity (RVNA) titre ≥ 0.5 IU/mL, the WHO recommended level. None of the patients who were followed until the end of the study (28/30 patients) developed rabies infection through day 84. See Section 5.1 Pharmacodynamic Properties, Clinical trials.
Adverse events that occurred in ≥ 3.3% of patients within the first 14 days of KamRAB and the first rabies vaccination administration are listed in Adverse Effects (Undesirable Effects).
The clinical trial conducted in the paediatric population is described in Pharmacodynamic Properties, Clinical trials. Additional evidence to support the use of KamRAB in children comes from Real World Evidence. Based on claims data, 172 children from the United States of America (≤ 17 years) were treated with KamRAB between 2018-2020. Based on Center for Disease Control (CDC) data, no children in the United States of America treated with postexposure prophylaxis have been reported to have had rabies between 2018 and April 2021.
Effects on laboratory tests.
After injection of immunoglobulin, a transitory rise of the various passively transferred antibodies in the patient's blood may result in misleading positive results of serologic tests after KamRAB administration.
Passive transmission of antibodies to erythrocyte antigens, e.g. A, B, and D, may interfere with serologic tests for red cell antibodies such as the antiglobulin test (Coombs' test).4.5 Interactions with Other Medicines and Other Forms of Interactions
Rabies virus vaccines.
Do not administer additional (repeat) doses of KamRAB once vaccination has been initiated, since additional doses of KamRAB may interfere with the immune response to the vaccine.
Do not administer in the same anatomical site(s) or in the same syringe as rabies vaccine.
Live attenuated virus vaccines.
KamRAB contains other antibodies that may interfere with the response to live vaccines such as measles, mumps, polio or rubella. Avoid immunization with live virus vaccines within 3 months after KamRAB administration, or in the case of measles vaccine, within 4 months after KamRAB administration.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available. It is not known whether KamRAB can affect reproductive capacity.
(Category B2)
KamRAB has not been studied in pregnant women. Therefore, the risk of major birth defects and miscarriage in pregnant women who are exposed to KamRAB is unknown. Animal developmental or reproduction toxicity studies have not been conducted with KamRAB.
There is no information regarding the presence of KamRAB in human milk, the effects on the breastfed infant, or the effects on milk production. The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for KamRAB and any potential adverse effects on the breastfed infant from KamRAB or from the underlying maternal condition.4.7 Effects on Ability to Drive and Use Machines
KamRAB has no known potential for impairment of driving ability, mental ability, or ability to operate machinery.
4.8 Adverse Effects (Undesirable Effects)
KamRAB was evaluated in three single-centre, controlled clinical trials. Subjects in the clinical studies of KamRAB were healthy adults, primarily white and ranged in age from 18 to 72 years. A total of 160 subjects were treated in these three studies, including 91 subjects who received single intramuscular doses of KamRAB (20 IU/kg) with or without rabies vaccine.
Table 1 summarises adverse events (assessed by the investigator as related or unrelated to study treatment) occurring in ≥ 3% of subjects in the clinical trials of KamRAB.
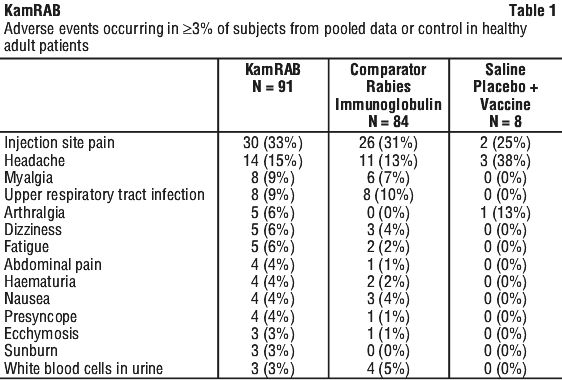
Adverse reactions in adult population.
The following convention has been utilised for the classification of undesirable effects: very common (≥ 1/10), common (≥ 1/100 and < 1/10), uncommon (≥ 1/1000 and < 1/100), rare (≥ 1/10,000 and < 1/1000), very rare (< 1/10,000).
Gastrointestinal disorders.
Common: Diarrhoea, Vomiting.
General system disorders.
Common: Asthenia, Malaise.
Hepatobiliary disorders.
Common: Liver function test abnormal.
Infections and infestations.
Common: Injection site hematoma.
Metabolism and nutrition disorders.
Common: Decrease appetite.
Musculoskeletal and connective tissue disorders.
Common: Musculoskeletal stiffness.
Nervous system disorders.
Common: Paraesthesia.
Skin and subcutaneous tissue disorders.
Common: Eczema, Pruritus.
Vascular disorders.
Common: Syncope.
KamRAB was also evaluated in a two-centre, open-label clinical trial in 30 paediatric patients exposed or possibly exposed to rabies virus. They ranged in age from 0.5 to 14.9 years. Study treatment included a single dose of KamRAB (20 IU/kg) and active rabies vaccine on Days 0, 3, 7 and 14 administered as per CDC Advisory Committee on Immunization Practices (ACIP) recommendations for rabies post-exposure prophylaxis.
Table 2 summarises the adverse events that occurred in > 5% of patients in the paediatric clinical trial within 14 days of receipt of KamRAB and the first dose of the rabies vaccine.
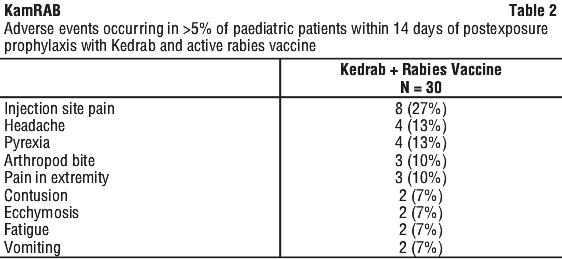
Adverse reactions in paediatric population.
The following convention has been utilised for the classification of undesirable effects: very common (≥ 1/10), common (≥ 1/100 and < 1/10), uncommon (≥ 1/1000 and < 1/100), rare (≥ 1/10,000 and < 1/1000), very rare (< 1/10,000).
Gastrointestinal disorders.
Common: Oral pain.
General disorders and administration site conditions.
Common: Injection site erythema, Injection site oedema.
Injury, poisoning and procedural complications.
Common: Wound complication.
Musculoskeletal and connective tissue disorders.
Common: Myalgia.
Psychiatric disorders.
Common: Insomnia (was reported as occurring after 14 days of administration).
For safety information with respect to transmissible agents, see Section 4.4 Special Warnings and Precautions for Use.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
There is no specific antidote for overdose and treatment should be symptomatic.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: immune sera and immunoglobulins; ATC code: J06BB05.
Mechanism of action.
Rabies is a zoonotic disease caused by RNA viruses in the family Rhabdoviridae, genus Lyssavirus.
Virus is typically present in the saliva of rabid mammals and is transmitted primarily through a bite. KamRAB is infiltrated into the inoculation site (i.e. at the beginning of anti-rabies PEP) to previously unvaccinated persons, to provide immediate passive rabies virus neutralising antibody protection until the patient’s immune system responds to vaccination by actively producing antibodies.
A protective threshold for RVNA has never been established. However, the WHO has generally accepted a RVNA of at least 0.5 IU/mL measured 14 days after initiation of PEP as protective. By comparison, the ACIP at the CDC recommends complete neutralisation of rabies virus at a 1:5 serum dilution by a rapid fluorescent focus inhibition test (RFFIT) from 1 to 2 weeks after prophylaxis; this corresponds to RVNA ~0.1-0.2 IU/mL. In support of these recommendations, there has been almost no documented clinical disease when the current rabies PEP regimen is administered appropriately.
Clinical trials.
The efficacy of KamRAB administered concurrently with rabies vaccine was studied in a single-centre, randomised, comparator rabies immunoglobulin-controlled clinical study (Study 003). Study subjects were healthy adults 18 to 72 years of age who were without significant acute or chronic illness. A total of 118 subjects (59 per treatment group) received intramuscular KamRAB or comparator rabies immunoglobulin at a dose of 20 IU/kg on Day 0, and rabies vaccine on Days 0, 3,
7, 14 and 28. The mean age of study subjects was 45 years; subjects were, predominantly white (93%), and 64% were women. The efficacy variable was RVNA, as assessed by RFFIT, on Day 14. Efficacy analyses were performed on the As-Treated Population, which comprised the 116 study subjects who received KamRAB or comparator rabies immunoglobulin and at least 3 of the 5 doses of rabies vaccine before Day 14.
Efficacy, considered when RVNA titre is 0.5 IU/mL or higher on Day 14 (as established by the WHO), was met by 56/57 subjects (98.2%) in the KamRAB group and 59/59 subjects in the comparator rabies immunoglobulin group. The lower limit of the 90% CI was greater than the pre-specified noninferiority margin of -10%; thus, KamRAB was non-inferior to comparator rabies immunoglobulin. See Table 3.
 KamRAB was also evaluated in a two-centre, open-label clinical trial in 30 paediatric patients (Study 004) exposed or possibly exposed to rabies virus for whom post-exposure prophylaxis was indicated. The patients were treated with KamRAB at a dose of 20 IU/kg on Day 0 and active rabies vaccine on Days 0, 3, 7, and 14 as per ACIP recommendations for rabies post-exposure prophylaxis. The patients ranged in age from 0.5 to 14.9 years, 46.7% were females, 6.7% were Asian, 23.3% were Black and 70% were White, 10% were Latino. The efficacy variables were RVNA as assessed by RFFIT on Day 14 and occurrence of rabies disease through Day 84 after administration of KamRAB. Efficacy analyses were performed on the As-Treated Population, which comprised all 30 study patients.
KamRAB was also evaluated in a two-centre, open-label clinical trial in 30 paediatric patients (Study 004) exposed or possibly exposed to rabies virus for whom post-exposure prophylaxis was indicated. The patients were treated with KamRAB at a dose of 20 IU/kg on Day 0 and active rabies vaccine on Days 0, 3, 7, and 14 as per ACIP recommendations for rabies post-exposure prophylaxis. The patients ranged in age from 0.5 to 14.9 years, 46.7% were females, 6.7% were Asian, 23.3% were Black and 70% were White, 10% were Latino. The efficacy variables were RVNA as assessed by RFFIT on Day 14 and occurrence of rabies disease through Day 84 after administration of KamRAB. Efficacy analyses were performed on the As-Treated Population, which comprised all 30 study patients.
In the As-Treated Population, the geometric mean (SD) Day 14 RVNA titre was 18.89 (31.61) IU/mL and the median Day-14 RVNA titre was 8.81 (range 0.21 - 153.62) IU/mL. Of the 30 treated paediatric patients, 28 patients (93.3%) had a Day 14 RVNA titre ≥ 0.5 IU/mL, the WHO recommended level. None of the 28/30 patients who were followed for the duration of the study developed rabies infection through day 84.
5.2 Pharmacokinetic Properties
A randomised, single-dose, two-period, two-treatment, two-sequence, double-blind, crossover study assessed the pharmacokinetics of KamRAB. Twenty-six healthy volunteer subjects were randomised to receive a single IM injection of 20 IU/kg rabies immunoglobulin on two separate occasions (KamRAB or Comparator rabies immunoglobulin). Subjects received the second treatment (A or B) following the 42-day test period and a 21-day washout period. Single dose IM injection of KamRAB resulted in maximum plasma RVNA levels of 0.25 IU/mL. The median Tmax was 7 days (range: 3 - 14 days). The elimination half-life was approximately 17.9 days.
Additionally, a prospective, randomised, double-blind, non-inferiority, study evaluated the pharmacokinetics, safety and effectiveness of simulated post-exposure prophylaxis with KamRAB with co-administration of active rabies vaccine in 118 healthy subjects. Subjects were randomised into two treatment groups (59 per treatment group) to receive intramuscular KamRAB or comparator rabies immunoglobulin at a dose of 20 IU/kg on Day 0, and rabies vaccine on Days 0, 3, 7, 14 and 28. The geometric mean of the peak plasma RVNA was 39.9 IU/mL and 36.2 IU/mL for KamRAB and comparator rabies immunoglobulin respectively. For both treatment groups, the median Tmax was 14 days (range: 14 - 49 days). The half-lives were 48.6 hours and 52.7 hours for KamRAB and comparator rabies immunoglobulin respectively (Table 4).
A plot of plasma rabies virus neutralizing antibody titre concentration versus time (Figure 1) demonstrated that, in both treatment groups, plasma rabies virus neutralizing antibody concentrations declined in a biphasic manner after the absorption phase was complete.
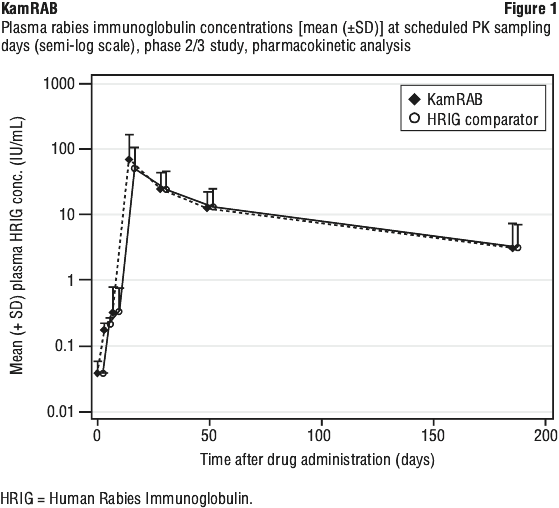
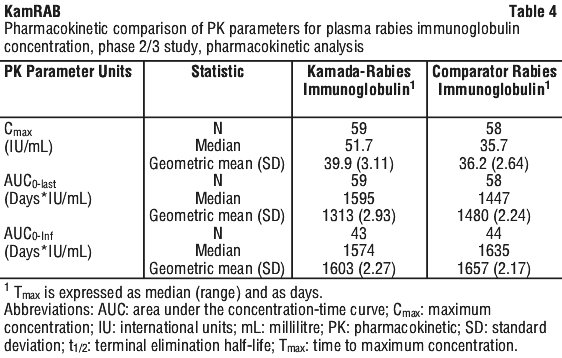
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
No data available.6 Pharmaceutical Particulars
6.1 List of Excipients
Glycine, water for injection, sodium hydroxide (for pH adjustment).
6.2 Incompatibilities
KamRAB should not be mixed with the rabies vaccine or injected in the same syringe with the rabies vaccine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
In-use stability.
KamRAB may be stored at room temperatures not exceeding 25°C for up to one month. Use within one month after removal from refrigeration, do not return to refrigeration.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze).
6.5 Nature and Contents of Container
2 mL and 10 mL vials (Type I borosilicate clear glass), closed with a stopper (chlorobutyl or bromobutyl) and seal (aluminium) fitted with a flip-off cap (polypropylene).
Pack size of 1 vial.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
No data available.
CAS number.
No data available.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.



 KamRAB was also evaluated in a two-centre, open-label clinical trial in 30 paediatric patients (Study 004) exposed or possibly exposed to rabies virus for whom post-exposure prophylaxis was indicated. The patients were treated with KamRAB at a dose of 20 IU/kg on Day 0 and active rabies vaccine on Days 0, 3, 7, and 14 as per ACIP recommendations for rabies post-exposure prophylaxis. The patients ranged in age from 0.5 to 14.9 years, 46.7% were females, 6.7% were Asian, 23.3% were Black and 70% were White, 10% were Latino. The efficacy variables were RVNA as assessed by RFFIT on Day 14 and occurrence of rabies disease through Day 84 after administration of KamRAB. Efficacy analyses were performed on the As-Treated Population, which comprised all 30 study patients.
KamRAB was also evaluated in a two-centre, open-label clinical trial in 30 paediatric patients (Study 004) exposed or possibly exposed to rabies virus for whom post-exposure prophylaxis was indicated. The patients were treated with KamRAB at a dose of 20 IU/kg on Day 0 and active rabies vaccine on Days 0, 3, 7, and 14 as per ACIP recommendations for rabies post-exposure prophylaxis. The patients ranged in age from 0.5 to 14.9 years, 46.7% were females, 6.7% were Asian, 23.3% were Black and 70% were White, 10% were Latino. The efficacy variables were RVNA as assessed by RFFIT on Day 14 and occurrence of rabies disease through Day 84 after administration of KamRAB. Efficacy analyses were performed on the As-Treated Population, which comprised all 30 study patients.
