What is in this leaflet
This leaflet answers some common questions about Karvezide. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have benefits and risks. Your doctor has weighed the benefits of you taking this medicine against the risks it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What Karvezide is used for
Karvezide is used to lower high blood pressure, which doctors call hypertension.
Everyone has blood pressure. This pressure helps get your blood all around your body. Your blood pressure may be different at different times of the day, depending on how busy or worried you are. You have hypertension (high blood pressure) when your blood pressure stays higher than is needed, even when you are calm and relaxed.
There are often no symptoms of high blood pressure. The only way of knowing that you have high blood pressure is to have your blood pressure checked on a regular basis.
If high blood pressure is not treated it can lead to serious health problems, such as stroke, heart disease and kidney failure.
How Karvezide works
Karvezide contains irbesartan and hydrochlorothiazide. Both medicines reduce blood pressure in different ways. Irbesartan belongs to a group of medicines known as angiotensin-II receptor antagonists. Angiotensin-II is a substance produced in the body that causes blood vessels to narrow. Irbesartan blocks angiotensin-II and therefore widens your blood vessels, making it easier for your heart to pump blood throughout your body. This helps to lower your blood pressure.
Hydrochlorothiazide belongs to the class of medicines known as diuretics. Diuretics cause an increase in the volume of urine. They also help with lowering blood pressure particularly when combined with other blood pressure reducing medicines.
Your doctor may have prescribed Karvezide for another reason. Ask your doctor if you have any questions about why Karvezide has been prescribed for you.
This medicine is only available with a doctor's prescription.
There is no evidence that this medicine is addictive.
Before you take Karvezide
When you must not take it
Do not take Karvezide if:
- you are pregnant (or think you may be pregnant) or are planning to become pregnant.
Karvezide may affect your developing baby if you take it during pregnancy. - you are breast-feeding or are planning to breast-feed.
- you are allergic to irbesartan or hydrochlorothiazide or to sulfonamide derived medicines (commonly known as sulfur drugs), or to any of the ingredients listed under at the end of this leaflet.
- you are diabetic or have kidney problems and are being treated with an ACE inhibitor, any aliskiren-containing medicines or a group of medicines known as AIIRAs (medicines also used to treat high blood pressure).
- you are not producing urine.
- the packaging is torn or shows signs of tampering.
- the expiry date on the pack has passed.
If you take this medicine after the expiry date has passed, it may not work.
If you are not sure if you should start taking Karvezide, talk to your doctor.
Karvezide should not be given to children.
Before you start to take it
Tell your doctor if have any allergies to:
- any of the ingredients listed at the end of this leaflet including lactose
- any other medicines, including sulfonamide derived medicines (commonly known as sulfur drugs)
- any other substances , such as foods, preservatives or dyes
Tell your doctor if you are pregnant or intend to become pregnant.
Tell your doctor if you are breast-feeding or plan to breastfeed.
Tell your doctor if you have or have had any medical conditions, especially the following:
- recent excessive vomiting or diarrhoea or think you are dehydrated
- kidney problems, or have had a kidney transplant or dialysis
- heart problems
- liver problems, or have had liver problems in the past
- diabetes
- gout or have had gout in the past
- lupus erythematosus
- high or low levels of potassium or sodium or other electrolytes in your blood
- primary aldosteronism
- you are strictly restricting your salt intake
- you have had a sympathectomy
- you have been taking diuretics
- you have a history of allergy or asthma
- you have had skin cancer or you develop an unexpected skin lesion during the treatment. Treatment with hydrochlorothiazide, particularly long term use with high doses, may increase the risk of some types of skin and lip cancer (non-melanoma skin cancer). Protect your skin from sun exposure and UV rays while taking Avapro HCT.
Tell your doctor if you plan to have surgery (even at the dentist) that needs a general anaesthetic.
If you have not told your doctor about any of the above, tell them before you take Karvezide.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
It is especially important that you tell your doctor if you are taking or plan to take any of the following:
- other medicines for high blood pressure
- other fluid tablets or diuretics
- lithium or lithium-containing medicines (for example lithium carbonate)
- potassium tablets
- potassium-containing salt substitutes
- anti-inflammatory medicines such as non-steroidal anti-inflammatory agents (for example diclofenac, ibuprofen) and COX-2 inhibitors (for example celecoxib) used to relieve pain, swelling and other symptoms of inflammation including arthritis. Taking Karvezide and an anti-inflammatory medicine alone or with a thiazide diuretic (fluid tablet) may damage your kidneys. It may also reduce the effect Karvezide has on lowering blood pressure
- alcoholic drinks
- sleeping tablets
- strong pain killing medicines such as codeine or morphine
- medicines for diabetes (oral tablets or capsules or insulins)
- calcium supplements or medicines containing calcium
- Vitamin D
- medicines for gout
- powder or granules used to help reduce cholesterol (cholestyramine or colestipol hydrochloride)
- heart medicines such as digoxin or antiarrhythmic medicines (for example sotalol hydrochloride)
- corticosteroid medicines such as prednisone, cortisone or ACTH
- medicines used to treat cancer (cytotoxic medicines)
- amantadine, a medicine used to treat Parkinson's disease or to prevent influenza
- anticholinergic medicines, these can be used to treat Parkinson's disease, to relieve stomach cramps or spasms or used to prevent travel sickness
- carbamazepine, an anticonvulsant medication
- medicines used during surgery
- medicines used in an emergency situation such as adrenaline
Your doctor will decide whether your treatment needs to be altered or whether you should have check-ups more frequently.
Your doctor and pharmacist may have more information on medicines to be careful with or avoid while taking Karvezide.
How to take Karvezide
How much to take:
Karvezide will usually be prescribed by your doctor if previous treatment does not produce a sufficient drop in your blood pressure. Your doctor will tell you how to switch from your previous treatment to Karvezide.
The usual dose of Karvezide is one tablet a day. The full blood pressure lowering effect should be reached 6-8 weeks after beginning treatment. If your blood pressure is not satisfactorily reduced with Karvezide, your doctor may prescribe another medicine to be taken with Karvezide.
How to take it
Swallow the tablet whole with a glass of water.
It does not matter whether you take Karvezide tablets before or after food.
When to take it
Take Karvezide at about the same time each day. Taking your tablets at the same time each day will have the best effect. It will also help you remember when to take the tablets.
How long to take it
Continue taking Karvezide until your doctor tells you to stop.
Karvezide helps control your high blood pressure but does not cure it. Therefore Karvezide must be taken every day.
To help you remember to take your tablets each day, Karvezide tablets are supplied in a Calendar pack with the foil backing marked with the days of the week. This is just a way to help you to remember to take your tablets. All of the tablets in the pack are the same.
When you start a new strip of tablets, take the tablet marked "START". On the next day, take the tablet marked with the relevant day of the week.
Continue taking your tablets each day until all of the tablets are taken. Commence the next strip at "START" and continue as before.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, and go back to taking your tablets as you would normally.
Do not take a double dose to make up for the dose you missed. This may increase the chance of you getting any unwanted side effects.
If you have trouble remembering to take your tablets, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or Poisons Information Centre (telephone: 13 11 26) or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else may have taken too much Karvezide.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
If you take too many Karvezide tablets, you will probably feel lightheaded or dizzy.
While you are using Karvezide
Things you must do
Tell any other doctors, dentists and pharmacists who are treating you that you are taking Karvezide.
If you become pregnant while taking Karvezide tell your doctor immediately.
Have your blood pressure checked when your doctor tells you to, to make sure Karvezide is working.
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are taking Karvezide.
If you plan to have surgery or other treatment (even at the dentist) that needs an anaesthetic, make sure that you tell your doctor or dentist that you are taking Karvezide. Your blood pressure may drop suddenly.
Make sure you drink enough water during exercise and hot weather when you are taking Karvezide, especially if you sweat a lot. If you do not drink enough water while taking Karvezide, you may faint or feel light-headed or sick. This is because your body does not have enough fluid and your blood pressure is low. If you continue to feel unwell, tell your doctor.
If you have excessive vomiting and/or diarrhoea while taking Karvezide, tell your doctor. You may lose too much water and salt and your blood pressure may drop too much.
If you feel light headed or dizzy after taking your first dose of Karvezide, or when your dose is increased, tell your doctor immediately.
Tell your doctor if you experience an increased sensitivity of the skin to the sun with symptoms of sunburn (such as redness, itching, swelling, blistering) occurring more quickly than normal
Things you must not do
Do not give Karvezide tablets to anyone else, even if they have the same condition as you.
Do not take Karvezide to treat any other complaints unless your doctor or pharmacist tells you to.
Do not stop taking Karvezide, or lower the dosage, without checking with your doctor.
Things to be careful of
If you feel light-headed, dizzy or faint when getting out of bed or standing up, get up slowly. Standing up slowly, especially when you get up from bed or chairs, will help your body get used to the change in position and blood pressure. If this problem continues or gets worse, talk to your doctor.
Be careful driving or operating machinery until you know how Karvezide affects you. As with many other medicines used to treat high blood pressure, Karvezide may cause dizziness or light-headedness in some people. If this occurs do not drive. Make sure you know how you react to Karvezide before you drive a car, operate machinery or do anything else that could be dangerous if you are dizzy or light-headed.
If you drink alcohol, dizziness or light-headedness may be worse.
The hydrochlorothiazide contained in this medicine could produce a positive analytical result in an antidoping test.
Things that would be helpful for lowering your blood pressure
Some self-help measures suggested below may help your condition. Talk to your doctor or pharmacist about these measures and for more information.
- Alcohol -
your doctor may advise you to limit your alcohol intake. - Weight -
if you are overweight, your doctor may suggest losing some weight to help lower your blood pressure and help lessen the amount of work your heart has to do. Some people may need a dietician's help to lose weight. - Diet -
your doctor may advise you to eat a healthy low-fat diet which includes plenty of fresh vegetables, fruit, bread, cereals and fish. - Salt -
your doctor may advise you to watch the amount of salt in your diet. To reduce your salt intake you should avoid using salt in cooking or at the table. - Exercise -
regular exercise helps to reduce blood pressure and helps get the heart fitter, but it is important not to overdo it. Walking is good exercise, but try to find a route that is reasonably flat. Before starting any exercise, ask your doctor about the best kind of programme for you. - Smoking -
your doctor may advise you to stop smoking or at least cut down.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Karvezide.
Karvezide helps most people with high blood pressure, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious. Most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- headache
- dizziness or light-headedness (vertigo)
- unusual tiredness or weakness, fatigue
- pain in the stomach or gut nausea and/or vomiting
- sexual problems
These are generally mild and do not normally require treatment to be interrupted.
Tell your doctor immediately if you notice any of the following:
- skin rash or itchiness
- aching muscles or sore joints, not caused by exercise
- muscle pain or weakness
- buzzing, ringing or other persistent noise in the ears
- changes in heart rhythm
- you are not urinating (passing water) as much as normal
- numbness or tingling in fingers or toes
- painful, swollen joints that may be symptoms of gout
- fainting
- yellowing of the skin and/or eyes, also known as jaundice
- symptoms that may occur if you have developed diabetes, such as excessive thirst, passing greatly increased amounts of urine, increased appetite with weight loss, feeling tired, drowsy, weak, depressed, irritable and generally unwell
- symptoms that may indicate kidney disease such as passing little or no urine, drowsiness, nausea, vomiting, breathlessness, loss of appetite and weakness
- symptoms that may indicate high potassium levels in the blood such as nausea, diarrhoea, muscle weakness and changes in heart rhythm
- symptoms that may indicate liver disease such as nausea, vomiting, loss of appetite, feeling generally unwell, fever, itching, yellowing of the skin and eyes and dark coloured urine
- symptoms that may indicate low platelet count such as easy or excessive bruising, bleeding from gums or nose, prolonged bleeding from cuts and blood in urine or stools
- disturbances in your vision (such as blurred vision and seeing haloes around lights) and eye pain.
These are serious side effects. Skin rash and itchiness may be symptoms of an allergic reaction.
You may need medical attention. These side effects are not common.
If any of the following happen, stop taking Karvezide and either tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- swelling to the face, lips, tongue or throat, which may cause difficulty in swallowing or breathing.
- severe and a sudden onset of pinkish, itchy swellings on the skin, also called hives or nettlerash.
These are very serious side effects. You may need urgent medical attention or hospitalisation.
These side effects are very rare.
Other side effects not listed above may occur in some patients. Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
After using Karvezide
Storage
Keep your Karvezide tablets in the blister pack until it is time to take them. If you take the tablets out of the blister pack they will not keep well.
Keep Karvezide tablets in a cool dry place where the temperature stays below 25°C. Do not store Karvezide or any other medicine in the bathroom near a sink. Do not leave them near a radiator, in a car on a hot day or on a window sill. Heat and dampness can destroy some medicines.
Keep all medicines out of the reach of children. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking Karvezide or the tablets have passed their expiry date, ask your pharmacist what to do with any tablets that are left over.
Product description
What it looks like
Karvezide 150/12.5 tablets - peach, oval shaped film-coated tablet with a heart shape imprinted on one side and "2875" engraved on the other side. Blister pack sizes include: 3*, 5*, 7*, 14*, 28*, 30, 56* and 98* tablets. AUST R 101831.
Karvezide 300/12.5 tablets - peach, oval shaped film-coated tablet with a heart shape imprinted on one side and "2876" engraved on the other side. Blister pack sizes include: 3*, 5*, 7*, 14*, 28*, 30, 56* and 98* tablets. AUST R 101833.
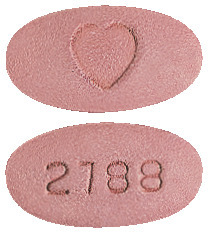
Karvezide 300/25 tablets - pink, oval shaped film-coated tablet with a heart shape imprinted on one side and "2788" engraved on the other side. Blister pack sizes include: 3*, 5*, 7*, 14*, 28*, 30, 56* and 98* tablets. AUST R 129442.
* presentations currently not-marketed.
Active Ingredients
Karvezide 150/12.5 tablets - 150 mg Irbesartan and 12.5 mg Hydrochlorothiazide per tablet.
Karvezide 300/12.5 tablets - 300 mg Irbesartan and 12.5 mg Hydrochlorothiazide per tablet.
Karvezide 300/25 tablets - 300 mg Irbesartan and 25 mg Hydrochlorothiazide per tablet.
Other Ingredients
Karvezide 150/12.5 & 300/12.5 tablets also contain carnauba wax, croscarmellose sodium, hypromellose, lactose, iron oxide red CI 77491, iron oxide yellow CI 77492, macrogol 3000, magnesium stearate, microcrystalline cellulose, silicon dioxide, and titanium dioxide.
Karvezide 300/25 tablets also contain carnauba wax, croscarmellose sodium, hypromellose, lactose, iron oxide red CI 77491, iron oxide yellow CI 77492, iron oxide black CI 77499, macrogol 3350, magnesium stearate, microcrystalline cellulose, silicon dioxide, pregelatinised maize starch, and titanium dioxide.
Manufacturer
Karvezide is supplied in Australia by
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park, NSW 2113.
Karvezide is a registered sanofi-aventis trademark.
This leaflet was prepared in September 2019.
karvezide-ccdsv15-cmiv12-09sep19
Published by MIMS November 2019



 Adverse reactions (clinical events probably or possibly related to therapy as determined by the clinical investigator) that occurred in more than 2 hypertensive patients when they were taking irbesartan/ hydrochlorothiazide and no additional study medications in premarketing clinical trials involving 2700 subjects, and that were not reported in the tabulation of adverse events, are listed in the following section.
Adverse reactions (clinical events probably or possibly related to therapy as determined by the clinical investigator) that occurred in more than 2 hypertensive patients when they were taking irbesartan/ hydrochlorothiazide and no additional study medications in premarketing clinical trials involving 2700 subjects, and that were not reported in the tabulation of adverse events, are listed in the following section. The empiric formula is C25H28N6O and the molecular weight, 428.5.
The empiric formula is C25H28N6O and the molecular weight, 428.5. The empiric formula is C7H8ClN3O4S2 and the molecular weight, 297.7.
The empiric formula is C7H8ClN3O4S2 and the molecular weight, 297.7.