1. Why am I using Kesimpta?
Kesimpta contains the active ingredient ofatumumab. Ofatumumab is a type of protein called a monoclonal antibody designed to recognize and attach to a target called CD20 on the surface of certain types of white blood cells which are part of the immune system (so called B-cells).
Kesimpta is used for the treatment of adults with relapsing forms of multiple sclerosis.
By targeting and removing the B-cells, Kesimpta helps to reduce their activity and thereby reduces the chance of having a relapse, relieves symptoms and slows down the progression of multiple sclerosis.
2. What should I know before I use Kesimpta?
Warnings
Do not use Kesimpta if:
- you are allergic to ofatumumab, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- are at risk of hepatitis B infection (all patients will have a blood test before starting Kesimpta)
- have an infection, especially if it is severe. Kesimpta must not be used until a severe infection is better
- have a weakened immune system and are at a higher risk of getting an infection. Kesimpta must not be used until your immune system is stronger
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
You should avoid becoming pregnant while using Kesimpta and for 6 months after you stop using it. If you become pregnant or think you are pregnant, tell your doctor right away.
Contraception
To avoid becoming pregnant, you should use contraception while using Kesimpta and for 6 months after you stop using it.
Use in children
- It is not known whether Kesimpta is safe and effective in children.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Kesimpta and affect how it works.
- Medicines that lower or modify the immune system including other medicines used to treat multiple sclerosis such as ocrelizumab, cladribine, fingolimod, natalizumab, teriflunomide, mitoxantrone, or dimethyl fumarate
- vaccines
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Kesimpta.
4. How do I use Kesimpta?
How much to use
- Inject 20mg under the skin on week 0, week 1 and week 2. There is no injection at week 3.
- At week 4 and then every month, inject 20mg under the skin.
- Follow the instructions provided and use Kesimpta until your doctor tells you to stop.
When to use Kesimpta
- Kesimpta is injected under the skin on week 0, week 1 and week 2. There is no injection at week 3. At week 4 and then every month inject under the skin.
How to use Kesimpta
- Kesimpta is injected under the skin.
- Follow the “Instructions for Use” leaflet in the pack.
If you forget to use Kesimpta
Kesimpta should be used regularly at the same time each week on weeks 0, 1 and 2 and then each month from week 4 onwards.
If you have missed a dose of Kesimpta, you should inject your missed dose as soon as possible without waiting until the next scheduled dose. Any following doses should be administered as per the recommended schedule.
Do not inject a double dose to make up for the dose you missed.
If you use too much Kesimpta
If you think that you have used too much Kesimpta, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Kesimpta?
Things you should do
- Have a blood test to check for Hepatitis B
- Tell your doctor if you plan to receive a vaccine
- Avoid becoming pregnant while using Kesimpta and for 6 months after you stop using it
Call your doctor straight away if you:
- Have injection related reactions or injection site reactions
- Have any type of infection
- Believe your multiple sclerosis is getting worse (e.g. weakness or eye changes) or if you notice any new or unusual symptoms – this can be serious.
Remind any doctor, dentist or pharmacist you visit that you are using Kesimpta.
Things you should not do
- Do not stop using this medicine suddenly.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Kesimpta affects you.
The effects of Kesimpta on a person’s ability to drive and use machines has not been tested.
Looking after your medicine
- Store between 2 to 8°C. Refrigerate. Do not freeze. Keep in the original package.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
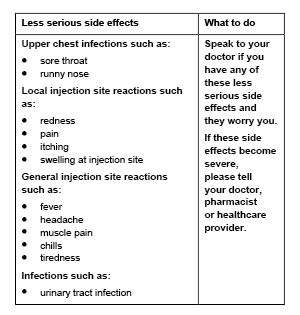
Serious side effects
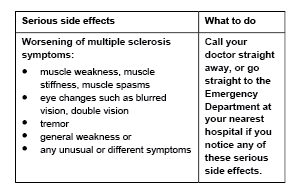
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell. Other side effects not listed here may occur in some people.
Some side effects (for example decrease in specific proteins in the blood (immunoglobulins M) which help protect against infection) can only be found when your doctor does tests from time to time to check your progress.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Kesimpta contains

Do not administer this medicine if you are allergic to any of these ingredients.
What Kesimpta looks like
Kesimpta is supplied as a 20 mg/0.4 mL pre-filled syringe or a 20 mg/0.4 mL pre-filled pen.*
This product contains only one pre-filled pen or pre-filled syringe per pack.
The single-use solution for injection is sterile, preservative-free, clear to slightly opalescent, and colorless to slightly brownish-yellow (Aust R 330617; AUST R 330601).
*Not all products may be marketed
Who distributes Kesimpta
Kesimpta is supplied in Australia by:
Novartis Pharmaceuticals Australia Pty Limited
(ABN 18 004 244 160)
54 Waterloo Road
Macquarie Park NSW 2113
Telephone 1 800 671 203
Website: www.novartis.com.au
This leaflet was prepared in March 2021.
Published by MIMS April 2021



 The efficacy results for both studies are summarized in Table 4, and Figure 1.
The efficacy results for both studies are summarized in Table 4, and Figure 1.

