What is in this leaflet
This leaflet answers some common questions about KEYTRUDA. It does not contain all the available information.
It does not take the place of talking to your doctor.
All medicines have risks and benefits. Your doctor has weighed the risks of you using KEYTRUDA against the benefits they expect it will have for you.
If you have any concerns about being given this medicine, ask your doctor.
Keep this leaflet. You may need to read it again.
What KEYTRUDA is used for
KEYTRUDA is used to treat:
- a kind of skin cancer called melanoma in adults.
- a kind of lung cancer called non-small cell lung cancer in adults.
- a kind of head and neck cancer called head and neck squamous cell carcinoma in adults.
- a kind of cancer called classical Hodgkin Lymphoma in adults.
- a kind of cancer called primary mediastinal B-cell lymphoma in adults and children.
- a kind of cancer called urothelial carcinoma, including bladder cancer in adults.
- a kind of cancer in adults and children that can occur in any part of the body and is shown by laboratory tests to be microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR).
- a kind of cancer called colon or rectal cancer in adults that is shown by a laboratory test to be MSI-H or dMMR
- a kind of uterine cancer called endometrial carcinoma
- a kind of kidney cancer called renal cell carcinoma in adults.
It is not known if KEYTRUDA is safe and effective in children with MSI-H or dMMR cancers of the brain or spinal cord (central nervous system cancers).
KEYTRUDA contains the active substance pembrolizumab.
Adults may get KEYTRUDA if their cancer has spread or cannot be taken out by surgery.
Adults get KEYTRUDA after they had surgery to remove melanoma to help prevent their cancer from coming back.
KEYTRUDA may be given in combination with other anti-cancer medicines. It is important that you also read the Consumer Medicine Information for these other medicines. If you have any questions about these specific medicines, please ask your doctor.
This medicine is available only with a doctor's prescription.
KEYTRUDA works by helping your immune system fight your cancer.
Ask your doctor if you have any questions about why KEYTRUDA has been prescribed for you.
Before you are given KEYTRUDA
KEYTRUDA can be used in children with primary mediastinal B-cell lymphoma.
Before you are given KEYTRUDA
Tell your doctor if you:
- have a disease of your immune system like Crohn's, ulcerative colitis or lupus
- had an organ transplant (like a kidney transplant) or a bone marrow (stem cell) transplant that used donor stem cells (allogeneic)
- have pneumonia or swelling of your lungs (called pneumonitis)
- have liver damage
Pregnancy
- If you are pregnant, think you may be pregnant or are planning to have a baby, tell your doctor.
- KEYTRUDA can cause harm or death to your unborn baby.
- You must use effective contraception while you are being treated with KEYTRUDA and for at least 4 months after the last dose of KEYTRUDA if you are a woman who could become pregnant.
Breast-feeding
- If you are breastfeeding, tell your doctor.
- Do not breastfeed while taking KEYTRUDA.
Taking other medicines
Tell your doctor if you
- are taking any other medicines, including medicines that you buy without a prescription from your pharmacy, supermarket or health food shop
- take other medicines that make your immune system weak. Examples of these may include steroids, such as prednisone.
How KEYTRUDA is given
- Your doctor will give you KEYTRUDA through an IV for about 30 minutes.
- Most people get KEYTRUDA every 3 weeks or every 6 weeks, depending on the dose you are given.
- Your doctor will decide how many treatments you need.
If you miss a dose
- Call your doctor right away to reschedule your appointment.
- It is very important that you do not miss a dose of this medicine.
While you are being treated with KEYTRUDA
Things you must do
Tell any other doctors, dentists, and pharmacists who are treating you that you are being given KEYTRUDA.
If you are about to be started on any new medicine, tell your doctor, dentist or pharmacist that you are being given KEYTRUDA.
Side Effects
Like all medicines, KEYTRUDA can cause side effects, although not everybody gets them. Your doctor will discuss these with you and will explain the risks and benefits of your treatment.
When you get KEYTRUDA, you can have some serious side effects.
These side effects can sometimes become life-threatening and can lead to death. These side effects may happen anytime during treatment or even after your treatment has ended. You may experience more than one side effect at the same time.
If you have any of the following symptoms, call or see your doctor right away.
- Signs and symptoms of lung problems
- shortness of breath
- chest pain
- coughing - Signs and symptoms of problems with your intestines
- diarrhoea or more bowel movements than usual
- your stools are black, tarry, sticky or have blood or mucus
- severe stomach pain or tenderness - Signs and symptoms of liver problems
- nausea or vomiting
- feeling less hungry
- pain on the right side of your stomach
- your skin looks yellow
- the whites of your eyes look yellow
- dark urine
- you bleed or bruise more easily than normal - Signs and symptoms of kidney problems
- changes in the amount or colour of your urine - Signs and symptoms of hormone gland problems (especially the thyroid, pituitary, and adrenal glands)
- rapid heart beat
- weight loss
- increased sweating
- weight gain
- hair loss
- feeling cold
- constipation
- your voice gets deeper
- muscle aches
- dizziness or fainting
- headaches that will not go away or unusual headache - Signs and symptoms of blood sugar problems
- feeling more hungry or thirsty
- needing to urinate more often
- weight loss - Signs and symptoms of skin problems
- rash
- itching
- skin blistering, peeling or sores
- ulcers in mouth or in lining of nose, throat, or genital area - Signs and symptoms of problems in other organs
- muscle pain or weakness
- changes in eyesight
- stomach area pain with nausea and vomiting (pancreatitis)
- confusion, fever, memory problems, or seizures (encephalitis)
- swollen lymph nodes, rash or tender lumps on skin, cough, or eye pain (sarcoidosis)
- shortness of breath, irregular heartbeat, feeling tired, or chest pain (myocarditis)
- inflammation of the heart muscle, which may present as shortness of breath, irregular heartbeat, feeling tired, or chest pain (pericarditis)
- inflammation of the covering of the heart and accumulation of fluid around the heart (pericardial effusion)
- inflammation of the nerves that may cause pain, weakness, and paralysis in the arms and legs (peripheral neuropathy)
- pain, numbness, tingling, or weakness in the arms or legs; bladder or bowel problems including needing to urinate more frequently, urinary incontinence, difficulty urinating and constipation (myelitis) - Signs and symptoms of infusion (IV) reactions
- shortness of breath
- itching or rash
- dizziness
- fever - There are possible side effects of treatment with KEYTRUDA in people who have received a transplant
- Rejection of a transplanted organ.
People who have had an organ transplant may have an increased risk of organ transplant rejection. Your doctor should tell you what signs and symptoms you should report and monitor you, depending on the type of organ transplant that you have had.
- Complications, including graft-versus-host-disease (GVHD), in people with bone marrow (stem cell) transplant that uses donor stem cells (allogeneic).
These complications can be severe and can lead to death. They may occur if you had this kind of transplant in the past or if you get it in the future. Your doctor will monitor you for the following signs and symptoms: skin rash, liver inflammation, abdominal pain, and diarrhoea.
The following side effects have been reported in clinical trials:
Very common (may affect more than 1 in 10 people)
- diarrhoea, nausea
- itching, rash
- joint pain
- back pain
- feeling tired
- cough
- patches of skin which have lost colour
- stomach pain
- decreased sodium levels in the blood
- fever
- infections of the upper respiratory tract
- a decreased number of white blood cells (which are important in fighting infection) in patients with primary mediastinal B-cell lymphoma
The following side effects have been reported in more than 1 in 5 people when KEYTRUDA was given in combination with chemotherapy:
- hair loss
- feeling tired
- diarrhoea
- decrease in white blood cell count
- joint pain
- rash
- swelling of the lining of the digestive system (for example mouth, intestines)
- mouth sores
- decrease in red blood cell count
- nausea
- constipation
- vomiting
Common side effects when KEYTRUDA is given in combination with lenvatinib include:
- feeling tired
- high blood pressure
- joint and muscle pain
- diarrhoea
- decreased appetite
- low levels of thyroid hormone
- nausea
- mouth sores
- vomiting
- weight loss
- stomach-area (abdominal) pain
- headache
- constipation
- urinary tract infection
- hoarseness
- bleeding
- low magnesium level
- blisters or rash on the palms of your hands and soles of your feet
- shortness of breath
- cough
- rash
The most common side effects when KEYTRUDA is given in combination with axitinib are:
- diarrhoea
- fatigue
- high blood pressure
- liver problems
- low levels of thyroid hormone
- decreased appetite
- blisters or rash on the palms of your hands and soles of your feet
- nausea
- mouth sores or swelling of the lining of the mouth, nose, eyes, throat, intestines, or vagina
- hoarseness
- rash
- cough
- constipation
The most common side effects when KEYTRUDA is given to children are:
- fever
- vomiting
- fatigue
- constipation
- stomach pain
- nausea
Less common side effects can happen.
Also, your doctor may do blood tests to check for side effects.
KEYTRUDA may cause other side effects that are not listed. For more information, ask your doctor.
If you have any side effect that bothers you or that does not go away, tell your doctor.
Ask your doctor to answer any questions you may have.
Storage
It is unlikely that you will be asked to store KEYTRUDA yourself. It will usually be stored in the pharmacy or on the ward.
Product Description
What it looks like
KEYTRUDA powder for injection comes as a white to off-white powder in a glass vial. KEYTRUDA concentrated injection comes as a clear to slightly opalescent, colourless to slightly yellow solution in a glass vial.
Ingredients
Active ingredient:
- pembrolizumab
Inactive ingredients:
- Histidine
- Histidine hydrochloride monohydrate
- Sucrose
- Polysorbate 80
- Water for Injections
Supplier
KEYTRUDA is supplied in Australia by:
Merck Sharp & Dohme (Australia) Pty Limited
Level 1, Building A, 26 Talavera
Road, Macquarie Park NSW 2113,
AUSTRALIA
This leaflet was prepared 22 February 2021.
Australian Register Numbers:
AUST R 226597 - KEYTRUDA 50mg Powder for injection
AUST R 263932 - KEYTRUDA 100mg/4mL concentrated injection
RCN000013027
Published by MIMS April 2021



 Incidences of pneumonitis in individual studies in patients with melanoma or non-small cell lung cancer treated with Keytruda as monotherapy ranged from 1.6% to 5.8%.
Incidences of pneumonitis in individual studies in patients with melanoma or non-small cell lung cancer treated with Keytruda as monotherapy ranged from 1.6% to 5.8%.
 Table 6 summarises the adverse events that occurred in at least 10% of patients treated with Keytruda in KEYNOTE-002. The most common adverse event (reported in at least 20% of patients) was pruritus. (See Table 7.)
Table 6 summarises the adverse events that occurred in at least 10% of patients treated with Keytruda in KEYNOTE-002. The most common adverse event (reported in at least 20% of patients) was pruritus. (See Table 7.)
 Overall, the safety profile was similar across all doses and between patients previously treated with ipilimumab and patients naïve to treatment with ipilimumab.
Overall, the safety profile was similar across all doses and between patients previously treated with ipilimumab and patients naïve to treatment with ipilimumab.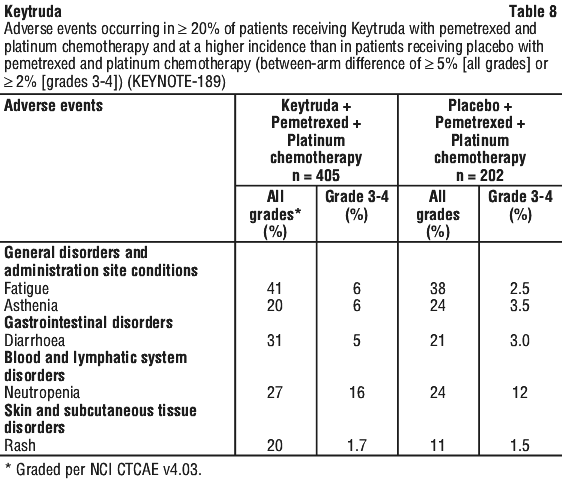





 Clinically relevant adverse reactions in < 10% of patients who received Keytruda included herpes virus infection (9%), pneumonia (8%), oropharyngeal pain (8%), hyperthyroidism (5%), hypersensitivity (4.1%), infusion reactions (3.4%), altered mental state (2.7%), and in 1.4% each, uveitis, myocarditis, thyroiditis, febrile neutropenia, sepsis, and tumour flare.
Clinically relevant adverse reactions in < 10% of patients who received Keytruda included herpes virus infection (9%), pneumonia (8%), oropharyngeal pain (8%), hyperthyroidism (5%), hypersensitivity (4.1%), infusion reactions (3.4%), altered mental state (2.7%), and in 1.4% each, uveitis, myocarditis, thyroiditis, febrile neutropenia, sepsis, and tumour flare.
 Clinically relevant adverse reactions in < 10% of patients who received Keytruda included infusion reactions (9%), hyperthyroidism (3%), pneumonitis (3%), uveitis and myositis (1% each), and myelitis and myocarditis (0.5% each).
Clinically relevant adverse reactions in < 10% of patients who received Keytruda included infusion reactions (9%), hyperthyroidism (3%), pneumonitis (3%), uveitis and myositis (1% each), and myelitis and myocarditis (0.5% each). Hyperbilirubinemia occurred in less than 15% of patients on KEYNOTE-087 (10% all grades, 2.4% grade 3-4).
Hyperbilirubinemia occurred in less than 15% of patients on KEYNOTE-087 (10% all grades, 2.4% grade 3-4). Clinically relevant adverse reactions (< 20%) include pyrexia (18%), dry skin (17%), vomiting (12%), pneumonitis/ILD (10%), hypothyroidism (10%), blurred vision (6%), infusion site extravasation (2%), and myositis (0.5%).
Clinically relevant adverse reactions (< 20%) include pyrexia (18%), dry skin (17%), vomiting (12%), pneumonitis/ILD (10%), hypothyroidism (10%), blurred vision (6%), infusion site extravasation (2%), and myositis (0.5%).




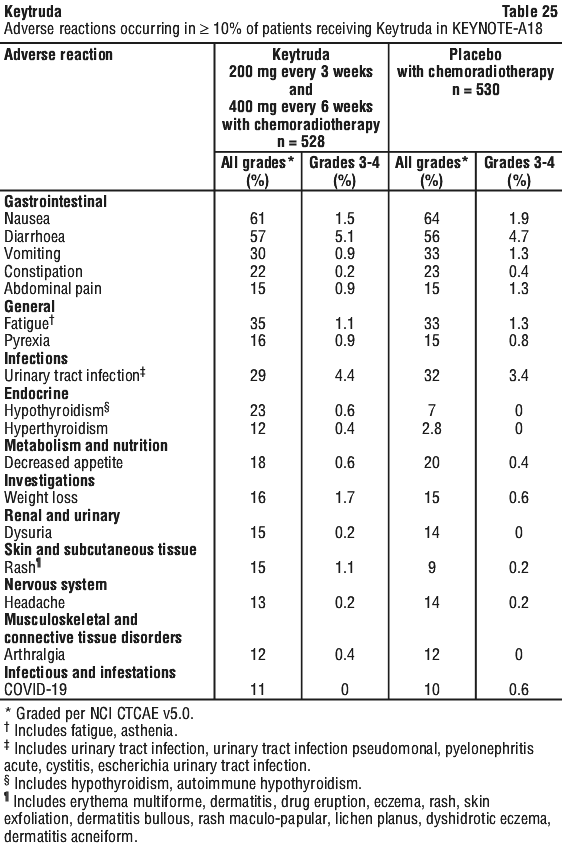












 The final analysis was performed after all patients had at least 21 months of follow-up. The final OS analysis was performed after 383 patient events (119 for Keytruda 10 mg/kg every 3 weeks, 122 for Keytruda 10 mg/kg every 2 weeks and 142 for ipilimumab). The OS HRs vs. ipilimumab were 0.68 (95% CI: 0.53, 0.86; p < 0.001) for patients treated with Keytruda 10 mg/kg every 3 weeks and 0.68 (95% CI: 0.53, 0.87; p < 0.001) for patients treated with Keytruda 10 mg/kg every 2 weeks. The OS rate at 18 months and 24 months were 62% and 55% respectively for Keytruda 10 mg/kg every 3 weeks, 60% and 55% respectively for Keytruda 10 mg/kg every 2 weeks, and 47% and 43% respectively for ipilimumab. At the final analysis, a long-term PFS analysis was performed based on 566 patient events (183 for Keytruda 10 mg/kg every 3 weeks, 181 for Keytruda 10 mg/kg every 2 weeks and 202 for ipilimumab). The PFS HRs vs. ipilimumab were 0.61 (95% CI: 0.50, 0.75) for patients treated with Keytruda 10 mg/kg every 3 weeks and 0.61 (95% CI: 0.50, 0.75) for patients treated with Keytruda 10 mg/kg every 2 weeks. (See Figures 1 and 2.) The percentage of responders with an ongoing response at 18 months was 68% for Keytruda 10 mg/kg every 3 weeks, 71% for Keytruda 10 mg/kg every 2 weeks and 70% for ipilimumab.
The final analysis was performed after all patients had at least 21 months of follow-up. The final OS analysis was performed after 383 patient events (119 for Keytruda 10 mg/kg every 3 weeks, 122 for Keytruda 10 mg/kg every 2 weeks and 142 for ipilimumab). The OS HRs vs. ipilimumab were 0.68 (95% CI: 0.53, 0.86; p < 0.001) for patients treated with Keytruda 10 mg/kg every 3 weeks and 0.68 (95% CI: 0.53, 0.87; p < 0.001) for patients treated with Keytruda 10 mg/kg every 2 weeks. The OS rate at 18 months and 24 months were 62% and 55% respectively for Keytruda 10 mg/kg every 3 weeks, 60% and 55% respectively for Keytruda 10 mg/kg every 2 weeks, and 47% and 43% respectively for ipilimumab. At the final analysis, a long-term PFS analysis was performed based on 566 patient events (183 for Keytruda 10 mg/kg every 3 weeks, 181 for Keytruda 10 mg/kg every 2 weeks and 202 for ipilimumab). The PFS HRs vs. ipilimumab were 0.61 (95% CI: 0.50, 0.75) for patients treated with Keytruda 10 mg/kg every 3 weeks and 0.61 (95% CI: 0.50, 0.75) for patients treated with Keytruda 10 mg/kg every 2 weeks. (See Figures 1 and 2.) The percentage of responders with an ongoing response at 18 months was 68% for Keytruda 10 mg/kg every 3 weeks, 71% for Keytruda 10 mg/kg every 2 weeks and 70% for ipilimumab.
 At the final analysis, a long-term PFS analysis was performed based on 466 PFS events (150 for Keytruda 2 mg/kg every 3 weeks; 144 for Keytruda 10 mg/kg every 3 weeks and 172 for chemotherapy). The PFS HRs vs. chemotherapy were 0.58 (95% CI: 0.46, 0.73) for patients treated with Keytruda 2 mg/kg every 3 weeks and 0.47 (95% CI: 0.37, 0.60 for patients treated with Keytruda 10 mg/kg every 3 weeks (Figure 3).
At the final analysis, a long-term PFS analysis was performed based on 466 PFS events (150 for Keytruda 2 mg/kg every 3 weeks; 144 for Keytruda 10 mg/kg every 3 weeks and 172 for chemotherapy). The PFS HRs vs. chemotherapy were 0.58 (95% CI: 0.46, 0.73) for patients treated with Keytruda 2 mg/kg every 3 weeks and 0.47 (95% CI: 0.37, 0.60 for patients treated with Keytruda 10 mg/kg every 3 weeks (Figure 3).
 Results for patients previously treated with ipilimumab (n = 84) and naïve to treatment with ipilimumab (n = 52) who received 10 mg/kg of Keytruda every 3 weeks were similar to those seen in patients who received 2 mg/kg of Keytruda every 3 weeks.
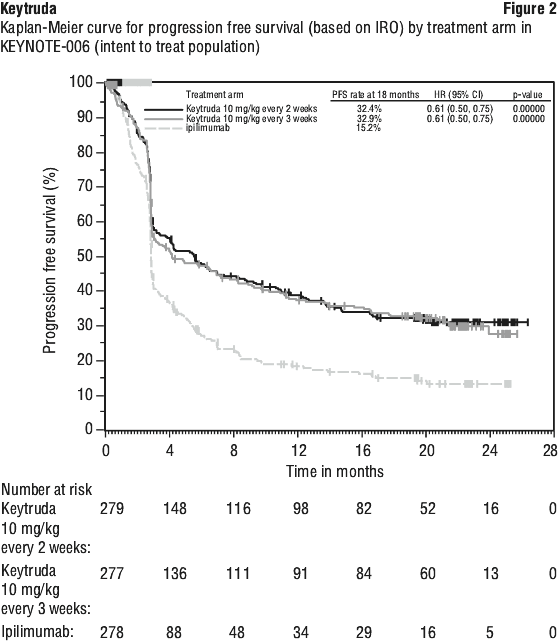
Results for patients previously treated with ipilimumab (n = 84) and naïve to treatment with ipilimumab (n = 52) who received 10 mg/kg of Keytruda every 3 weeks were similar to those seen in patients who received 2 mg/kg of Keytruda every 3 weeks. A pre-specified sensitivity analysis of RFS that included new primary melanomas was consistent with the primary RFS analysis, with an HR of 0.64 (95% CI: 0.46, 0.88).
A pre-specified sensitivity analysis of RFS that included new primary melanomas was consistent with the primary RFS analysis, with an HR of 0.64 (95% CI: 0.46, 0.88).

 For patients in the ITT population, the RFS rate at 42 months was 60% in the Keytruda arm and 41% in the placebo arm (HR was 0.59 [95% CI: 0.49, 0.70]).
For patients in the ITT population, the RFS rate at 42 months was 60% in the Keytruda arm and 41% in the placebo arm (HR was 0.59 [95% CI: 0.49, 0.70]).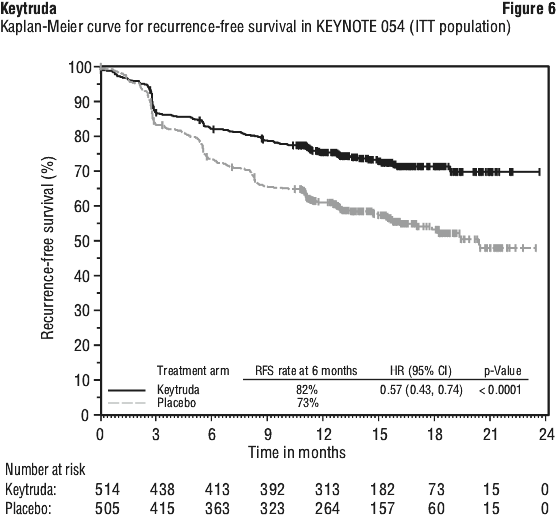
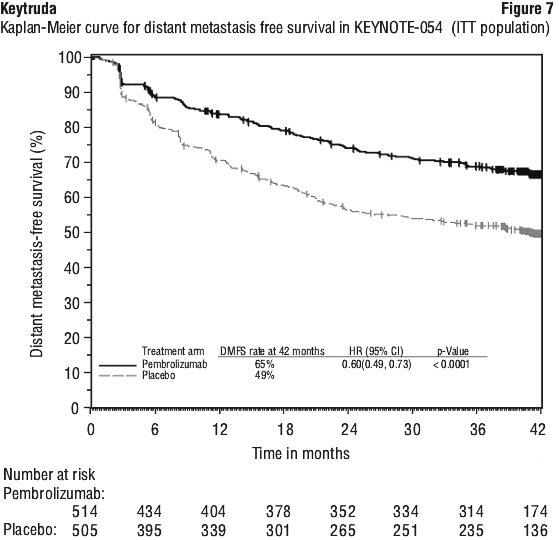
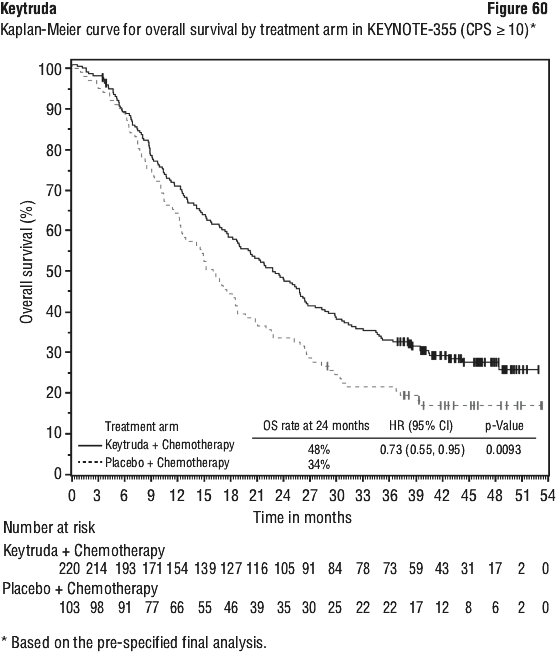
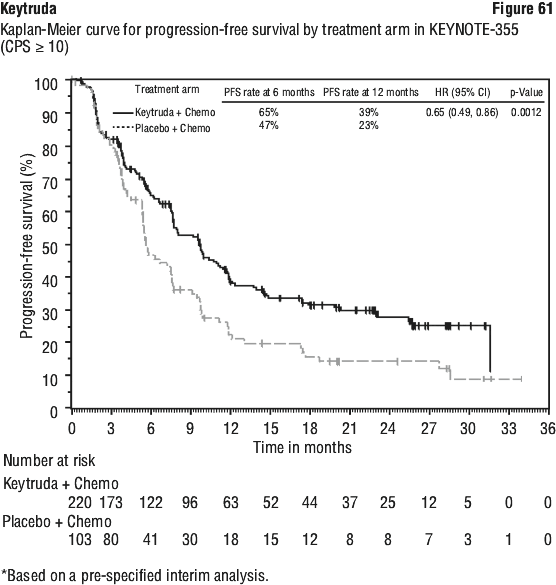
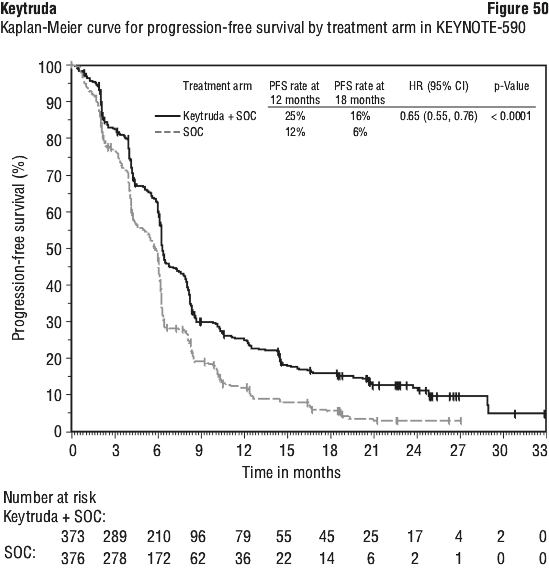
 The final OS analysis was performed at a median duration of follow-up of 18.8 months after 421 patient events (258 for Keytruda combination arm and 163 for the placebo plus chemotherapy arm). Median OS was 22.0 months (95% CI: 19.5, 24.5) for the Keytruda combination arm and 10.6 months (95% CI: 8.7, 13.6) for the placebo plus chemotherapy arm. The OS HR was 0.56 (95% CI: 0.46, 0.69; p < 0.00001). At final analysis, a PFS analysis was performed based on 534 patient events (337 for the Keytruda combination arm and 197 for the placebo plus chemotherapy arm). The median PFS was 9.0 months (95% CI: 8.1, 10.4) for the Keytruda combination arm and 4.9 months (95% CI: 4.7, 5.5) for the placebo plus chemotherapy arm. The PFS HR was 0.49 (95% CI: 0.41, 0.59, p < 0.00001). See Figure 8 and Figure 9.
The final OS analysis was performed at a median duration of follow-up of 18.8 months after 421 patient events (258 for Keytruda combination arm and 163 for the placebo plus chemotherapy arm). Median OS was 22.0 months (95% CI: 19.5, 24.5) for the Keytruda combination arm and 10.6 months (95% CI: 8.7, 13.6) for the placebo plus chemotherapy arm. The OS HR was 0.56 (95% CI: 0.46, 0.69; p < 0.00001). At final analysis, a PFS analysis was performed based on 534 patient events (337 for the Keytruda combination arm and 197 for the placebo plus chemotherapy arm). The median PFS was 9.0 months (95% CI: 8.1, 10.4) for the Keytruda combination arm and 4.9 months (95% CI: 4.7, 5.5) for the placebo plus chemotherapy arm. The PFS HR was 0.49 (95% CI: 0.41, 0.59, p < 0.00001). See Figure 8 and Figure 9.
 Patient-reported outcomes were assessed using the EORTC QLQ-C30 and EORTC QLQ-LC13. Exploratory analyses of patients receiving pembrolizumab combination therapy showed stable EORTC QLQ-C30 Global Health Status/QoL at week 12 and week 21 vs declines in patients receiving placebo plus chemotherapy. There was a trend toward a prolonged time to deterioration in the EORTC QLQ-LC13/QLQ-C30 endpoint of cough, dyspnoea or chest pain observed for patients receiving pembrolizumab combination therapy.
Patient-reported outcomes were assessed using the EORTC QLQ-C30 and EORTC QLQ-LC13. Exploratory analyses of patients receiving pembrolizumab combination therapy showed stable EORTC QLQ-C30 Global Health Status/QoL at week 12 and week 21 vs declines in patients receiving placebo plus chemotherapy. There was a trend toward a prolonged time to deterioration in the EORTC QLQ-LC13/QLQ-C30 endpoint of cough, dyspnoea or chest pain observed for patients receiving pembrolizumab combination therapy. The final OS analysis was performed at a median duration of follow-up of 14.3 months after 365 patient events (168 for Keytruda combination arm and 197 for placebo plus chemotherapy arm). Median OS was 17.1 months (95% CI: 14.4, 19.9) for the Keytruda combination arm and 11.6 months (95% CI: 10.1, 13.7) for the placebo plus chemotherapy arm. The OS HR was 0.71 (95% CI: 0.58, 0.88; p = 0.0006). At final analysis, a PFS analysis was performed based on 469 patient events (217 for the Keytruda combination arm and 252 for the placebo plus chemotherapy arm). The median PFS was 8.0 months (95% CI: 6.3, 8.4) for the Keytruda combination arm and 5.1 months (95% CI: 4.3, 6.0) for the placebo plus chemotherapy arm. The PFS HR was 0.57 (95% CI: 0.47, 0.69, p < 0.0001). See Figure 10 and Figure 11.
The final OS analysis was performed at a median duration of follow-up of 14.3 months after 365 patient events (168 for Keytruda combination arm and 197 for placebo plus chemotherapy arm). Median OS was 17.1 months (95% CI: 14.4, 19.9) for the Keytruda combination arm and 11.6 months (95% CI: 10.1, 13.7) for the placebo plus chemotherapy arm. The OS HR was 0.71 (95% CI: 0.58, 0.88; p = 0.0006). At final analysis, a PFS analysis was performed based on 469 patient events (217 for the Keytruda combination arm and 252 for the placebo plus chemotherapy arm). The median PFS was 8.0 months (95% CI: 6.3, 8.4) for the Keytruda combination arm and 5.1 months (95% CI: 4.3, 6.0) for the placebo plus chemotherapy arm. The PFS HR was 0.57 (95% CI: 0.47, 0.69, p < 0.0001). See Figure 10 and Figure 11.

 The results of all efficacy outcome measures in the subgroup of patients with PD-L1 TPS ≥ 20% NSCLC were intermediate between the results of those with PD-L1 TPS ≥ 1% and those with PD-L1 TPS ≥ 50%. In a pre-specified exploratory subgroup analysis for patients with TPS 1-49% NSCLC, the median OS was 13.4 months (95% CI: 10.7, 18.2) for the pembrolizumab group and 12.1 months (95% CI: 11.0, 14.0) in the chemotherapy group, with an HR of 0.92 (95% CI: 0.77, 1.11).
The results of all efficacy outcome measures in the subgroup of patients with PD-L1 TPS ≥ 20% NSCLC were intermediate between the results of those with PD-L1 TPS ≥ 1% and those with PD-L1 TPS ≥ 50%. In a pre-specified exploratory subgroup analysis for patients with TPS 1-49% NSCLC, the median OS was 13.4 months (95% CI: 10.7, 18.2) for the pembrolizumab group and 12.1 months (95% CI: 11.0, 14.0) in the chemotherapy group, with an HR of 0.92 (95% CI: 0.77, 1.11). In KEYNOTE-042, a higher number of deaths within 4 months of treatment initiation followed by a long-term survival benefit was observed with pembrolizumab monotherapy compared to chemotherapy (see Figure 12).
In KEYNOTE-042, a higher number of deaths within 4 months of treatment initiation followed by a long-term survival benefit was observed with pembrolizumab monotherapy compared to chemotherapy (see Figure 12). The final OS analysis was performed at a median follow-up of 25 months after 169 patient events (73 for Keytruda and 96 for chemotherapy). Median OS was 30.0 months (95% CI: 18.3, NA) for Keytruda and 14.2 months (95% CI: 9.8, 19.0) for chemotherapy. The OS HR was 0.63 (95% CI: 0.47, 0.86; p = 0.002). See Figure 14.
The final OS analysis was performed at a median follow-up of 25 months after 169 patient events (73 for Keytruda and 96 for chemotherapy). Median OS was 30.0 months (95% CI: 18.3, NA) for Keytruda and 14.2 months (95% CI: 9.8, 19.0) for chemotherapy. The OS HR was 0.63 (95% CI: 0.47, 0.86; p = 0.002). See Figure 14.
 The improved benefit as assessed by PFS, OS, ORR, and response duration for Keytruda as compared to chemotherapy in the population studied was associated with improvements in health-related quality of life (HRQoL). The change from baseline to week 15 showed a meaningful improvement in the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ) C30 global health status/QoL score for patients receiving Keytruda compared to chemotherapy (difference in LS means = 7.82; 95% CI: 2.85, 12.79; two-sided p = 0.002). The time to deterioration in the EORTC QLQ-LC13 composite endpoint of cough, dyspnoea, and chest pain was prolonged for patients receiving Keytruda compared to chemotherapy (HR = 0.66; 95% CI: 0.44, 0.97; two-sided p = 0.029), where deterioration is defined as a confirmed 10-point or greater score decrease from baseline in any one of these three symptoms.
The improved benefit as assessed by PFS, OS, ORR, and response duration for Keytruda as compared to chemotherapy in the population studied was associated with improvements in health-related quality of life (HRQoL). The change from baseline to week 15 showed a meaningful improvement in the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire (EORTC QLQ) C30 global health status/QoL score for patients receiving Keytruda compared to chemotherapy (difference in LS means = 7.82; 95% CI: 2.85, 12.79; two-sided p = 0.002). The time to deterioration in the EORTC QLQ-LC13 composite endpoint of cough, dyspnoea, and chest pain was prolonged for patients receiving Keytruda compared to chemotherapy (HR = 0.66; 95% CI: 0.44, 0.97; two-sided p = 0.029), where deterioration is defined as a confirmed 10-point or greater score decrease from baseline in any one of these three symptoms.

 Efficacy results were similar for the 2 mg/kg and 10 mg/kg Keytruda arms. Efficacy results for OS were consistent regardless of the age of tumour specimen (new versus archival).
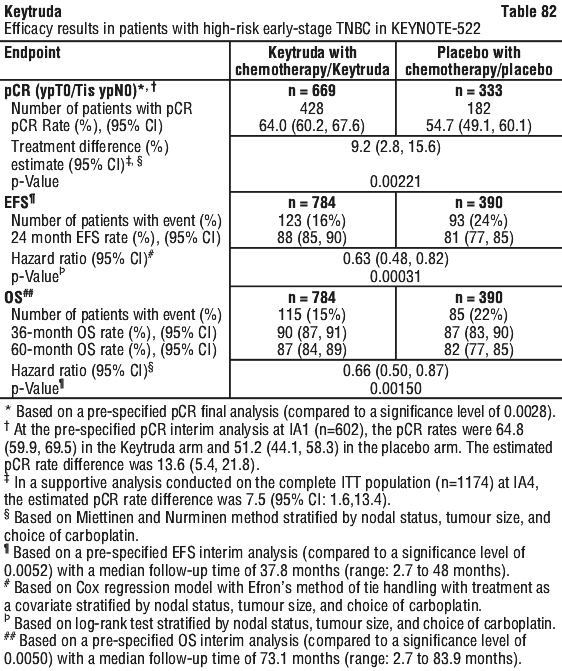
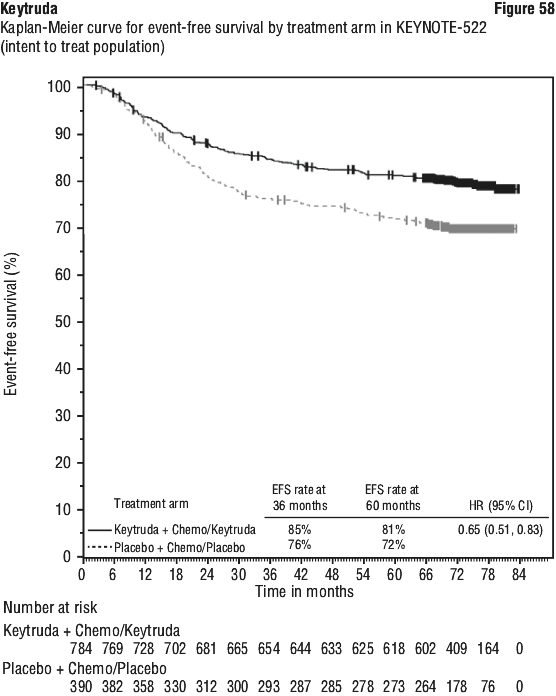
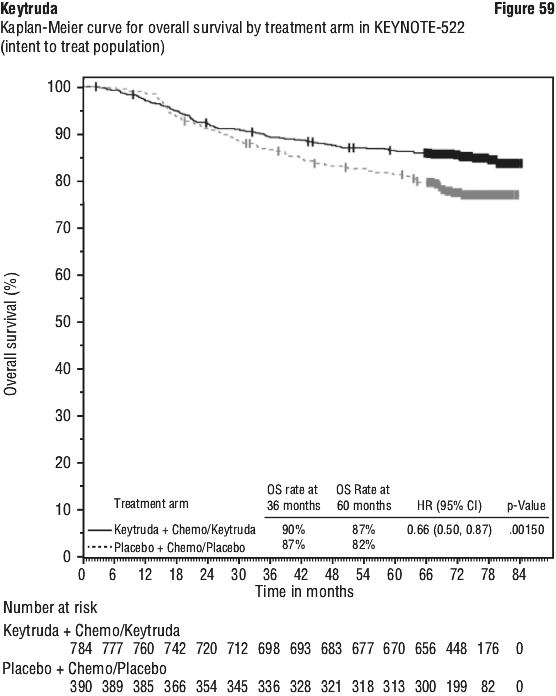
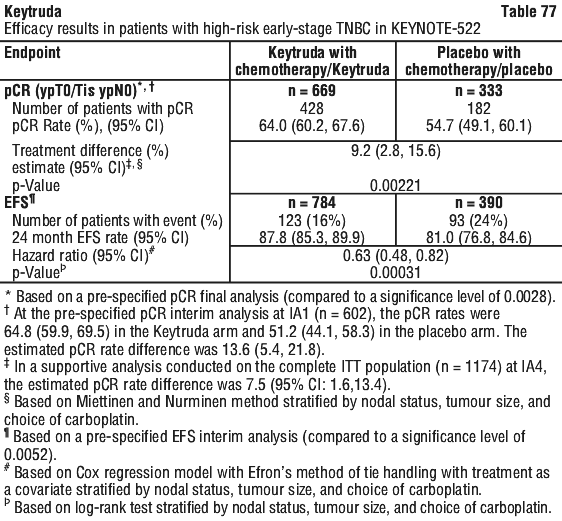
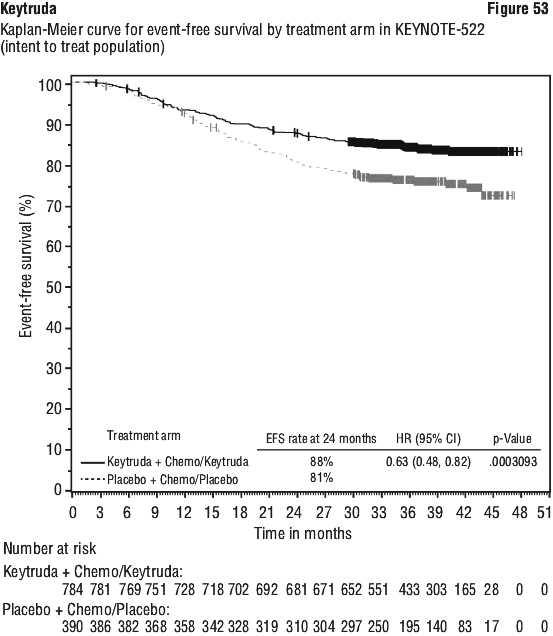
Efficacy results were similar for the 2 mg/kg and 10 mg/kg Keytruda arms. Efficacy results for OS were consistent regardless of the age of tumour specimen (new versus archival). A significant difference in EFS was demonstrated at interim analysis 1 (IA1). EFS analysis was repeated at IA2; after 422 patient events (174 for the Keytruda arm and 248 for the placebo arm). Median EFS at IA2 was 47.2 months (95% CI: 32.9, NR) for the Keytruda arm and 18.3 months (95% CI: 14.8, 22.1) for the placebo arm. The EFS HR at IA2 was 0.59 (95% CI: 0.48, 0.72). The EFS Kaplan-Meier curve for IA2 is presented in Figure 18 rather than the primary EFS analysis.
A significant difference in EFS was demonstrated at interim analysis 1 (IA1). EFS analysis was repeated at IA2; after 422 patient events (174 for the Keytruda arm and 248 for the placebo arm). Median EFS at IA2 was 47.2 months (95% CI: 32.9, NR) for the Keytruda arm and 18.3 months (95% CI: 14.8, 22.1) for the placebo arm. The EFS HR at IA2 was 0.59 (95% CI: 0.48, 0.72). The EFS Kaplan-Meier curve for IA2 is presented in Figure 18 rather than the primary EFS analysis.




 Figure 21 and Figure 22 show the Kaplan-Meier curves for OS for the epithelioid and non-epithelioid histological subgroups based on a pre-specified exploratory analysis. Due to its exploratory nature, the subgroup analysis by histology should be interpreted with caution.
Figure 21 and Figure 22 show the Kaplan-Meier curves for OS for the epithelioid and non-epithelioid histological subgroups based on a pre-specified exploratory analysis. Due to its exploratory nature, the subgroup analysis by histology should be interpreted with caution.



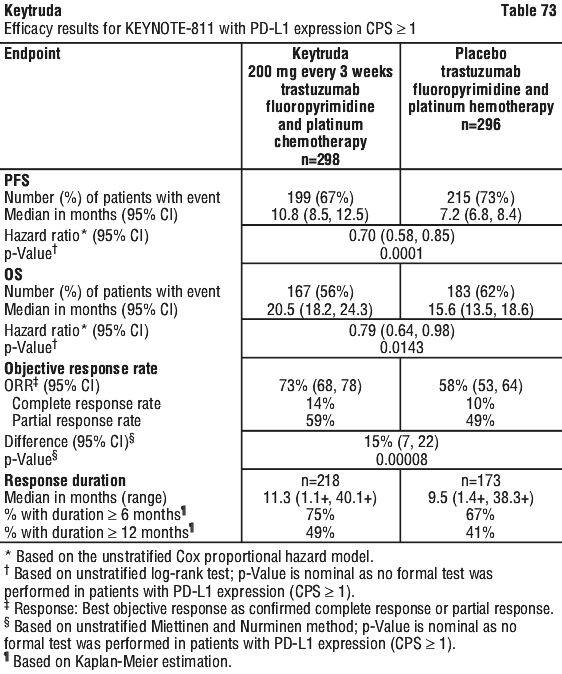
 While OS results were not mature at this interim analysis, with 76% of pre-specified OS events in the CPS ≥ 1 population, no trend towards a detriment was observed.
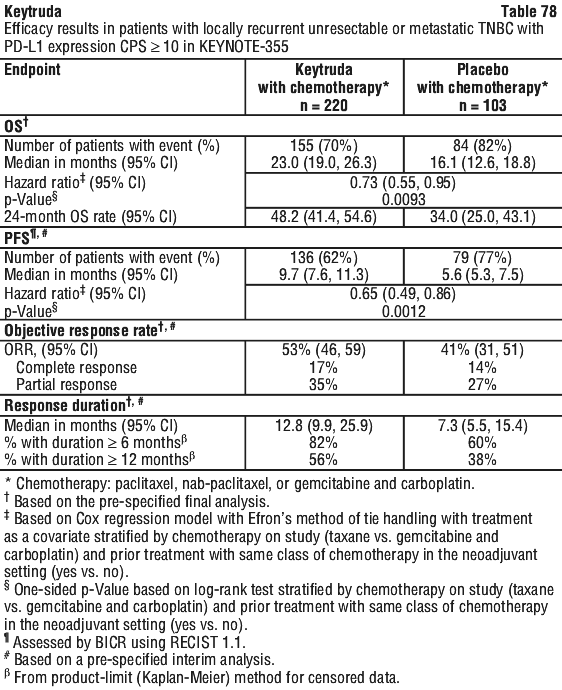
While OS results were not mature at this interim analysis, with 76% of pre-specified OS events in the CPS ≥ 1 population, no trend towards a detriment was observed. In KEYNOTE-048, OS HRs for patients randomised to Keytruda in combination with chemotherapy, compared with cetuximab in combination with chemotherapy, were similar for all populations regardless of PD-L1 expression in a pre-specified interim analysis: ITT (HR 0.77, 95% CI: 0.63, 0.93), CPS ≥ 1 (HR 0.71, 95% CI: 0.57, 0.88), CPS ≥ 20 (HR 0.69, 95% CI: 0.51, 0.94). A positive association was observed between increasing PD-L1 expression and treatment benefit. The trial also demonstrated a statistically significant improvement in OS for the patients expressing PD-L1 CPS ≥ 1 and CPS ≥ 20. The OS HRs at final analysis were CPS ≥ 1 (0.65, 95% CI: 0.53, 0.80), CPS ≥ 20 (0.60, 95% CI: 0.45, 0.82).
In KEYNOTE-048, OS HRs for patients randomised to Keytruda in combination with chemotherapy, compared with cetuximab in combination with chemotherapy, were similar for all populations regardless of PD-L1 expression in a pre-specified interim analysis: ITT (HR 0.77, 95% CI: 0.63, 0.93), CPS ≥ 1 (HR 0.71, 95% CI: 0.57, 0.88), CPS ≥ 20 (HR 0.69, 95% CI: 0.51, 0.94). A positive association was observed between increasing PD-L1 expression and treatment benefit. The trial also demonstrated a statistically significant improvement in OS for the patients expressing PD-L1 CPS ≥ 1 and CPS ≥ 20. The OS HRs at final analysis were CPS ≥ 1 (0.65, 95% CI: 0.53, 0.80), CPS ≥ 20 (0.60, 95% CI: 0.45, 0.82).

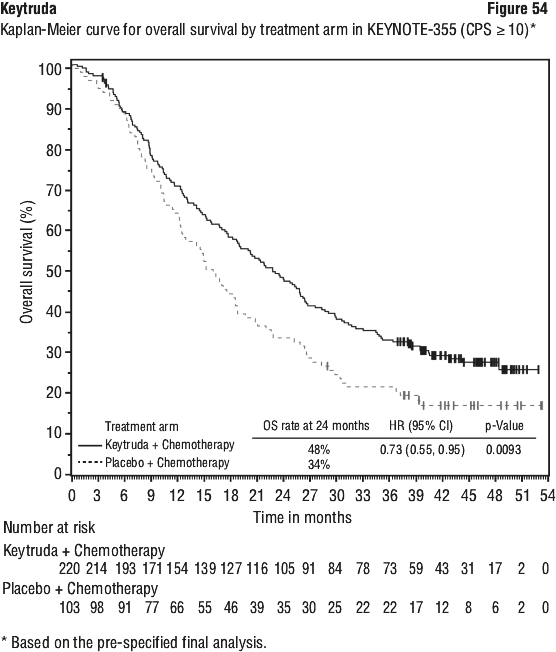
 Additional OS analyses based on PD-L1 expression (CPS ≥ 1 and CPS ≥ 20) were performed in KEYNOTE-048. The trial demonstrated a statistically significant improvement in OS at the protocol-specified interim analysis for patients randomised to Keytruda monotherapy compared to standard treatment for PD-L1 expression CPS ≥ 1 and CPS ≥ 20. OS for patients who had PD-L1 CPS ≥ 1 or CPS ≥ 20 for Keytruda monotherapy compared to standard treatment is summarised in Table 54.
Additional OS analyses based on PD-L1 expression (CPS ≥ 1 and CPS ≥ 20) were performed in KEYNOTE-048. The trial demonstrated a statistically significant improvement in OS at the protocol-specified interim analysis for patients randomised to Keytruda monotherapy compared to standard treatment for PD-L1 expression CPS ≥ 1 and CPS ≥ 20. OS for patients who had PD-L1 CPS ≥ 1 or CPS ≥ 20 for Keytruda monotherapy compared to standard treatment is summarised in Table 54.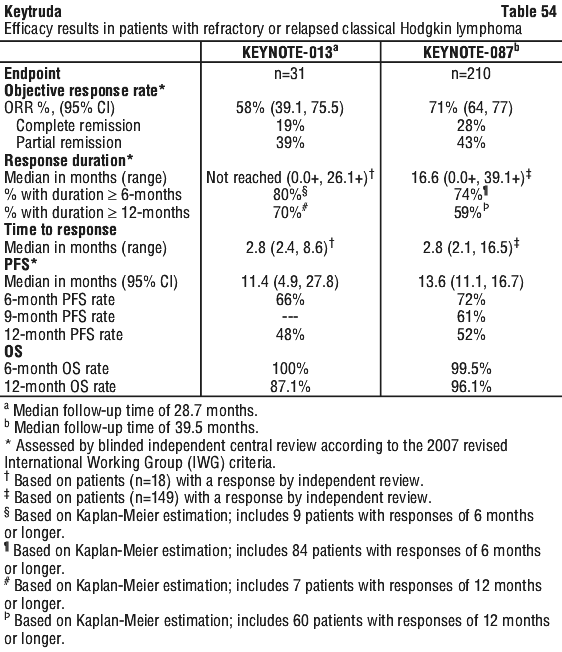



 Patient-reported outcomes (PROs) were assessed using EORTC QLQ-C30. A prolonged time to deterioration in EORTC QLQ C30 global health status/QoL was observed for patients treated with pembrolizumab compared to BV (HR 0.40; 95% CI: 0.22-0.74). Over 24 weeks of follow-up, patients treated with pembrolizumab had an improvement in global health status/QoL compared to BV which showed a decline (difference in Least Square (LS) means = 8.60; 95% CI: 3.89, 13.31; nominal two-sided p = 0.0004). These results should be interpreted in the context of the open-label study design and therefore taken cautiously.
Patient-reported outcomes (PROs) were assessed using EORTC QLQ-C30. A prolonged time to deterioration in EORTC QLQ C30 global health status/QoL was observed for patients treated with pembrolizumab compared to BV (HR 0.40; 95% CI: 0.22-0.74). Over 24 weeks of follow-up, patients treated with pembrolizumab had an improvement in global health status/QoL compared to BV which showed a decline (difference in Least Square (LS) means = 8.60; 95% CI: 3.89, 13.31; nominal two-sided p = 0.0004). These results should be interpreted in the context of the open-label study design and therefore taken cautiously.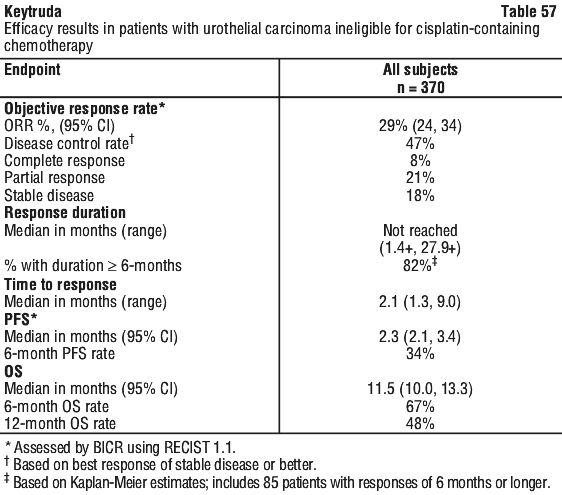 The improved benefit as assessed by ORR, CRR, and response duration in the KEYNOTE-087 population was accompanied by overall improvements in health-related quality of life (HRQoL) as assessed using the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) and the European Quality of Life Five Dimensions Questionnaire (EQ-5D). Relative to subjects with stable disease or progressive disease, subjects with a complete or partial response had the largest improvement and the highest proportion with a 10 point or greater increase in their EORTC QLQ-C30 global health status/QoL score, as well as, had the largest improvement in their EQ-5D utility and VAS scores from baseline to week 12.
The improved benefit as assessed by ORR, CRR, and response duration in the KEYNOTE-087 population was accompanied by overall improvements in health-related quality of life (HRQoL) as assessed using the European Organization for Research and Treatment of Cancer Quality of Life Questionnaire Core 30 (EORTC QLQ-C30) and the European Quality of Life Five Dimensions Questionnaire (EQ-5D). Relative to subjects with stable disease or progressive disease, subjects with a complete or partial response had the largest improvement and the highest proportion with a 10 point or greater increase in their EORTC QLQ-C30 global health status/QoL score, as well as, had the largest improvement in their EQ-5D utility and VAS scores from baseline to week 12.



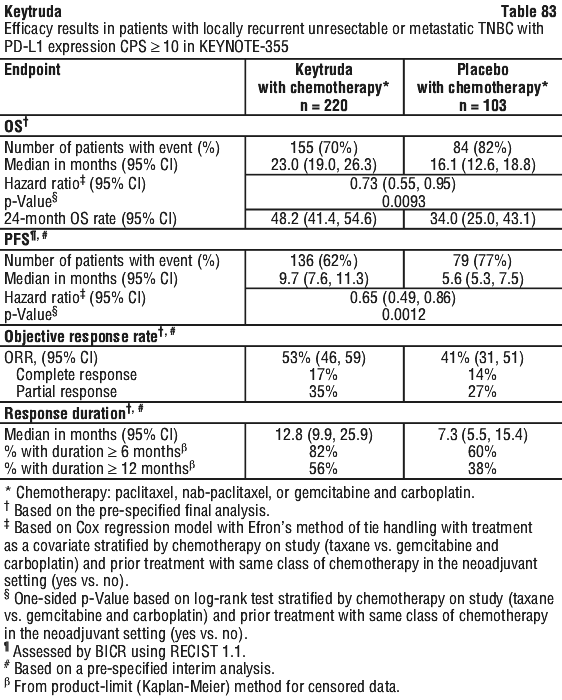
 The final ORR analysis was performed 9.9 months after the interim analysis with 106 ORR events for all patients [median follow-up of 11.4 months (range: 0.1, 41.2 months)]. ORR was 29% (95% CI: 24, 34) and 47% (95% CI: 38, 57), respectively, for all subjects and subjects with CPS ≥ 10. The complete and partial response rates were 9% and 20%, respectively, in all subjects and 20% and 27%, respectively, in subjects with CPS ≥ 10. At the final analysis among the responding patients, the median response duration was 30.1 months (range 1.4+ to 35.9+ months) in all subjects (n = 106) and not reached (range 1.4+ to 35.4+ months) in subjects with CPS ≥ 10 (n = 52). Responses of 6 months or longer (based on Kaplan-Meier estimation) were 81% and 82%, respectively for all subjects and subjects with CPS ≥ 10.
The final ORR analysis was performed 9.9 months after the interim analysis with 106 ORR events for all patients [median follow-up of 11.4 months (range: 0.1, 41.2 months)]. ORR was 29% (95% CI: 24, 34) and 47% (95% CI: 38, 57), respectively, for all subjects and subjects with CPS ≥ 10. The complete and partial response rates were 9% and 20%, respectively, in all subjects and 20% and 27%, respectively, in subjects with CPS ≥ 10. At the final analysis among the responding patients, the median response duration was 30.1 months (range 1.4+ to 35.9+ months) in all subjects (n = 106) and not reached (range 1.4+ to 35.4+ months) in subjects with CPS ≥ 10 (n = 52). Responses of 6 months or longer (based on Kaplan-Meier estimation) were 81% and 82%, respectively for all subjects and subjects with CPS ≥ 10. At the interim analysis, median duration of response was not reached in the Keytruda arm (range 1.6+ to 15.6+ months) and was 4.3 months (range: 1.4+ to 15.4+ months) in the chemotherapy arm. At the time of the analysis, responses were ongoing in 41 and 14 patients at 6 and 12 months, respectively, in the Keytruda arm, and 7 and 3 patients at 6 and 12 months, respectively, in the chemotherapy arm.
At the interim analysis, median duration of response was not reached in the Keytruda arm (range 1.6+ to 15.6+ months) and was 4.3 months (range: 1.4+ to 15.4+ months) in the chemotherapy arm. At the time of the analysis, responses were ongoing in 41 and 14 patients at 6 and 12 months, respectively, in the Keytruda arm, and 7 and 3 patients at 6 and 12 months, respectively, in the chemotherapy arm. Patient-reported outcomes (PROs) were assessed using EORTC QLQ-C30. A prolonged time to deterioration in EORTC QLQ-C30 global health status/QoL was observed for patients treated with pembrolizumab compared to investigator's choice chemotherapy (HR 0.70; 95% CI 0.55-0.90). Over 15 weeks of follow-up, patients treated with pembrolizumab had stable global health status/QoL, while those treated with investigator's choice chemotherapy had a decline in global health status/QoL. These results should be interpreted in the context of the open-label study design and therefore taken cautiously.
Patient-reported outcomes (PROs) were assessed using EORTC QLQ-C30. A prolonged time to deterioration in EORTC QLQ-C30 global health status/QoL was observed for patients treated with pembrolizumab compared to investigator's choice chemotherapy (HR 0.70; 95% CI 0.55-0.90). Over 15 weeks of follow-up, patients treated with pembrolizumab had stable global health status/QoL, while those treated with investigator's choice chemotherapy had a decline in global health status/QoL. These results should be interpreted in the context of the open-label study design and therefore taken cautiously.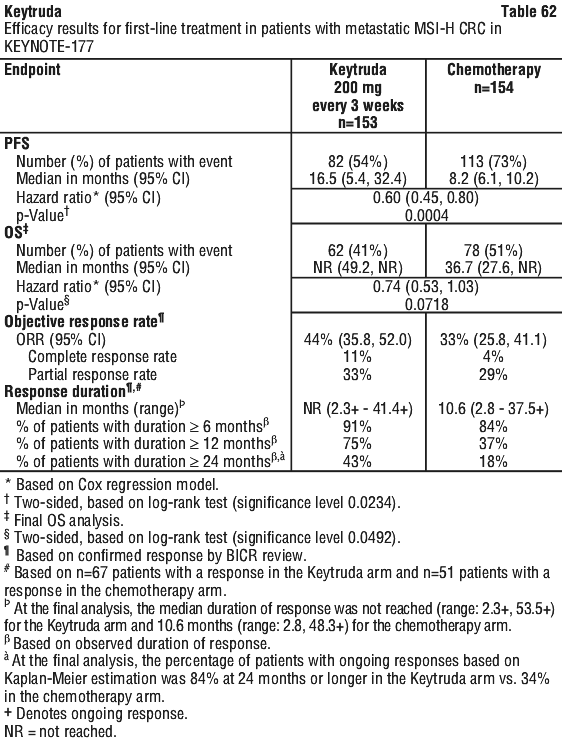 Results remained similar at a final analysis, when the median follow-up time was 69 months. Out of the 96 patients, 44 eventually underwent radical cystectomy at various times after discontinuation of Keytruda (range: 1.4 to 46.5 months). The rate of pathological upstaging to muscle invasive disease or non-organ confined disease at the time of radical cystectomy was 14% (n=6).
Results remained similar at a final analysis, when the median follow-up time was 69 months. Out of the 96 patients, 44 eventually underwent radical cystectomy at various times after discontinuation of Keytruda (range: 1.4 to 46.5 months). The rate of pathological upstaging to muscle invasive disease or non-organ confined disease at the time of radical cystectomy was 14% (n=6).
 Among the 7 paediatric patients from KEYNOTE-051 (6 with brain cancer, 1 with abdominal adenocarcinoma), 1 patient with brain cancer had a response per irRECIST.
Among the 7 paediatric patients from KEYNOTE-051 (6 with brain cancer, 1 with abdominal adenocarcinoma), 1 patient with brain cancer had a response per irRECIST. At the final analysis, the PFS and ORR results were consistent with the primary analysis.
At the final analysis, the PFS and ORR results were consistent with the primary analysis.


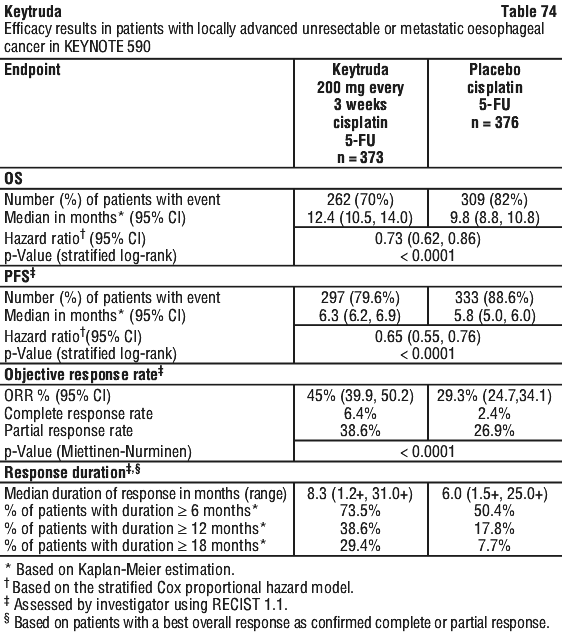
 There was no statistically significant improvement in progression-free survival, objective response rate or duration of response in patients receiving pembrolizumab plus gemcitabine and cisplatin or placebo plus gemcitabine and cisplatin.
There was no statistically significant improvement in progression-free survival, objective response rate or duration of response in patients receiving pembrolizumab plus gemcitabine and cisplatin or placebo plus gemcitabine and cisplatin.


 The protocol-specified final OS analysis with approximately 16 months of additional follow-up duration from the interim analysis (overall median follow-up time of 14.7 months [range: 0.3 to 43.0 months]) was performed without statistical inferential testing, which was not required after the success criteria for study hypotheses was met at the pre-specified interim analysis for OS. The median OS for Keytruda in combination with lenvatinib was 18.0 months (95% CI: 14.9, 20.5) compared to 12.2 months (95% CI: 11.0, 14.1) for doxorubicin or paclitaxel, with an HR of 0.70 (95% CI: 0.58, 0.83). At the time of the protocol-specified final OS analysis, the median PFS for Keytruda in combination with lenvatinib was 6.7 months (95% CI: 5.6, 7.4) compared to 3.8 months (95% CI: 3.6, 5.0) for doxorubicin or paclitaxel, with an HR of 0.60 (95% CI: 0.50, 0.72).
The protocol-specified final OS analysis with approximately 16 months of additional follow-up duration from the interim analysis (overall median follow-up time of 14.7 months [range: 0.3 to 43.0 months]) was performed without statistical inferential testing, which was not required after the success criteria for study hypotheses was met at the pre-specified interim analysis for OS. The median OS for Keytruda in combination with lenvatinib was 18.0 months (95% CI: 14.9, 20.5) compared to 12.2 months (95% CI: 11.0, 14.1) for doxorubicin or paclitaxel, with an HR of 0.70 (95% CI: 0.58, 0.83). At the time of the protocol-specified final OS analysis, the median PFS for Keytruda in combination with lenvatinib was 6.7 months (95% CI: 5.6, 7.4) compared to 3.8 months (95% CI: 3.6, 5.0) for doxorubicin or paclitaxel, with an HR of 0.60 (95% CI: 0.50, 0.72).
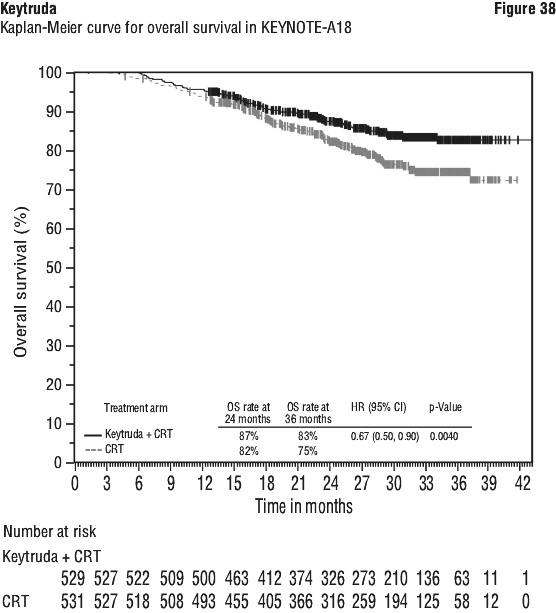


 At IA2, PFS and OS hazard ratios (HR; with 95% CI) by stage stratum were as follows:
At IA2, PFS and OS hazard ratios (HR; with 95% CI) by stage stratum were as follows: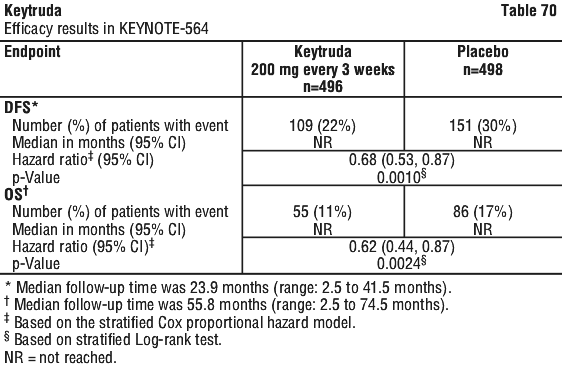

 The protocol-specified final OS analysis was performed at a median duration of follow-up of 37.7 months after 418 patient events (193 in the Keytruda and axitinib arm and 225 in the sunitinib arm). Median OS was 45.7 months (95% CI: 43.6, NA) in the Keytruda and axitinib arm and 40.1 months (95% CI: 34.3, 44.2) in the sunitinib arm. The OS HR was 0.73 (95% CI: 0.60, 0.88). The 12-month OS rates were 90% (95% CI: 86, 92) in the Keytruda and axitinib arm and 79% (95% CI: 75, 83) in the sunitinib arm. The 24-month OS rates were 74% (95% CI: 70, 78) in the Keytruda and axitinib arm and 66% (95% CI: 61, 70) in the sunitinib arm. The 36-month OS rates were 63% (95% CI: 58, 67) in the Keytruda and axitinib arm and 54% (95% CI: 49, 58) in the sunitinib arm. The 42-month OS rates were 58% (95% CI: 53, 62) in the Keytruda and axitinib arm and 49% (95% CI: 44, 53) in the sunitinib arm. At final analysis, a PFS analysis was performed based on 587 patient events (286 in the Keytruda and axitinib arm and 301 in the sunitinib arm). The median PFS was 15.7 months (95% CI: 13.6, 20.2) in the Keytruda and axitinib arm and 11.1 months (95% CI: 8.9, 12.5) in the sunitinib arm. The PFS HR was 0.68 (95% CI: 0.58, 0.80).
The protocol-specified final OS analysis was performed at a median duration of follow-up of 37.7 months after 418 patient events (193 in the Keytruda and axitinib arm and 225 in the sunitinib arm). Median OS was 45.7 months (95% CI: 43.6, NA) in the Keytruda and axitinib arm and 40.1 months (95% CI: 34.3, 44.2) in the sunitinib arm. The OS HR was 0.73 (95% CI: 0.60, 0.88). The 12-month OS rates were 90% (95% CI: 86, 92) in the Keytruda and axitinib arm and 79% (95% CI: 75, 83) in the sunitinib arm. The 24-month OS rates were 74% (95% CI: 70, 78) in the Keytruda and axitinib arm and 66% (95% CI: 61, 70) in the sunitinib arm. The 36-month OS rates were 63% (95% CI: 58, 67) in the Keytruda and axitinib arm and 54% (95% CI: 49, 58) in the sunitinib arm. The 42-month OS rates were 58% (95% CI: 53, 62) in the Keytruda and axitinib arm and 49% (95% CI: 44, 53) in the sunitinib arm. At final analysis, a PFS analysis was performed based on 587 patient events (286 in the Keytruda and axitinib arm and 301 in the sunitinib arm). The median PFS was 15.7 months (95% CI: 13.6, 20.2) in the Keytruda and axitinib arm and 11.1 months (95% CI: 8.9, 12.5) in the sunitinib arm. The PFS HR was 0.68 (95% CI: 0.58, 0.80).

 The final OS analysis was conducted at an updated data cut-off date of 31 July 2022 (median OS follow-up 49.4 months). The median duration of treatment was 22.6 months (range: 2 days - 62.1 months) in the Keytruda in combination with lenvatinib arm and 7.8 months (range: 3 days - 57.5 months) in the sunitinib arm. Median OS for Keytruda in combination with lenvatinib was 53.7 months (95% CI: 48.7, NE) compared to 54.3 months (95% CI: 40.9, NE) for sunitinib, with a HR 0.79 (95% CI: 0.63, 0.99). See Figure 45. Subsequent to their participation in KEYNOTE-581, 55% of patients in the sunitinib arm and 16% of patients in the Keytruda plus lenvatinib arm received subsequent systemic anti-PD-1/PD-L1 therapy. OS analyses were not adjusted to account for subsequent therapies.
The final OS analysis was conducted at an updated data cut-off date of 31 July 2022 (median OS follow-up 49.4 months). The median duration of treatment was 22.6 months (range: 2 days - 62.1 months) in the Keytruda in combination with lenvatinib arm and 7.8 months (range: 3 days - 57.5 months) in the sunitinib arm. Median OS for Keytruda in combination with lenvatinib was 53.7 months (95% CI: 48.7, NE) compared to 54.3 months (95% CI: 40.9, NE) for sunitinib, with a HR 0.79 (95% CI: 0.63, 0.99). See Figure 45. Subsequent to their participation in KEYNOTE-581, 55% of patients in the sunitinib arm and 16% of patients in the Keytruda plus lenvatinib arm received subsequent systemic anti-PD-1/PD-L1 therapy. OS analyses were not adjusted to account for subsequent therapies.


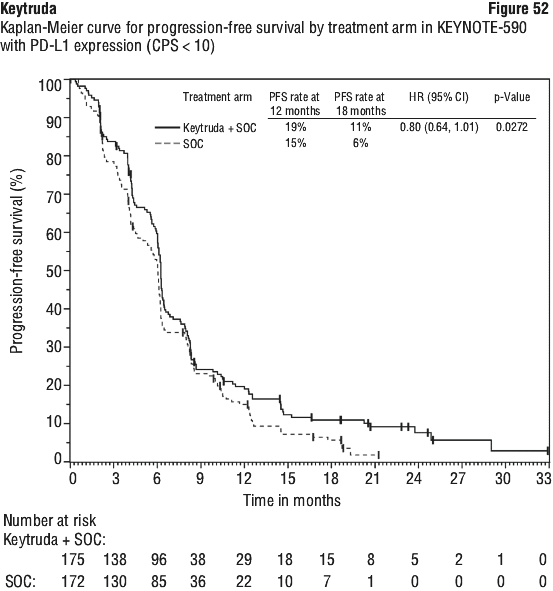
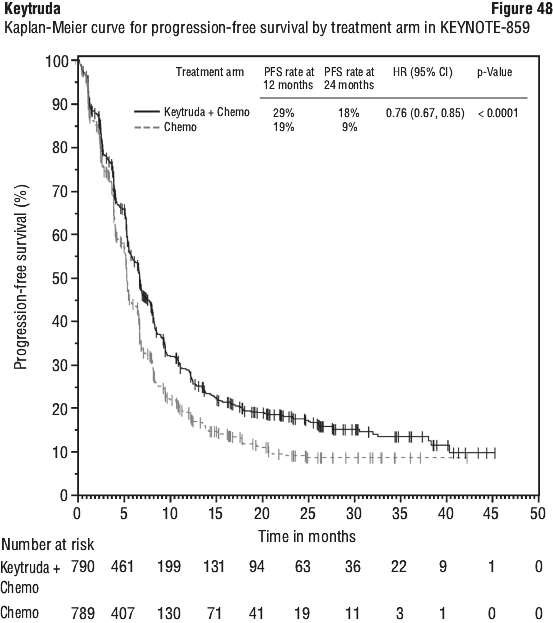 In an exploratory subgroup analysis in patients with PD-L1 CPS < 1 (n=344): The median OS was 12.7 months (95% CI: 11.4, 15.0) for the Keytruda arm and 12.2 months (95% CI: 9.5, 14.0) for the placebo arm, with a HR of 0.92 (95% CI: 0.73, 1.17); The median PFS was 7.2 months (95% CI: 6.0, 8.5) for the Keytruda arm and 5.8 months (95% CI: 5.4, 6.9) for the placebo arm, with a HR of 0.90 (95% CI: 0.70, 1.15). See Figure 49.
In an exploratory subgroup analysis in patients with PD-L1 CPS < 1 (n=344): The median OS was 12.7 months (95% CI: 11.4, 15.0) for the Keytruda arm and 12.2 months (95% CI: 9.5, 14.0) for the placebo arm, with a HR of 0.92 (95% CI: 0.73, 1.17); The median PFS was 7.2 months (95% CI: 6.0, 8.5) for the Keytruda arm and 5.8 months (95% CI: 5.4, 6.9) for the placebo arm, with a HR of 0.90 (95% CI: 0.70, 1.15). See Figure 49.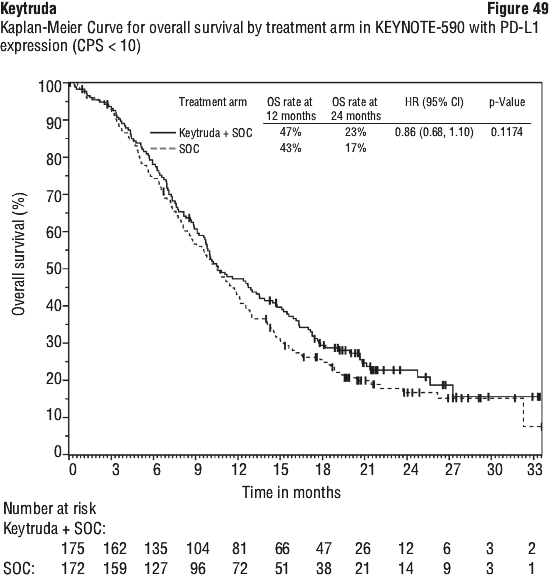
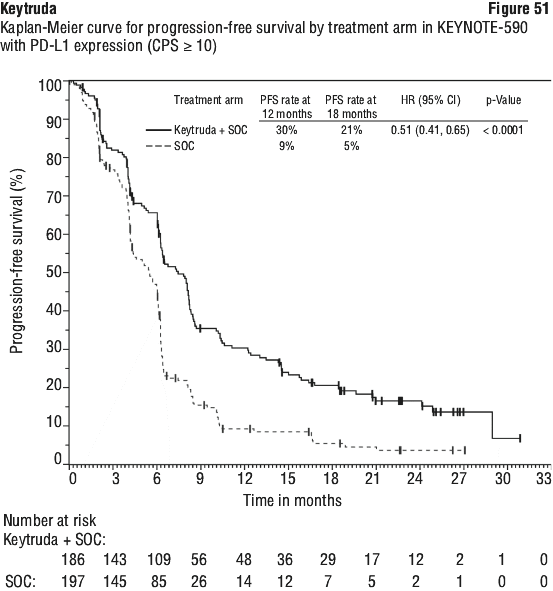
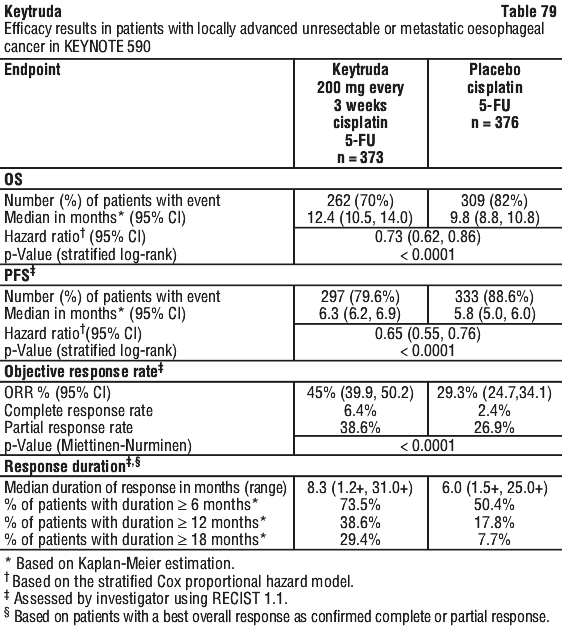
 In a pre-specified formal test of OS in patients with PD-L1 CPS ≥ 10 (n = 383), the median was 13.5 months (95% CI: 11.1, 15.6) for the Keytruda arm and 9.4 months (95% CI: 8.0, 10.7) for the placebo arm, with a HR of 0.62 (95% CI: 0.49, 0.78; p-Value < 0.0001). In an exploratory analysis, in patients with PD-L1 CPS < 10 (n = 347), the median OS was 10.5 months (95% CI: 9.7, 13.5) for the Keytruda arm and 10.6 months (95% CI: 8.8, 12.0) for the placebo arm, with a HR of 0.86 (95% CI: 0.68, 1.10).
In a pre-specified formal test of OS in patients with PD-L1 CPS ≥ 10 (n = 383), the median was 13.5 months (95% CI: 11.1, 15.6) for the Keytruda arm and 9.4 months (95% CI: 8.0, 10.7) for the placebo arm, with a HR of 0.62 (95% CI: 0.49, 0.78; p-Value < 0.0001). In an exploratory analysis, in patients with PD-L1 CPS < 10 (n = 347), the median OS was 10.5 months (95% CI: 9.7, 13.5) for the Keytruda arm and 10.6 months (95% CI: 8.8, 12.0) for the placebo arm, with a HR of 0.86 (95% CI: 0.68, 1.10).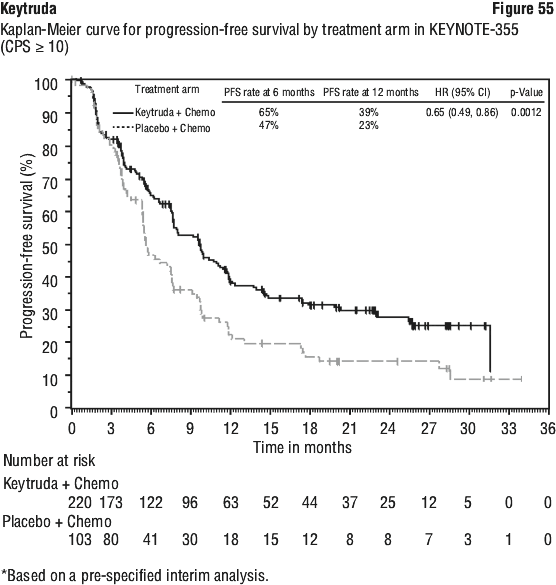
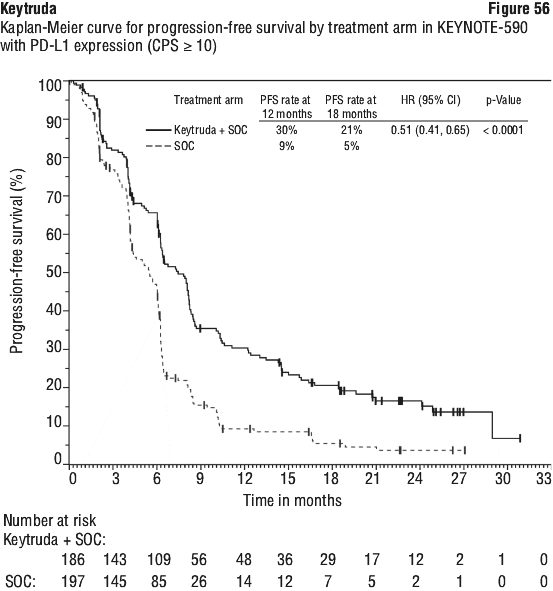
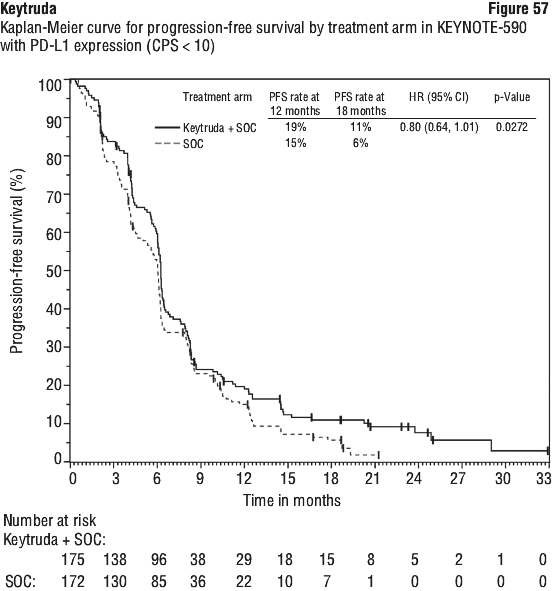
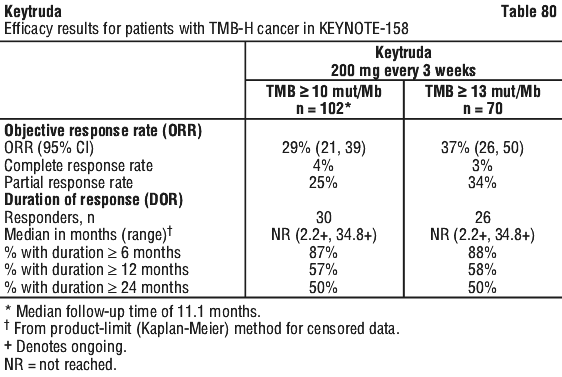
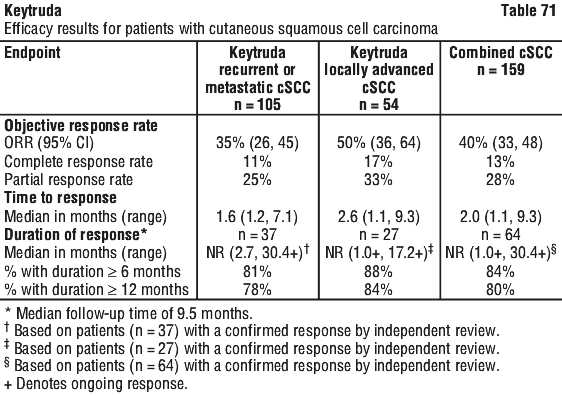
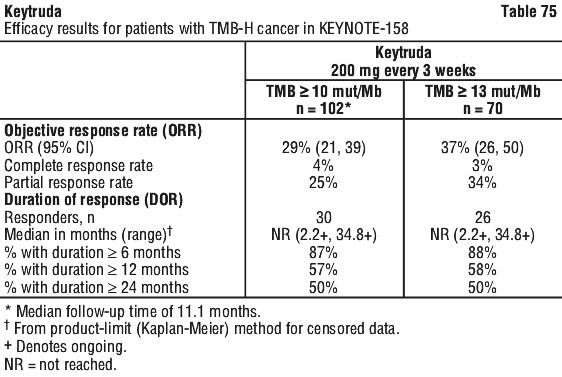
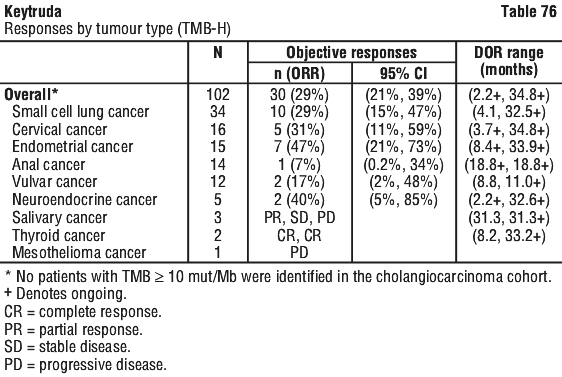
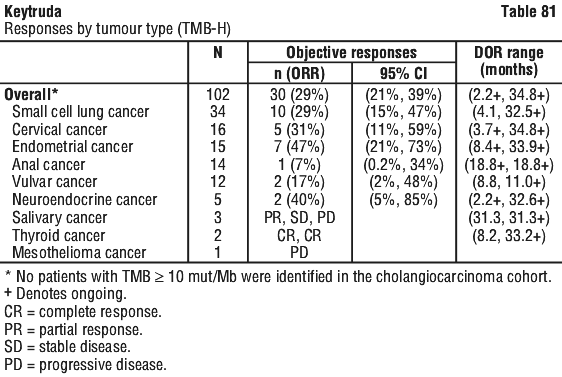 In an exploratory analysis in 32 patients enrolled in KEYNOTE-158 whose cancer had TMB ≥ 10 mut/Mb and < 13 mut/Mb, the ORR was 13% (95% CI: 4%, 29%), including two complete responses and two partial responses.
In an exploratory analysis in 32 patients enrolled in KEYNOTE-158 whose cancer had TMB ≥ 10 mut/Mb and < 13 mut/Mb, the ORR was 13% (95% CI: 4%, 29%), including two complete responses and two partial responses.