What is in this leaflet
This leaflet answers some common questions about KYTRIL tablets. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking KYTRIL against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What KYTRIL is used for
KYTRIL contains the active ingredient granisetron.
It belongs to a group of medicines called anti-emetics.
KYTRIL is used to stop you feeling sick (nauseous) or being sick (vomiting). It is especially useful when you need to have medical treatment that may cause you to feel or be sick, for example, chemotherapy.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
It is available only with a doctor's prescription.
Before you take KYTRIL
When you must not take it
Do not take KYTRIL if you have an allergy to:
- any medicine containing granisetron
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or breathing difficulty; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
Do not take KYTRIL after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if:
- you have allergies to any other medicines, foods, preservatives or dyes.
- you have had an allergic reaction with other medicines used to prevent or treat nausea and vomiting, such as ondansetron and tropisetron. You may have an allergic reaction to KYTRIL as well.
- you have severe constipation.
- you are pregnant or plan to become pregnant or are breast-feeding.
Your doctor can discuss with you the risks and benefits of taking KYTRIL if you are pregnant or breast-feeding. - you have used KYTRIL before, and you became unwell.
You may need to be given another medicine instead. - you have a heart condition related to changes in the rhythm or rate of your heart beat.
- you are intolerant to lactose or galactose or your body does not absorb glucose or galactose well.
If you have not told your doctor about any of the above, tell him/her before you start taking KYTRIL.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and KYTRIL may interfere with each other. These include:
- phenobarbitone, a medicine used to treat epilepsy.
Phenobarbitone may be affected by KYTRIL or may affect how well KYTRIL works. You may need different amounts of your medicine, or you may need to take a different medicine. - medicines that can affect the serotonin levels in your body. These may include: some antibiotics, medicines used to treat depression, medicines that treat or prevent pain, some medicines to treat Parkinson's disease, medicines to treat obesity; and medicines used to treat attention hyperactivity disorder.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking KYTRIL.
How to take KYTRIL
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
How much to take
Your doctor or pharmacist will tell you how many KYTRIL tablets to take each day.
Chemotherapy Patients
For the prevention of nausea and vomiting associated with chemotherapy, the usual adult dose is one 2 mg tablet once a day, taken within 1 hour before the start of chemotherapy, and then daily for up to one week following chemotherapy.
Radiotherapy Patients
For the prevention of nausea and vomiting associated with radiotherapy, the usual adult dose of KYTRIL is one 2 mg tablet taken within 1 hour before the start of radiotherapy.
How to take it
Swallow the tablets whole with a full glass of water. Do not crush or chew the tablets.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take this medicine before or after food.
How long to take it
Continue taking KYTRIL tablets for as long as your doctor tells you.
Your doctor will tell you when your treatment should be stopped.
If you forget to take it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
If you think that you or anyone else may have taken too much KYTRIL, immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Keep telephone numbers for these places handy.
If you are not sure what to do, contact your doctor or pharmacist.
While you are taking KYTRIL
Things you must do
If you are about to start a new medicine, remind your doctor and pharmacist that you are taking KYTRIL.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking KYTRIL.
Tell your doctor immediately if you become pregnant while taking this medicine.
Tell your doctor if, for any reason, you have not taken your medicine exactly as prescribed. Otherwise, your doctor may think that it was not effective and change your treatment unnecessarily.
Tell your doctor if you feel the tablets are not helping your condition.
Keep all of your doctor's appointments so that your progress can be checked.
Things you must not do
Do not take KYTRIL to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dose without checking with your doctor.
Do not take any other medicines whether they require a prescription or not without first telling your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how KYTRIL affects you.
Be careful when drinking alcohol while you are taking KYTRIL. Your doctor may suggest that you avoid alcohol or reduce the amount of alcohol you drink while you are taking KYTRIL.
If you drink alcohol, dizziness or light-headedness may be worse.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking KYTRIL.
This medicine helps most people with nausea and vomiting, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- headache
- unusual tiredness or weakness
- dizziness or light headedness
- nervousness
- drowsiness
- difficulty in sleeping
- abdominal pain
- constipation
- diarrhoea
- altered taste
- fever
- skin rash.
The above list includes the more common side effects of your medicine.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- chest pain
- changes in your heart beat
- severe dizziness or fainting
- seizures
- symptoms of serotonin syndrome such as fever, sweating, fast heartbeat, agitation or confusion and loss of muscle coordination, which may lead to loss of consciousness
- symptoms of an allergic reaction, such as shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are rare.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using KYTRIL
Storage
Keep your tablets in the pack until it is time to take them. If you take the tablets out of the pack they may not keep well.
Keep your tablets in a cool dry place where the temperature stays below 30°C.
Do not store KYTRIL tablets, or any other medicine, in a bathroom or near a sink.
Do not leave KYTRIL tablets in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep KYTRIL tablets where children cannot reach them. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What KYTRIL looks like
KYTRIL 2 mg tablets are triangular, and white, with "K2" engraved on one side.
KYTRIL 2 mg tablets come in a:
- pack containing 1 tablet; or
- pack containing 5 tablets
KYTRIL is also available as an injection that is given by a healthcare professional.
Ingredients
Active Ingredients
Each 2 mg KYTRIL tablet contains 2 mg of granisetron.
Inactive Ingredients
- microcrystalline cellulose
- sodium starch glycollate
- hypromellose
- lactose
- magnesium stearate
- macrogol 400
- polysorbate 80
- titanium dioxide.
This medicine does not contain sucrose, gluten, tartrazine or any other azo dyes.
Sponsor
Atnahs Pharma Australia Pty Ltd
Level 10
10 Shelley Street,
SYDNEY, NSW, 2000, Australia
Supplier
KYTRIL is supplied by:
Clinect Pty Ltd
120 - 132 Atlantic Drive
Keysborough VIC 3173
Australia
Customer enquiries: 1 800 899 005
Please check with your pharmacist for the latest Consumer Medicine Information.
Australian Registration Number (AUST R)
2 mg tablet: 60872.
This leaflet was prepared on 15 September 2017
Published by MIMS December 2017

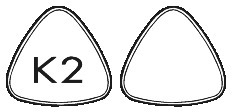
 Table 2 gives the comparative frequencies of the five commonly reported adverse events (> 3%) in patients receiving Kytril injection 40 microgram/kg, in single day chemotherapy trials. These patients received chemotherapy, primarily cisplatin, and intravenous fluids during the 24 hour period following Kytril injection administration.
Table 2 gives the comparative frequencies of the five commonly reported adverse events (> 3%) in patients receiving Kytril injection 40 microgram/kg, in single day chemotherapy trials. These patients received chemotherapy, primarily cisplatin, and intravenous fluids during the 24 hour period following Kytril injection administration. In the absence of a placebo group, there is uncertainty as to how many of these events should be attributed to Kytril, except for headache, which was clearly more frequent than in comparison groups.
In the absence of a placebo group, there is uncertainty as to how many of these events should be attributed to Kytril, except for headache, which was clearly more frequent than in comparison groups.
