1 Name of Medicine
Molnupiravir.
2 Qualitative and Quantitative Composition
Each capsule contains 200 mg of molnupiravir.
Molnupiravir is a white to off-white solid that is soluble in water.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Lagevrio is available as a 'Swedish Orange' opaque capsule with the corporate logo and "82" printed with white ink. Each capsule is approximately 21.7 mm in length.
4.1 Therapeutic Indications
Lagevrio (molnupiravir) has provisional approval for the treatment of adults with COVID-19 who do not require initiation of oxygen due to COVID-19 and who are at increased risk for hospitalisation or death [see Section 5.1 Pharmacodynamic Properties, Clinical trials].
The decision to approve this indication has been made on the basis of the analysis of efficacy and safety data from a Phase 3 trial. Continued approval of this indication depends on additional data.
4.2 Dose and Method of Administration
The recommended dose of Lagevrio in adult patients is 800 mg (four 200 mg capsules) taken orally every 12 hours for 5 days, with or without food.
The safety and efficacy of Lagevrio when administered for periods longer than 5 days have not been established.
Lagevrio should be administered as soon as possible after a diagnosis of COVID-19 has been made and within 5 days of symptom onset in adults who are at risk for progression to severe COVID-19, including hospitalisation or death.
In women of childbearing potential, health care providers should discuss the chance that they may be pregnant and consider the need for a pregnancy test.
Missed dose.
If the patient misses a dose of Lagevrio within 10 hours of the time it is usually taken, the patient should take it as soon as possible and resume the normal dosing schedule. If a patient misses a dose by more than 10 hours, the patient should not take the missed dose and instead take the next dose at the regularly scheduled time. The patient should not double the dose to make up for a missed dose.
Paediatric patients.
Safety and efficacy of Lagevrio have not been established in patients less than 18 years of age, therefore use in paediatric patients is not recommended [see Section 4.4 Special Warnings and Precautions for Use, Paediatric use; Section 5.2 Pharmacokinetic Properties, Special populations].
Elderly patients.
No dose adjustment of Lagevrio is recommended for geriatric patients [see Section 4.4 Special Warnings and Precautions for Use, Use in the elderly; Section 5.2 Pharmacokinetic Properties, Special populations].
Renal impairment.
No dose adjustment of Lagevrio is required in patients with renal impairment [see Section 5.2 Pharmacokinetic Properties, Special populations].
Hepatic impairment.
No dose adjustment of Lagevrio is recommended in patients with hepatic impairment [see Section 5.2 Pharmacokinetic Properties, Special populations].4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients listed in Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
Paediatric use.
Safety and efficacy of Lagevrio have not been established in patients less than 18 years of age, therefore use in paediatric patients is not recommended [see Section 4.2 Dose and Method of Administration, Paediatric patients; Section 5.2 Pharmacokinetic Properties, Paediatric population; Section 5.3 Preclinical Safety Data, General toxicity].
Use in the elderly.
In MOVe-OUT, there was no difference in safety and tolerability between patients > 65 years of age and younger patients who were treated with molnupiravir. No dose adjustment is recommended based on age. Based on population PK analysis, PK of N4-hydroxycytidine (NHC) was similar in geriatric patients compared to younger patients [see Section 4.2 Dose and Method of Administration, Elderly patients].
Hypersensitivity.
Hypersensitivity reactions have been reported with Lagevrio [see Section 4.8 Adverse Effects (Undesirable Effects), Post-marketing experience]. If signs or symptoms of a clinically significant hypersensitivity reaction occur, immediately discontinue Lagevrio and initiate appropriate medications and/or supportive care.4.5 Interactions with Other Medicines and Other Forms of Interactions
No drug interactions have been identified based on the limited available data.
Clinical drug-drug interaction trials of Lagevrio with concomitant medications have not been conducted. Molnupiravir is hydrolyzed to NHC prior to reaching systemic circulation. Uptake and metabolism of NHC are mediated by the same pathways involved in endogenous pyrimidine metabolism. NHC is not a substrate of major drug metabolizing enzymes or transporters. Neither molnupiravir nor NHC are inhibitors or inducers of major drug metabolizing enzymes or transporters. Therefore, the potential for molnupiravir or NHC to interact with concomitant medications is considered unlikely.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There were no effects on female or male fertility in rats at oral doses up to 500 mg/kg/day, with NHC exposures (based on AUC) of approximately 2 and 6 times respectively, the exposure in humans at the recommended human dose (RHD).
(Category D)
The use of Lagevrio is not recommended during pregnancy.
Advise women of childbearing potential to use effective contraception for the duration of treatment and for 4 days after the last dose of Lagevrio (molnupiravir). Based on animal data, Lagevrio may cause fetal harm when administered to pregnant women. There are no available data on the use of Lagevrio in pregnant women to evaluate the risk of major birth defects, miscarriage or adverse maternal or fetal outcomes.
In an animal reproduction study, oral administration of molnupiravir to pregnant rats during the period of organogenesis resulted in embryofetal lethality and teratogenicity at 1000 mg/kg/day (8 times the human NHC exposures at the recommended human dose (RHD)) and reduced fetal growth at ≥ 500 mg/kg/day (3 times the human NHC exposure at the RHD). Oral administration of molnupiravir to rabbits during the period of organogenesis resulted in reduced fetal body weights at 750 mg/kg/day (18 times the human NHC exposure at the RHD).
In a pre- and post-natal developmental study, molnupiravir was administered orally to female rats at doses up to 500 mg/kg/day from GD6 through lactation day 20. There were no effects on gestation and postnatal development of offspring at up to 500 mg/kg/day (1.6 times the human NHC exposure at the RHD).
It is unknown whether molnupiravir or any of the metabolites of molnupiravir are present in human milk, affect human milk production, or have effect on the breastfed infant.
Based on the potential for adverse reactions on the infant from Lagevrio, breastfeeding is not recommended during treatment and for 4 days after the last dose of Lagevrio.
NHC was detected in the plasma of nursing pups (~ 0.01 - 0.09% of mean plasma concentrations in maternal rats) from lactating rats administered molnupiravir.
Contraception.
There is no data available in relation to whether molnupiravir affects sperm. It is recommended that men who are sexually active with a partner of childbearing potential use an adequate form of contraception during and 3 months after treatment with molnupiravir.
It is recommended that sexually active women of childbearing potential use contraception during and for 4 days after treatment with molnupiravir.4.7 Effects on Ability to Drive and Use Machines
Lagevrio is predicted to have no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials experience.
The safety of Lagevrio was evaluated based on an analysis of a Phase 3 double-blind trial (MOVe-OUT) in which 1,411 non-hospitalised subjects with COVID-19 were randomised and treated with Lagevrio (N=710) or placebo (N=701) for up to 5 days and followed through Day 29. Adverse events were those reported while subjects were on study intervention or within 14 days of study intervention completion/discontinuation [see Section 5.1 Pharmacodynamic Properties, Clinical trials].
Discontinuation of study intervention due to an adverse event occurred in 1% of subjects receiving molnupiravir and 3% of subjects receiving placebo. Serious adverse events occurred in 7% of subjects receiving molnupiravir and 10% receiving placebo; none was considered drug-related by the investigator and most were COVID-19 related. Adverse events leading to death occurred in < 1% of the subjects receiving molnupiravir and 2% of subjects receiving placebo.
The most common adverse reactions in the molnupiravir treatment group in MOVe-OUT are presented in Table 1, all of which were Grade 1 (mild) or Grade 2 (moderate).

Laboratory abnormalities.
Selected laboratory abnormalities reported through Day 29 are presented in Table 2.

Post-marketing experience.
The following adverse reactions have been identified during post-marketing use of Lagevrio. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Gastrointestinal disorders.
Vomiting.
Immune system disorders.
Hypersensitivity [see Section 4.4 Special Warnings and Precautions for Use].
Skin and subcutaneous tissue disorders.
Angioedema, erythema, pruritus, rash, urticaria.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
There is no human experience of overdosage with Lagevrio. Treatment of overdose with Lagevrio should consist of general supportive measures including the monitoring of the clinical status of the patient. Haemodialysis is not expected to result in effective elimination of NHC.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Antivirals for systemic use.
ATC code: Not yet assigned.
Mechanism of action.
Lagevrio is a prodrug that is metabolised to the cytidine nucleoside analogue NHC, which distributes into cells where it is phosphorylated to form the pharmacologically active ribonucleoside triphosphate (NHC-TP). NHC-TP incorporation into viral RNA by the viral RNA polymerase, results in an accumulation of errors in the viral genome leading to inhibition of replication. This mechanism of action is known as viral error catastrophe.
Clinical trials.
Clinical data are based on data from 1,433 randomised subjects in the Phase 3 MOVe-OUT trial. MOVe-OUT is a randomised, placebo-controlled, double-blind clinical trial studying molnupiravir for the treatment of non-hospitalised patients with mild to moderate COVID-19 (defined by SpO2 > 93%, respiratory rate < 30 breaths per minute, heart rate < 125 beats per minute) who are at risk for progressing to severe COVID-19 and/or hospitalisation. Eligible subjects were 18 years of age and older and had one or more pre-defined risk factors for disease progression: over 60 years of age, diabetes, obesity (BMI ≥ 30), chronic kidney disease, serious heart conditions, chronic obstructive pulmonary disease, or active cancer. The study included symptomatic subjects not vaccinated against SARS-CoV-2 and who had laboratory confirmed SARS-CoV-2 infection and symptom onset within 5 days of randomisation. Subjects were randomised 1:1 to receive 800 mg of molnupiravir or placebo orally twice daily for 5 days.
At baseline, in all randomised subjects, the median age was 43 years (range: 18 to 90); 17% of subjects were over 60 years of age and 3% were 75 years of age or older; 49% of subjects were male; 57% were White, 5% Black or African American, 3% Asian, 50% Hispanic or Latino. The majority of subjects were enrolled from sites in Latin America (46%) and Europe (33%); 12% were enrolled in Africa, 6% were enrolled in North America and 3% were enrolled in Asia. Forty-eight percent of subjects received molnupiravir or placebo within 3 days of COVID-19 symptom onset. The most common risk factors were obesity (74%), over 60 years of age (17%), and diabetes (16%). Among 792 subjects (55% of total randomised population) with available baseline SARS-CoV-2 variant/clade identification results, 58% were infected with Delta (B.1.617.2 and AY lineages), 20% were infected with Mu (B.1.621), 11% were infected with Gamma (P.1), and the remainder were infected with other variants/clades. Overall, baseline demographic and disease characteristics were well balanced between the treatment arms.
Table 3 provides the results of the primary endpoint (the percentage of subjects who were hospitalised or died through Day 29 due to any cause).
 An exploratory analysis assessing the primary endpoint in prespecified subgroups is provided in Figure 1. For the majority of subgroups, the point estimate favours molnupiravir.
An exploratory analysis assessing the primary endpoint in prespecified subgroups is provided in Figure 1. For the majority of subgroups, the point estimate favours molnupiravir.
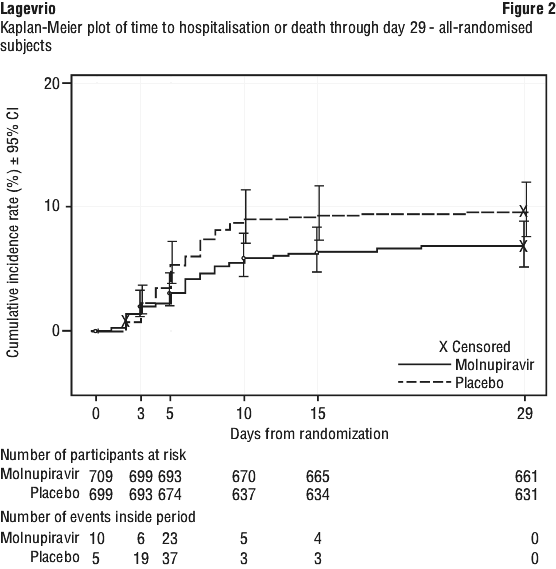
 The Kaplan-Meier curve shows time to hospitalisation or death, and results are consistent with the primary results (Figure 2).
The Kaplan-Meier curve shows time to hospitalisation or death, and results are consistent with the primary results (Figure 2).
 Higher percentages of subjects reported sustained improvement or resolution in most self-reported COVID-19 signs and symptoms, as recorded on a daily symptom diary, in the molnupiravir group compared to the placebo group (Figure 3).
Higher percentages of subjects reported sustained improvement or resolution in most self-reported COVID-19 signs and symptoms, as recorded on a daily symptom diary, in the molnupiravir group compared to the placebo group (Figure 3).

Microbiology.
The relationship between NHC and intracellular NHC-TP with antiviral efficacy has not been evaluated clinically.
Antiviral activity.
NHC was active in cell culture assays against SARS-CoV-2 (USA-WA1/2020 isolate) with 50% effective concentrations (EC50) ranging between 0.67 to 2.66 microM in A-549 cells and 0.32 to 2.03 microM in Vero E6 cells. NHC had similar antiviral activity against SARS-CoV-2 variants Alpha (B.1.1.7), Beta (B.1.351), Gamma (P.1), Delta (B.1.617.2), Lambda (C.37), Mu (B.1.621) and Omicron (BA.1.1.529/BA.1, BA.1.1, BA.2, BA.4 and BA.5) with mean EC50 values of 0.55-2.95 microM.
Resistance.
No amino acid substitutions in SARS-CoV-2 associated with resistance to NHC have been identified in clinical trials. Studies to evaluate selection of resistance to NHC with SARS-CoV-2 in cell culture showed a low likelihood of resistance development to NHC. Following 30 passages in cell culture, no substantial change in susceptibility (< 2-fold) was observed and no NHC resistance-associated amino acid substitutions were identified.
Cross-resistance.
NHC retained activity in vitro against virus with polymerase substitutions (e.g. F480L, V557L and E802D) associated with decreased remdesivir sensitivity.
Viral RNA rebound.
Post-treatment increases in SARS-CoV-2 RNA shedding levels (i.e. viral RNA rebound) in nasopharyngeal samples were observed on Day 10, Day 15, and/or Day 29 in a subset of Lagevrio and placebo recipients in the Phase 3 MOVe-OUT trial. Approximately 1% of both Lagevrio and placebo recipients had evidence of recurrent COVID-19 symptoms coinciding with a rebound in viral RNA levels in nasopharyngeal samples.
Post-treatment viral RNA rebound was not associated with the primary clinical outcome of hospitalisation or death through Day 29 following the single 5-day course of Lagevrio treatment. Post-treatment viral RNA rebound also was not associated with the detection of cell culture infectious virus in nasopharyngeal swab samples.
5.2 Pharmacokinetic Properties
Molnupiravir is a 5'-isobutyrate prodrug that is hydrolysed to NHC prior to reaching systemic circulation. The pharmacokinetics of NHC are similar in healthy subjects and patients with COVID-19.
The pharmacokinetics of NHC at steady-state following administration of 800 mg molnupiravir every 12 hours are provided in Table 4.

Absorption.
Following twice daily oral administration of 800 mg molnupiravir, the median time to peak plasma NHC concentrations (Tmax) was 1.5 hours.
Effect of food.
In healthy subjects, the administration of a single 200 mg dose of molnupiravir with a high-fat meal resulted in a 35% reduction in Cmax, but AUC was not significantly affected. Molnupiravir can be taken with or without food.
Distribution.
NHC does not bind to plasma proteins.
Metabolism.
Molnupiravir is hydrolysed by carboxylesterases to NHC prior to reaching systemic circulation. Uptake and metabolism of NHC are mediated by the same pathways involved in endogenous pyrimidine metabolism. NHC is not a substrate of major drug metabolizing enzymes or transporters.
Neither molnupiravir nor NHC are inhibitors or inducers of major drug metabolizing enzymes or transporters.
Excretion.
The effective half-life of NHC is approximately 3.3 hours.
The fraction of dose excreted as NHC in the urine was ≤ 3% in healthy participants.
Special populations.
Paediatric population.
The pharmacokinetics of molnupiravir in paediatric patients less than 18 years of age have not been evaluated.
Renal impairment.
Renal clearance is not a meaningful route of elimination for NHC. No dose adjustment of Lagevrio is required in patients with any degree of renal impairment is needed. In a population PK analysis, mild or moderate renal impairment did not have a meaningful impact on the PK of NHC. While the PK of NHC has not been evaluated in patients with eGFR less than 30 mL/min/1.73 m2 or on dialysis, severe renal impairment and end-stage renal disease (ESRD) are not expected to have a significant effect on NHC exposure [see Section 4.2 Dose and Method of Administration, Renal impairment].
Hepatic impairment.
The PK of molnupiravir and NHC has not been evaluated in patients with hepatic impairment. Preclinical data indicate that hepatic elimination is not expected to be a major route of NHC elimination; therefore, hepatic impairment is unlikely to affect NHC exposure. No dose adjustment in patients with hepatic impairment is needed [see Section 4.2 Dose and Method of Administration, Hepatic impairment].
Gender, race and age.
Population PK analysis showed that age, gender, race and ethnicity do not meaningfully influence the PK of NHC.
5.3 Preclinical Safety Data
General toxicity.
Reversible, dose-related bone marrow toxicity (decreased cellularity) affecting all haematopoietic cell lines was observed in dogs at ≥ 17 mg/kg/day (0.4 times the human NHC exposure at the RHD). Mild decreases in peripheral blood cell and platelet counts were seen after 7 days of molnupiravir treatment progressing to more severe haematological changes after 14 days of treatment.
Neither bone marrow nor haematological toxicity was observed in a 1-month toxicity study in mice up to 2000 mg/kg/day (19 times the human NHC exposure at the RHD) and a 3-month toxicity study in rats up to 1000 mg/kg/day (9 and 15 times the human NHC exposure at the RHD in female and male rats, respectively).
Bone and cartilage toxicity, consisting of an increase in the thickness of physeal and epiphyseal growth cartilage with decreases in trabecular bone was observed in the femur and tibia of rapidly growing rats in a 3-month toxicity study at ≥ 500 mg/kg/day (5 times the human NHC exposure at the RHD). There was no bone or cartilage toxicity in a 1-month toxicity study in rapidly growing rats up to 500 mg/kg/day (4 and 8 times the human NHC exposure at the RHD in female and male rats, respectively), in dogs dosed for 14 days up to 50 mg/kg/day (2 times the human NHC exposure at the RHD), or in a 1-month toxicity study in mice up to 2000 mg/kg/day (19 times the human NHC exposure at the RHD). Growth cartilage is not present in mature skeletons, therefore the bone and cartilage findings are not relevant for adult humans but may be relevant for paediatric patients.
Genotoxicity.
Molnupiravir and NHC were positive in the in vitro bacterial reverse mutation assay (Ames assay) with and without metabolic activation. Molnupiravir was negative for induction of chromosomal damage in an in vitro micronucleus assay (with and without metabolic activation). Molnupiravir was negative for induction of chromosomal damage in the in vivo rat micronucleus assay. In the in vivo Pig-a mutagenicity assay, the results were equivocal. In the in vivo Big Blue (cII Locus) transgenic rodent assay, molnupiravir did not induce increased mutation rates. Based on the totality of the genotoxicity data, molnupiravir is of low risk for genotoxicity or mutagenicity in clinical use.
Carcinogenicity.
Molnupiravir was not carcinogenic in a 6-month oral carcinogenicity study in RasH2 transgenic (Tg.RasH2) mice at any dose tested (30, 100 or 300 mg/kg/day).6 Pharmaceutical Particulars
6.1 List of Excipients
Each Lagevrio capsule contains the following inactive ingredients: Croscarmellose sodium, hyprolose, magnesium stearate, microcrystalline cellulose, purified water.
The capsule shell contains: Hypromellose, iron oxide red, titanium dioxide.
The white ink contains: Tert-butyl alcohol, ethanol absolute, isopropyl alcohol, potassium hydroxide, propylene glycol, purified water, shellac, strong ammonia solution, titanium dioxide.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store Lagevrio below 30°C.
Store Lagevrio in the original bottle.
6.5 Nature and Contents of Container
HDPE bottle containing 40 capsules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
Molnupiravir is the 5'-isobutyrate prodrug of the antiviral ribonucleoside analogue NHC.
The chemical name for molnupiravir is {(2R,3S,4R,5R)-3,4-Dihydroxy- 5-[(4Z)-4-(hydroxyimino)-2-oxo-3,4-dihydropyrimidin- 1(2H)-yl}oxolan-2-yl]methyl 2-methylpropanoate.
It has an empirical formula of C13H19N3O7 and its molecular weight is 329.31 g/mol.
Its structural formula is:
 Molnupiravir is a white to off-white solid that is soluble in water.
Molnupiravir is a white to off-white solid that is soluble in water.
CAS number.
2492423-29-5.7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Summary Table of Changes




 An exploratory analysis assessing the primary endpoint in prespecified subgroups is provided in Figure 1. For the majority of subgroups, the point estimate favours molnupiravir.
An exploratory analysis assessing the primary endpoint in prespecified subgroups is provided in Figure 1. For the majority of subgroups, the point estimate favours molnupiravir. The Kaplan-Meier curve shows time to hospitalisation or death, and results are consistent with the primary results (Figure 2).
The Kaplan-Meier curve shows time to hospitalisation or death, and results are consistent with the primary results (Figure 2). Higher percentages of subjects reported sustained improvement or resolution in most self-reported COVID-19 signs and symptoms, as recorded on a daily symptom diary, in the molnupiravir group compared to the placebo group (Figure 3).
Higher percentages of subjects reported sustained improvement or resolution in most self-reported COVID-19 signs and symptoms, as recorded on a daily symptom diary, in the molnupiravir group compared to the placebo group (Figure 3).

 Molnupiravir is a white to off-white solid that is soluble in water.
Molnupiravir is a white to off-white solid that is soluble in water.