1 Name of Medicine
Chlormethine hydrochloride.
2 Qualitative and Quantitative Composition
Each gram of gel contains chlormethine hydrochloride equivalent to 160 micrograms of chlormethine.
For the full list of excipients, see Section 6.1.
3 Pharmaceutical Form
Gel.
Clear, colourless gel.
4.1 Therapeutic Indications
Ledaga is indicated for the topical treatment of mycosis fungoides-type cutaneous T-cell lymphoma (MF-type CTCL) in adult patients.
4.2 Dose and Method of Administration
Treatment with Ledaga should be initiated by an appropriately experienced physician.
Posology.
A thin film of Ledaga should be applied once daily to affected areas of the skin.
Treatment with Ledaga should be stopped for any grade of skin ulceration or blistering, or moderately severe or severe dermatitis (e.g. marked skin redness with oedema). Upon improvement, treatment with Ledaga can be restarted at a reduced frequency of once every 3 days. If reintroduction of treatment is tolerated for at least 1 week, the frequency of application can be increased to every other day for at least 1 week and then to once-daily application if tolerated.
Elderly.
The dosing recommendation for elderly patients (≥ 65 years old) is the same as for younger adult patients (see Section 4.8).
Paediatric population.
The safety and efficacy of Ledaga in children aged 0 to 18 years have not been established. No data are available.
Method of administration.
Ledaga is for topical application to the skin.
The following instructions should be followed by patients or caregivers when applying Ledaga:
Patients must wash hands thoroughly with soap and water immediately after handling or applying Ledaga. Patients should apply Ledaga to affected areas of the skin. In case of Ledaga exposure to non-affected areas of the skin, patients should wash the exposed area with soap and water.
Caregivers must wear disposable nitrile gloves when applying Ledaga to patients. Caregivers should remove gloves carefully (turning them inside out during the removal to avoid contact with Ledaga) and wash hands thoroughly with soap and water after removal of gloves. If there is accidental skin exposure to Ledaga, caregivers must immediately wash exposed areas thoroughly with soap and water for at least 15 minutes. Remove and wash contaminated clothing.
The opening of the tube is covered with a foil safety seal. The cap should be used to puncture the seal. The tube should not be used and the pharmacist should be contacted if the seal is missing, punctured, or lifted.
Ledaga should be applied immediately or within 30 minutes after removal from the refrigerator. The tube should be returned to the refrigerator immediately after each use. With clean hands, the tube should be placed back into the original box and the box should be placed in the supplied transparent, sealable, plastic bag for storage in the refrigerator.
Ledaga should be applied to completely dry skin at least 4 hours before or 30 minutes after showering or washing. The patient should allow treated areas to dry for 5 to 10 minutes after application before covering with clothing. Occlusive (air- or water-tight) dressings should not be used on areas of the skin where Ledaga was applied.
Emollients (moisturisers) or other topical products may be applied to the treated areas 2 hours before or 2 hours after application of Ledaga.
Fire, flame, and smoking must be avoided until Ledaga has dried.4.3 Contraindications
Hypersensitivity to chlormethine or to any of the excipients listed, see Section 6.1.
4.4 Special Warnings and Precautions for Use
Mucosal or eye exposure.
Contact with mucous membranes, especially those of the eyes, must be avoided. Exposure of mucous membranes such as the oral mucosa or nasal mucosa causes pain, redness, and ulceration, and these may be severe. Exposure of the eyes to chlormethine causes pain, burns, inflammation, photophobia, and blurred vision. Blindness and severe irreversible anterior eye injury may occur.
Patients should be advised that if any mucous membrane exposure occurs:
irrigation should be performed immediately for at least 15 minutes with copious amounts of water (or sodium chloride 9 mg/mL (0.9%) solution for injection, or a balanced salt ophthalmic irrigating solution may be used if there is eye exposure), and
medical care should be obtained immediately (including ophthalmological consultation if there is eye exposure).
Local skin reactions.
Patients should be assessed during treatment for skin reactions such as dermatitis (e.g. redness, swelling, inflammation), pruritus, blisters, ulceration, and skin infections. The face, genitalia, anus, and intertriginous skin are at increased risk of skin reactions to topical chlormethine. Therefore, administration of Ledaga in these areas should be avoided.
For dose modification information in case of skin reactions, see Section 4.2.
Hypersensitivity.
Hypersensitivity reactions, including isolated cases of anaphylaxis, have been reported in the literature after the use of topical formulations of chlormethine (see Section 4.3; Section 4.8).
Skin cancer.
Skin-directed therapies for MF-type CTCL have been associated with secondary skin cancers, although the specific contribution of chlormethine has not been established. Patients should be monitored for development of skin cancers during and after discontinuation of treatment with chlormethine.
Secondary exposure to Ledaga.
Direct skin contact with Ledaga should be avoided in individuals other than the patient. Direct skin contact with Ledaga should also be avoided in non-affected areas in patients.
Risks of secondary exposure may include skin reactions, mucosal injury, and skin cancers. Recommended application instructions should be followed to prevent secondary exposure (see Section 4.2).
Excipients.
The medicinal product contains propylene glycol and butylhydroxytoluene, which may cause skin irritation (e.g. contact dermatitis). In addition, butylhydroxytoluene has been reported to cause irritation to the eyes and mucous membranes.
Use in the elderly.
The safety profile observed in elderly patients was consistent with that in the overall patient population. No dose adjustments are required (see Section 4.2).
Paediatric use.
The safety of Ledaga in children aged 0 to 18 years has not been established. No data are available.
Effects on laboratory tests.
Clinical laboratory safety data were monitored throughout the two clinical studies and no trend toward abnormal values were noted following topical administration.4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction studies have been performed.
4.6 Fertility, Pregnancy and Lactation
Ledaga is not recommended in women of childbearing potential not using contraception.
Effects on fertility.
Female patients of reproductive potential should be advised to use effective contraception during treatment with Ledaga. A barrier method of contraception should be used to avoid direct exposure of reproductive organs to Ledaga.
Males with female partners of reproductive potential should be advised to use effective contraception during treatment with Ledaga. A barrier method of contraception should be used to avoid direct exposure of reproductive organs to Ledaga.
Adverse effects on fertility have been observed with chlormethine after systemic administration in animals. Fertility was impaired in male rats with intravenous administration at doses ≥ 0.25 mg/kg every 2 weeks, and in mice (treated males paired with treated females) with intraperitoneal administration at 0.5 mg/kg/day for 4 days. The relevance to humans receiving topical chlormethine is unknown.
(Category B3)
Ledaga is not recommended during pregnancy. Based on case reports in humans, findings in animal reproduction studies, its mechanism of action, and genotoxicity findings, chlormethine may cause fetal harm. There are case reports of children born with malformations in pregnant women systemically administered chlormethine.
Chlormethine was teratogenic and embryo-lethal after a single subcutaneous administration to animals. Advise women to avoid becoming pregnant while using Ledaga. If this medicine is used during pregnancy or if the patient becomes pregnant while taking this medicine, the patient should be apprised of the potential hazard to a fetus.
Chlormethine has been shown to cause fetal malformations, embryofetal lethality and fetal growth retardation in mice and rats after a single injection at 1-2.5 mg/kg. Animal embryofetal development studies involving topical administration of chlormethine have not been performed.
Breastfeeding during treatment with Ledaga is not recommended because of the potential for topical or systemic exposure to Ledaga through exposure to the mother's skin and the potential for serious adverse reactions in the breastfed child from chlormethine. There are no data on the presence of chlormethine or its metabolites in human milk, the effects of the drug on the breastfed child, or the effects of the drug on milk production.4.7 Effects on Ability to Drive and Use Machines
Ledaga has no or negligible influence on the ability to drive or use machines.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile.
In a randomised-controlled trial (n=128 exposed to Ledaga for a median duration of 52 weeks), the most frequent adverse reactions to Ledaga were skin related: dermatitis (54.7%; e.g. skin irritation, erythema, rash, urticaria, skin-burning sensation, pain of the skin), pruritus (20.3%), skin infections (11.7%), skin ulceration and blistering (6.3%), and skin hyperpigmentation (5.5%). Cutaneous hypersensitivity reactions were reported in 2.3% of the treated patients.
Tabulated list of adverse events in controlled trial.
See Table 1.
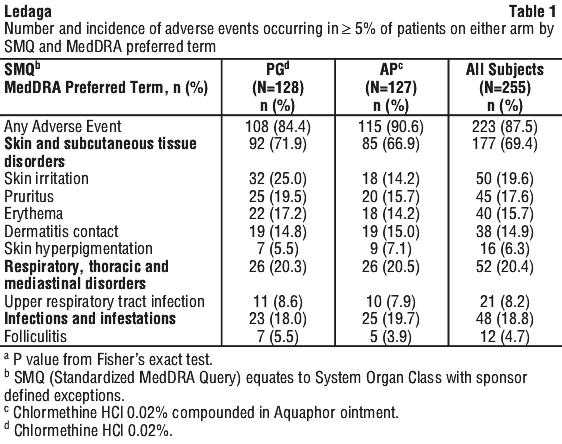
Elderly population.
In the controlled clinical trial, 31% (79/255) of the study population were aged 65 years or older. The safety profile observed in elderly patients was consistent with that in the overall patient population.
Postmarketing.
An observational post-marketing study was undertaken in the United States. Non-serious AEs assessed as related to Chlormethine Gel were experienced by 83 or 298 patients (27.9%) in the Chlormethine Gel plus any other treatment group. No serious adverse events were assessed to be related to chlormethine gel.
Of the skin and subcutaneous tissue disorder AEs related to Chlormethine Gel, dermatitis was assessed to be related in 37 patients (12.4%), pruritus in 22 patients (7.4%) and skin irritation in 21 patients (7.0%). These rates are lower than in the registration study and which may be due to: widespread concomitant use of topical corticosteroids; periods of less frequent dosing; most patients (254/298=85.2%) were already using mechlorethamine gel for > 30 days at enrolment. Dermatitis reactions are known to occur more frequently early in treatment.
Based on the evaluation of the cumulative safety data from all global post-marketing sources, no new risks or signals have been identified with the chlormethine gel formulation.
Reporting of suspected adverse reactions.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
No cases of overdose after cutaneous use of Ledaga were reported during the clinical development programme or post-marketing period. Management of overdose should consist of washing the exposed area with water.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Antineoplastic agents, nitrogen mustard analogues, ATC code: L01AA05.
Mechanism of action.
Chlormethine is a bifunctional alkylating agent that reacts with DNA to form cross-links, inducing the death of rapidly proliferating cells.
Clinical trials.
The efficacy and safety of Ledaga were assessed in a randomised, multicentre, observer-blinded, active-controlled, non-inferiority clinical trial (Study 201) of 260 adult patients with Stage IA (141), IB (115), and IIA (4) MF-type CTCL who had received at least one prior skin-directed therapy. Qualifying prior therapies included topical corticosteroids, phototherapy, topical bexarotene, and topical nitrogen mustard. Patients were not required to be refractory to or intolerant of prior therapies. Patients were stratified based on stage (IA vs IB and IIA) and then randomised to receive either Ledaga (equivalent to 0.02% chlormethine HCl) or the comparator (a petroleum-based 0.02% chlormethine HCl ointment).
Study medicinal product was to be applied topically once daily for 12 months. Dosing could be suspended or continued at reduced frequency in the case of skin reactions. The median daily usage of Ledaga was 1.8 g. The maximum individual daily usage in the trial was 10.5 g of gel (i.e. 2.1 mg of chlormethine HCl).
The primary efficacy endpoint in Study 201 was the Composite Assessment of Index Lesion Severity (CAILS) response rate. The CAILS score is obtained by adding the severity score of each of the following categories for up to 5 index lesions: erythema, scaling, plaque elevation, and surface area. Severity was graded from 0 (none) to 8 (severe) for erythema and scaling; 0 to 3 for plaque elevation; and 0 to 9 for surface area. Assessment was undertaken by a blinded observer. A response was defined as an at least 50% improvement in the baseline CAILS score, confirmed at a subsequent visit at least 4 weeks later. A complete response was defined as a confirmed CAILS score of 0. A partial response was defined as an at least 50% reduction in the baseline CAILS score. Non-inferiority was considered to have been demonstrated if the lower bound of the 95% confidence interval around the ratio of response rates (Ledaga/comparator) was greater than or equal to 0.75. The CAILS score was adjusted by removal of the pigmentation score and simplification of the plaque elevation scale.
As the main secondary endpoint, patients were also evaluated using the Severity Weighted Assessment Tool (SWAT), which was based on an assessment of all lesions. The SWAT score is derived by measuring each involved area as a percentage of total body surface area (% BSA) and multiplying it by a severity weighting factor (1=patch, 2=plaque, 3=tumour or ulcer). The response criteria were the same as for CAILS.
Efficacy was evaluated in the Intent-To-Treat (ITT) population, which included all 260 randomised patients [Table 2], and in the Efficacy Evaluable (EE) population, which included 185 patients who were treated for at least 6 months with no major protocol deviations.
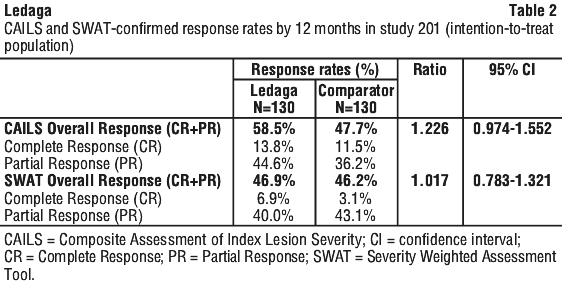 The ratio of response and the 95% confidence interval in the ITT population were 1.226 (0.974-1.552) for CAILS and 1.017 (0.783-1.321) for SWAT and were consistent with those in the EE population for both the overall CAILS and SWAT responses.
The ratio of response and the 95% confidence interval in the ITT population were 1.226 (0.974-1.552) for CAILS and 1.017 (0.783-1.321) for SWAT and were consistent with those in the EE population for both the overall CAILS and SWAT responses.
Reductions in mean CAILS scores were observed as early as at 4 weeks, with further reductions observed with continuing therapy.
In the EE population, the percentage of patients who achieved a confirmed response by CAILS was similar between disease stages IA (79.6%) and IB-IIA (73.2%).
Results in other secondary endpoints (response in percentage of body surface area affected, time to first confirmed CAILS response, duration of first confirmed CAILS response and time to disease progression) were consistent with those for CAILS and SWAT.
A small number of subjects (6.3%, 8/128) treated with Ledaga utilised topical corticosteroids. Thus, the safety of the concomitant use of Ledaga with topical corticosteroids has not yet been established.
5.2 Pharmacokinetic Properties
Patients who received Ledaga in Study 201 had no measurable concentrations of chlormethine in blood samples collected 1, 3 and 6 hours post-application on Day 1, and at the first month visit.
Similarly, patients who received chlormethine gel 0.04% in a follow-up study (Study 202) had no measurable concentrations of chlormethine or its degradation product (half-mustard) in blood collected 1 hour post-application on Day 1 or after 2, 4, or 6 months of treatment.
5.3 Preclinical Safety Data
Genotoxicity.
Chlormethine was shown to be genotoxic in multiple assays, including for mutagenicity in bacteria (Ames test), chromosomal aberrations in vitro (in cultured human lymphocytes) and for clastogenicity in vivo (mouse bone marrow micronucleus test). Covalent binding to DNA is the key mechanism for the desired cytotoxic action of chlormethine.
Carcinogenicity.
Chlormethine has been shown to be carcinogenic in rodents after subcutaneous and intravenous injection, and with topical dermal administration. Dermal application of chlormethine to mice at a dose of 12 to 15 mg/kg/week for 20 weeks resulted in skin tumours (squamous cell carcinomas and skin papillomas). There were no reports of systemic tumours after topical administration of chlormethine.6 Pharmaceutical Particulars
6.1 List of Excipients
Diethylene glycol monoethyl ether, propylene glycol, isopropyl alcohol, glycerol, lactic acid, hyprolose, sodium chloride, menthol, disodium edetate, butylhydroxytoluene.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
Unopened tube.
5 years in the freezer (-15°C to -25°C).
After defrosting.
Store at 2°C to 8°C for up to 60 days (Refrigerate. Do not freeze).
Ledaga should be removed from the refrigerator just prior to application and returned to the refrigerator immediately after each use in its box inside the child-resistant, transparent, sealable, plastic bag.
6.4 Special Precautions for Storage
Store and transport at -15°C to -25°C (deep freeze).
For storage conditions after defrosting Ledaga, see Section 6.3.
6.5 Nature and Contents of Container
Ledaga is provided in a white aluminium tube with an inner lacquer and an aluminium seal and a white polypropylene screw cap. Each tube contains 60 g of gel.
6.6 Special Precautions for Disposal
Ledaga is a cytotoxic medicinal product.
Caregivers must wear nitrile gloves when handling Ledaga. Patients and caregivers must wash hands after handling Ledaga.
Ledaga is an alcohol-based product and is flammable. The recommended application instructions should be followed (see Section 4.2).
Unused refrigerated Ledaga should be discarded after 60 days, together with the plastic bag.
Any unused medicinal product or waste material, including the plastic bag and the nitrile gloves used for application, must be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
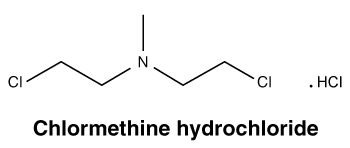 Molecular Formula: C5H11C12N.HCl.
Molecular Formula: C5H11C12N.HCl.
Molecular Weight: 192.51.
CAS number.
55-86-7.7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Summary Table of Changes



 The ratio of response and the 95% confidence interval in the ITT population were 1.226 (0.974-1.552) for CAILS and 1.017 (0.783-1.321) for SWAT and were consistent with those in the EE population for both the overall CAILS and SWAT responses.
The ratio of response and the 95% confidence interval in the ITT population were 1.226 (0.974-1.552) for CAILS and 1.017 (0.783-1.321) for SWAT and were consistent with those in the EE population for both the overall CAILS and SWAT responses. Molecular Formula: C5H11C12N.HCl.
Molecular Formula: C5H11C12N.HCl.