1 Name of Medicine
Levonorgestrel and ethinylestradiol.
2 Qualitative and Quantitative Composition
Leveth 150/30 ED is a combined oral contraceptive tablet containing the synthetic progesterone, levonorgestrel and the synthetic estrogen, ethinylestradiol.
Each yellow active tablet in Leveth 150/30 ED contains ethinylestradiol 30 microgram and levonorgestrel 150 microgram.
Excipients with known effect.
Lactose (present in both yellow active tablet and white placebo tablet).
For the full list of excipients see Section 6.1 List of Excipients.3 Pharmaceutical Form
Active tablet.
Plain, round, yellow film-coated tablet.
Placebo tablet.
Plain, round, white film-coated tablet.4.1 Therapeutic Indications
Oral contraception.
4.2 Dose and Method of Administration
Leveth 150/30 ED tablets are intended for oral administration.
Dosage.
How to take Leveth 150/30 ED. COCs, when taken correctly, have a failure rate of approximately 1% per year. The failure rate may increase when pills are missed or taken incorrectly.
One tablet is to be taken daily. The tablets must be taken in the order directed on the packaging at about the same time every day, with some liquid as needed. Daily tablet taking should be continuous for 28 consecutive days, starting with a tablet corresponding to that day of the week from the red section of the pack.
If a woman starts on a Monday, Tuesday, Wednesday, Thursday or Friday, her first tablet is a white placebo one, while if she starts on a Saturday or Sunday her first tablet will be a yellow active one. Thereafter, one tablet is taken daily, following the arrows marked on the pack, until all tablets have been taken. Each subsequent pack is started the day after the last tablet of the previous pack. Withdrawal bleeding usually starts on day 2 to 3 after starting the white placebo tablets (last row) and may not have finished before the next pack is started.
How to start Leveth 150/30 ED. No preceding hormonal contraceptive use (in the past month).
Tablet-taking has to start on day 1 of the woman's natural cycle (i.e. the first day of her menstrual bleeding), but during the first additional non-hormonal contraceptive methods must be used for the first 14 days of tablet-taking.
When changing pills. Changing from a COC or vaginal ring.
The woman should start Leveth 150/30 ED on the day after the last active tablet (the last tablet containing the active substances) of her previous COC.
In case a vaginal ring has been used, the woman should start taking Leveth 150/30 ED on the day of removal.
Changing from a progestogen-only method (minipill, injection, implant) or from a progestogen releasing intrauterine system (IUS).
The woman may switch:
from the minipill on any day;
from an implant or IUS on the day of removal;
when the next injection would be due for an injectable;
but in all of these cases the woman should be advised to additionally use a barrier method for the first 14 days of tablet-taking.
Following first-trimester abortion. The woman may start immediately. Additional non-hormonal contraceptive measures are necessary for the first 14 days.
After childbirth or second-trimester abortion. Women should be advised to start 21 to 28 days after delivery or second-trimester abortion. Additional non-hormonal contraceptive methods are necessary for the next 14 days. If intercourse has already occurred, pregnancy should be excluded before the actual start of COC use or the woman has to wait for her first menstrual period.
For breastfeeding women, see Section 4.6 Fertility, Pregnancy and Lactation, Use in lactation.
Additional contraceptive precautions. When additional contraceptive precautions are required, the woman should be advised either to abstain from sex or to use a barrier method of contraception, such as a cap (or diaphragm) plus spermicide, or for her partner to use a condom. Rhythm methods should not be advised as the COC disrupts the cyclical changes associated with the natural menstrual cycle e.g. changes in temperature and cervical mucus.
How to shift periods or how to delay a period. To delay a period the woman should continue with another pack of Leveth 150/30 ED by missing the white placebo tablets from the current pack, and starting with the yellow active tablets from the next pack as soon as the yellow active tablets from the current pack are finished. The extension can be carried on for as long as wished until the end of the second pack. During the extension the woman may experience breakthrough bleeding or spotting. Regular intake of Leveth 150/30 ED is then resumed after the white placebo tablet phase.
To shift her period to another day of the week than the woman is used to with her current scheme, she can be advised to shorten her forthcoming white placebo tablet phase by as many days as she likes. The shorter the interval, the higher the risk that she does not have a withdrawal bleed and will experience breakthrough bleeding and spotting during the second pack (just as when delaying a period).
How to manage reduced reliability. When COCs are taken according to the directions for use, the occurrence of pregnancy is highly unlikely. However, the reliability of oral contraceptives may be reduced under the following circumstances.
Management of missed tablets. Missed pills from the last row of the blister are placebo tablets and thus can be disregarded. However, they should be discarded to avoid unintentionally prolonging the placebo tablet phase. The following advice only refers to missed yellow active tablets (rows 1 - 3 of the blister).
If the woman is less than 12 hours late in taking any yellow active tablet, contraceptive protection is not reduced. The woman should take the tablet as soon as she remembers and should take further tablets at the usual time.
If the woman is more than 12 hours late in taking any yellow active tablet, contraceptive protection may be reduced. The more tablets are missed and the closer they are to the white placebo tablet phase, the higher the risk of a pregnancy.
The management of missed tablets can be guided by the following two basic rules:
1. 'Active tablet'-taking must never be discontinued for longer than 7 days.
2. Seven days of uninterrupted 'active tablet'-taking are required to attain adequate suppression of the hypothalamic-pituitary-ovarian-axis.
Accordingly, the following advice can be given in daily practice:
Week 1 of the active tablets.
If the woman is more than 12 hours late in taking any yellow active tablet (or several active tablets) from the pack, she should take the last missed yellow active tablet as soon as she remembers, even if this means taking two tablets in one day at the same time, and then continue to take tablets at the normal time. Additional contraceptive precautions such as a condom should be used for the next 7 days.
If intercourse took place in the preceding 7 days, the possibility of a pregnancy should be considered.
Week 2 of the active tablets.
The woman should take the last missed yellow active tablet as soon as she remembers, even if this means taking two yellow active tablets at the same time. She then continues to take tablets at her usual time. Provided that the woman has taken her tablets correctly in the 7 days preceding the first missed yellow active tablet, there is no need to use extra contraceptive precautions. However, if this is not the case, or if she missed more than one yellow active tablet, the woman should be advised to use extra precautions for 7 days.
Week 3 of the active tablets.
The risk of reduced reliability is imminent because of the forthcoming white placebo tablet interval. However, by adjusting the tablet-intake schedule, reduced contraceptive protection can still be prevented. By adhering to either of the following two options, there is therefore no need to use extra contraceptive precautions, provided that in the 7 days preceding the first missed yellow active tablet the woman has taken all tablets correctly. If this is not the case, the woman should be advised to follow the first of these two options and to use extra precautions for the next 7 days as well.
1. The woman should take the last missed yellow active tablet as soon as she remembers, even if this means taking two yellow active tablets at the same time. She then continues to take tablets at her usual time until the yellow active tablets are taken. The 7 white placebo tablets must be discarded. The next pack must be started right away. The user is unlikely to have a withdrawal bleed until the end of the yellow active tablets of the second pack, but she may experience spotting or breakthrough bleeding on tablet-taking days.
2. The woman may also be advised to discontinue taking the yellow active tablets from the current pack. She should then have a tablet-free interval of up to 7 days, including the days she missed tablets, and subsequently continue with the next pack.
If the woman missed tablets and subsequently has no withdrawal bleed in the hormone-free white tablet phase, the possibility of a pregnancy should be considered.
Advice in case of gastrointestinal disturbances. In case of severe gastrointestinal disturbances, absorption may not be complete and additional contraceptive measures should be taken.
If vomiting occurs within 3 - 4 hours after taking an active tablet, the advice concerning management of missed tablets (above) is applicable. If the woman does not want to change her normal tablet-taking schedule, she has to take the extra tablet(s) needed from another pack.4.3 Contraindications
COCs should not be used in the presence of any of the conditions listed below. Should any of the conditions appear for the first-time during COC use, the product should be stopped immediately.
Presence or risk of venous thromboembolism (VTE) (see Section 4.4 Special Warnings and Precautions for Use):
current VTE (on anticoagulants) or history of deep venous thrombosis [DVT] or pulmonary embolism [PE];
known hereditary or acquired predisposition for venous thromboembolism, such as APC-resistance (including Factor V Leiden), antithrombin-III-deficiency, protein C deficiency, protein S deficiency;
major surgery with prolonged immobilisation;
a high risk of venous thromboembolism due to the presence of multiple risk factors.
Presence or risk of arterial thromboembolism (ATE) (see Section 4.4 Special Warnings and Precautions for Use):
current ATE or history of ATE (e.g. myocardial infarction or stroke) or prodromal condition (e.g. angina pectoris or transient ischaemic attack [TIA]);
known hereditary or acquired predisposition for arterial thromboembolism, such as hyperhomocysteinaemia and antiphospholipid-antibodies (e.g. anticardiolipin-antibodies and lupus anticoagulant);
history of migraine with focal neurological symptoms;
a high risk of arterial thromboembolism due to multiple risk factors or to the presence of one serious risk factor such as: diabetes mellitus with vascular symptoms, severe hypertension, severe dyslipoproteinaemia.
Pancreatitis or a history thereof if associated with severe hypertriglyceridaemia.
Presence or history of severe hepatic disease as long as liver function values have not returned to normal.
Presence or history of liver tumours (benign or malignant).
Leveth 150/30 ED is contraindicated for concomitant use with the medicinal products glecaprevir, pibrentasvir, sofosbuvir, velpatasvir, voxilaprevir, ombitasvir, paritaprevir or dasabuvir and combinations of these (see Section 4.4 Special Warnings and Precautions for Use; Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Known or suspected malignant conditions of the genital organs, the breasts, or other organs, if sex steroid influenced.
Undiagnosed vaginal bleeding.
Known or suspected pregnancy.
Hypersensitivity to any of the ingredients contained in Leveth 150/30 ED tablets.
4.4 Special Warnings and Precautions for Use
If any of the conditions/risk factors mentioned below are present, the benefits of Leveth 150/30 ED should be weighed against the possible risks for each individual woman and discussed with the woman before she decides to start taking it. In the event of aggravation, exacerbation or first appearance of any of these conditions or risk factors, the woman should contact her doctor. The doctor should then decide on whether Leveth 150/30 ED should be discontinued.
Circulatory disorders.
Epidemiological studies have suggested an association between the use of combined oral contraceptives (COCs) containing ethinylestradiol and an increased risk of arterial and venous thrombotic and thromboembolic diseases such as myocardial infarction, stroke, deep venous thrombosis (DVT) and pulmonary embolism (PE). These events occur rarely in average-risk woman.
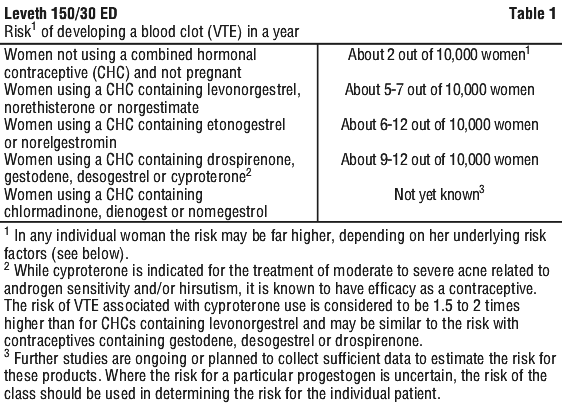
Risk of venous thromboembolism (VTE). The use of any combined hormonal contraceptive (CHC) increases the risk of VTE compared with no use. The woman should be advised that her VTE risk is highest in the first ever year of use and that there is some evidence that the risk is increased when a CHC is re-started after a break in use of 4 weeks or more.
It is important that women understand that VTE associated with CHC use is rare in average-risk women (see Table 1). The risk in pregnancy (5 - 20 per 10,000 women over 9 months) and the risk in the post-partum period (45 - 65 per 10,000 women over 12 weeks) is higher than that associated with CHC use.
Combined hormonal contraceptive (CHC) in Table 1 refers to oral contraceptives with a low estrogen dose (< 50 microgram ethinylestradiol). An additional increase in VTE risk for CHCs containing ≥ 50 microgram ethinylestradiol cannot be excluded.
The decision to use any product other than one with the lowest VTE risk should be taken only after a discussion with the woman to ensure she understands the risk of VTE with CHCs, and how her current risk factors influence this risk.
Products that contain the progestogens levonorgestrel, such as Leveth 150/30 ED, norgestimate or norethisterone are associated with the lowest risk of VTE.
 The increased risk of VTE during the postpartum period must be considered if re-starting Leveth 150/30 ED (see Section 4.2 Dose and Method of Administration; Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy, Use in lactation).
The increased risk of VTE during the postpartum period must be considered if re-starting Leveth 150/30 ED (see Section 4.2 Dose and Method of Administration; Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy, Use in lactation).
VTE may be life threatening or may have a fatal outcome (in 1 - 2% of cases).
Extremely rarely, thrombosis has been reported to occur in CHC users in other blood vessels e.g. hepatic, mesenteric, renal, cerebral or retinal veins and arteries.
The risk of venous thromboembolic events in CHC users may increase substantially in a woman with additional risk factors, particularly if there are multiple risk factors (see list below).
Leveth 150/30 ED is contraindicated if a woman has multiple risk factors that put her at high risk of venous thrombosis. If a woman has more than one risk factor, it is possible that the increase in risk is greater than the sum of the individual factors - in this case her total risk of VTE should be considered. If the balance of benefits and risks is considered to be negative a CHC should not be prescribed.
When considering risk/benefit, the doctor should take into account that the adequate treatment of a condition may reduce the associated risk of thrombosis.
Risk factors for VTE.
Obesity (body mass index over 30 kg/m2). Risk increases substantially as BMI rises.
Prolonged immobilisation, major surgery, any surgery to the legs or pelvis, neurosurgery or major trauma.
Temporary immobilisation including air travel > 4 hours can also be a risk factor for VTE, particularly in women with other risk factors.
Positive family history (i.e. venous thromboembolism ever in a sibling or parent especially at a relatively early age e.g. before 50).
Biochemical factors that may be indicative of hereditary or acquired predisposition for VTE include Activated Protein C (APC) resistance (including Factor V Leiden), antithrombin-III deficiency, protein C deficiency, protein S deficiency.
Other medical conditions associated with VTE: cancer; systemic lupus erythematosus; haemolytic uraemic syndrome; chronic inflammatory bowel disease (e.g. Crohn's disease or ulcerative colitis); sickle cell disease.
Increasing age, particularly above 35 years.
Smoking.
In women at risk of prolonged immobilisation (including major surgery, any surgery to the legs or pelvis, neurosurgery, or major trauma), it is advisable to discontinue use of Leveth 150/30 ED (in the case of elective surgery at least four weeks in advance) and not resume until two weeks after complete remobilisation. Another method of contraception should be used to avoid unintentional pregnancy. Antithrombotic treatment should be considered if Leveth 150/30 ED has not been discontinued in advance.
If a hereditary predisposition to VTE is suspected, the woman should be referred to a specialist for advice before deciding about any CHC use.
There is no consensus about the possible role of varicose veins and superficial thrombophlebitis in VTE.
Symptoms of VTE (deep vein thrombosis and pulmonary embolism).
Women should be informed of the symptoms of VTE and be advised to seek urgent medical attention if VTE symptoms develop and to inform the healthcare professional that she is taking a CHC.
Symptoms of deep vein thrombosis (DVT) can include: unilateral swelling of the leg and/or foot or along a vein in the leg; pain or tenderness in the leg which may be felt only when standing or walking; increased warmth in the affected leg; red or discoloured skin on the leg.
Symptoms of pulmonary embolism (PE) can include: sudden onset of unexplained shortness of breath or rapid breathing; sudden coughing which may be associated with haemoptysis; sharp chest pain or sudden severe pain in the chest which may increase with deep breathing; severe light headedness or dizziness; rapid or irregular heartbeat.
Some of these symptoms (e.g. "shortness of breath", "coughing") are non-specific and might be misinterpreted as more common or less severe events (e.g. respiratory tract infections).
Other signs of vascular occlusion can include: sudden pain, swelling and slight blue discoloration of an extremity.
If the occlusion occurs in the eye symptoms can range from painless blurring of vision which can progress to loss of vision. Sometimes loss of vision can occur almost immediately.
Risk of arterial thromboembolism (ATE). Epidemiological studies have associated the use of CHCs with an increased risk for arterial thromboembolism (e.g. myocardial infarction, angina pectoris, stroke or TIA).
Arterial thromboembolic events may be fatal.
The risk of arterial thromboembolic complications in CHC users increases in women with risk factors. Leveth 150/30 ED is contraindicated if a woman has one serious or multiple risk factors for ATE that puts her at high risk of arterial thrombosis. If a woman has more than one risk factor, it is possible that the increase in risk is greater than the sum of the individual factors - in this case her total risk should be considered. If the balance of benefits and risks is considered to be negative a CHC should not be prescribed.
Risk factors for ATE.
Increasing age, particularly above 35 years.
Smoking.
Hypertension.
Obesity.
Positive family history (arterial thromboembolism ever in a sibling or parent especially at relatively early age e.g. below 50).
Biochemical factors that may be indicative of hereditary or acquired predisposition for ATE include: hyperhomocysteinaemia and antiphospholipid antibodies (e.g. anticardiolipin antibodies, and lupus anticoagulant).
Migraine.
Other medical conditions associated with adverse vascular events: diabetes mellitus; hyperhomocysteinaemia; valvular heart disease; atrial fibrillation; dyslipoproteinaemia; systemic lupus erythematosus.
Women should be advised not to smoke if they wish to use a CHC. Women over 35 years who continue to smoke should be strongly advised to use a different method of contraception.
If a hereditary predisposition is suspected, the woman should be referred to a specialist for advice before deciding about any CHC use.
An increase in frequency or severity of migraine during CHC use (which may be prodromal of a cerebrovascular event) may be a reason for immediate discontinuation.
Symptoms of ATE.
Women should be informed of the symptoms of ATE and be advised to seek urgent medical attention if ATE symptoms develop and to inform the healthcare professional that she is taking a CHC.
Symptoms of a stroke can include: sudden numbness or weakness of the face, arm or leg, especially on one side of the body; sudden trouble walking, dizziness, loss of balance or coordination; sudden confusion, slurred speech or aphasia; sudden partial or complete loss of vision; diplopia; sudden, severe or prolonged headache with no known cause; loss of consciousness or fainting with or without seizure.
Temporary symptoms suggest the event is a transient ischaemic attack (TIA).
Symptoms of myocardial infarction (MI) can include: pain, discomfort, pressure, heaviness, sensation of squeezing or fullness in the chest, arm, or below the breastbone; discomfort radiating to the back, jaw, throat, arm, stomach; feeling of being full, having indigestion or choking; sweating, nausea, vomiting or dizziness; extreme weakness, anxiety, or shortness of breath; rapid or irregular heartbeats.
Tumours.
The most important risk factor for cervical cancer is persistent as human papilloma virus (HPV) infection. Some epidemiological studies have indicated that long-term use of COCs may further contribute to this increased risk but there continues to be controversy about the extent to which this finding is attributable to confounding effects e.g. cervical screening and sexual behaviour, including use of barrier contraceptives.
A meta-analysis from 54 epidemiological studies reported that there is a slightly increased relative risk (RR = 1.24) of having breast cancer diagnosed in women who are currently taking COCs. The excess risk gradually disappears during the course of the 10 years after cessation of COC use. Because breast cancer is rare in women under 40 years of age, the excess number of breast cancer diagnoses in current and recent COC users is small in relation to the overall risk of breast cancer. These studies do not provide evidence for causation. The observed pattern of increased risk may be due to an earlier diagnosis of breast cancer in COC users, the biological effects of COCs or a combination of both. The breast cancers diagnosed in ever-users tend to be less advanced clinically than the cancers diagnosed in never-users.
In rare cases, benign liver tumours, and even more rarely, malignant liver tumours, have been reported in users of COCs. In isolated cases, these tumours have led to life-threatening intra-abdominal haemorrhages. A liver tumour should be considered in the differential diagnosis when severe upper abdominal pain, liver enlargement or signs of intra-abdominal haemorrhage occur in women taking COCs.
Malignancies may be life-threatening or may have a fatal outcome.
Other conditions.
Women with hypertriglyceridaemia, or a family history thereof, may be at an increased risk of pancreatitis when taking COCs.
Although small increases in blood pressure have been reported in many women taking COCs, clinically relevant increases are rare. However, if a sustained clinically significant hypertension develops during the use of a COC, then it is prudent for the doctor to withdraw the COC and treat the hypertension. Where considered appropriate, COC use may be resumed if normotensive values can be achieved with antihypertensive therapy.
The following conditions have been reported to occur or deteriorate with both pregnancy and COC use, but the evidence of an association with COC use is inconclusive: jaundice and/or pruritus related to cholestasis; gallstone formation; porphyria; systemic lupus erythematosus; haemolytic uraemic syndrome; Sydenham's chorea; herpes gestationis; and otosclerosis-related hearing loss.
In women with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema.
Acute or chronic disturbances of liver function may necessitate the discontinuation of COC use until markers of liver function return to normal. Recurrence of cholestatic jaundice which occurred first during pregnancy or previous use of sex steroids necessitates the discontinuation of COCs.
Although COCs may have an effect on peripheral insulin resistance and glucose tolerance, there is no evidence for a need to alter the therapeutic regimen in women with diabetes taking low dose COCs (containing < 50 microgram ethinylestradiol). However, women with diabetes should be carefully observed while taking COCs.
Crohn's disease and ulcerative colitis have been associated with COC use.
Chloasma may occasionally occur, especially in women with a history of chloasma gravidarum. Women with a tendency to chloasma should avoid exposure to the sun or ultraviolet radiation whilst taking COCs.
Each yellow active tablet contains 84.32 mg lactose and each white placebo tablet contains 89.50 mg lactose anhydrous. Patients with rare hereditary problems of galactose intolerance, Lapp lactase deficiency or glucose-galactose malabsorption who are on a lactose-free diet should take this amount into consideration.
Medical examination/consultation.
A complete medical history and physical examination should be taken prior to the initiation or reinstitution of COC use, guided by Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use, and should be repeated periodically during the use of COCs. In general, an annual examination is recommended. Periodic medical assessment is also of importance because contraindications (e.g. a transient ischaemic attack, etc.) or risk factors (e.g. a family history of venous or arterial thrombosis) may appear for the first time during the use of a COC. The frequency and nature of these assessments should be based on established practice guidelines and be adapted to the individual woman but should generally include special reference to blood pressure, breasts, abdomen and pelvic organs, including cervical cytology, and relevant laboratory tests.
Sexually transmitted infections (STIs) including human immunodeficiency virus (HIV) infections and AIDS.
Leveth 150/30 ED is intended to prevent pregnancy. It does not protect against sexually transmitted infections (STIs), including HIV infections (AIDS). The woman should be advised that additional barrier contraceptive measures are needed to prevent transmission of STIs.
Reduced efficacy.
The efficacy of COCs may be reduced in the event of missed yellow active tablets, vomiting or diarrhoea during active tablet taking (see Section 4.2 Dose and Method of Administration) or concomitant medication (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Reduced cycle control.
With all COCs, irregular bleeding (spotting or breakthrough bleeding) may occur, especially during the first months of use. Therefore, the evaluation of any irregular bleeding is only meaningful after an adaptation interval of about three cycles. If bleeding irregularities persist or occur after previously regular cycles, then non-hormonal causes should be considered and adequate diagnostic measures are indicated to exclude malignancy or pregnancy. These may include curettage.
In some women withdrawal bleeding may not occur during the placebo tablet interval. If the COC has been taken according to the directions described in Section 4.2 Dose and Method of Administration, it is unlikely that the woman is pregnant. However, if the COC has not been taken according to these directions prior to the first missed withdrawal bleed or if two withdrawal bleeds are missed, pregnancy must be ruled out before COC use is continued.
Alanine transaminase (ALT) elevations.
In patients treated with hepatitis C antiviral medications including glecaprevir, pibrentasvir, ombitasvir, paritaprevir or dasabuvir, ALT elevations may occur in women using ethinylestradiol-containing medications such as CHCs. Prescribers should consult the relevant antiviral medicine product safety information. Patients taking a CHC should therefore be switched to an alternative method of contraception (e.g. progestogen-only contraception or non-hormonal methods) prior to starting therapy.
Use in renal impairment.
COCs have not been specifically studied in renally-impaired patients.
Use in hepatic impairment.
COCs are contraindicated in women with severe hepatic disease as long as liver function values have not returned to normal (see Section 4.3 Contraindications).
Use in the elderly.
COCs are not indicated after menopause.
Paediatric use.
COCs are only indicated after menarche.
Effects on laboratory tests.
The use of contraceptive steroids may influence the results of certain laboratory tests, including biochemical parameters of liver, thyroid, adrenal and renal function, plasma levels of carrier proteins, e.g. corticosteroid binding globulin and lipid/lipoprotein fractions, parameters of carbohydrate metabolism and parameters of coagulation and fibrinolysis. Changes generally remain within the normal laboratory range.4.5 Interactions with Other Medicines and Other Forms of Interactions
Note.
The prescribing information of concomitant medications should be consulted to identify potential interactions.
Interactions between oral contraceptives and other medicines may lead to breakthrough bleeding and/or oral contraceptive failure. Enzyme Induction (increase of hepatic metabolism).
Interactions can occur with medicines that induce microsomal enzymes (e.g. cytochrome P450 enzymes, CYP34A, which can result in increased clearance of sex hormones and may lead to breakthrough bleeding and/or contraceptive failure.
Enzyme induction can already be observed after a few days of treatment. Maximal enzyme induction is generally seen within a few weeks. After the cessation of drug therapy enzyme induction may be sustained for about 4 weeks.
Women prescribed any of these medicines should temporarily use a barrier method in addition to the COC or choose another method of contraception. The barrier method should be used during the time of concomitant medicine administration and for 28 days after their discontinuation. If the period in which the barrier method is used runs beyond the end of the active tablets in the COC pack, the white placebo tablets should be omitted and the next COC pack started.
Women taking interacting medications on a chronic basis should consider another method of contraception.
Antibiotics (interference with enterohepatic circulation).
Some clinical reports suggest that enterohepatic circulation of estrogens may decrease when certain antibiotic agents are given, which may reduce ethinylestradiol concentrations (e.g. penicillins, tetracyclines).
Women prescribed antibiotics (except rifampicin and griseofulvin) should use the barrier method until 7 days after completing a course of antibiotics. If the period in which the barrier method is used runs beyond the end of the active tablets in the COC pack, the white placebo tablets should be omitted and the next COC pack started.
Substances increasing the clearance of COCs (diminished efficacy of COCs by enzyme-induction), e.g.
Phenytoin, barbiturates, primidone, carbamazepine, rifampicin and possibly also oxcarbazepine, topiramate, felbamate, griseofulvin and herbal medicines containing St John's wort (Hypericum perforatum).
Substances with variable effects on the clearance of COCs, e.g.
When co-administered with COCs, many human immunodeficiency virus (HIV)/hepatitis C virus (HCV) protease inhibitors (e.g. ritonavir), non-nucleoside reverse transcriptase inhibitors (e.g. nevirapine) can increase or decrease plasma concentration of estrogen or progestogen. These changes may be clinically relevant in some cases and combinations of them have been reported to potentially affect hepatic metabolism.
Substances decreasing the clearance of COCs (enzyme inhibitors).
Strong and moderate CYP3A4 inhibitors such as azole antifungals (e.g. itraconazole, voriconazole, fluconazole), verapamil, macrolides (e.g. clarithromycin, erythromycin), diltiazem and grapefruit juice can increase plasma concentrations of the estrogen or the progestin or both.
Etoricoxib doses of 60 to 120 mg/day have been shown to increase plasma concentrations of ethinylestradiol 1.4 to 1.6-fold, respectively when taken concomitantly with a combined hormonal contraceptive containing 0.035 mg ethinylestradiol.
Influence of levonorgestrel/ethinylestradiol on other medicines.
Oral contraceptives may affect the metabolism of certain other medicines. Accordingly, plasma and tissue concentrations may either increase (e.g. cyclosporin) or decrease (e.g. lamotrigine).
In vitro, ethinylestradiol is a reversible inhibitor of CYP2C19, CYP1A1 and CYP1A2 as well as a mechanism-based inhibitor of CYP3A4/5, CYP2C8, and CYP2J2. In clinical studies, administration of a hormonal contraceptive containing ethinylestradiol lead to no, or a weak increase in CYP3A4 substrates (e.g. midazolam) and a weak (e.g. theophylline) to moderate (e.g. melatonin, tizanidine) increase of CYP1A2 substrates.
Pharmacodynamic interactions.
Co-administration of ethinylestradiol-containing medicinal products with direct-acting antiviral (DAA) medicinal products containing ombitasvir, paritaprevir, or dasabuvir, and combinations of these has been shown to be associated with increases in alanine aminotransferase (ALT) levels to greater than 20 times the upper limit of normal in healthy female subjects and HCV infected women (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use). ALT elevations have also been observed with HCV anti-viral medicinal products including glecaprevir/pibrentasvir. Patients taking a CHC should therefore be switched to an alternative method of contraception (e.g. progestogen-only contraception or non-hormonal methods) prior to starting therapy.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category B3)
COCs are contraindicated during pregnancy. If pregnancy occurs during treatment, further intake must be stopped immediately.
Epidemiological studies have found no significant effects on foetal development in children born to women who used COCs prior to pregnancy, nor a teratogenic effect when COCs were taken inadvertently during early pregnancy.
Lactation may be influenced by COCs as they may reduce the quantity and change the composition of breast milk. Small amounts of the contraceptive steroids and/or their metabolites may be excreted with the milk. Therefore, the use of COCs should generally not be recommended until the nursing mother has completely weaned her child.4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration. However, adverse effects of Leveth 150/30 ED include dizziness which could affect the ability to drive or use machines (see Section 4.8 Adverse Effects (Undesirable Effects)).
4.8 Adverse Effects (Undesirable Effects)
Various adverse reactions have been associated with oral contraceptive use. The most commonly reported adverse reactions with Leveth 150/30 ED are nausea, abdominal pain, increased weight, headache, depressed mood, altered mood, breast pain and breast tenderness. They occur in ≥ 1% of users.
Serious adverse reactions are arterial and venous thromboembolism.
The most serious reactions associated with the use of oral contraceptives are discussed under Section 4.4 Special Warnings and Precautions for Use.
In the event of aggravation, exacerbation or first appearance of any of these conditions or risk factors, the woman should contact her doctor. The doctor should then decide on whether its use should be discontinued.
The following adverse reactions have been reported in users of COCs and the association has been neither confirmed nor refuted.
Genital tract.
Changes in vaginal discharge (e.g. due to vaginitis).
Breast.
Breast tenderness, breast pain, breast hypertrophy and breast discharge.
Gastrointestinal tract.
Nausea, diarrhoea, abdominal pain and vomiting.
Skin.
Various skin disorders (e.g. acne, hirsutism, alopecia, rash, urticaria, erythema nodosum, erythema multiforme).
Eyes.
Contact lens intolerance, cataract.
CNS.
Headache, migraine, depressive moods, mood altered and change in libido.
Metabolic.
Fluid retention, and change in body weight.
Body as a whole.
Hypersensitivity reaction.
Vascular disorders.
Venous and arterial thromboembolic events (peripheral deep venous occlusion, thrombosis, and embolism/pulmonary vascular occlusion, thrombosis, embolism, and infarction/myocardial infarction/cerebral infarction and stroke not specified as haemorrhagic).
In women with hereditary angioedema exogenous estrogens may induce or exacerbate symptoms of angioedema.
(Also see Section 4.4 Special Warnings and Precautions for Use, Effects on laboratory tests).
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.4.9 Overdose
There have been no reports of serious deleterious effects from overdose. Symptoms that may occur in this case are: nausea, vomiting and withdrawal bleeding. The last may even occur in girls before their menarche, if they have accidentally taken the medicinal product. There are no antidotes and further treatment should be symptomatic.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
The contraceptive effect of COCs is based on the interaction of various factors. The primary mechanisms are inhibition of ovulation (by suppression of gonadotropins) and changes in the cervical secretion (blocking the entry of sperm into the uterus).
As well as protection against pregnancy, COCs have several positive properties which, next to the negative properties (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)), can be useful in deciding on the method of birth control. For the majority of users, the cycle is more regular, the menstruation is often less painful and bleeding is lighter.
The latter may result in a decrease in the occurrence of iron deficiency. In addition, with the higher-dosed COCs (> 35 microgram ethinylestradiol), there is evidence of a reduced risk of fibrocystic tumours of the breasts, ovarian cysts, pelvic inflammatory disease, ectopic pregnancy, endometrial and ovarian cancer and a decreased incidence and severity of acne. These additional benefits have only been established in case control and cohort studies. Results from randomised control trials are not available. Whether this also applies to lower-dosed COCs remains to be confirmed.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Levonorgestrel.
Absorption.
Orally administered levonorgestrel is rapidly and completely absorbed. Peak serum concentrations of about 3 - 4 nanogram/mL are reached at about 1 hour after single ingestion. Levonorgestrel is almost completely bioavailable after oral administration. In a study comparing the AUC level following 90 microgram levonorgestrel administration orally with same dose administered intravenously in 18 healthy women, the absolute bioavailability obtained was 82%.
Distribution.
Levonorgestrel is bound to serum albumin and to sex hormone binding globulin (SHBG). Only 1.3% of the total serum drug concentration is present as free steroid, approximately 64% is specifically bound to SHBG and about 35% is non-specifically bound to albumin. The ethinylestradiol-induced increase in SHBG influences the proportion of levonorgestrel bound to the serum proteins, causing an increase of the SHBG-bound fraction and a decrease of the albumin-bound fraction. The apparent volume of distribution of levonorgestrel is about 184 L after single administration.
Metabolism.
Levonorgestrel is extensively metabolised. The major metabolites in plasma are the unconjugated and conjugated forms of 3α, 5β-tetrahydrolevonorgestrel. Additionally, based on in vitro and in vivo studies, CYP3A4 is the main enzyme involved in the oxidative metabolism of levonorgestrel. The clearance rate from serum is approximately 1.3 - 1.6 mL/min/kg.
Excretion.
Levonorgestrel serum levels decrease in two phases, which are characterised by half-lives of approximately 1 hour and about 20 hours, respectively. Levonorgestrel is not excreted in unchanged form. Its metabolites are excreted at about equal parts via urine and faeces. The half-life of metabolite excretion is about 1 day.
Steady state conditions.
Following daily ingestion, drug serum levels increase about 3 - 4 fold reaching steady state conditions during the second half of a treatment cycle. Levonorgestrel pharmacokinetics are influenced by SHBG levels, which are increased about 1.7 fold after daily oral administration of levonorgestrel/ ethinylestradiol tablets. This effect leads to a reduction of the clearance rate to about 0.7 mL/min/kg at steady-state.
Ethinylestradiol.
Absorption.
Orally administered ethinylestradiol is rapidly and almost completely absorbed. Peak serum concentrations of about 95 picogram/mL are reached within 1 - 2 hours. Absolute bioavailability, as a result of pre-systemic conjugation and first pass metabolism, is approximately 60%.
Distribution.
Ethinylestradiol is highly but non-specifically bound to serum albumin (approximately 98.5%) and induces an increase in the serum concentrations of SHBG. An apparent volume of distribution of approximately 5 L/kg was reported.
Metabolism.
Ethinylestradiol is subject to pre-systemic conjugation in both small bowel mucosa and the liver. Ethinylestradiol is primarily metabolised by aromatic hydroxylation, but a wide variety of hydroxylated and methylated metabolites are formed. These are present as free metabolites and as conjugates with glucuronides and sulfate. The metabolic clearance rate is approximately 5 mL/min/kg.
Excretion.
Ethinylestradiol serum levels decrease in two phases; the terminal disposition phase is characterised by a half-life of approximately 24 hours. Ethinylestradiol is not excreted as unchanged drug. Ethinylestradiol metabolites are excreted at a urinary to biliary ratio of 4:6. The half-life of metabolite excretion is approximately 1 day.
Steady state conditions.
Ethinylestradiol serum concentrations increase slightly after daily oral administration of levonorgestrel/ethinylestradiol tablets. The maximum concentrations are approximately 114 picogram/mL at the end of a treatment cycle. According to the variable half-life of the terminal disposition phase from serum and the daily ingestion, steady-state serum levels of ethinylestradiol will be reached after about one week.
5.3 Preclinical Safety Data
Genotoxicity.
There is limited evidence available in the literature suggesting that estrogens may be weakly genotoxic at high doses. Ethinylestradiol was negative in studies for DNA-adduct formation in cultured human liver slices and in assays for gene mutations (bacterial or mammalian cells in vitro) and gave equivocal results in assays for chromosomal damage (clastogenic effects were not consistently seen and occurred at high doses).
The genotoxic potential of levonorgestrel has not been fully investigated, although limited data available to date suggest that it did not appear to be genotoxic.
Carcinogenicity.
Long-term continuous administration of natural and synthetic estrogens in certain animal species increases the frequency of carcinomas of the breast, uterus, cervix, vagina, testis and liver. A long-term study with levonorgestrel in dogs showed an increased incidence of mammary tumours, although a similar effect was not apparent in studies in mice, rats or monkeys. The occurrence of these mammary tumours in dogs may be due in part to a hormonal feedback mechanism. The clinical relevance of these findings is uncertain.
Numerous epidemiological studies have been conducted to determine the incidence of breast, endometrial, ovarian and cervical cancer in women taking COCs. Some of these studies have shown an increased relative risk of breast cancer in certain subgroups of COC users. Women with a strong family history of breast cancer or who have breast nodules, fibrocystic disease or abnormal mammograms should be monitored with particular care. Benign hepatic adenomas have been found to be associated with the use of oral contraceptives. Although benign, hepatic adenomas may rupture and cause death through intra-abdominal haemorrhage. Some epidemiological studies also suggest that COC use has been associated with an increase in the risk of cervical intraepithelial neoplasia in some populations of women, although there continues to be controversy about the extent to which this finding is attributable to the confounding effects of sexual behaviour and other factors, such HPV. It must also be borne in mind that sexual steroids can promote the growth of certain hormone-dependent tissues and tumours (see Section 4.4 Special Warnings and Precautions for Use).6 Pharmaceutical Particulars
6.1 List of Excipients
Each yellow active tablet contains the following inactive ingredients: lactose monohydrate, povidone, crospovidone, magnesium stearate, Opadry II complete film coating system 85F32450 yellow.
Each white placebo tablet in Leveth 150/30 ED contains: lactose, povidone, magnesium stearate, Opadry II complete film coating system 85F18422 white.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Each blister platform (PVC/PVdC/Al) contains 21 active tablets and 7 placebo tablets (28 tablets). Blister platforms are packed within a carton.
Each carton contains 1 x 28 or 4 x 28 tablets.
Not all pack sizes may be available.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Levonorgestrel is a white or almost white, odourless or almost odourless, crystalline powder. It is practically insoluble in water; slightly soluble in alcohol, acetone, and ether; soluble in chloroform; and sparingly soluble in methylene chloride.
Ethinylestradiol is a white to creamy white, odourless, crystalline powder. It is practically insoluble in water and soluble in alcohol, chloroform, ether, vegetable oils and aqueous solutions of alkali hydroxides.
Chemical structure.
Levonorgestrel.
 Chemical name: 13β-Ethyl-17β-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one.
Chemical name: 13β-Ethyl-17β-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one.
Molecular formula: C21H28O2.
Molecular weight: 312.5.
Ethinylestradiol.
 Chemical name: 19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17β-diol.
Chemical name: 19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17β-diol.
Molecular formula: C20H24O2.
Molecular weight: 296.4.
CAS number.
Levonorgestrel.
797-63-7.
Ethinylestradiol.
57-63-6.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes




 The increased risk of VTE during the postpartum period must be considered if re-starting Leveth 150/30 ED (see Section 4.2 Dose and Method of Administration; Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy, Use in lactation).
The increased risk of VTE during the postpartum period must be considered if re-starting Leveth 150/30 ED (see Section 4.2 Dose and Method of Administration; Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy, Use in lactation). Chemical name: 13β-Ethyl-17β-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one.
Chemical name: 13β-Ethyl-17β-hydroxy-18,19-dinor-17α-pregn-4-en-20-yn-3-one. Chemical name: 19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17β-diol.
Chemical name: 19-Nor-17α-pregna-1,3,5(10)-trien-20-yne-3,17β-diol.