1 Name of Medicine
Levothyroxine sodium.
2 Qualitative and Quantitative Composition
Each Levothox tablet contains levothyroxine sodium as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Oral tablets.
Levothox 25 microgram.
Round, orange coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "1" on one side and plain on the other side.
Levothox 50 microgram.
Round, white coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "2" on one side and plain on the other side.
Levothox 75 microgram.
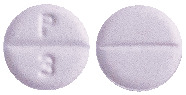
Round, violet coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "3" on one side and plain on the other side.
Levothox 88 microgram.
Round, olive coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "4" on one side and plain on the other side.
Levothox 100 microgram.
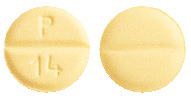
Round shaped, yellow coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "14" on one side and plain on the other side.
Levothox 112 microgram.
Round, rose coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "6" on one side and plain on the other side.
Levothox 125 microgram.
Round, brown coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "7" on one side and plain on the other side.
Levothox 137 microgram.
Round, turquoise coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "8" on one side and plain on the other side.
Levothox 150 microgram.
Round, blue coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "9" on one side and plain on the other side.
Levothox 175 microgram.
Round, lilac coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "10" on one side and plain on the other side.
Levothox 200 microgram.
Round, pink coloured, uncoated tablets, with a break line on both sides. Debossed with "P" and "11" on one side and plain on the other side.4.1 Therapeutic Indications
Levothox is indicated for the management of demonstrated thyroid hormone deficiency.
Levothox is also used to suppress thyrotropin (TSH) for the management of TSH-responsive tumours of the thyroid.
4.2 Dose and Method of Administration
Levothyroxine is best taken as a single daily dose first thing in the morning. It should be taken with water and on an empty stomach, and at least 30 minutes and preferably 60 minutes before the intake of any food or other medications. Levothyroxine is best ingested in the fasting state, as food will impair absorption.
The dose should be individualised on the basis of clinical response and biochemical tests. Regular monitoring of TSH and levothyroxine is recommended when starting therapy or changing the dose.
Adults.
Initial dose.
Commence with levothyroxine 50 to 100 microgram daily. Increase the daily dose by 25 to 50 microgram according to response at not less than 4-weekly intervals, up to 100 to 200 microgram daily.
In patients aged 60 years and over and in those with ischaemic heart disease, levothyroxine therapy should normally be initiated with low doses (25 or 50 microgram/day).
Wherever possible, whole tablets should be taken (e.g. if the dose required is 150 microgram then 1 x 100 microgram tablet and 1 x 50 microgram tablet should be taken, rather than 1 ½ x 100 microgram tablets).
When there is no contraindication to full thyroxine replacement, a TSH level of about 1 mU/L with a serum thyroxine level in the high-normal range, usually indicates optimal therapy.
Maintenance doses.
Adults.
100 to 200 microgram per day.
Children.
See Table 1.

4.3 Contraindications
Known hypersensitivity to thyroxine, which has been described rarely.
Untreated hyperthyroidism.
Uncorrected primary or secondary adrenal insufficiency.
Thyrotoxicosis.
Acute myocardial infarction uncomplicated by hypothyroidism.
Acute myocarditis.
Acute pancarditis.
During pregnancy, combination of levothyroxine and antithyroid agents for the treatment of hyperthyroidism is contraindicated. (see Section 4.6 Fertility, Pregnancy and Lactation).
4.4 Special Warnings and Precautions for Use
Do not interchange Levothox and other levothyroxine containing products. If a decision is made to switch from another levothyroxine product to Levothox, then prescribers should be aware that dose adjustment may be required; TSH should be monitored.
Initiation of therapy.
In the elderly or patients with ischaemic heart disease, Levothox should not be initiated at more than 50 microgram/day, and dose then be gradually increased. (See Section 4.2 Dose and Method of Administration).
Presence of cardiac disorder.
Extreme caution is required in patients with a cardiovascular disorder. In the event of cardiovascular effects, the dosage of Levothox should be lowered. Even smaller initial dosage (e.g. 12.5 to 25 microgram/day) should be used with increments of not more than 25 microgram/day at not less than two weeks intervals. If this routine is not tolerated because of angina, increments should be further reduced with prolongation of the intervals between changes. The use of a β-blocker may help to control angina.
Cortisone deficiency.
Corticosteroid replacement therapy must precede initiation of Levothox therapy to avoid Addisonian crisis in such conditions as hypopituitarism and adrenal insufficiency.
Effects on bone mineral density.
In women, long-term levothyroxine sodium therapy has been associated with increased bone resorption, thereby decreasing bone mineral density, especially in post-menopausal women on greater than replacement doses or in women who are receiving suppressive doses of levothyroxine sodium. The increased bone resorption may be associated with increased serum levels and urinary excretion of calcium and phosphorus, elevations in bone alkaline phosphate and suppressed serum parathyroid hormone levels. Therefore, it is recommended that patients receiving levothyroxine sodium be given the minimum dose necessary to achieve and desired clinical and biochemical response.
Diabetes.
Caution is also required when levothyroxine is given to hypothyroid patients with diabetes mellitus or diabetes insipidus, as it may cause the required dosage of insulin and oral antidiabetic agents to be increased. Careful monitoring of diabetic control is recommended, especially when Levothox therapy is initiated, changed or discontinued. Adjustments in the dosage of these agents should only be made accordingly if necessary.
Hyperthyroidism.
Lower doses of levothyroxine may be required in patients with a history of hyperthyroidism, as such patients may have residual autonomous thyroid function.
Thyrotoxicosis.
Patients who have thyrotoxicosis who are being treated with anti-thyroid medication, may have increased sensitivity to levothyroxine.
Long-standing hypothyroidism and myxoedema.
Caution is also required for patients with long-standing hypothyroidism or myxoedema, as they are more sensitive to thyroid hormones.
Levothyroxine should not be used for the treatment of obesity or weight loss.
In euthyroid patients, doses within the range of daily hormonal requirements are ineffective for weight reduction. Larger doses may produce serious or even life threatening manifestations of toxicity, particularly when given in association with sympathomimetic amines such as those used for anorectic effects.
Malabsorption syndromes.
Levothyroxine absorption is decreased in patients with malabsorption syndromes. It is advised to treat the malabsorption condition to ensure effective levothyroxine treatment with regular levothyroxine dose.
Use in hepatic impairment.
In spite of the major involvement of the liver in levothyroxine metabolism, there is no evidence that dosage should be modified in the presence of cirrhosis. However, thyroid function tests may be influenced and need careful interpretation.
Use in renal impairment.
There is no evidence that levothyroxine dosage should be modified in the presence of renal failure. However, thyroid function tests may be influenced and need careful interpretation.
Use in the elderly.
These patients may be more sensitive to the effects of thyroid hormones. Levothox should be gradually introduced in the elderly and in those with long-standing hypothyroidism, so that any sudden increases in metabolic demands may be avoided. Individualisation of dosage is recommended and caution is required, as occult cardiac disease may be present.
Paediatric use.
Studies performed have not yet demonstrated paediatric-specific problems that would limit the usefulness of thyroid hormones in children. However, neonates should be carefully observed for evidence of altered thyroid functions. This caution is required, as the infant pituitary gland is relatively insensitive to negative feedback effects of thyroid hormones. The parents of children who are receiving the thyroid agent need to be aware that partial loss of hair may occur during the first few months of therapy. However, this effect is usually transient and subsequent regrowth usually occurs.
Haemodynamic parameters should be monitored when levothyroxine therapy is initiated in very low birth weight preterm neonates as circulatory collapse may occur due to the immature adrenal function.
Effects on laboratory tests.
Biotin may interfere with thyroid immunoassays that are based on a biotin/streptavidin interaction, leading to either falsely decreased or falsely increased test results. The risk of interference increases with higher doses of biotin.
When interpreting results of laboratory tests, possible biotin interference has to be taken into consideration, especially if a lack of coherence with the clinical presentation is observed in patients taking biotin-containing products, laboratory personnel should be informed when a thyroid function test is requested. Alternative tests not susceptible to biotin interference should be used, if available.4.5 Interactions with Other Medicines and Other Forms of Interactions
Oral anticoagulants, coumarin or indandione derivative e.g. warfarin.
Depending on the thyroid status of the patient, concurrent use of oral anticoagulants with levothyroxine will increase the therapeutic effects of oral anticoagulants. Therefore, an increase in the dosage of levothyroxine may necessitate a decrease in the oral anticoagulant dosage. Adjustment of oral anticoagulant dosage is recommended on the basis of prothrombin time. Patients should be observed closely for adverse effects.
SSRIs e.g. sertraline.
The effects of levothyroxine in hypothyroid patients may be decreased by concomitant use of sertraline, therefore resulting in an increase in levothyroxine requirements.
Insulin and antidiabetic agents e.g. sulfonylurea.
Levothyroxine may increase the required dosage of insulin and other oral antidiabetic drugs. Therefore, careful monitoring of diabetic control is recommended.
Beta-adrenergic blocking agents e.g. propranolol.
These agents may decrease the peripheral conversion of thyroxine to tri-iodothyronine.
Ion-exchange resins e.g. cholestyramine, sodium polystyrene sulphonate or colestipol.
Due to concurrent use of ion-exchange resins with levothyroxine, the effects of levothyroxine may be reduced due to the resin binding to levothyroxine in the gastrointestinal tract, causing a delay or impairment in levothyroxine absorption. An interval of 4 to 5 hours between the administration of the two medications is recommended.
Corticosteroids e.g. prednisolone and dexamethasone.
The clearance of corticosteroids may be increased in hyperthyroid patients, and decreased in hypothyroid patients, solely due to the administration, changes in dosage and discontinuation of levothyroxine. Therefore, corticosteroid dosage may need to be adjusted.
Oestrogen.
In patients with a non-functioning thyroid gland, oestrogen may increase the serum thyroxine-binding globulin, therefore generating an increase in levothyroxine requirements.
Antiepileptics e.g. phenytoin, carbamazepine and barbiturates.
These agents may increase the hepatic degradation of levothyroxine, therefore resulting in an increase in levothyroxine requirements.
Ritonavir.
Ritonavir may interact with levothyroxine, therefore resulting in an increase in levothyroxine requirements.
Antimalarials.
The combined use of chloroquine and proguanil may increase the hepatic degradation of levothyroxine, therefore resulting in an increase in levothyroxine requirements.
Antibacterials e.g. rifampicin and ciprofloxacin.
Rifampicin may increase the hepatic degradation of levothyroxine, therefore resulting in an increase in levothyroxine requirements.
Oral ciprofloxacin may decrease the absorption of levothyroxine. An interval of 6 hours between the administration of the two medications is recommended.
Androgens and anabolic steroids.
Androgens may decrease the concentration of the serum thyroxine-binding globulin, therefore generating a decrease in levothyroxine requirements.
Ketamine.
Cautious administration of ketamine is recommended in patients on levothyroxine therapy, as marked hypertension and tachycardia may occur.
Lithium.
Due to the direct action of lithium on the thyroid gland, inhibition of thyroid hormones may result, leading to clinical hypothyroidism.
Tricyclic antidepressants.
Due to concurrent use with levothyroxine, an increase in the sensitivity to catecholamines may occur, therefore increasing the therapeutic and toxic effects of both drugs.
Sympathomimetics.
Due to concurrent use with levothyroxine, there may be an increase in the effects of both drugs, which may lead to a risk of coronary insufficiency.
Digoxin.
Levothyroxine may reduce the clinical effects of digoxin.
Medicines that (partially) inhibit the peripheral transformation of T4 to T3.
Propranolol, amiodarone, lithium, iodide, oral contrast agents, propylthiouracil and glucocorticoids can occasionally decrease the peripheral conversion of thyroxine to tri-iodothyronine. However, any dose adjustment should be based on TSH levels.
Weight loss drugs.
Orlistat may decrease levothyroxine absorption which may result in hypothyroidism. To avoid this orlistat and levothyroxine should be administered at least 4 hours apart. Regular monitoring for changes in thyroid function is required.
Tyrosine kinase inhibitors.
Treatment with tyrosine kinase inhibitors (e.g. imatinib and sunitinib) has been associated with increased levothyroxine requirements in patients with hypothyroidism.
Statins.
There have been reports that some HMG-CoA reductase inhibitors (statins) such as simvastatin and lovastatin may increase thyroid hormone requirements in patients receiving levothyroxine treatment. It is not known whether this is the case with all statins. Thus, during concomitant use of statins, close monitoring of thyroid function and appropriate adjustment of the levothyroxine dose may be required.
St John's wort.
Medicinal products containing St John's Wort (Hypericum perforatum L.) may increase the hepatic clearance of levothyroxine, which may lead to reduced serum concentrations of thyroid hormone. Therefore, patients receiving thyroid replacement therapy may need an increase in the thyroid hormone dose when these medicinal products are used concomitantly.
Biotin-containing products.
Biotin may interfere with immunoassays for assessing thyroid function based on a biotin/streptavidin interaction, thus leading to falsely decreased or falsely increased test results (see Section 4.4 Special Warnings and Precautions for Use).
In addition.
Levothyroxine can enhance the clinical effects of pentobarbitone and dihydrotachysterol. Therefore, the adjustment of dosage may be necessary.
The clinical effect of levothyroxine can be reduced by soya flour, sucralfate, calcium-, aluminium-, magnesium-, iron supplements, lanthanum, sevelamer, and proton pump inhibitors - which interfere with absorption from the gastrointestinal tract. If these substances are taken, then their ingestion should be separated by several hours from the ingestion of levothyroxine.
Soy-containing compounds and high fibre diets can decrease the intestinal absorption of levothyroxine. Therefore, a dosage adjustment of levothyroxine may be necessary, in particular at the beginning or after termination of nutrition with soy supplements.
Co-administration of tamoxifen, methadone and 5-fluorouracil may increase the serum concentration of thyroxine-binding globulin and thus increase levothyroxine requirements.
Thyroid function tests can be modified, without changes in clinical effect of levothyroxine, by some NSAIDs, salicylates, diazepam, heparin and fenclofenac.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There is no information available on the possible effects of levothyroxine on human fertility.
(Category A)
If overt hypothyroidism is diagnosed during pregnancy, thyroid function test results should be normalised as rapidly as possible. In newly-diagnosed hypothyroidism in pregnancy, levothyroxine dosage should be titrated rapidly, for example 1.5-2.0 microgram/kg/day may be required for initial replacement. If hypothyroidism has been diagnosed before pregnancy, levothyroxine therapy should be optimised before conception and monitored during pregnancy by measurement of serum TSH and levothyroxine levels. The levothyroxine dose commonly needs incremental adjustments by 4-6 weeks of gestation and may require a 25-40% increase in dosage. It is recommended that those levels should be re-evaluated every 3 to 4 weeks during the first and second trimesters, with levothyroxine dosage changes as appropriate. The requirement is likely to decrease post-partum.
Monitoring of TSH concentrations can give guidance. Thyroxine-binding globulin (TBG) increases during pregnancy and therefore total T4 and T3 may appear to be elevated. Measurement of free T4 and T3 may be more appropriate. There is contradictory evidence concerning the passage of T4 and T3 across the placenta but it is unlikely that the fetus is at risk. Clinical experience does not indicate any adverse effects on the fetus when levothyroxine is administered during pregnancy.
During pregnancy, combination of levothyroxine and antithyroid agents for the treatment of hyperthyroidism is contraindicated.
Australian categorisation definition of Category A.
Drugs which have been taken by a large number of pregnant women and women of childbearing age without any proven increase in the frequency of malformations or other direct or indirect harmful effects on the fetus having been observed.
Women who are breast feeding should continue to take Levothox. In euthyroid women, breast milk contains negligible amounts of thyroid hormone.4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Individual patients vary in response to both the maintenance dose of levothyroxine and to the size and frequency of dose increments. Too large an increment or too high a replacement dose can lead to manifestations of thyrotoxicosis which include:
Cardiovascular.
Chest pain, increased blood pressure, tachycardia, cardiac arrhythmias, palpitations, angina pectoris, myocardial ischaemia, myocardial infarction, cardiac failure, death.
Nervous system.
Irritability, anxiety, nervousness, agitation, restlessness, tremors, headache, poor concentration, affect lability, sleep disturbance, insomnia, mania, psychosis, psychotic depression, seizures, petit mal status epilepticus, benign intracranial hypertension (especially in children).
Gastrointestinal system.
Abdominal pain, nausea, diarrhoea, vomiting, malabsorption.
Immune system.
Hypersensitivity reactions such as rash, pruritus, anaphylactic reactions.
Skin.
Warmth, erythema, telangiectasia, hyperhidrosis, alopecia, hyperpigmentation, angioedema, rash, urticaria.
Respiratory system.
Increased minute ventilation, tachypnoea, and dyspnoea.
Neuromuscular system.
Myopathy, lid lag, muscle weakness, muscle spasm, epiphyses premature fusion (in children).
Reproductive system.
Amenorrhoea, menstruation irregular, decreased libido, gynaecomastia (in male), infertility.
Metabolic.
Pyrexia, glucose intolerance, weight loss, premature craniosynostosis (in children), TRH suppression, temperature intolerance, flushing, fatigue and increased appetite.
Endocrine system.
Hyperthyroidism.
Investigations.
Decreased bone mineral density.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Within three to six days after ingestion, any or all of the symptoms and signs listed (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)) may become evident. They may progress to "thyroid storm", with hyperpyrexia, convulsions, heart failure, coma and subsequent death.
Treatment of overdose.
Early treatment has included gastric lavage, induced emesis and ingestion of activated charcoal. Oxygen may need to be administered and ventilation may need to be maintained. Treatment is usually symptomatic and supportive. Measures to control fever, hypoglycemia or fluid loss should be initiated as necessary. Of various adrenergic β-blockers, propranolol has been used commonly to control cardiac arrhythmia and other manifestations. Reserpine, guanethidine, and digoxin have also been used. Exchange transfusion has been recommended for progressive deterioration.
When overdose does occur, there must be an extended follow-up period as symptoms may be delayed for several days due to the gradual peripheral conversion of thyroxine to tri-iodothyronine.5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Levothox is used as thyroid replacement therapy for the treatment of hypothyroidism. The principal pharmacological effect of thyroid hormones is to increase the metabolic rate of body tissues. Thyroid hormones are also involved in the regulation of cell growth and differentiation. Thyroxine is the major component of normal secretions of the thyroid gland; therefore, it is the essential determinant of normal thyroid function.
Clinical trials.
Comparison to reference registered products (Eutroxsig/Oroxine).
A single dose, 2-period, 2-treatment, crossover study compared the T4 levels of 31 healthy, fasting adults after a supra-therapeutic dose of levothyroxine (600 microgram) versus the same dose of Oroxine (600 microgram). For uncorrected total T4, the results for the ratio of levothyroxine/Oroxine were AUC0-t=89.1% (90% CI: 84.7%, 93.7%); Cmax=86.2% (82.1%, 90.6%). This suggests that levothyroxine may be less bioavailable than Oroxine.
5.2 Pharmacokinetic Properties
Absorption.
Levothyroxine sodium is variably but adequately absorbed from the gastrointestinal tract following oral administration. Approximately 50 to 75% of levothyroxine sodium is absorbed. Studies in humans indicate that levothyroxine sodium is absorbed from the jejunum and ileum, and in the duodenum. Fasting will only increase the extent of absorption, whereas malabsorption will only cause a decrease in absorption. Following the initiation of therapy, levothyroxine sodium has a slow onset of action as the peak therapeutic effect occurs between 3 to 4 weeks. It also has a long duration of action occurring between 1 to 3 weeks, even following the discontinuance of the drug.
Distribution.
Levothyroxine sodium apparently undergoes enterohepatic circulation. Once inside the circulation, levothyroxine is extensively protein bound, principally to thyroxine-binding globulin (TBG) and also to a lesser extent to thyroxine-binding pre-albumin (TBPA) or to albumin. Levothyroxine is distributed into most body tissues and fluids with the highest concentration occurring in the liver and kidneys. A minimal amount of levothyroxine is distributed into breast milk.
Metabolism.
Levothyroxine sodium has a plasma half-life in euthyroidism of about 6 to 7 days. In hypothyroidism, the half-life is prolonged between 9 to 10 days. However, the half-life is reduced between 3 to 4 days in hyperthyroidism. Levothyroxine is primarily metabolised in the liver and in the kidney to tri-iodothyronine. Approximately 40% of levothyroxine sodium is metabolised to the inactive reverse tri-iodothyronine, which both undergo further deiodination to inactive metabolites. About 85% of the levothyroxine sodium metabolised daily is deiodinated.
Excretion.
Levothyroxine is reported to undergo enterohepatic recycling and is excreted in faeces.
5.3 Preclinical Safety Data
Genotoxicity.
No data available.
Carcinogenicity.
Animal studies to determine the carcinogenic or mutagenic potential of thyroid agents have not been established. However, there is epidemiological evidence against the use of thyroid supplements enhancing the risk of breast cancer.6 Pharmaceutical Particulars
6.1 List of Excipients
Levothox tablets contain the following excipients: microcrystalline cellulose, light magnesium oxide, sodium starch glycollate and sodium stearylfumarate. They also contain Lake Blend colouring agents as below:
25 microgram tablets.
Sunset yellow FCF Aluminium lake.
75 microgram tablets.
Lake Blend LB-505008 Purple (ID#110747).
88 microgram tablets.
Lake Blend LB-510028 Green (ID#117692).
100 microgram tablets.
Lake Blend LB-520044 Yellow (ID#117693).
112 microgram tablets.
Lake Blend LB-540042 Pink (ID#117694).
125 microgram tablets.
Lake Blend LB-575003 Brown (ID#110743).
137 microgram tablets.
Brilliant Blue FCF Aluminium Lake.
150 microgram tablets.
Indigo Carmine Aluminium Lake.
175 microgram tablets.
Lake Blend LB-500017 Purple (ID#117689).
200 microgram tablets.
Allura Red AC Aluminium Lake.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Store in the original container. Keep container tightly closed.
6.5 Nature and Contents of Container
Levothox is approved for supply in blister packs of 100 tablets. Blisters are made of an amber, thermoformable PVC/EVOH/ACLAR film, and a hard tempered aluminium foil with a heat seal lacquer. Tablets are also approved for supply in HDPE bottle packs with child resistant closures of 90 and 200 tablets.*
* All tablet strengths and pack sizes may not be available in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
Levothyroxine sodium, is a monosodium salt of the levo isomer of thyroxine, the principal secretion of the thyroid gland. It has the following chemical structure:
 It has the molecular formula C15H10I4NNaO4 and a molecular weight of 798.86.
It has the molecular formula C15H10I4NNaO4 and a molecular weight of 798.86.
CAS number.
55-03-8 (anhydrous form).7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Summary Table of Changes





 It has the molecular formula C15H10I4NNaO4 and a molecular weight of 798.86.
It has the molecular formula C15H10I4NNaO4 and a molecular weight of 798.86.