1 Name of Medicine
Lidocaine hydrochloride.
2 Qualitative and Quantitative Composition
Lidocaine Accord 1%.
1 mL contains 10 mg lidocaine hydrochloride.
1 ampoule of 2 mL solution for injection contains 20 mg lidocaine hydrochloride.
1 ampoule of 5 mL solution for injection contains 50 mg lidocaine hydrochloride.
1 ampoule of 10 mL solution for injection contains 100 mg lidocaine hydrochloride.
1 vial of 20 mL solution for injection contains 200 mg lidocaine hydrochloride.
Lidocaine Accord 2%.
1 mL contains 20 mg lidocaine hydrochloride.
1 ampoule of 2 mL solution for injection contains 40 mg lidocaine hydrochloride.
1 ampoule of 5 mL solution for injection contains 100 mg lidocaine hydrochloride.
1 ampoule of 10 mL solution for injection contains 200 mg lidocaine hydrochloride.
1 vial of 20 mL solution for injection contains 400 mg lidocaine hydrochloride.
For the full list of excipients, see Section 6.1 List of Excipients.3 Pharmaceutical Form
Lidocaine Accord is a clear, colourless solution for injection.
4.1 Therapeutic Indications
Lidocaine Accord solutions are indicated for the production of local or regional anaesthesia by the following techniques: infiltration; intravenous regional anaesthesia; peripheral nerve block; epidural block; subarachnoid block.
4.2 Dose and Method of Administration
Lidocaine Accord solutions contain no antimicrobial agent. The product is for single use in one patient only. Discard any residue.
The lowest dosage and volume that results in effective anaesthesia should be used and should be based on the status of the patient and the type of regional anaesthesia intended.
Lidocaine should be administered with great caution to patients with impaired cardiovascular function as they may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by these drugs.
Adult.
See Table 1.
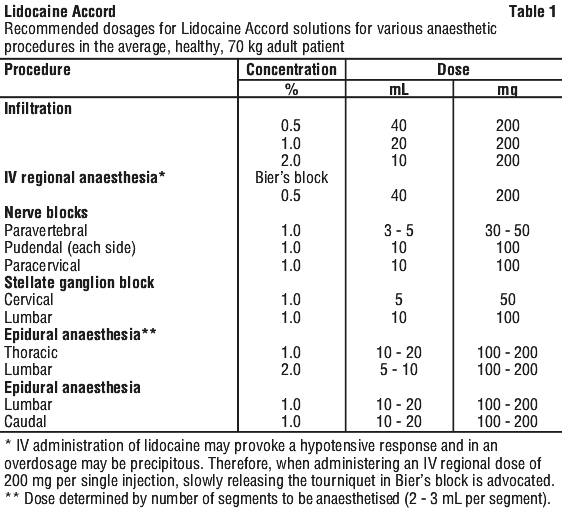 Note.
Note. 1. Recommended doses.
The above suggested concentrations and volumes serve only as a guide. Toxic doses vary widely between patients and toxic effects may occur after any local anaesthetic procedure.
Careful observation of the patient must therefore be maintained. It is recommended that the dose of lidocaine at any one time should not exceed 3 mg/kg. However, the dose administered must be tailored to the individual patient and procedure, and the maximum doses here quoted should be used as a guide only.
2. Hypotension.
During thoracic and lumbar epidural anaesthesia, a marked fall in blood pressure and/or intercostal paralysis may be seen, possibly due to the use of excessive doses, improper positioning of the patient or accidental disposition of the anaesthetic within the subarachnoid space. Hypotension and bradycardia may occur as a result of sympathetic blockade.
3. Test dose.
For epidural anaesthesia, a 3 - 5 mL test dose of a local anaesthetic solution preferably containing up to 15 micrograms of adrenaline (lidocaine with adrenaline is available from other brands) should be administered.
Verbal contact and repeated monitoring of heart rate and blood pressure should be maintained for 5 minutes after the test dose after which, in the absence of signs of subarachnoid or intravascular injection, the main dose may be administered.
Use of a test dose containing adrenaline may have further advantages in that an intravascular injection of adrenaline will be quickly recognised by an increase in heart rate, usually within about 40 seconds. To detect this, the heart rate and rhythm should be monitored with an electrocardiogram.
Prior to administration of the total dose, aspiration should be repeated. The main dose should be injected slowly, with continual assessment of the patient. If toxic symptoms or signs occur, the injection should be stopped immediately.
Use in children.
(See Section 4.4 Special Warnings and Precautions for Use).
For children, a reduced dosage based on body weight or surface area should be used. The dosage should be calculated for each patient individually and modified in accordance with the physician's experience and knowledge of the patient.
In order to minimise the possibility of toxic effects, the use of lidocaine 0.5% or 1% solutions is recommended for most anaesthetic procedures involving paediatric patients.
In children, early signs of local anaesthetic toxicity may be difficult to detect in cases where the block is given during general anaesthesia.
Use in elderly.
(See Section 4.4 Special Warnings and Precautions for Use).
A reduction in dosage may be necessary for elderly patients especially those with compromised cardiovascular and/or hepatic function.
In epidural anaesthesia, a smaller dose may provide adequate anaesthesia.
With impaired hepatic function.
(See Section 4.4 Special Warnings and Precautions for Use).
Although lidocaine is metabolised by the liver, dosage reduction for local anaesthesia is probably not warranted. However, caution should be exercised with repeated doses.
With impaired renal function.
(See Section 4.4 Special Warnings and Precautions for Use).
Impairment of renal function is unlikely to affect lidocaine clearance in the short term (24 hours). However, toxicity due to accumulation may develop with prolonged or repeated administration.4.3 Contraindications
Allergy or hypersensitivity to amide type local anaesthetics or to any excipients. Detection of suspected hypersensitivity by skin testing is of limited value.
Local anaesthetics are contraindicated for epidural and spinal anaesthesia in patients with uncorrected hypotension or coagulation disorders or in patients receiving anti coagulation treatment.
Local anaesthetic techniques must not be used when there is inflammation and/or sepsis in the region of the proposed injection and/or in the presence of septicaemia.
General contraindications related to epidural anaesthesia, regardless of the local anaesthetic used, should be taken into account.
4.4 Special Warnings and Precautions for Use
When any local anaesthetic agent is used, resuscitative equipment and drugs, including oxygen, should be immediately available in order to manage possible adverse reactions involving the cardiovascular, respiratory or central nervous systems. Because of the possibility of hypotension and bradycardia following major blocks, an IV cannula should be inserted before the local anaesthetic is injected. Delay in proper management of dose-related toxicity, under-ventilation from any cause and/or altered sensitivity may lead to the development of acidosis, cardiac arrest and death.
Injection should always be made slowly with frequent aspirations to avoid inadvertent intravascular injection which can produce cerebral symptoms even at low doses.
Although intra-articular continuous infusions of local anaesthetics following arthroscopic and other surgical procedures is an unapproved use, there have been post-marketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in paediatric and adult patients following intra-articular continuous infusions of local anaesthetics with and without adrenaline for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are not associated with these findings. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2nd month after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement. Therefore, Lidocaine Accord should not be used for post-operative intra-articular continuous infusion.
Careful and constant monitoring of cardiovascular and respiratory vital signs and the patient's state of consciousness should be accomplished after each local anaesthetic injection. It should be kept in mind that at such times restlessness, anxiety, tinnitus, dizziness, blurred vision, tremors, depression or drowsiness may be early warning signs of CNS toxicity.
Low molecular weight heparins and heparinoids (spinal/epidural haematomas) - When neuraxial anaesthesia (epidural/spinal anaesthesia) is employed, patients anticoagulated or scheduled to be anticoagulated with low molecular weight heparins or heparinoids are at risk of developing an epidural or spinal haematoma which can result in long-term or permanent paralysis. The risk of these events is increased by the use of indwelling epidural catheters, traumatic or repeated epidural/spinal puncture, and the concomitant use of drugs affecting haemostasis such as NSAID, platelet inhibitors or other anticoagulants. Patients should be frequently monitored for signs and symptoms of neurological impairment.
The safety and effectiveness of Lidocaine Accord depends on proper dosage, correct technique and adequate precautions. Standard textbooks should be consulted regarding specific techniques and precautions for various regional anaesthetic procedures.
The lowest dosage that results in effective anaesthesia should be used (see Section 4.2 Dose and Method of Administration). Repeated injection of Lidocaine Accord may cause accumulation of lidocaine or its metabolites and result in toxic effects.
Tolerance to elevated blood levels varies with the status of the patient. Elderly, young or debilitated patients, including those with advanced liver disease or severe renal dysfunction, should be given reduced doses commensurate with their age and physical condition.
Lidocaine should be given with great caution to patients with epilepsy, impaired cardiac conduction, bradycardia, severe shock or digitalis intoxication. Lidocaine should also be administered with great caution to patients with impaired cardiovascular function as they may be less able to compensate for functional changes associated with the prolongation of AV conduction produced by these drugs. In patients with Stokes-Adams syndrome or Wolff-Parkinson-White syndrome extreme care should be taken to avoid accidental arterio-venous injection.
Hypokalaemia, hypoxia and disorders of acid-base balance should be corrected before treatment with lidocaine.
Central nerve blocks may cause cardiovascular depression, especially in the presence of hypovolaemia. Epidural anaesthesia should be used with caution in patients with impaired cardiovascular function. Epidural anaesthesia may lead to hypotension and bradycardia. Hypotension should be treated promptly with a sympathomimetic intravenously and repeated as necessary.
Local anaesthetics should be given with great caution (if at all) to patients with pre-existing abnormal neurological pathology, e.g. myasthenia gravis. Use with extreme caution in epidural, caudal and spinal anaesthesia when there are serious diseases of the CNS or of the spinal cord, e.g. meningitis, spinal fluid block, cranial or spinal haemorrhage, tumours, poliomyelitis, syphilis, tuberculosis or metastatic lesions of the spinal cord.
Inadvertent intravascular or subarachnoid injection of small doses of local anaesthetics injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses.
Clinicians who perform retrobulbar blocks should be aware that there have been reports of cardiovascular collapse and apnoea following the use of local anaesthetic injections for retrobulbar block. Prior to retrobulbar block, necessary equipment, drugs and personnel should be immediately available as with all other regional procedures. Retrobulbar injections may very occasionally reach the subarachnoid space, causing temporary blindness, cardiovascular collapse, apnoea, convulsions etc. These must be diagnosed and treated promptly.
Retro- and peribulbar injections of local anaesthetics carry a low risk of persistent ocular muscle dysfunction. The primary causes include trauma and/or local toxic effects on muscles or nerves. The severity of such tissue reactions is related to the degree of trauma, the concentration of the local anaesthetic and the duration of exposure of the tissue to the local anaesthetic. For this reason, as with all local anaesthetics, the lowest effective concentration and dose of local anaesthetic should be used. Vasoconstrictors may aggravate tissue reactions and should be used only when indicated.
Foetal bradycardia/tachycardia frequently follows paracervical block and may be associated with foetal acidosis and hypoxia. Occasional cases of perinatal morbidity and mortality have been reported. When the recommended dose is exceeded the risk of foetal bradycardia increases. Careful monitoring of the foetal heart rate is necessary.
Lidocaine should be used with caution in patients with known drug sensitivities.
Patients being treated with anti-arrhythmic drugs class III (e.g. amiodarone) should be under close surveillance and ECG monitoring since cardiac effects may be additive.
Lidocaine Accord solutions for injection are probably porphyrinogenic and should only be prescribed to patients with acute porphyria on strong or urgent indications. Appropriate precautions should be taken for all porphyric patients.
There have been reports of lidocaine-induced hypersensitivity reactions leading to Kounis syndrome.
Methemoglobinemia.
Cases of methemoglobinemia have been reported in association with local anaesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinaemia may occur immediately or may be delayed some hours after exposure, and are characterised by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methaemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue lidocaine injection and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e. oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
Inform patients that use of local anaesthetics may cause methemoglobinaemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, grey, or blue coloured skin (cyanosis); headache; rapid heart rate; shortness of breath; light-headedness; or fatigue.
Patients who are administered local anaesthetics are at increased risk of developing methemoglobinaemia when concurrently exposed to the following medicines, which could include other local anaesthetics:
Nitrates/nitrites such as nitric oxide, nitroglycerin, sodium nitroprusside, nitrous oxide.
Local anaesthetics such as articaine, benzocaine, bupivacaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine.
Antineoplastic agents such as cyclophosphamide monohydrate, flutamide, hydroxycarbamide (hydroxyurea), ifosfamide, rasburicase.
Antibiotics such as dapsone, nitrofurantoin, para-aminosalicylic acid, sulphonamides.
Antimalarials such as chloroquine phosphate, primaquine.
Anticonvulsants such as phenobarbital (phenobarbitone), phenytoin, sodium valproate.
Other drugs such as paracetamol, metoclopramide, quinine, sulfasalazine.
Use in hepatic impairment.
Since lidocaine is metabolised in the liver and excreted via the kidneys, the possibility of drug accumulation should be considered in patients with hepatic and/or renal impairment (see Section 4.2 Dose and Method of Administration).
Use in renal impairment.
Since lidocaine is metabolised in the liver and excreted via the kidneys, the possibility of drug accumulation should be considered in patients with hepatic and/or renal impairment (see Section 4.2 Dose and Method of Administration).
Use in the elderly.
See Section 4.2 Dose and Method of Administration, Use in elderly.
Paediatric use.
See Section 4.2 Dose and Method of Administration, Use in children.
Effects on laboratory tests.
Creatinine.
Creatinine measurements in patients with therapeutic plasma levels of lidocaine are about 15 - 35% higher when measured by an enzymatic method versus the Jaffé method. This appears to be due to assay interference from N-ethylglycine, a metabolite of lidocaine.
Creatine kinase.
The intramuscular injection of lidocaine may result in an increase in creatine kinase levels for up to 48 hrs. This may interfere with the diagnosis of myocardial infarction.4.5 Interactions with Other Medicines and Other Forms of Interactions
Anti-arrhythmic drugs.
Local anaesthetics of the amide type, such as lidocaine, should be used with caution in patients receiving other local anaesthetics or agents structurally related to amide-type local anaesthetics e.g. certain anti arrhythmic drugs such as disopyramide, procainamide, mexilitene since potentiation of cardiac effects may occur. Specific interaction studies with lidocaine and anti-arrhythmic drugs class III (e.g. amiodarone) have not been performed, but caution should be advised (see Section 4.4 Special Warnings and Precautions for Use).
Amiodarone.
Amiodarone has been reported to reduce the clearance of lidocaine in two case reports, although a small prospective study of combined therapy on lidocaine pharmacokinetics found no change in clearance or other pharmacokinetic factor. This combination has been reported to precipitate seizures and to lead to severe sinus bradycardia and a long sinoatrial arrest. Until more experience with concurrent use of lidocaine and amiodarone becomes available, patients receiving the combination should be monitored carefully.
Beta adrenoreceptor antagonists.
Propranolol and metoprolol reduce the metabolism of IV administered lidocaine and the possibility of this effect with other beta adrenergic blockers should be kept in mind. If these drugs are administered concurrently, the patient should be closely observed for signs of lidocaine toxicity.
Cimetidine.
Cimetidine reduces the clearance of IV administered lidocaine and toxic effects due to high serum lidocaine levels have been reported when these two drugs have been administered concurrently.
Anticonvulsive agents.
Phenytoin and other antiepileptic drugs such as phenobarbitone, primidone and carbamazepine appear to enhance the metabolism of lidocaine but the significance of this effect is not known. Phenytoin and lidocaine have additive cardiac depressant effects.
Inhalational anaesthetics.
Lidocaine decreases the minimum effective concentration of inhalational anaesthetics, e.g. nitrous oxide.
Skeletal muscle relaxants.
Lidocaine and skeletal muscle relaxants, e.g. suxamethonium, lead to excessive neuromuscular blockade; therefore, this combination must be used with caution.
Structurally related local anaesthetics.
Lidocaine should be used with caution in patients receiving agents structurally related to local anaesthetics.
Alcohol.
Acute, severe alcohol intoxication can centrally depress the cardiovascular system and may thereby prolong lidocaine elimination half-life.
Alkaline solutions.
The solubility of lidocaine is limited at pH values above 7.0. This must be taken into consideration if adding an alkaline solution since precipitation might occur at higher pH values.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
No data available.
(Category A)
The safe use of lidocaine during pregnancy has not been established. Although lidocaine has been used extensively for surgical procedures during pregnancy with no reports of ill effects to mother or foetus, there are no adequate or well-controlled studies in pregnant women of the effect of lidocaine on the developing foetus.
Lidocaine has been effectively used for obstetrical analgesia and adverse effects on the course of labour or delivery are rare. After epidural administration of lidocaine to women in labour, lidocaine crosses the placental barrier. However, concentrations in umbilical veins are lower than those found in the maternal circulation. It has been suggested that blood glucose levels should be checked in newborns after obstetric regional anaesthesia.
Note.
Paracervical blocks may be associated with foetal bradycardia (see Section 4.4 Special Warnings and Precautions for Use).
Lidocaine passes into breast milk. The amount of lidocaine appearing in breast milk from a nursing mother receiving parenteral lidocaine is unlikely to lead to a significant accumulation of the parent drug in the breast fed infant. The remote possibility of an idiosyncratic or allergic reaction in the breast fed infant from lidocaine remains to be determined.4.7 Effects on Ability to Drive and Use Machines
Depending on dosage, local anaesthetics may have a very mild effect on mental function and may temporarily impair locomotion and coordination.
4.8 Adverse Effects (Undesirable Effects)
Adverse experiences following the administration of lidocaine are similar in nature to those observed with other amide local anaesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage or rapid absorption, or may result from a hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient.
Pronounced acidosis or hypoxia may increase the risk or severity of toxic reactions. Such reactions are systemic and may involve the central nervous system and/or the cardiovascular system.
Inadvertent subarachnoid injection during anaesthetic procedures may lead to cardiovascular collapse, CNS depression and respiratory arrest.
Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
Central nervous system.
CNS manifestations are excitatory and/or depressant and may be characterised by light-headedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, hyperacusis, blurred vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and/or arrest, agitation, difficulty swallowing, paraesthesia circumoral, numbness of the tongue and slurred speech.
The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest. Drowsiness following administration of lidocaine is usually an early sign of a high blood level of the drug and may occur as a result of rapid absorption. In unconscious patients, circulatory collapse should be watched for as CNS effects may not be apparent, as an early manifestation of toxicity may in some cases progress to frank convulsions and ultimately lead to respiratory depression and/or arrest. It is crucial to have resuscitative equipment and anticonvulsant drugs available to manage such patients. (See Section 4.9 Overdose, Treatment of overdosage.)
Cardiovascular.
Cardiovascular manifestations are usually depressant and are characterised by bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest.
Cardiac arrhythmias and hypertension have also been observed. Methemoglobinaemia can occur following IV administration.
Cardiovascular toxic effects are generally preceded by signs of toxicity in the central nervous system, unless the patient is receiving a general anaesthetic or is heavily sedated with drugs such as a benzodiazepine or a barbiturate. In rare cases, cardiac arrest has occurred without prodromal CNS effects.
In children, early signs of local anaesthetic toxicity may be difficult to detect in cases where the block is given during general anaesthesia.
Haemodynamic.
Regional anaesthesia may lead to maternal hypotension.
Allergic.
Allergic reactions are characterised by cutaneous lesions, urticaria, oedema or anaphylactoid reactions/shock.
Allergy to amide type local anaesthetics is rare.
The detection of sensitivity by skin testing is of doubtful value.
Neurologic.
The incidences of adverse reactions associated with the use of local anaesthetics may be related to the total dose of local anaesthetic administered and are also dependent on the particular drug used, the route of administration and the physical status of the patient.
Neurological reactions following regional nerve blocks have included persistent numbness, paraesthesia and other sensory disturbances.
In a prospective review of 10,440 patients who received lidocaine for spinal anaesthesia, the incidences were reported to be about 3% each for positional headaches, hypotension and backache; 2% for shivering; and less than 1% each for peripheral nerve symptoms, nausea, respiratory inadequacy and double vision. Many of these observations may be related to local anaesthetic techniques, with or without a contribution from the local anaesthetic.
In the practice of caudal or lumbar epidural block, occasional unintentional penetration of the subarachnoid space by the catheter may occur. Subsequent adverse effects may depend partially on the amount of drug administered subdurally.
These may include spinal block of varying magnitude (including total spinal block), hypotension secondary to spinal block, loss of bladder and bowel control and loss of perineal sensation and sexual function. Persistent motor, sensory and/or autonomic (sphincter control) deficit of some lower spinal segments with slow recovery (several months) or incomplete recovery have been reported in rare instances when caudal or lumbar epidural block has been attempted. Backache and headache have also been noted following use of these anaesthetic procedures.
Peripheral nerve injury and arachnoiditis have been observed.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at http://www.tga.gov.au/reporting-problems.4.9 Overdose
Symptoms.
Acute emergencies associated with the use of local anaesthetics are generally related to high plasma levels or to unintended subarachnoid injection of the local anaesthetic solution (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
With accidental intravascular injections, the toxic effect will be obvious within 1-3 min. With overdosage, peak plasma concentrations may not be reached for 20-30 min depending on the site of injection and toxic signs will be delayed. Toxic reactions mainly involve the central nervous and cardiovascular systems.
In children, early signs of local anaesthetic toxicity may be difficult to detect in cases where the block is given during general anaesthesia.
Symptoms of acute toxicity.
Central nervous system toxicity is a graded response with symptoms and signs of escalating severity. The first symptoms are circumoral paraesthesia, numbness of the tongue, light-headedness, hyperacusis and tinnitus. Visual disturbance and muscular tremors are more serious and precede the onset of generalised convulsions. These signs must not be mistaken for neurotic behaviour.
Unconsciousness and grand mal convulsions may follow. These may last from a few seconds to several minutes. Hypoxia and hypercarbia occur rapidly following convulsions due to the increased muscular activity, together with the interference with normal respiration and loss of the airway. In severe cases, apnoea may occur. Acidosis increases the toxic effects of local anaesthetics.
Recovery is due to redistribution of the local anaesthetic drug from the central nervous system and metabolism. Recovery may be rapid unless large amounts of the drug have been injected.
Cardiovascular toxicity indicates a more severe situation. Hypotension, bradycardia, decreased cardiac output, heart block, arrhythmia and even ventricular arrhythmias, ventricular fibrillation and cardiac arrest may occur as a result of huge systemic concentrations of local anaesthetics.
Cardiovascular toxic effects are generally preceded by signs of toxicity in the central nervous system, unless the patient is receiving a general anaesthetic or is heavily sedated with drugs such as a benzodiazepine or a barbiturate. In rare cases, cardiac arrest has occurred without prodromal CNS effects.
Treatment of overdosage.
If signs of acute systemic toxicity appear injection of the local anaesthetic should be stopped immediately. If convulsions occur then immediate attention is required for the maintenance of a patent airway and assisted or controlled ventilation with oxygen, via a positive airway pressure delivery system mask. Adequacy of the circulation should then be evaluated, bearing in mind that drugs used to treat convulsions depress the circulation when administered intravenously.
Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, appropriate anticonvulsant medication such as an ultra-short acting barbiturate (e.g. thiopentone) or a benzodiazepine (e.g. diazepam) may be administered IV. The clinician should be familiar with these anticonvulsant drugs prior to use of local anaesthetics.
Suxamethonium will stop the muscle convulsions rapidly but will require tracheal intubation and controlled ventilation, and should only be used by those familiar with these procedures.
If ventricular fibrillation or cardiac arrest occurs, effective cardiovascular resuscitation treatment must be instituted and maintained for a prolonged period if necessary. Optimal oxygenation and ventilation, and circulatory support as well as treatment of acidosis are of vital importance.
If cardiovascular depression occurs (hypotension, bradycardia), appropriate treatment with intravenous fluids, vasopressor, chronotropic and or inotropic agents should be considered. Children should be given doses commensurate with age and weight.
Dialysis is of negligible value in the treatment of acute overdosage with lidocaine.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
Lidocaine is classed as a membrane stabilising agent and is a local anaesthetic of the amide type.
Lidocaine, like other local anaesthetics, causes a reversible blockade of impulse propagation along nerve fibres by preventing the inward movement of sodium ions through the nerve membrane. Local anaesthetics of the amide type are thought to act within the sodium channels of the nerve membrane.
Local anaesthetic drugs may have similar effects on excitable membranes in the brain and myocardium. If excessive amounts of drug reach the systemic circulation rapidly, symptoms and signs of toxicity will appear, emanating mainly from the central nervous system and cardiovascular systems.
Central nervous system toxicity usually precedes the cardiovascular effects as it occurs at lower plasma concentrations. Direct effects of local anaesthetics on the heart include slow conduction, negative inotropism and eventually cardiac arrest.
Indirect cardiovascular effects, e.g. hypotension and bradycardia, may occur after epidural or spinal administration depending on the extent of the concomitant sympathetic block.
Clinical trials.
No data available.
5.2 Pharmacokinetic Properties
Absorption.
Lidocaine has a rapid onset and a medium duration of action. The onset of action is 1-5 minutes following infiltration and 5-15 minutes following other types of administration.
The rate of absorption depends upon the dose, the route of administration and the vascularity of the injection site. Intercostal blocks give the highest peak plasma concentrations (approximately 1.5 microgram/mL for every 100 mg injected), while abdominal subcutaneous injections give the lowest (approx. 0.5 microgram/mL per 100 mg injected). Epidural and major nerve block produce peak plasma levels intermediate between these.
The addition of adrenaline considerably slows the absorption of lidocaine, although the rate also depends on the site of injection. Peak plasma concentrations are reduced by 50% following subcutaneous injection, by 30% following epidural injection and by 20% following intercostal block if adrenaline 5 microgram/mL is added.
Absorption of lidocaine from the epidural space occurs in 2 phases; the first phase is in the order of 9 minutes and the second is approximately 82 minutes. The slow absorption is the rate-limiting step in the elimination of lidocaine, which also explains why the apparent elimination half-life following epidural injection is longer than after intravenous administration.
Distribution.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 microgram of free base/mL, 60 - 80% of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the α1-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers by passive diffusion. Since the degree of plasma protein binding in the foetus is less than in the mother, although free lidocaine concentrations will be the same, the total plasma concentration will be greater in the mother.
Metabolism.
Approximately 90% of a parenteral dose of lidocaine is rapidly metabolised in the liver by de-ethylation to form monoethylglycinexylidide (MEGX) and glycinexylidide (GX) followed by cleavage of the amide bond to form xylidine and 4-hydroxyxylidine which are excreted in the urine. Less than 10% of a dose is excreted unchanged in the urine.
The principal metabolites, MEGX and GX also possess pharmacological activity. The rate of metabolism of lidocaine appears to be limited by liver blood flow which may be reduced in patients after acute myocardial infarction and/or congestive heart failure. The rate of lidocaine metabolism may also be reduced in patients with liver or hepatic tissue necrosis, possibly because of altered perfusion.
Excretion.
Lidocaine has a total plasma clearance of 0.95 L/min, a volume of distribution at steady state of 91 L, an elimination half-life of 1.6 hr and an estimated hepatic extraction ratio of 0.65.
The duration of action depends upon the concentration used, the dose given, the nerves to be blocked and the status of the patient. The 2% solution will produce an effect for 1½ - 2 hrs when given epidurally, and up to 5 hrs when given as a peripheral nerve block. When used in a 1% concentration there is less effect on motor nerve fibres and the duration of effect is shorter.
5.3 Preclinical Safety Data
Genotoxicity.
The genotoxic potential of 2,6-xylidine has been studied with mixed results: Positive results were reported in assays for gene mutations (weakly positive in the Ames test with metabolic activation and in the mouse lymphoma assay) and chromosomal damage (chromosomal aberrations in Chinese hamster ovary cells at concentrations at which the drug precipitated from solution). No evidence of genotoxicity was found in in vivo assays for chromosomal damage (micronucleus assay) and DNA damage (unscheduled DNA synthesis). Covalent binding studies of DNA from liver and ethmoid turbinates in rats indicate that 2,6-xylidine may be genotoxic under certain conditions in vivo.
Carcinogenicity.
A two-year oral toxicity study of 2,6-xylidine, a metabolite of lidocaine, has shown that in both male and female rats, 2-6-xylidine in daily doses of 900 mg/m2 (150 mg/kg) resulted in carcinomas and adenomas of the nasal cavity. No nasal tumours were observed in the low dose (15 mg/kg or control animals). In addition, the compound also caused subcutaneous fibromas and or fibrosarcomas in male and female rats (significant at 150 mg/kg).6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, sodium hydroxide, hydrochloric acid, water for injections.
6.2 Incompatibilities
Local anaesthetics react with certain metals and cause the release of their respective ions which, if injected, may cause severe local irritation. Adequate precautions should be taken to avoid prolonged contact between Lidocaine Accord solutions and metal surfaces such as metal bowls, cannulae and syringes with metal parts.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
Solutions showing discolouration and unused portions of solutions from ampoules and single dose vials should be discarded.
Surface sterilisation using pure, undiluted isopropyl alcohol (91%) or 70% ethyl alcohol (USP) may be carried out if desired.
6.5 Nature and Contents of Container
Lidocaine Accord 1%.
Lidocaine Accord 1% is available in four strengths: 20 mg/2 mL, 50 mg/5 mL and 100 mg/10 mL in Type I glass ampoules in packs of 20. The 200 mg/20 mL is in Type I glass vials in packs of 1 or 5.
Lidocaine Accord 2%.
Lidocaine Accord 2% is available in four strengths: 40 mg/2 mL, 100 mg/5 mL and 200 mg/10 mL in Type I glass ampoules in packs of 20. The 400 mg/20 mL is in Type I glass vials in packs of 1 or 5.
Not all presentations and pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.

CAS number.
The CAS number for lidocaine hydrochloride monohydrate (AAN) is 6108-05-0 and lidocaine hydrochloride is 73-78-9.7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Summary Table of Changes




