What is in this leaflet
This leaflet answers some of the common questions people ask about LOSEC. It does not contain all the information that is known about LOSEC.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor will have weighed the risks of you taking LOSEC against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What LOSEC is used for
Reflux Oesophagitis
LOSEC is used to treat the symptoms of reflux oesophagitis or reflux disease. This can be caused by "washing back" (reflux) of food and acid from the stomach into the food pipe (oesophagus).
Reflux can cause a burning sensation in the chest rising up to the throat, also known as heartburn.
LOSEC is also taken to help stop reflux oesophagitis coming back or relapsing.
Peptic Ulcers
LOSEC is used to treat peptic ulcers. Depending on the position of the ulcer it is called a gastric or duodenal ulcer. A gastric ulcer occurs in the stomach. A duodenal ulcer occurs in the duodenum which is the tube leading out from the stomach.
These ulcers can be caused by too much acid being made in the stomach.
LOSEC is also used to help stop gastric or duodenal ulcers coming back.
Peptic Ulcers Associated with Helicobacter pylori Infection
Most people who have a peptic ulcer also have a bacterium called Helicobacter pylori in their stomach.
When LOSEC is taken with antibiotics, they work to kill the bacterium and let your ulcer heal. You may need further treatment with antibiotics.
Peptic Ulcers Associated with Non-steroidal Anti-Inflammatory Drugs (NSAIDs)
Some peptic ulcers are caused by taking medicines called non-steroidal anti-inflammatory drugs (NSAIDs), a type of medicine used to treat pain or inflammation.
LOSEC is also used to heal and prevent ulcers associated with NSAIDs.
Zollinger-Ellison Syndrome
LOSEC is also used to treat a rare condition called Zollinger-Ellison syndrome, where the stomach produces large amounts of acid, much more than in ulcers or reflux disease.
How LOSEC Works
LOSEC is a type of medicine called a proton-pump inhibitor. It works by decreasing the amount of acid made by the stomach, to give relief of symptoms and allow healing to take place. This does not stop food being digested in the normal way.
Follow all directions given to you by your doctor carefully. They may differ from the information contained in this leaflet.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may prescribe this medicine for another reason.
There is no evidence that LOSEC is addictive.
This medicine is only available on prescription.
Before you take LOSEC
When you must not take it
Do not take LOSEC if you have an allergy to:
- omeprazole or any ingredient listed at the end of this leaflet
- any medicine containing a proton-pump inhibitor
Some of the symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing; swelling of the face, lips, tongue or other parts of the body; rash, itching or hives on the skin.
Do not take LOSEC if you are also taking cilostazol. Please check with your doctor or pharmacist if you are taking cilostazol. This medicine will be affected by LOSEC.
Do not take LOSEC after the use by (expiry) date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should take this medicine, talk to your doctor.
Before you start to take it
You must tell your doctor if you have:
- allergies to any other medicines, foods, dyes or preservatives
- any problems with your liver
- any other medical conditions
- been diagnosed with osteoporosis
- if you have ever had a skin reaction after treatment with a medicine similar to LOSEC that reduces stomach acid
Do not take LOSEC if you are pregnant or breastfeeding unless your doctor says so. Ask your doctor about the risks and benefits involved. It is not known if it is safe for you to take LOSEC while you are pregnant. It may affect your baby.
It is not known if your baby can take in LOSEC from breast milk if you are breastfeeding.
Taking other medicines
Do not take LOSEC if you are taking the following medicine:
- cilostazol - a medicine used to treat intermittent claudication
Tell your doctor if you are taking any other medicines including any that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with LOSEC tablets. These include:
- phenytoin - a medicine used to treat epilepsy or fits
- warfarin and clopidogrel - medicines used to prevent blood clots
- digoxin - a medicine used to treat heart conditions
- diazepam - a medicine used to treat anxiety and some other conditions
- St John's wort - a herbal remedy used to treat mood disorders
- ketoconazole, itraconazole, voriconazole - medicines used to treat fungal infection
- clarithromycin or rifampicin - medicines used to treat infections
- atazanavir and nelfinavir - medicines used to treat viral infections such as HIV
- tacrolimus and mycophenolate mofetil - medicines used to assist in organ transplants
- methotrexate - a medicine used to treat arthritis and some types of cancer
- erlotinib or related medicines used to treat cancer
These medicines may be affected by LOSEC or may affect how well it works. You may need different amounts of your medicine or you may need to take different medicines. Your doctor can tell you what to do if you are taking any other medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
If you have not told your doctor about any of these things, tell them before you take LOSEC.
How to take LOSEC
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the label, ask your doctor or pharmacist for help.
How to take it
Take one LOSEC tablet each day, unless your doctor has told you otherwise.
Adults:
The dose of LOSEC is usually 20 mg a day. The dose may vary from 10 mg to 40 mg a day depending on what condition you are being treated for and how severe it is.
Children (1 year or older):
The dose of LOSEC is 10 mg a day for children 10 to 20 kg. This dose may be increased to 20 mg if required. For children more than 20 kg the dose is 20 mg a day. This dose may be increased to 40 mg if required.
Swallow LOSEC whole with a glass of water. Do not crush or chew the tablets. If the tablets are chewed or crushed, they will not work properly.
If you have difficulty swallowing the tablets
- Place the tablet in half a glass of non-carbonated water or fruit juice. Mineral water, carbonated fruit juices, or other liquids are not suitable.
- Gently mix the tablet and liquid by stirring, taking care not to crush the tablet.
- Stir until the tablet disperses into little pellets.
- Drink the liquid with the pellets immediately, or within 30 minutes. Do not chew the pellets
- Rinse the glass with half a glass of water and drink.
When to take it
Take LOSEC at about the same time each day. Keeping a regular time for taking LOSEC will help to remind you to take it.
LOSEC can be taken with food or on an empty stomach.
How long to take it
Keep taking LOSEC for as long as your doctor recommends. In most patients, LOSEC relieves symptoms rapidly and healing is usually complete within 4 weeks. Continue taking LOSEC for as long as your doctor tells you to.
If you forget to take it
If you forget to take a dose, take it as soon as you remember, and then go back to taking it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose that you missed. If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much (Overdose)
Telephone your doctor or the Poisons Information Centre (13 11 26) or go to Accident and Emergency at your nearest hospital immediately if you think that you or anyone else may have taken too much LOSEC even if there are no signs of discomfort or poisoning.
While you are taking LOSEC
Things you must do
Take LOSEC exactly as your doctor has prescribed.
If you are about to start any new medicine, remind your doctor and pharmacist that you are taking LOSEC.
Tell all doctors, dentists and pharmacists who are treating you that you are taking LOSEC.
Tell your doctor if you become pregnant while you are taking LOSEC.
Tell your doctor if your symptoms return. Although LOSEC can heal ulcers successfully, it may not prevent them recurring at a later date.
If you need to have any medical tests while you are taking LOSEC, tell your doctor. It may affect the results of some tests.
Things you must not do
Do not take LOSEC to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or change the dosage without checking with your doctor. If you stop taking it suddenly or change the dose, your condition may worsen or you may have unwanted side effects.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking LOSEC.
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- constipation
- nausea or vomiting
- diarrhoea
- headache
- skin rash, itchy skin
- wind
- stomach pain
- dizziness
- dry or sore mouth
These side effects are usually mild.
Tell your doctor immediately if you notice any of the following:
- muscle pain or weakness, joint pain
- "pins and needles"
- changes in sleep patterns
- mood changes, confusion or depression
- blurred vision
- increase in breast size (males)
- fever
- increased bruising
- increased sweating
- hair loss
- tremor
These are serious side effects that may require medical attention. Serious side effects are rare.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital.
- swelling of the face, lips, mouth, tongue or throat which may cause difficulty in breathing
- shortness of breath or difficulty in breathing
- severe skin reaction which may include rash, itching, redness, blistering or peeling of the skin
- ulcers, blisters or bleeding of the lips, eyes, mouth, nose and genitals
- blood in the urine
- swelling of hands, feet or ankles
- signs of liver inflammation including yellowing of the skin or eyes, feeling generally unwell, nausea, vomiting, loss of appetite
- skin reaction, especially in sun-exposed areas, with joint pain
These are very serious side effects. You may need urgent medical attention or hospitalisation. These side effects are rare.
Occasionally, LOSEC may be associated with changes in your liver or blood, which may require your doctor to do certain blood tests.
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may occur in some people.
Other problems are more likely to arise from the ulcer itself rather than the treatment.
For this reason, contact your doctor immediately if you notice any of the following:
- pain or indigestion that occurs during treatment with LOSEC
- you begin to vomit blood or food
- you pass black (blood-stained) motions
Tell your doctor if your reflux symptoms return after you stop taking LOSEC.
After using it
Storage
Keep your LOSEC in the blister pack until it is time to take them. If you take LOSEC out of the blister pack they will not keep well.
Keep it in a cool, dry place where the temperature stays below 25°C.
Do not store it or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking LOSEC or the tablets have passed their expiry date, ask your pharmacist what to do with any tablets you have left over.
Product description
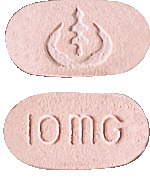
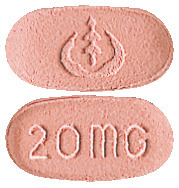
What LOSEC looks like
LOSEC 10 mg tablets are light pink, oblong shaped, marked with 10 mg on one side and a logo on the other side.
LOSEC 20 mg tablets are pink, oblong shaped, marked with 20 mg on one side and a logo on the other side.
Ingredients
Each LOSEC tablet contains omeprazole magnesium as the active ingredient equivalent to omeprazole 10 mg or 20 mg, plus the following inactive ingredients:
- Glyceryl monostearate
- Hyprolose
- Hypromellose
- Magnesium stearate
- Methacrylic acid copolymer
- Microcrystalline cellulose
- Synthetic paraffin
- Macrogol 6000
- Polysorbate 80
- Crospovidone
- Sodium stearylfumarate
- Purified talc
- Titanium dioxide
- Triethyl citrate
- Sodium hydroxide
- Sugar spheres (maize starch and sucrose)
The tablets are coloured with iron oxide red (CI77491) and/or iron oxide yellow (CI77492).
LOSEC are available in blister packs of 30 tablets.
LOSEC tablets do not contain gluten.
Sponsor
AstraZeneca Pty Ltd
ABN 54 009 682 311
66 Talavera Road
MACQUARIE PARK NSW 2113
Telephone: 1800 805 342
Australian Registration Numbers:
LOSEC 10 mg (blister pack) - AUST R 63414
LOSEC 20 mg (blister pack) - AUST R 63416
LOSEC 40 mg (blister pack) - AUST R 63418*
*non-marketed
This leaflet was prepared in March 2021.
LOSEC® is a registered trade mark of the AstraZeneca group of companies.
© AstraZeneca 2021
Doc ID-001944965 V10.0
Published by MIMS April 2021