What is in this Leaflet
This leaflet answers some common questions about LOZANOC Capsules. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking LOZANOC Capsules against the benefits this medicine is expected to have for you.
If you have any concerns about taking LOZANOC Capsules, ask your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What LOZANOC® Capsules are used for
LOZANOC Capsules are used to treat certain fungal infections which include the following:
- persistent infections of the nails, skin, hands, feet or groin;
- persistent candida (yeast) infections of the vagina;
- eye infections which have not responded to other treatment or which may be affecting vision;
- candida (yeast) infections of the mouth or throat in patients with lower resistance to disease;
- generalised infections.
LOZANOC works by killing or stopping the growth of the fungus that causes the infection.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
Before you take LOZANOC® Capsules
When you must not take it
Do not take LOZANOC Capsules if:
- you have an allergy to LOZANOC Capsules or any of the ingredients (see Product Description at the end of this Leaflet).
- you have a condition called heart failure (also called congestive heart failure or CHF), LOZANOC could make it worse.
- you are pregnant or may become pregnant.
If there is a chance you may become pregnant, talk to your doctor about the need for highly effective contraception. Once you have finished LOZANOC, contraception should be continued until you have had your next period. Tell your doctor immediately, if you do become pregnant.
Do not take LOZANOC Capsules with any of the following medicines:
- certain medicines for allergy or hay fever (e.g. terfenadine, astemizole, mizolastine);
- certain medicines used to treat angina and high blood pressure (e.g. bepridil, felodipine, nisoldipine, lercanidipine, ranolazine, eplerenone) and ivabradine, a heart rate lowering agent;
- cisapride, a drug used to treat gastric reflux;
- domperidone, an antiemetic used to treat nausea, vomiting, bloating and fullness;
- levomethadyl and methadone, which are opioids;
- antipsychotic medications, such as pimozide, lurasidone and sertindole;
- ticagrelor, an anticoagulant;
- halofantrine, a medicine used to treat malaria;
- isavuconazole, an antifungal medicine;
- naloxegol, a medicine used to reduce opioid constipation;
- avanafil, a drug used for erectile dysfunction and dapoxetine, used for premature ejaculation;
- eliglustat, used to treat Gaucher disease;
- irinotecan, a chemotherapy agent for colon cancer;
- certain medicines used to produce calmness or to help you sleep (midazolam oral or triazolam);
- certain medicines used to lower your cholesterol, known as HMGCoA reductase inhibitors, (e.g. simvastatin, lomitapide, lovastatin);
- dronedarone, dofetilide, quinidine or disopyramide (used to treat irregular heartbeats);
- dihydroergotamine or ergotamine (used to treat migraine);
- fesoterodine and solifenacin in patients with moderate to severe liver or kidney disease (these are medicines used to treat overactive bladder);
- colchicine in patients with severe liver or kidney disease (medicine used to treat gout and Behcets disease);
- telithromycin in patients with severe liver or kidney disease (an antibiotic used to treat pneumonia);
- ergometrine or methylergometrine (used to control bleeding).
Do not take LOZANOC Capsules after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions:
- liver or kidney problems;
- you have had an allergic reaction to other medicines used to treat fungal infections;
- heart problems;
- neutropenia (low levels of a type of white blood cells) or AIDS or an organ transplant patient;
- hearing problems;
- weakness, numbness and pain from nerve damage, usually in the hands and feet;
- you have cystic fibrosis.
Tell your doctor if you are breastfeeding or wish to breastfeed. Your doctor can discuss with you the risks and benefits involved.
If you have not told your doctor or pharmacist about any of the above, tell them before you start taking or LOZANOC Capsules.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including medicines you can buy without a prescription from a pharmacy, supermarket or health food shop.
In particular, LOZANOC capsules must not be taken with some medicines. Wait at least 2 weeks after stopping LOZANOC capsules before taking any of these medicines. Examples are:
- certain medicines for allergy or hay fever (e.g. terfenadine, astemizole, mizolastine);
- certain medicines used to treat angina and high blood pressure (e.g. bepridil, felodipine, nisoldipine, lercanidipine, ranolazine, eplerenone) and ivabradine, a heart rate lowering agent;
- cisapride, a drug used to treat gastric reflux;
- domperidone, an antiemetic used to treat nausea, vomiting, bloating and fullness;
- levomethadyl and methadone, which are opioids;
- antipsychotic medications, such as pimozide, lurasidone and sertindole;
- ticagrelor, an anticoagulant;
- halofantrine, a medicine used to treat malaria;
- isavuconazole, an antifungal medicine;
- naloxegol, a medicine used to reduce opioid constipation;
- avanafil, a drug used for erectile dysfunction and dapoxetine, used for premature ejaculation;
- eliglustat, used to treat Gaucher disease;
- irinotecan, a chemotherapy agent for colon cancer;
- certain medicines used to produce calmness or to help you sleep (midazolam or triazolam);
- certain medicines used to lower your cholesterol, known as HMGCoA reductase inhibitors, (e.g. simvastatin, lomitapide, lovastatin);
- dronedarone, dofetilide, quinidine or disopyramide (used to treat irregular heartbeats);
- dihydroergotamine or ergotamine (used to treat migraine);
- fesoterodine and solifenacin in patients with moderate to severe liver or kidney disease (these are medicines used to treat overactive bladder);
- colchicine in patients with severe liver or kidney disease (medicine used to treat gout and Behcets disease);
- telithromycin in patients with severe liver or kidney disease (an antibiotic used to treat pneumonia);
- ergometrine or methylergometrine (used to control bleeding).
Certain medicines are not recommended because they may be affected by LOZANOC Capsules or may affect how well LOZANOC Capsules work. Your doctor may need to adjust the dose or adapt your treatment.
Examples of these medicines are:
- alfuzosin, tamsulosin, silodosin (used to treat Benign Prostatic enlargement);
- alfentanil, buprenorphine, oxycodone, sufentanil (used in surgery for pain relief and to help anaesthesia);
- fentanyl, a strong medicine for pain;
- digoxin (used to treat heart failure);
- bedaquiline, delamanid, rifampicin, rifabutin or isoniazid (used to treat tuberculosis);
- ciprofloxacin, erythromycin, clarithromycin, (antibiotics);
- trimetrexate, used to treat a certain type of pneumonia;
- anticoagulants (used to slow blood clotting), such as apixaban, rivaroxaban, vorapaxar, coumarins and coumarin like medicines (eg warfarin) or dagibatran; phenytoin, phenobarbital or carbamazepine (used to treat fits);
- medicines taken for diabetes, such as repaglinide and saxagliptan;
- praziquantel, a worm medication;
- artemether-lumefantrine, quinine (used to treat malaria);
- bilastine, ebastine, rupatadine (used to treat allergies);
- eletriptan (used to treat migraine);
- certain antineoplastics such as axitinib, bosutinib, bortezomib, brentuximab vedotin, busulfan, carbazitaxel, cabozanitinib, ceritinib, cobimetinib, crizotinib, dabrafenib, dasatinib, docetaxel, erlotinib, gefitinib, imatinib, ibrutinib, idelalisib, ixabepilone, lapatinib, nilotinib, nintedanib, olaparib, panobinstat, pazopanib, ponatinib, regorafenib, ruxolitinib, sonidegib, sunitinib, trabectedin, trastuzumab emtansine, vandetanib, vinca alkaloids (used to treat certain cancers);
- alprazolam, aripiprazole, brotizolam, buipirone, cariprazine, haloperidol, midazolam i.v., perospirone, quetiapine, ramelteon, risperidone, suvorexant, zoplicone, (used to treat anxiety or help you sleep);
- certain medicines used to treat AIDS, such as cobicistat, darunavir (boosted), efavirenz, elvitegravir (boosted), fosamprenavir (ritonavirboosted), indinavir, maraviroc, nevirapine, saquinavir, tenofovir disoproxil fumarate, or ritonavir;
- simeprevir, boosted asunaprevir, boceprevir, daclatasvir, elbasvir/ grazoprevir, glecaprevir/pibrentasvir, ombitasvir/paritaprevir/ritonavir (with or without dasabuvir), telaprevir, vaniprevir (used to treat hepatitis C);
- aliskiren, bosentan, nadolol or riociguat (used to treat heart or blood pressure problems or hypertension);
- sildenafil or tadalafil (used to treat erectile dysfunction or pulmonary hypertension);
- certain calcium channel blockers (used to treat heart or blood pressure problems), such as diltiazem, verapamil or other dihydropyridines;
- contraceptives such as, dienogest or ulipristal;
- aprepitant, netupitant (used for nausea and vomiting during cancer treatment);
- Saccharolmyces boulardii, loperamide (used to treat diarrhoea);
- budesonide, ciclesonide, cyclosporin, dexamethasone, fluticasone, methylprednisolone, sirolimus, tacrolimus, temsirolimus or everolimus (used to help prevent organ transplant rejection or to treat certain problems with the immune system);
- atorvastatin (used to lower cholesterol);
- meloxicam (a non-steroidal anti-inflammatory drug);
- salmeterol, a respiratory drug;
- reboxetine or venlafaxine (used to treat depression);
- darifenacin, vardenafil, dutasteride, imidafenacin, oxybutynin, tolterodine or udenafil (used to treat urinary disorders);
- cinacalcet, to treat an over active parathyroid;
- alitretinoin (oral formulation), to treat eczema;
- cabergoline (used to treat Parkinson’s Disease);
- cannabinoids (used to treat nausea and vomiting, weight loss for patients with immune system problems and muscle spasms in patients with Multiple Sclerosis);
- galantamine (used to treat Alzheimer's disease)
- ivacaftor, lumacaftor/ ivacaftor (used to treat Cystic Fibrosis);
- guanfacine (used to treat attention deficit hyperactivity disorder and high blood pressure);
- conivaptan, tolvaptan (used to treat low blood sodium levels);
- mozavaptan, used to treat low blood sodium.353672-8
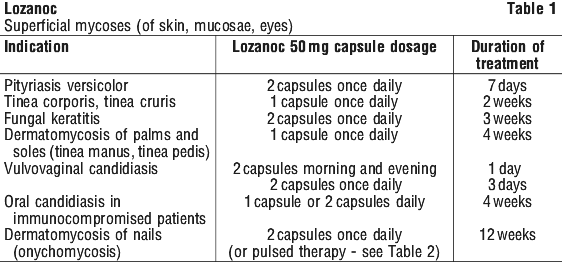
How to Take LOZANOC® Capsules
How much to take
Adults
LOZANOC Capsules are specially designed to give higher blood levels of active ingredient than other formulations of itraconazole capsules. A 50 mg capsule of LOZANOC is therapeutically equivalent to 100 mg of other brands of itraconazole capsules but the capsules are not interchangeable.
Only take as many LOZANOC capsules as you have been prescribed and ask your doctor or pharmacist if you are not sure.
The usual doses are shown below, but your doctor may decide to adjust them for your individual needs.
Tinea of body & groin:
1 capsule daily for 2 weeks.
Tinea of hands & feet:
1 capsule daily for 4 weeks.
Other skin infections:
2 capsules daily for 1 week.
Eye infections:
2 capsules daily for 3 weeks.
Vaginal infections:
2 capsules morning & evening for 1 day, or 2 capsules daily for 3 days.
Mouth infections:
1 to 2 capsules daily for 4 weeks.
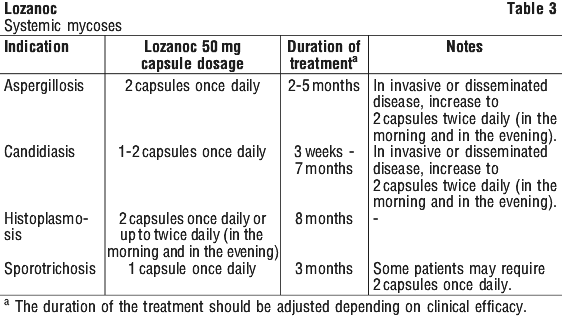
Systemic infections:
1 to 2 capsules once or twice daily for 3 weeks to 8 months, depending on the condition.
Nail infections:
Continuous nail therapy:
2 capsules once daily for 3 months.
Cyclic (pulse) nail therapy:
2 capsules twice daily for 1 week. After that, stop taking LOZANOC for 3 weeks. Then the cycle is repeated, once for fingernails and twice for toenail infections (with or without fingernail infections).
Children and Elderly
LOZANOC Capsules are not recommended for use in children and in the elderly.
How to take it
Always take LOZANOC exactly as your doctor has told you to.
Check with your doctor or pharmacist if you are not sure. LOZANOC Capsules can be taken before or after a meal.
How long to take it
Don't worry if you don't see an immediate improvement after your treatment.
- With skin infections, the marks or spots (lesions) typically disappear a few weeks after you finish the course. Although the medicine kills the fungus, the marks don't disappear until after new skin has grown.
- With nail infections, marks on the nail may take 6 to 9 months to disappear, because new nail needs to grow.
Ask your doctor or pharmacist if you're not sure whether the treatment is working.
If you forget to take it
Take the dose you missed as soon as you remember, and then continue to take it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the one you missed.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you have taken too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at your nearest hospital. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are taking LOZANOC® Capsules
Things you must do
Always follow your doctor's instructions carefully.
If you have to take LOZANOC Capsules continuously for more than 1 month, your doctor may ask you to have your blood checked regularly to make sure that your liver is not affected.
If you become pregnant while taking this medicine, tell your doctor immediately. If there is any chance of you becoming pregnant, talk to your doctor about the need for adequate contraception.
Tell any other doctors, dentists or pharmacists who treat you that you are taking this medicine.
Always complete the treatment as directed by your doctor, even if the signs of infection have gone.
Things you must not do
Do not take LOZANOC Capsules to treat any other complaint unless your doctor says so.
Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Things to be careful of
Be careful driving or operating machinery. You may feel dizzy while taking LOZANOC capsules. If you experience this or similar effects, you should avoid driving and using machines.
Make sure you know how you react to LOZANOC capsules before you drive a car, operate machinery or do anything else that could be dangerous if you are dizzy or lightheaded.
Side Effects
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some side effects. Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you experience any of the following:
- upset stomach, stomach pain or discomfort, nausea, vomiting, diarrhoea, constipation, an unpleasant taste in your mouth.
- shortness of breath, headache, dizziness, fever.
- a change in menstrual pattern.
- unusual hair loss or thinning.
- erectile dysfunction.
- muscle weakness or pain, painful joints.
Tell your doctor immediately if you notice any of the following as you may need urgent medical care:
- tingling, numbness or weakness in the hands or feet;
- swelling of hands ankles, feet, legs or abdomen;
- shortness of breath, unexpected weight gain, unusual fatigue, or begin to wake up at night;
- oversensitivity to sunlight;
- blurry or double vision, ringing in the ears;
- lose the ability to control your bladder or urinate much more than usual.
STOP taking LOZANOC Capsules and tell your doctor immediately or go to Accident and Emergency at your nearest hospital if any of the following happen:
- abnormal tiredness, loss of appetite, nausea, vomiting, dark urine, pale stools, yellowing of the skin or eyes.
- sudden signs of allergy such as rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body, shortness of breath or difficulty breathing, wheezing or trouble breathing.
- a severe skin disorder (widespread rashes with peeling skin and blisters in the mouth, eyes and genitals, or rashes with small pustules or blisters).
- you experience any hearing loss symptoms. In very rare cases, patients taking LOZANOC have reported temporary or permanent hearing loss.
Other side effects not listed above may also occur in some people. Tell your doctor if you notice any other effects.
After using LOZANOC® Capsules
Storage
Keep LOZANOC Capsules in the pack until it is time to take them. If you take the capsules out of the pack, they may not keep well.
Keep LOZANOC Capsules in a cool dry place where the temperature is below 25°C.
Keep your medicines where young children cannot reach them. A locked cupboard at least one-and-a-half metres (1.5 m) above the ground is good place to store medicines.
Do not store LOZANOC Capsules, or any other medicine, in the bathroom or near a sink. Do not leave medicines in the car or on window sills. Heat and dampness can destroy some medicines.
Disposal
If your doctor tells you to stop taking LOZANOC Capsules or your medicine has passed its expiry date, ask your pharmacist what to do with any medicine which may be left over.
Product Description
What it looks like
LOZANOC Capsules are blue opaque capsules with i-50 printed in black.
They are supplied in a blister pack or bottle pack.
Ingredients
LOZANOC Capsules contain 50 mg of itraconazole as the active ingredient. It also contains:
- hypromellose phthalate
- sodium starch glycollate (Type A)
- silicon dioxide
- magnesium stearate
- gelatin
- brilliant blue FCF
- titanium dioxide
- TekPrint SW-9008 Black Ink
LOZANOC Capsules do not contain lactose or gluten.
Sponsor
Mayne Pharma International Pty Ltd
1538 Main North Rd
Salisbury South, SA 5106
Australian Registration Number:
Blister pack AUST R 206361
Bottle pack AUST R 206360
This leaflet was prepared February 2020.
CMI Version Number: 5.0
Published by MIMS April 2020


 An alternative dosage regimen for dermatomycosis of nails (onychomycosis) is pulsed therapy. A pulse treatment consists of two capsules twice daily for one week. Two pulse treatments are recommended for fingernail infections, three pulse treatments for toenail infections. Pulse treatments are always separated by a 3 week drug free interval. Clinical response will become evident as the nail regrows, following discontinuation of the treatment.
An alternative dosage regimen for dermatomycosis of nails (onychomycosis) is pulsed therapy. A pulse treatment consists of two capsules twice daily for one week. Two pulse treatments are recommended for fingernail infections, three pulse treatments for toenail infections. Pulse treatments are always separated by a 3 week drug free interval. Clinical response will become evident as the nail regrows, following discontinuation of the treatment.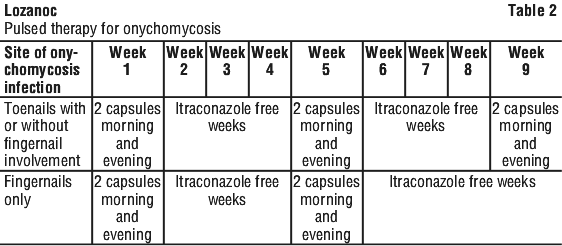
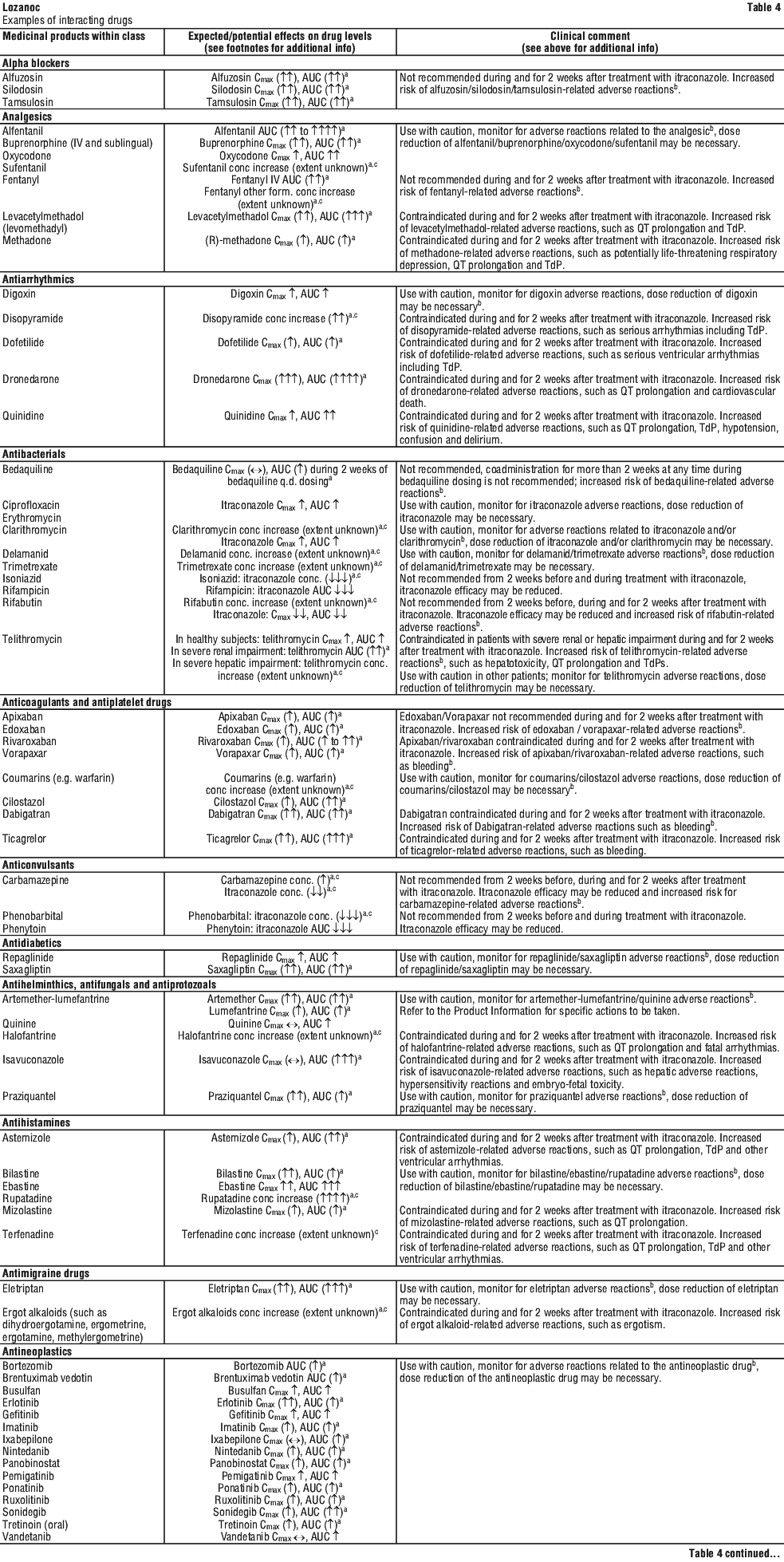
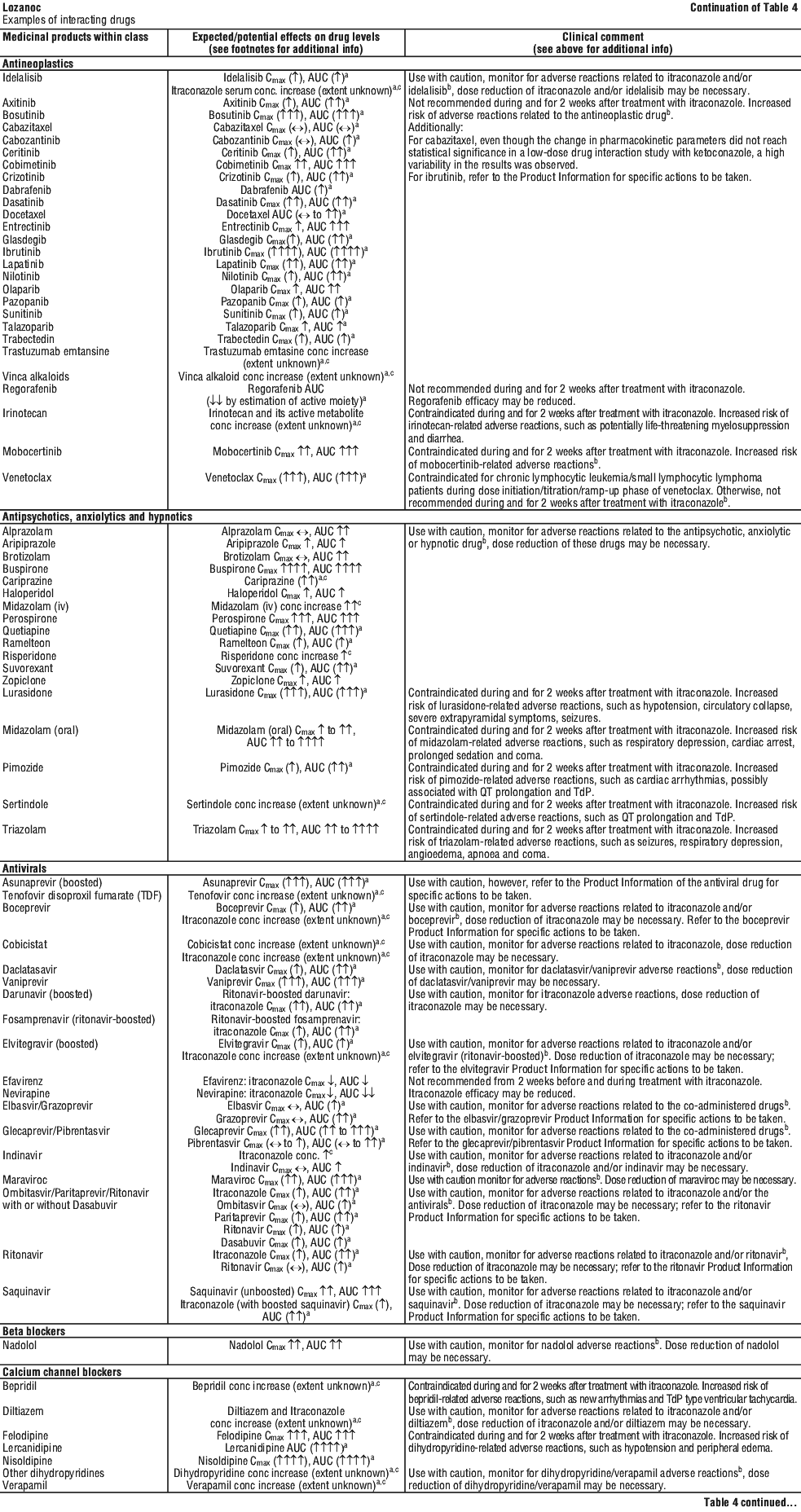
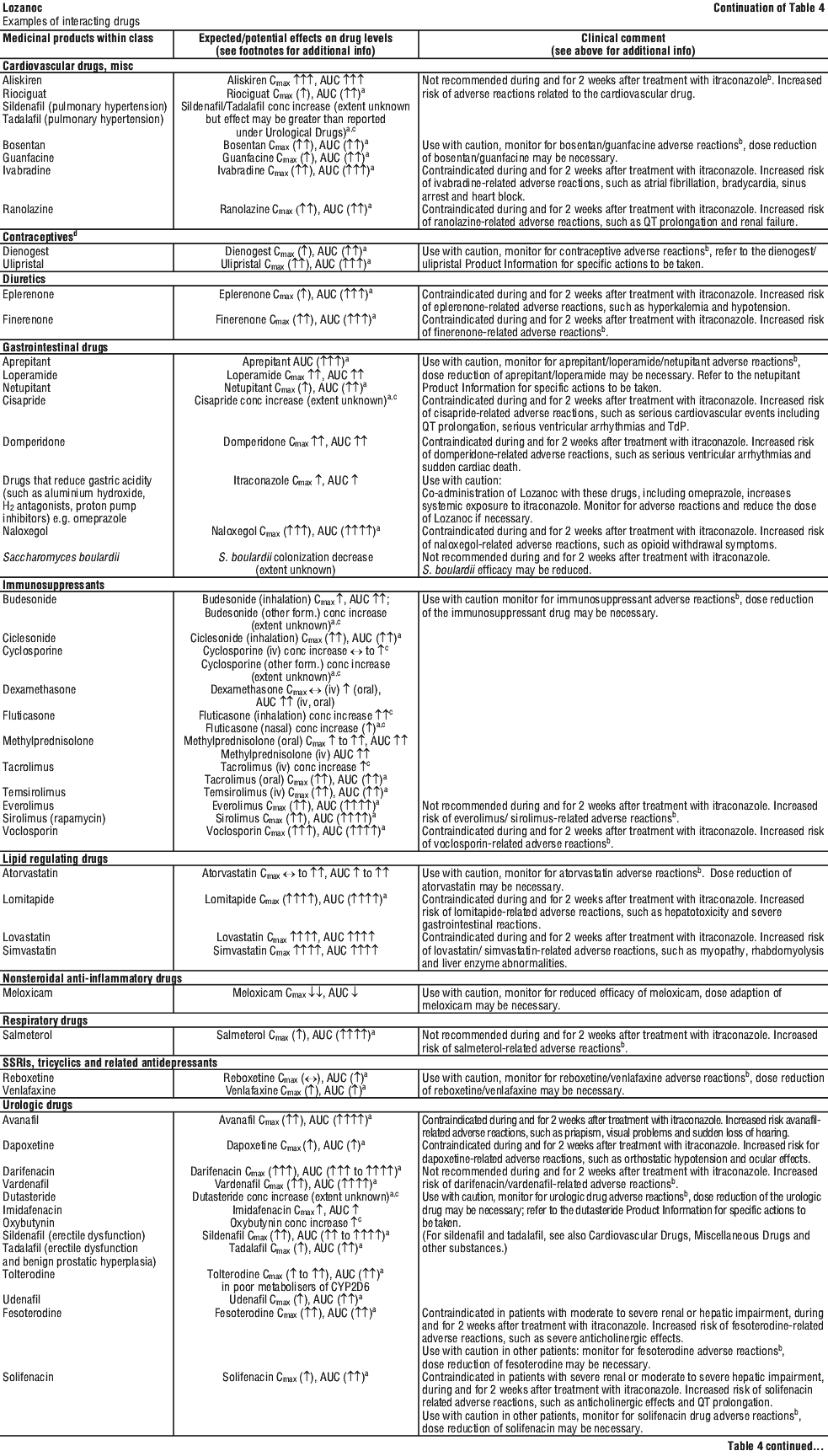
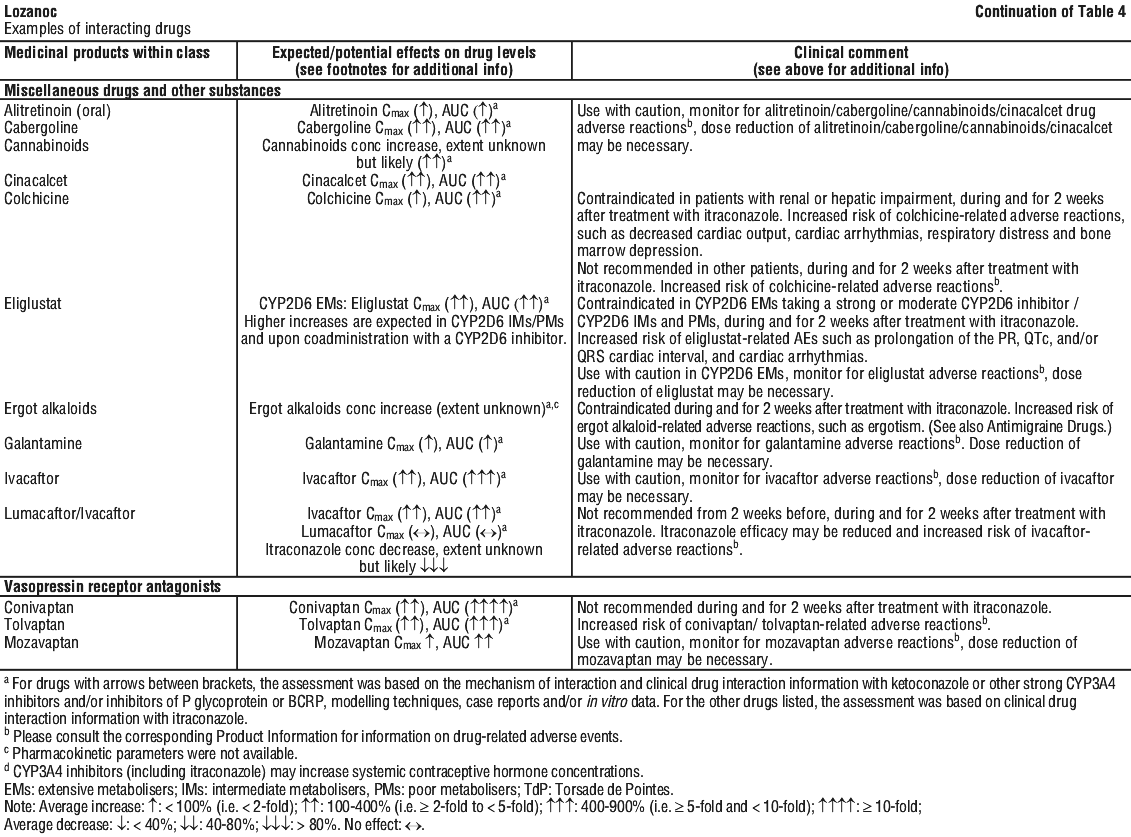 For the effect (middle column) the name of the parent drug is stated, even when the effect is related to the active moiety or the active metabolite of a prodrug.
For the effect (middle column) the name of the parent drug is stated, even when the effect is related to the active moiety or the active metabolite of a prodrug.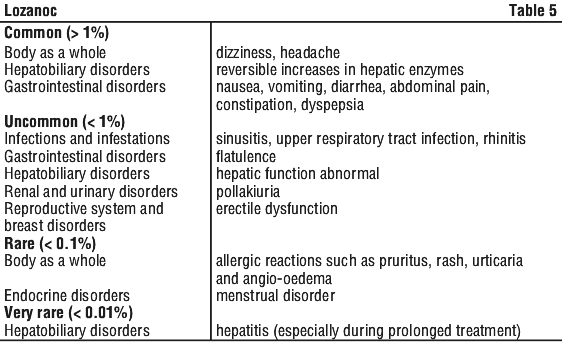 The following is a list of additional adverse effects associated with itraconazole. The adverse effects are related to the active substance and are not specifically formulation dependent. See Table 6.
The following is a list of additional adverse effects associated with itraconazole. The adverse effects are related to the active substance and are not specifically formulation dependent. See Table 6.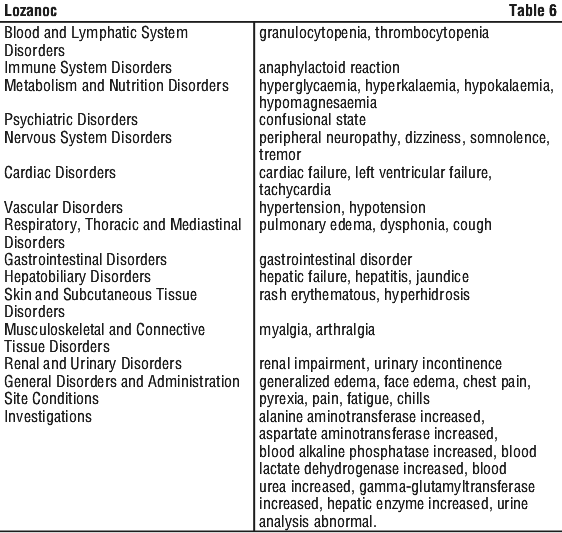
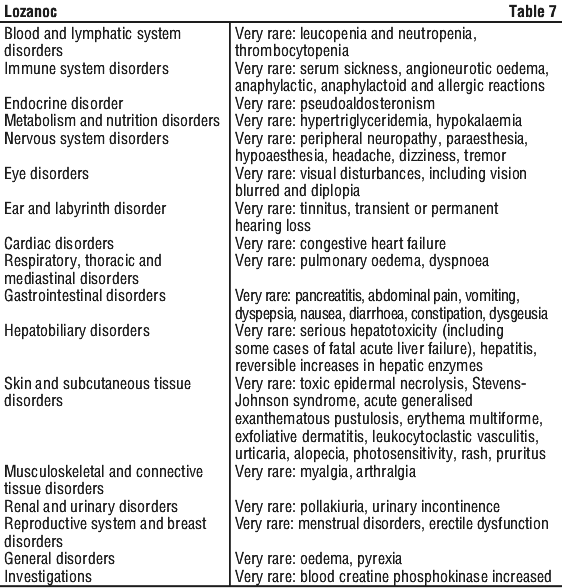
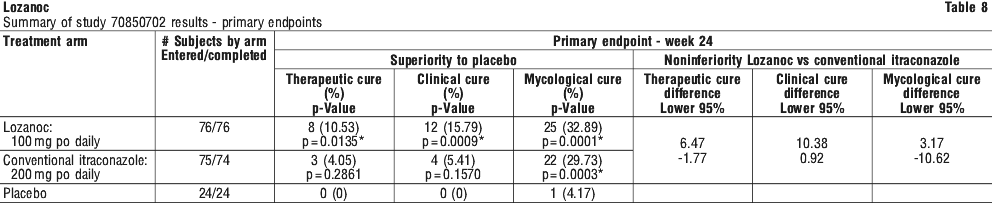 The following clinical trials were conducted with a conventional formulation of itraconazole. One capsule of Lozanoc 50 mg is therapeutically equivalent to one 100 mg capsule of conventional itraconazole capsules. The recommended dose for Lozanoc is therefore half the recommended dose for conventional itraconazole capsules.
The following clinical trials were conducted with a conventional formulation of itraconazole. One capsule of Lozanoc 50 mg is therapeutically equivalent to one 100 mg capsule of conventional itraconazole capsules. The recommended dose for Lozanoc is therefore half the recommended dose for conventional itraconazole capsules. Molecular formula: C35H38CI2N8O4.
Molecular formula: C35H38CI2N8O4.