1 Name of Medicine
Lurasidone hydrochloride.
2 Qualitative and Quantitative Composition
Each Lurasidone Sun tablet contains 20 mg, 40 mg, or 80 mg of lurasidone hydrochloride.
Lurasidone hydrochloride (HCl) is chemically identified as (3aR,4S,7R,7aS)-2-{(1R,2R)-2- [4-(1,2- benzisothiazol-3-yl) piperazin-1-ylmethyl] cyclohexylmethyl} hexahydro-4,7-methano-2H-isoindole- 1,3-dione hydrochloride. Lurasidone is an atypical antipsychotic belonging to the chemical class of benzisothiazol derivatives. It has antagonist activity on the dopamine 2 (D2) and serotonin (5‐HT)‐2A receptors. Lurasidone HCl (active entity) is a white to off‐white powder. It is very slightly soluble in water, practically insoluble or insoluble in 0.1 N HCl, slightly soluble in ethanol, sparingly soluble in methanol, practically insoluble or insoluble in toluene very slightly soluble in acetone and has a pKa of 7.6.
For the full list of excipients, see Section 6.1.
3 Pharmaceutical Form
Film‐coated tablet.
Lurasidone Sun 20 mg.
White to off‐white, round, film‐coated tablet, debossed with '578' on one side and plain on other side.
Lurasidone Sun 40 mg.
White to off‐white, round, film‐coated tablet, debossed with '684' on one side and plain on other side.
Lurasidone Sun 80 mg.
Pale green coloured, oval shaped, film‐coated tablet, debossed with '685' on one side and plain on other side.4.1 Therapeutic Indications
Lurasidone Sun is indicated for the treatment of schizophrenia in adults and adolescents (aged 13 to 17 years).
4.2 Dose and Method of Administration
Lurasidone Sun film‐coated tablets are intended for oral administration only.
The efficacy of lurasidone has been established at doses of 40, 80, 120 and 160 mg/day. The recommended starting dose is 40 mg once daily. Initial dose titration is not required. Patients should be treated with the lowest effective dose that provides optimal clinical response and tolerability, which is expected to be 40 mg or 80 mg once daily for most patients. Dose increase should be based on physician judgement and observed clinical response. In the six week controlled trials, there was no suggestion of added benefit with the 120 mg/day dose compared to 40 and 80 mg/day. In the pooled analyses, added benefit occurred at 160 mg/day compared to lower doses. Doses above 80 mg may be considered for certain patients based on individual clinical judgment. The maximum recommended dose is 160 mg/day.
Lurasidone Sun should be taken with food.
Children and adolescents.
The recommended starting dose of lurasidone in adolescent patients 13 to 17 years of age with schizophrenia is 40 mg/day. The maximum recommended dose in adolescent patients is 80 mg/day. Lurasidone was studied in adolescent patients 13 to 17 years of age with schizophrenia at daily doses of 40 mg and 80 mg. The safety and efficacy of lurasidone in children aged less than 13 years have not been established (see Section 4.4).
Due to difficulties associated with diagnosing schizophrenia in adolescents, diagnosis of schizophrenia in adolescents should be made by practitioners experienced in the diagnosis and management of adolescent psychiatric disorders.
The effectiveness of lurasidone has not been established in controlled studies for treatment duration of more than 6 weeks in adolescents. Therefore, if lurasidone is to be used for more than 6 weeks, the treating physician should periodically re‐evaluate the long‐term use of lurasidone for the individual patient.
Patients with renal impairment.
There are limited clinical data in patients with renal impairment. No dose adjustment for Lurasidone Sun is required in patients with mild (CrCl: 50 to 80 mL/min) renal impairment.
In patients with moderate (CrCl: 30 to < 50 mL/min) or severe (CrCl: < 30 mL/min) renal impairment, the recommended starting dose is 20 mg and the maximum dose should not exceed 80 mg once daily.
Patients with hepatic impairment.
There are limited clinical data in patients with hepatic impairment.
No dose adjustment for Lurasidone Sun is required in patients with mild hepatic impairment.
Dose adjustment is recommended in patients with moderate (Child‐Pugh Class B) hepatic impairment. The recommended starting dose is 20 mg. The dose in patients with moderate hepatic impairment should not exceed 80 mg.
Lurasidone Sun is not recommended in patients with severe (Child‐Pugh Class C) hepatic impairment.
Elderly patients.
No dose adjustment is necessary in elderly patients. Clinical studies of lurasidone in the treatment of schizophrenia did not include sufficient numbers of patients aged 65 and older to determine whether or not they respond differently from younger patients. In elderly patients with psychosis (65 to 85), lurasidone concentrations (20 mg/day) were similar to those in young subjects.
Dosing recommendations for older patients with normal renal function (CrCl ≥ 80 mL/min) are the same as for adults with normal renal function. However, as older patients may have diminished renal function, dose adjustments may be required according to their renal function status (see Section 4.2). Renal function and cardiovascular status should be assessed prior to commencing treatment with Lurasidone Sun.
Dose adjustment due to interactions.
If Lurasidone Sun is being prescribed and a moderate CYP3A4 inhibitor (e.g. diltiazem) is added to therapy, the Lurasidone Sun dose should be reduced to half of the original dose level.
Similarly, if a moderate CYP3A4 inhibitor is being prescribed and Lurasidone Sun is added to the therapy, the recommended starting dose of Lurasidone Sun is 20 mg per day, and the maximum dose of Lurasidone Sun is 80 mg per day.
Lurasidone Sun is contraindicated for use in combination with strong CYP3A4 inhibitors (e.g. ketoconazole, clarithromycin, ritonavir, and voriconazole) and strong CYP3A4 inducers (e.g. rifampin, St. John's wort, phenytoin, and carbamazepine).
Grapefruit and grapefruit juice should be avoided in patients taking Lurasidone Sun, as these may inhibit CYP3A4 and alter lurasidone concentrations.
Switching between antipsychotic medicinal products.
Due to different pharmacodynamic and pharmacokinetic profiles among antipsychotic medicinal products, supervision by a clinician is needed when switching to another antipsychotic product is considered medically appropriate.
Abuse.
Lurasidone has not been systematically studied in humans for its potential for abuse or physical dependence or its ability to induce tolerance. While clinical studies with lurasidone did not reveal any tendency for drug‐seeking behaviour, these observations were not systematic and it is not possible to predict the extent to which a CNS‐active drug will be misused, diverted and/or abused once it is marketed. Patients should be evaluated carefully for a history of drug abuse, and such patients should be observed carefully for signs of Lurasidone Sun misuse or abuse (e.g. development of tolerance, drug‐seeking behaviour, increases in dose).4.3 Contraindications
Lurasidone Sun (lurasidone HCl) is contraindicated in any patient with a known hypersensitivity to lurasidone HCl or any components in the formulation.
Lurasidone is contraindicated with strong CYP3A4 inhibitors (e.g. ketoconazole, clarithromycin, ritonavir, and voriconazole) and strong CYP3A4 inducers (e.g. rifampin, St. John's wort, phenytoin, and carbamazepine) (see Section 4.4).
4.4 Special Warnings and Precautions for Use
Increased mortality in elderly patients with dementia‐related psychosis.
In placebo‐controlled trials with similar atypical antipsychotics in elderly subjects with dementia‐related psychosis, there was a higher incidence of fatalities, compared to placebo‐treated subjects. Elderly patients with dementia‐related psychosis treated with atypical antipsychotics are at an increased risk of death compared to placebo. A meta‐analysis of seventeen placebo controlled trials with dementia-related behavioural disorders showed a risk of death in the drug‐treated patients of approximately 1.6 to 1.7 times that seen in placebo‐treated patients. The clinical trials included in the meta‐analysis were undertaken with olanzapine, aripiprazole, risperidone and quetiapine. Over the course of these trials averaging about 10 weeks in duration, the rate of death in drug‐treated patients was about 4.5%, compared to a rate of approximately 2.6% in the placebo group. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular (e.g. heart failure, sudden death) or infectious (e.g. pneumonia) in nature. Lurasidone is not approved for the treatment of elderly patients with dementia‐related psychosis or behavioural disorders.
Cerebrovascular adverse reactions, including stroke in elderly patients with dementia‐related psychosis.
In placebo‐controlled trials with risperidone, aripiprazole, and olanzapine in elderly subjects with dementia, there was a higher incidence of cerebrovascular adverse reactions (cerebrovascular accidents and transient ischemic attacks), including fatalities, compared to placebo‐treated subjects. Lurasidone Sun is not approved for the treatment of patients with dementia‐related psychosis.
Neuroleptic malignant syndrome.
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS), characterised by hyperthermia, muscle rigidity, autonomic instability, altered consciousness and elevated serum creatine phosphokinase levels, has been reported in association with administration of antipsychotic medicines, including lurasidone. Additional signs may include myoglobinuria (rhabdomyolysis) and acute renal failure. If a patient develops signs and symptoms indicative of NMS, or presents with unexplained high fever without additional clinical manifestations of NMS, all antipsychotic medicines, including lurasidone, must be discontinued.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. If reintroduced, the patient should be carefully monitored, since recurrences of NMS have been reported.
Seizures.
As with other antipsychotic medicines, lurasidone should be used cautiously in patients with a history of seizures or with conditions that lower the seizure threshold, e.g. Alzheimer's dementia.
Conditions that lower the seizure threshold may be more prevalent in patients 65 years or older.
Suicide.
The possibility of a suicide attempt is inherent in psychotic illness and close supervision of high‐risk patients should accompany treatment with an antipsychotic medicine. Prescriptions for lurasidone should be written for the smallest quantity of tablets consistent with good patient management in order to reduce the risk of overdose.
Weight gain.
Weight gain has been observed with atypical antipsychotic use. Clinical monitoring of weight is recommended.
Pooled data from short‐term, placebo‐controlled schizophrenia studies are presented in Table 1. The mean weight gain was 0.43 kg for lurasidone‐treated patients compared to ‐0.02 kg for placebo‐treated patients. The proportion of patients with a ≥ 7% increase in body weight (at Endpoint) was 4.8% for lurasidone‐treated patients versus 3.3% for placebo‐treated patients.
 In long‐term controlled studies, for patients who had normal BMI status at baseline (18.5 to < 25.0), the rate of clinically significant weight gain (≥ 7% increase in BMI) at month 12 was 12.4%, 34.5% and 5.6% and to study endpoint (LOCF) was 9.6%, 17.7% and 8.3% of the lurasidone, risperidone and quetiapine XR groups, respectively. For those who were overweight at baseline (BMI 25.0 to < 30.0), the rate of clinically significant weight gain at study endpoint was 6.3%, 14.1% and 9.5%, in patients given lurasidone, risperidone and quetiapine XR, respectively.
In long‐term controlled studies, for patients who had normal BMI status at baseline (18.5 to < 25.0), the rate of clinically significant weight gain (≥ 7% increase in BMI) at month 12 was 12.4%, 34.5% and 5.6% and to study endpoint (LOCF) was 9.6%, 17.7% and 8.3% of the lurasidone, risperidone and quetiapine XR groups, respectively. For those who were overweight at baseline (BMI 25.0 to < 30.0), the rate of clinically significant weight gain at study endpoint was 6.3%, 14.1% and 9.5%, in patients given lurasidone, risperidone and quetiapine XR, respectively.
Orthostatic hypotension, syncope and cardiovascular disease.
Lurasidone may cause orthostatic hypotension, perhaps due to its α1‐adrenergic receptor antagonism. Lurasidone should be used with caution in patients with known cardiovascular disease (e.g. heart failure, history of myocardial infarction, ischemia, or conduction abnormalities), cerebrovascular disease, or conditions that predispose the patient to hypotension (e.g. dehydration, hypovolemia, and treatment with antihypertensive medications). Monitoring of orthostatic vital signs should be considered in patients who are vulnerable to hypotension.
Caution should be exercised when lurasidone is prescribed in patients with known cardiovascular disease or family history of QT prolongation, hypokalaemia, and in concomitant use with other medicinal products thought to prolong the QT interval. Lurasidone has not been evaluated or used to any appreciable extent in patients with a recent history of myocardial infarction or unstable heart disease. Patients with these diagnoses were excluded from premarketing clinical trials. Due to the risk of orthostatic hypotension with lurasidone, caution should be observed in patients with known cardiovascular disease.
Electrocardiogram (ECG) measurements were taken at various time points during the lurasidone clinical trial program. No post‐baseline QT prolongations exceeding 500 msec were reported in patients treated with lurasidone. Within a subset of patients defined as having an increased cardiac risk, no potentially important changes in ECG parameters were observed. No cases of torsade de pointes or other severe cardiac arrhythmias were observed in the pre‐marketing clinical program.
The effects of lurasidone on the QT/QTc interval were evaluated in a dedicated QT study involving 87 clinically stable patients with schizophrenia or schizoaffective disorder, who were treated with lurasidone doses of 120 mg daily, 600 mg daily, or ziprasidone 160 mg daily. Holter monitor‐derived electrocardiographic assessments were obtained over an eight hour period at baseline and steady state. The maximum mean (upper 1‐sided, 95% CI) increases of baseline‐adjusted QTc intervals based on individual correction method (QTcI) were 0.36 (1.40) ms for lurasidone 120 mg and 1.69 (6.51) ms for lurasidone 600 mg. No patients treated with lurasidone experienced QTc increases > 60 msec from baseline, nor did any patient experience a QTc of > 500 msec.
Tardive dyskinesia.
Tardive dyskinesia is a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements that can develop in patients treated with antipsychotic medicines, including lurasidone. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic medicines differ in their potential to cause tardive dyskinesia is unknown.
The risk of development of tardive dyskinesia may be reduced by using the lowest effective dose. Lurasidone should not be continued in patients who have not responded to treatment. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that (1) is known to respond to antipsychotic medicines, and (2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient on lurasidone, discontinuation of lurasidone should be considered.
Venous thromboembolism.
Cases of venous thromboembolism (VTE) have been reported with antipsychotic medicinal products. Since patients treated with antipsychotics often present with acquired risk factors for VTE, all possible risk factors for VTE should be identified before and during treatment with lurasidone and preventive measures undertaken.
Hyperprolactinemia.
As with other medicines that antagonize dopamine D2 receptors, lurasidone elevates prolactin levels. Hyperprolactinemia may suppress hypothalamic GnRH, resulting in reduced pituitary gonadotrophin secretion. This, in turn, may inhibit reproductive function by impairing gonadal steroidogenesis in both female and male patients. Galactorrhea, amenorrhea, gynecomastia, and impotence have been reported with prolactin‐elevating compounds. Long‐standing hyperprolactinemia, when associated with hypogonadism, may lead to decreased bone density in both female and male patients.
Premenopausal women who develop secondary amenorrhoea of greater than six months duration should receive appropriate preventative therapy to avoid hypooestrogenic bone loss.
In short‐term, placebo‐controlled schizophrenia studies, the median change from baseline to endpoint in prolactin levels for lurasidone‐treated patients was +0.4 nanogram/mL and was ‐1.9 nanogram/mL in the placebo-treated patients. The median change from baseline to endpoint for males was +0.5 nanogram/mL and for females was ‐0.2 nanogram/mL. Median changes for prolactin by dose are shown in Table 2.
 The proportion of patients with prolactin elevations ≥ 5x upper limit of normal (ULN) was 2.8% for lurasidone‐treated patients versus 1.0% for placebo‐treated patients. The proportion of female patients with prolactin elevations ≥ 5x ULN was 5.7% for lurasidone‐treated patients versus 2.0% for placebo-treated female patients. The proportion of male patients with prolactin elevations ≥ 5x ULN was 1.6% versus 0.6% for placebo‐treated male patients.
The proportion of patients with prolactin elevations ≥ 5x upper limit of normal (ULN) was 2.8% for lurasidone‐treated patients versus 1.0% for placebo‐treated patients. The proportion of female patients with prolactin elevations ≥ 5x ULN was 5.7% for lurasidone‐treated patients versus 2.0% for placebo-treated female patients. The proportion of male patients with prolactin elevations ≥ 5x ULN was 1.6% versus 0.6% for placebo‐treated male patients.
Leukopenia, neutropenia and agranulocytosis.
Leukopenia/neutropenia has been reported during treatment with antipsychotic agents. Agranulocytosis (including fatal cases) has been reported with other agents in the class.
Possible risk factors for leukopenia/neutropenia include pre‐existing low white blood cell count (WBC) and history of drug induced leukopenia/neutropenia. Patients with a pre‐existing low WBC or a history of drug‐induced leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and lurasidone should be discontinued at the first sign of decline in WBC, in the absence of other causative factors.
Patients with neutropenia should be carefully monitored for fever or other symptoms or signs of infection and treated promptly if such symptoms or signs occur. Patients with severe neutropenia (absolute neutrophil count < 1000/mm3) should discontinue lurasidone and have their WBC followed until recovery.
Hyperglycemia and diabetes mellitus.
Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population. Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should be monitored regularly for worsening of glucose control. Patients treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. Pooled data from short‐term, placebo‐controlled schizophrenia studies are presented in Table 3.

Dyslipidemia.
Undesirable alterations in lipids have been observed in patients treated with atypical antipsychotics.
Pooled data from short‐term, placebo‐controlled schizophrenia studies are presented in Table 4.
 In long term controlled studies the rate of markedly abnormal metabolic parameters was similar between lurasidone, risperidone and quetiapine XR. For patients given any dose of lurasidone the rate of shift from normal to high total cholesterol was 2.2% and triglycerides was 6.2%.
In long term controlled studies the rate of markedly abnormal metabolic parameters was similar between lurasidone, risperidone and quetiapine XR. For patients given any dose of lurasidone the rate of shift from normal to high total cholesterol was 2.2% and triglycerides was 6.2%.
Body temperature regulation.
Disruption of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing lurasidone for patients who will be experiencing conditions that may contribute to an elevation in core body temperature, e.g. exercising strenuously, exposure to extreme heat, receiving concomitant medication with anticholinergic activity, or being subject to dehydration.
Dysphagia.
Oesophageal dysmotility and aspiration have been associated with the use antipsychotic medicines. Aspiration pneumonia is a common cause of morbidity and mortality in elderly patients, in particular those with advanced Alzheimer's dementia. Lurasidone and other antipsychotic medicines should be used cautiously in patients at risk for aspiration pneumonia.
Sleep apnoea.
Sleep apnoea and related disorders have been reported in patients treated with antipsychotic medicines, with or without prior history of sleep apnoea, and with or without concomitant weight‐gain. In patients who have a history of or are at risk for sleep apnoea, or who are concomitantly using central nervous system depressants, antipsychotic medicines including lurasidone should be used with caution.
Use in the elderly.
Clinical studies with lurasidone did not include sufficient numbers of patients aged 65 and older to determine whether or not they respond differently from younger patients. In elderly patients with psychosis (65 to 85), lurasidone concentrations (20 mg/day) were similar to those in young subjects. It is unknown whether dose adjustment is necessary on the basis of age alone.
Elderly patients with schizophrenia may have reduced renal function and co‐existing cardiovascular disease. In these patients, the starting dose should be reduced (see Section 4.2).
Elderly patients with dementia‐related psychosis treated with lurasidone are at an increased risk of death compared to placebo. Lurasidone is not approved for the treatment of patients with dementia‐related psychosis.
Paediatric use.
The safety and efficacy of lurasidone has been established in adolescents aged 13 to 17 years. The safety and efficacy of lurasidone in children aged less than 13 years have not yet been established. Lurasidone is not recommended in children aged less than 13 years.
Effects on laboratory tests.
See Section 4.8 Adverse Effects (Undesirable Effects), Clinical laboratory changes.4.5 Interactions with Other Medicines and Other Forms of Interactions
Given the primary central nervous system effects of lurasidone (see Section 4.8), lurasidone should be used with caution in combination with other centrally acting medicines and alcohol.
Effects on lurasidone.
Based on in vitro studies, lurasidone is not a substrate of CYP1A1, CYP1A2, CYP2A6, CYP4A11, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6 or CYP2E1 enzymes. This suggests that an interaction of lurasidone with medicines that are inhibitors or inducers of these enzymes is unlikely.
Grapefruit and grapefruit juice inhibits CYP3A4 and may increase the serum levels of lurasidone. It should not be taken with lurasidone.
Lurasidone is predominantly metabolized by CYP3A4; interaction of lurasidone with strong and moderate inhibitors or inducers of this enzyme has been observed (Table 5). Lurasidone should not be used in combination with strong inhibitors or inducers of this enzyme.

Effects on coadministered medicines digoxin (P‐gp substrate).
Coadministration of lurasidone (120 mg/day) at steady state with a single dose of digoxin (0.25 mg) increased Cmax and AUC(0‐24) for digoxin by approximately 9% and 13%, respectively relative to digoxin alone. Digoxin dose adjustment is not required when coadministered with lurasidone.
Lithium.
Coadministration of lurasidone (120 mg/day) and lithium (1200 mg/day) at steady state resulted in comparable mean lithium Cmax values on Day 4 (0.65 mmol/L) and Day 8 (0.75 mmol/L) and maintenance of the therapeutic range for lithium (0.6 to 1.2 mmol/L). No adjustment of lithium dose is required when coadministered with lurasidone.
Midazolam (CYP3A4 substrate).
Coadministration of lurasidone (120 mg/day) at steady state with a single dose of 5 mg midazolam increased midazolam Cmax and AUC(0‐24) by approximately 21% and 44%, respectively relative to midazolam alone. Midazolam dose adjustment is not required when coadministered with lurasidone.
Oral contraceptive (estrogen/progesterone).
Coadministration of lurasidone (40 mg/day) at steady state with an oral contraceptive (OC) containing ethinyl estradiol and norelgestimate resulted in equivalent AUC(0‐24) and Cmax of ethinyl estradiol and norelgestromin relative to OC administration alone. Also, sex hormone binding globulin levels were not meaningfully affected by coadministration of lurasidone and OC. Dose adjustment of OC dose is not required when coadministered with lurasidone.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
There was no effect on mating performance or fertility in male rats treated with lurasidone prior to and during mating at oral doses up to 150 mg/kg/day, corresponding to about 9 times the maximum recommended human dose (MRHD) based on body surface area.
In female rats given lurasidone prior to and during mating and in early pregnancy, oestrus was prolonged and mating was delayed at oral doses of 1.5‐150 mg/kg/day (0.1‐9 times the MRHD based on body surface area); the no‐effect dose was 0.1 mg/kg/day. At the 150 mg/kg/day dose, reductions were observed in the proportion of females mating, fertility and the number of corpora lutea, implantations and live fetuses per dam. These changes were reversed after a 14‐day treatment‐free period. The no-effect dose for reduced fertility was 15 mg/kg/day (approximately the MRHD based on body surface area).
(Category B1)
There are no adequate and well‐controlled studies in pregnant women. Patients should be advised to notify their physician if they become pregnant or intend to become pregnant during treatment with lurasidone. Lurasidone should be used in pregnancy only if the potential benefit justifies the potential risk to the fetus.
Human data.
Neonates exposed to antipsychotic medicines during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress and feeding disorder in these neonates. These complications have varied in severity; while in some cases symptoms have been self‐limited, in other cases neonates have required intensive care unit support and prolonged hospitalization.
Animal data.
No teratogenic or other adverse effects on fetuses were observed in studies in which lurasidone was administered during the period of organogenesis to rats and rabbits at respective oral doses up to 25 and 50 mg/kg/day, corresponding to 1.5 and 5 times, respectively, the MRHD based on body surface area. No effects on delivery or pup development were observed in rats given lurasidone from early gestation to weaning at oral doses up to 10 mg/kg/day (about half the MRHD based on body surface area).
Lurasidone and/or other metabolites were excreted in milk of rats during lactation and the same would be expected for human milk. Women receiving lurasidone should not breastfeed.4.7 Effects on Ability to Drive and Use Machines
Lurasidone, like other antipsychotics, has the potential to impair judgment, thinking or motor skills. Patients should not operate hazardous machinery, including motor vehicles, until they are reasonably certain that therapy with lurasidone does not affect them adversely.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit‐risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting‐problems.
The following findings are derived from a clinical study database for lurasidone consisting of 2,905 patients with schizophrenia exposed to one or more doses with a total experience of 985.3 patient‐years. Of these patients, 1,508 participated in short‐term, placebo‐controlled schizophrenia studies with doses of 20 mg, 40 mg, 80 mg, 120 mg or 160 mg once daily. A total of 769 lurasidone‐treated patients had at least 24 weeks and 371 lurasidone‐treated patients had at least 52 weeks of exposure.
The most common adverse events (incidence ≥ 5% and at least twice the rate of placebo) in patients treated with lurasidone were somnolence, akathisia, nausea and parkinsonism.
Adverse events reported with the use of lurasidone (incidence of 2% or greater, rounded to the nearest percent and lurasidone incidence greater than placebo) that occurred during acute therapy (up to 6weeks in patients with schizophrenia) are shown in Table 6.

Dose‐related adverse events.
Akathisia and extrapyramidal symptoms were dose‐related in adults with schizophrenia. The frequency of akathisia increased with dose up to 120 mg/day (5.6% for lurasidone 20 mg, 10.7% for lurasidone 40 mg, 12.3% for lurasidone 80 mg, and 22.0% for lurasidone 120 mg). Akathisia was reported by 7.4% (9/121) of adult patients receiving 160 mg/day. Akathisia occurred in 3.0% of adult subjects receiving placebo. The frequency of extrapyramidal symptoms increased with dose up to 120 mg/day (5.6% for lurasidone 20 mg, 11.5% for lurasidone 40 mg, 11.9% for lurasidone 80 mg, and 22.0% for lurasidone 120 mg).
Due to the observed dose‐related adverse effects, the recommended starting dose of lurasidone in adults (40 or 80 mg/day) should be utilized for initial treatment based on clinical evaluation. Dose increases should be based on physician judgment and observed clinical response.
Extrapyramidal symptoms.
In the short‐term, placebo‐controlled schizophrenia studies, for lurasidone‐treated adult patients, the incidence of reported events related to extrapyramidal symptoms (EPS), excluding akathisia and restlessness, was 13.5% versus 5.8% for placebo‐treated patients. The incidence of akathisia for lurasidone‐treated adult patients was 12.9% versus 3.0% for placebo‐treated adult patients. Incidence of EPS by dose is provided in Table 7.

Other adverse reactions observed during the premarketing evaluation of lurasidone.
Following is a list of other adverse reactions and laboratory investigations reported by adult patients treated with lurasidone at multiple doses of ≥ 20 mg once daily during any phase of a study within the database of 3,202 patients. The reactions listed are those that could be of clinical relevance, as well as reactions that are plausibly drug‐related on pharmacologic or other grounds. Reactions listed in Table 6 or those that appear elsewhere (see Section 4.8 Adverse Effects (Undesirable Effects)) are not included. Although the reactions reported occurred during treatment with lurasidone, they were not necessarily caused by it.
The following adverse reactions are classified by system organ class and are according to the following definitions: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1000 to < 1/100), rare (≥ 1/10,000 to < 1/1000), very rare (< 1 /10,000). Within each frequency grouping, adverse reactions are presented in order of decreasing seriousness.
Blood and lymphatic system disorders.
Uncommon: anaemia; rare: leukopenia, neutropenia.
Cardiac disorders.
Common: tachycardia; uncommon: angina pectoris, bradycardia; rare: A-V block 1st degree.
Ear and labyrinth disorders.
Uncommon: vertigo.
Eye disorders.
Common: vision blurred.
Gastrointestinal disorders.
Common: abdominal pain, diarrhoea; uncommon: dysphagia, gastritis.
General disorders and administrative site conditions.
Rare: sudden death.
Investigations.
Common: creatinine phosphokinase increased.
Metabolism and nutritional system disorders.
Common: decreased appetite; uncommon: hyponatremia.
Musculoskeletal and connective tissue disorders.
Rare: rhabdomyolysis.
Nervous system disorders.
Uncommon: dysarthria:, cerebrovascular accident, syncope, tardive dyskinesia; rare: neuroleptic malignant syndrome, seizure.
Psychiatric disorders.
Uncommon: abnormal dreams, panic attack, sleep disorder; rare: suicidal behaviour.
Renal and urinary disorders.
Uncommon: dysuria; rare: renal failure.
Reproductive system and breast disorders.
Uncommon: amenorrhea, dysmenorrhoea, erectile dysfunction rare: breast pain, galactorrhoea, breast enlargement.
Skin and subcutaneous tissue disorders.
Common: rash, pruritus; rare: angioedema.
Vascular disorders.
Common: hypertension; uncommon: orthostatic hypotension.
Paediatric population.
The following findings are based on the short‐term, placebo‐controlled pre‐marketing study for schizophrenia in which lurasidone was administered at daily doses ranging from 40 to 80 mg (n=326). The most common adverse events reported (incidence ≥ 5% and at least twice the rate of placebo) in patients treated with lurasidone were somnolence, nausea, akathisia, and vomiting.
Adverse events reported with the use of lurasidone (incidence of 2% or greater, rounded to the nearest percent and lurasidone incidence greater than placebo) that occurred during acute therapy (up to 6 weeks in adolescent patients with schizophrenia) are shown in Table 8.

Clinical laboratory changes.
Serum creatinine.
In short‐term, placebo‐controlled trials in adults, the mean change from baseline in serum creatinine was +0.05 mg/dL for lurasidone‐treated patients compared to +0.02 mg/dL for placebo-treated patients. A creatinine shift from normal to high occurred in 3.0% (43/1,453) of lurasidone-treated patients and 1.6% (11/681) on placebo (Table 9). The threshold for high creatinine value varied from > 0.79 to > 1.3 mg/dL based on the centralized laboratory definition for each study. See Table 10.


Adverse reactions observed post‐marketing.
As these reactions are reported voluntarily from a population of uncertain size, the incidence rate of these adverse reactions cannot be estimated (frequency unknown).
The following adverse reactions, classified by system organ class and frequency, have been identified with lurasidone in the post‐marketing period.
Immune system disorders.
Frequency unknown: hypersensitivity (hypersensitivity may include symptoms such as throat swelling, tongue swelling, urticaria, or symptoms of angioedema. Hypersensitivity may also include symptoms of severe cutaneous reactions such as dermatitis bullous, rash maculopapular, rash pustular, skin eruption and skin exfoliation).
Metabolism and nutritional system disorders.
Frequency unknown: hyponatraemia.
The following adverse reactions, classified by system organ class and frequency, have been identified with atypical antipsychotic medicines including lurasidone in the post‐marketing period.
Psychiatric disorders.
Frequency unknown: somnambulism (sleepwalking) and related behaviours including sleep‐related eating disorder.4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
There is no specific antidote to lurasidone, therefore, appropriate supportive measures should be instituted and close medical supervision and monitoring should continue until the patient recovers.
Cardiovascular monitoring should commence immediately, including continuous electrocardiographic monitoring for possible arrhythmias. If antiarrhythmic therapy is administered, disopyramide, procainamide, and quinidine carry a theoretical hazard of additive QT‐prolonging effects when administered in patients with an acute overdose of lurasidone. Similarly the alpha‐blocking properties of bretylium might be additive to those of lurasidone, resulting in problematic hypotension. Hypotension and circulatory collapse should be treated with appropriate measures. Adrenaline and dopamine should not be used or other sympathomimetics with beta agonist activity, since beta stimulation may worsen hypotension in the setting of lurasidone‐induced alpha blockade. In case of severe extrapyramidal symptoms, anticholinergic medication should be administered.
Administration of activated charcoal together with a laxative should be considered.
The possibility of obtundation, seizures, or dystonic reaction of the head and neck following overdose may create a risk of aspiration with induced emesis.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action.
The mechanism of action of lurasidone, as with other medicines having efficacy in schizophrenia, is not fully understood. However, based on its receptor pharmacology, it is believed that the efficacy of lurasidone is mediated mainly through antagonist activity at dopamine D2 and 5‐hydroxytryptamine (5‐HT, serotonin) 5‐HT2A receptors.
In vitro receptor binding studies revealed that lurasidone binds with high affinity at human D2 receptors (Ki=0.994 nanoM) and 5‐HT2A (Ki=0.47 nanoM) and 5‐HT7 (Ki=0.495 nanoM) receptors, with moderate affinity at human α2C adrenergic receptors (Ki=10.8 nanoM), D3 receptors (Ki=15.7 nanoM) and 5‐HT1A (Ki=6.38 nanoM) receptors, and with weak affinity at human D4.4 (Ki=29.7 nanoM) and α2A (Ki = 40.7 nanoM) and α1A (Ki = 35.7 nanoM) adrenergic receptors. Lurasidone exhibits little or no affinity for human histamine H1 and muscarinic M1 receptors (IC50 > 1,000 nanoM). Lurasidone is a partial agonist at 5‐HT1A receptors but is believed to act as an antagonist at all the other receptors.
Lurasidone doses ranging from 10 to 80 mg administered to healthy subjects produced a dose‐dependent reduction in the binding of 11C‐raclopride, a D2/D3 receptor ligand, in the caudate, putamen and ventral striatum detected by positron emission tomography.
After single administration at doses of 20 and 40 mg in a quantitative electroencephalographic evaluation, lurasidone decreased the threshold of flicker discrimination in the flicker test without affecting GFP value in any frequency band in electroencephalography.
Clinical trials.
The efficacy of lurasidone in the treatment of adults with schizophrenia was established in five short‐term (6‐week), placebo‐controlled, studies in adult patients (mean age of 38.4 years, range 18‐72) who met DSM‐IV criteria for schizophrenia. An active control arm (olanzapine or quetiapine XR) was included in two studies to assess assay sensitivity.
Several instruments were used for assessing psychiatric signs and symptoms in these studies:
1. Positive and Negative Syndrome Scale (PANSS), is a multi‐item inventory of general psychopathology used to evaluate the effects of drug treatment in schizophrenia. PANSS total scores may range from 30 to 210.
2. Brief Psychiatric Rating Scale derived (BPRSd), derived from the PANSS, is a multi‐item inventory primarily focusing on positive symptoms of schizophrenia, whereas the PANSS includes a wider range of positive, negative and other symptoms of schizophrenia. BPRSd scores may range from 18 to 126.
3. The Clinical Global Impression severity scale (CGI‐S) is a validated clinician‐rated scale that measures the subject's current illness state on a 1 to 7‐point scale.
The endpoint associated with each instrument is change from baseline in the total score to the end of Week 6. These changes are then compared to placebo changes for the lurasidone and control groups.
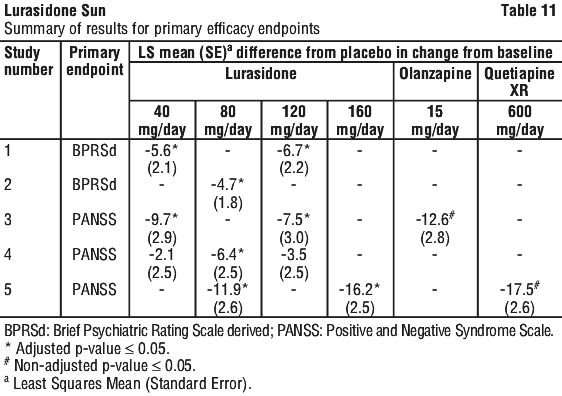
The results of the studies follow (Table 11):
1. In a 6‐week, placebo‐controlled trial (N=145) involving two fixed doses of lurasidone (40 or 120 mg/day), both doses of lurasidone at Endpoint were superior to placebo on the BPRSd total score, and the CGI‐S.
2. In a 6‐week, placebo‐controlled trial (N=180) involving a fixed dose of lurasidone (80 mg/day), lurasidone at Endpoint was superior to placebo on the BPRSd total score, and the CGI‐S.
3. In a 6‐week, placebo and active‐controlled trial (N=473) involving two fixed doses of lurasidone (40 or 120 mg/day) and an active control (olanzapine), both lurasidone doses and the active control at Endpoint were superior to placebo on the PANSS total score, and the CGI‐S.
4. In a 6‐week, placebo‐controlled trial (N=489) involving three fixed doses of lurasidone (40, 80 or 120 mg/day), only the 80 mg/day dose of lurasidone at Endpoint was superior to placebo on the PANSS total score, and the CGI‐S.
5. In a 6‐week, placebo and active‐controlled trial (N=482) involving two fixed doses of lurasidone (80 or 160 mg/day) and an active control (quetiapine XR), both lurasidone doses and the active control at Endpoint were superior to placebo on the PANSS total score, and the CGI‐S.
 Examination of population subgroups based on age (there were few patients over 65), gender and race did not reveal any clear evidence of differential responsiveness.
Examination of population subgroups based on age (there were few patients over 65), gender and race did not reveal any clear evidence of differential responsiveness.
An analysis of adult patients with a ≥ 30% reduction from baseline PANSS score (clinical response analysis) was performed in three of these studies. The placebo response rate was around 35% across the studies and the response rates for lurasidone 40 mg, 80 mg and 120 mg were all around 50%, giving a 15% difference in response rates from placebo and a NNT of 6.7 for one patient to achieve a clinically significant improvement. One study assessed efficacy of the 160 mg dose and 120 patients were given this dose. The response rate for 160 mg lurasidone was 63%, a NNT of approximately 3.6. There was limited evidence of dose response for doses between 40 mg and 80 mg.
Adolescents.
The efficacy of lurasidone in the treatment of schizophrenia in adolescent patients (13 to 17 years of age) was evaluated in one 6‐week, placebo‐controlled study of patients (N=327) who met DSM‐IV criteria for schizophrenia [D1050301]. Both the 80 mg and 40 mg doses of lurasidone demonstrated superiority over placebo on the PANSS total score after 6 weeks of double‐blind treatment (Table 12).

Maintenance of effect.
A double‐blind study compared flexibly dosed lurasidone (40 to 160 mg daily) with flexibly dosed quetiapine XR (200 to 600 mg daily) for up to 12 months in adult patients with schizophrenia who had shown a clinical response to lurasidone in a short‐term study. The mean daily dose of lurasidone was 125.5 mg and of quetiapine XR was 629.6 mg. Relapses were reported in 21% of subjects given lurasidone and in 27% given quetiapine XR. The probability of relapse by month 12 was 23.7% and 33.6% for lurasidone and quetiapine, respectively. The relapse hazard ratio of lurasidone versus quetiapine XR was 0.728 (95% CI: 0.41, 1.29).
5.2 Pharmacokinetic Properties
The activity of lurasidone is primarily due to the parent drug. The pharmacokinetics of lurasidone is dose-proportional within a total daily dose range of 20 mg to 160 mg. Steady‐state concentrations of lurasidone are reached within 7 days of starting lurasidone. Following administration of 40 mg the mean (%CV) elimination half‐life was 18 (7) hours.
Absorption.
Lurasidone is absorbed and reaches peak serum concentrations in approximately 1‐3 hours. It is estimated that 9‐19% of an administered dose is absorbed. In a food effect study, lurasidone mean Cmax and AUC were about 3‐times and 2‐times, respectively, when administered with food compared to the levels observed under fasting conditions. Lurasidone exposure was not affected as meal size was increased from 350 to 1000 calories and was independent of meal fat content (see Section 4.2).
Distribution.
Following administration of 40 mg of lurasidone, the mean (%CV) apparent volume of distribution was 6173 (17.2) L. Lurasidone is highly bound (~99%) to serum proteins.
Metabolism.
Lurasidone is metabolised mainly via CYP3A4. The major biotransformation pathways are oxidative N‐dealkylation, hydroxylation of norbornane ring, and S‐oxidation. Lurasidone is metabolised into two nonmajor active metabolites (ID‐14283 and ID‐14326) and two major non‐active metabolites (ID‐20219 and ID‐20220). Lurasidone and its metabolites ID‐14283, ID‐14326, ID‐20219 and ID‐20220 correspond to approximately 11.4, 4.1, 0.4, 24 and 11% respectively, of serum radioactivity respectively. Lurasidone is a single isomer form, which does not appear to undergo conversion to other enantiomers on metabolism.
Excretion.
Total excretion of radioactivity in urine and faeces combined was approximately 89%, with about 80% recovered in faeces and 9% recovered in urine, after a single dose of [14C]‐labeled lurasidone. Following administration of 40 mg the mean (%CV) apparent clearance was 3,902 (18.0) mL/min.
Special populations.
Renal impairment.
After administration of a single dose of 40 mg lurasidone to 27 patients with mild (n=9; CrCl: 50 to 80 mL/min), moderate (n=9; CrCl: 30 to < 50 mL/min) and severe (n= 9; CrCl: < 30 mL/min) renal impairment, mean Cmax increased by 1.4‐, 1.9‐ and 1.5‐fold, respectively, and mean AUC(0‐∞) increased by 1.5‐, 1.9‐ and 2.0‐fold, respectively, compared to healthy matched subjects (n=9).
Hepatic impairment.
The exposure to lurasidone is increased in patients with Child‐Pugh Class A and B hepatic impairment with mean Cmax increased by 1.3‐ and 1.2‐fold, respectively and mean AUC(0‐∞) increased by 1.5‐ and 1.7‐fold, respectively compared to healthy matched subjects. The pharmacokinetics of lurasidone has not been adequately established in patients with severe hepatic impairment and lurasidone is not recommended in these patients.
Elderly.
Limited data have been collected in patients ≥ 65 years. Of the data collected, similar exposure was obtained compared with subjects < 65 years.
Paediatrics.
The pharmacokinetics of lurasidone, in paediatric patients 6‐17 years of age was similar to those in adults. There were no clinically relevant differences between genders in the pharmacokinetics of lurasidone in patients with schizophrenia.
5.3 Preclinical Safety Data
Genotoxicity.
Lurasidone was not genotoxic in the bacterial reverse mutation (Ames) test, the in vitro chromosomal aberration test in Chinese hamster lung cells, or the in vivo mouse bone marrow micronucleus test.
Carcinogenicity.
Lurasidone was administered orally for 24 months at doses of 30, 100, 300, or 650 (the high dose was reduced from 1,200 in males) mg/kg/day to ICR mice and 3, 12, or 36 (high dose reduced from 50) mg/kg/day to Sprague‐Dawley rats.
In the mouse study, there were increased incidences of malignant mammary gland tumours and pituitary gland adenomas in females at all doses; the lowest dose tested produced plasma levels (AUC in females) approximately equal to those in humans receiving the maximum recommended human dose (MRHD) of 160 mg/day. No increases in tumours were seen in male mice up to the highest dose tested, which produced plasma levels (AUC) 14 times those in humans receiving the MRHD.
In rats, an increased incidence of mammary gland carcinomas was seen in females at the two higher doses; the no‐effect dose of 3 mg/kg produced plasma levels (AUC) 0.4 times those in humans receiving the MRHD. No increases in tumours were seen in male rats up to the highest dose tested, which produced plasma levels (AUC) 6 times those in human receiving the MRHD.
Proliferative and/or neoplastic changes in the mammary and pituitary glands of rodents have been observed following chronic administration of antipsychotic medicines and are considered to be prolactin mediated. The relevance of this increased incidence of prolactin‐mediated pituitary or mammary gland tumours in rodents in terms of human risk is unknown.
To date, neither clinical studies nor epidemiological studies have shown an association between chronic administration of these medicines and mammary tumorigenesis. However, since tissue culture experiments indicate that approximately one‐third of human breast cancers are prolactin‐dependent in vitro, lurasidone should be used cautiously in patients with previously detected breast cancer or in patients with pituitary tumours.6 Pharmaceutical Particulars
6.1 List of Excipients
Mannitol, alginic acid, pregelatinised starch, croscarmellose sodium, silicon dioxide, magnesium strearate, Opadry complete film coating system 03B580015 White (ARTG PI No. 138475) (20 mg and 40 mg tablets) and Opadry complete film coating system 03B510036 Green (ARTG PI No. 138474) (80 mg tablet).
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Lurasidone Sun tablets are supplied in OPA/Al/PVC blister packs of 30.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
 Molecular formula: C28H36N4O2S [free base].
Molecular formula: C28H36N4O2S [free base].
C28H36N4O2S.HCl [HCl salt].
Molecular weight: 492.67 [free base].
529.14 [HCl salt].
CAS number.
367514‐88‐3.7 Medicine Schedule (Poisons Standard)
S4 - Prescription only medicine.
Summary Table of Changes


 In long‐term controlled studies, for patients who had normal BMI status at baseline (18.5 to < 25.0), the rate of clinically significant weight gain (≥ 7% increase in BMI) at month 12 was 12.4%, 34.5% and 5.6% and to study endpoint (LOCF) was 9.6%, 17.7% and 8.3% of the lurasidone, risperidone and quetiapine XR groups, respectively. For those who were overweight at baseline (BMI 25.0 to < 30.0), the rate of clinically significant weight gain at study endpoint was 6.3%, 14.1% and 9.5%, in patients given lurasidone, risperidone and quetiapine XR, respectively.
In long‐term controlled studies, for patients who had normal BMI status at baseline (18.5 to < 25.0), the rate of clinically significant weight gain (≥ 7% increase in BMI) at month 12 was 12.4%, 34.5% and 5.6% and to study endpoint (LOCF) was 9.6%, 17.7% and 8.3% of the lurasidone, risperidone and quetiapine XR groups, respectively. For those who were overweight at baseline (BMI 25.0 to < 30.0), the rate of clinically significant weight gain at study endpoint was 6.3%, 14.1% and 9.5%, in patients given lurasidone, risperidone and quetiapine XR, respectively. The proportion of patients with prolactin elevations ≥ 5x upper limit of normal (ULN) was 2.8% for lurasidone‐treated patients versus 1.0% for placebo‐treated patients. The proportion of female patients with prolactin elevations ≥ 5x ULN was 5.7% for lurasidone‐treated patients versus 2.0% for placebo-treated female patients. The proportion of male patients with prolactin elevations ≥ 5x ULN was 1.6% versus 0.6% for placebo‐treated male patients.
The proportion of patients with prolactin elevations ≥ 5x upper limit of normal (ULN) was 2.8% for lurasidone‐treated patients versus 1.0% for placebo‐treated patients. The proportion of female patients with prolactin elevations ≥ 5x ULN was 5.7% for lurasidone‐treated patients versus 2.0% for placebo-treated female patients. The proportion of male patients with prolactin elevations ≥ 5x ULN was 1.6% versus 0.6% for placebo‐treated male patients.
 In long term controlled studies the rate of markedly abnormal metabolic parameters was similar between lurasidone, risperidone and quetiapine XR. For patients given any dose of lurasidone the rate of shift from normal to high total cholesterol was 2.2% and triglycerides was 6.2%.
In long term controlled studies the rate of markedly abnormal metabolic parameters was similar between lurasidone, risperidone and quetiapine XR. For patients given any dose of lurasidone the rate of shift from normal to high total cholesterol was 2.2% and triglycerides was 6.2%.





 Examination of population subgroups based on age (there were few patients over 65), gender and race did not reveal any clear evidence of differential responsiveness.
Examination of population subgroups based on age (there were few patients over 65), gender and race did not reveal any clear evidence of differential responsiveness.
 Molecular formula: C28H36N4O2S [free base].
Molecular formula: C28H36N4O2S [free base].