What is in this leaflet
This leaflet answers some common questions about Madopar tablets and capsules.
It does not contain all the available information.
It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Madopar against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Madopar is used for
The name of your medicine is Madopar. It contains two active ingredients called levodopa and benserazide.
Madopar belongs to a group of medicines called antiparkinsonian agents and is used for the treatment of Parkinson's disease.
Antiparkinsonian agents work on the central nervous system. The symptoms of Parkinson's disease are caused by a deficiency of a natural substance in the brain called dopamine. Madopar helps to replace this substance. By improving muscle control, Madopar allows more normal movements of the body.
The symptoms of patients suffering from Parkinson's disease can be reduced by taking this medicine. Madopar does not, however, cure the disease, since the cause of the dopamine deficiency within the brain is not removed.
Your doctor, however, may have prescribed Madopar for another purpose.
Ask your doctor if you have any questions about why Madopar has been prescribed for you.
This medicine is available only with a doctor's prescription.
Before you take Madopar
Do not take Madopar if:
Do not take Madopar if you have an allergy to:
- any medicine containing levodopa or benserazide
- any of the ingredients listed at the end of this leaflet
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not take Madopar if:
- you have taken a non-selective monoamine oxidase inhibitor (MAO-I) medicine such as tranylcypromine or phenelzine within the last 2 weeks
- you have taken a combination of a selective MAO-A inhibitor such as moclobemide with a selective MAO-B inhibitor such as selegiline or rasagiline
- you have untreated heart, liver, kidney, lung, blood or endocrine (hormonal) disease
- you have narrow (closed) angle glaucoma (high pressure in the eye)
- you are being treated for severe mental illness (active psychosis or serious psychoneurosis)
- you have melanoma or suspected melanoma
- you are under the age of 30
- you are being treated for Huntington's chorea or intention tremor
Do not take Madopar after the expiry date (EXP) printed on the pack or if the packaging is torn or shows signs of tampering. If you take this medicine after the expiry date has passed, it may not work as well.
If you are not sure if you should be taking Madopar, talk to your doctor.
You must tell your doctor if:
- you are allergic to any other medicines, foods, dyes or preservatives
- you have any other health problems including:
- convulsions (fits)
- diabetes
- stomach ulcers
- feeling down (depression) or other psychological disorders
- osteoporosis or osteomalacia
- irregular heart beat
- wide-angle glaucoma (high pressure in the eye)
- any compulsive behaviour disorder
- onset of sudden sleep disorder
- other illnesses
- you plan to have surgery
- you are pregnant or plan to become pregnant.
Madopar should not be taken during pregnancy. You should use effective contraception to avoid becoming pregnant while you are being treated with Madopar.
- you are breast-feeding or plan to breast-feed.
Your doctor will discuss the risks and benefits of taking Madopar when breast-feeding.
If you have not told your doctor about any of the above, tell them before you start taking Madopar.
When taken with certain foods
Madopar may not work as well as usual when taken with a protein rich meal (e.g. meat).
Taking other medicines
Tell your doctor if you are taking any other medicines including any that you have bought without a prescription from a pharmacy, supermarket or health food shop.
Some medicines and Madopar may interfere with each other. These medicines include:
- sympathomimetics such as cough and cold medicines containing ephedrine or pseudoephedrine, adrenaline, noradrenaline, isoprenaline, dexamphetamine, asthma preparations, epi-pens
- blood pressure lowering medications such as metoprolol, atenolol
- other antiparkinsonian medications such as anticholinergics, amantadine, selegiline, bromocriptine and dopamine agonists
- medicines containing iron
- medicines for relief of nausea such as metoclopramide
- some medications used to treat mental illness, particularly the phenothiazines, tricyclic antidepressants and butyrophenone derivatives such as chlorpromazine, fluphenazine, prochlorperazine, thioridazine, trifluoperazine or haloperidol
- general anaesthetics (medicines that put you to sleep before an operation), particularly cyclopropane and halothane. If you know you are going to have an operation, you should stop Madopar 2-3 days beforehand. You should discuss this with your doctor first.
- opioids such as morphine, pethidine, methadone and codeine (may be present in some cough and cold or pain relieving medicines)
- antacids (medicines for heartburn, indigestion) should not be taken at the same time as Madopar HBS, as they interfere with the controlled release characteristics of Madopar HBS. Antacids can be taken at other times of the day.
These medicines may be affected by Madopar or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor or pharmacist has more information on the medicines to be careful with or to avoid while taking Madopar.
If you have not told your doctor about any of the above, tell them before you start taking Madopar.
Ask your doctor or pharmacist if you are not sure about any of the medicines on this list.
How to take Madopar
Follow all directions given to you by your doctor or pharmacist carefully. This may differ from the information contained in this leaflet.
How much to take
Your doctor will tell you how many Madopar tablets or capsules to take each day.
The dose depends on your individual symptoms and your response to the medicine. The dose of Madopar has to be carefully adjusted for each person. If you take too little Madopar your symptoms may not be controlled. If you take too much Madopar, you may experience unwanted effects. It may take several weeks before the best dose for you is reached.
Remember this is just a guide. This guide can be used for Madopar capsules or tablets.
Treatment with Madopar is usually started with lower doses. This may be increased each week until you have reached your required dose. Your doctor will decide which dose is best for you.
Do not stop taking Madopar without first discussing with your doctor.
Madopar HBS (slow release) Capsules
Madopar HBS is different to other Madopar products. It takes longer to start working but its effects last longer.
If you change from normal Madopar to Madopar HBS, you start on the same number of capsules or tablets, but this is gradually increased by your doctor.
Some people who change to Madopar HBS capsules might feel a little worse at first. This should only be temporary, but speak to your doctor if anything worries you.
When you take Madopar HBS, your doctor may tell you to take a Madopar capsule or Madopar Rapid (dispersible) tablet in the morning to help maintain better control of your symptoms.
How to take it
Madopar Capsules
Madopar capsules should be swallowed whole with a glass of water or non-alcoholic drink.
Madopar HBS Capsules
Madopar HBS capsules should be swallowed whole and not chewed, opened or dissolved in liquid.
Madopar Rapid (dispersible) Tablets
Madopar Rapid tablets are for people who have difficulty swallowing whole tablets or who require the medicine to work quickly. They should be mixed with a quarter of a glass (at least 25-50mL) of water. The tablets will disperse into granules to give a milky-looking liquid which you should drink within 30 minutes. Remember to stir the liquid immediately before taking because the granules can settle at the bottom. The tablets do not completely dissolve.
Madopar Tablets
Madopar tablets can be broken across the score line. Any tablets which do not break cleanly along the line should not be taken.
Do not chew capsules or tablets.
When to take it
If possible, Madopar should be taken either 30 minutes before a meal or one hour after a meal. However, some patients may prefer to take Madopar with food.
If you have stomach upsets which can occur in the early stages of treatment with Madopar, try taking Madopar with a small snack (e.g. a biscuit) or liquid. Tell your doctor if anything worries you.
How long to take Madopar
When you start Madopar you will need to take it long term. You may need at least 6 months before deciding if Madopar is working for you.
Continue taking Madopar until your doctor tells you to stop.
If you forget to take Madopar
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for one you have missed. This may increase the chance of you getting an unwanted side effect.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you are not sure what to do, ask your doctor or pharmacist.
In case of an overdose
If you think that you or anyone else may have taken too much Madopar, immediately telephone your doctor or Poisons Information Centre (Australia telephone 13 11 26; New Zealand telephone 0800 764 766) for advice or go to Accident and Emergency at your nearest hospital.
Do this even if there are no signs of discomfort or poisoning.
You may need urgent medical attention.
Keep telephone numbers for these places handy.
If you take an overdose of Madopar, you will experience some or all of the symptoms described under Side Effects, but these symptoms may be more severe.
While you are taking Madopar
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Madopar.
Tell any other doctors, dentists and pharmacists who are treating you that you are taking Madopar.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine.
Tell your doctor if you become pregnant whilst taking Madopar. Madopar should not be taken during pregnancy. You should use effective contraception to avoid becoming pregnant while you are being treated with Madopar.
Tell your doctor if, for any reason, you have not taken your medicine exactly as prescribed. Otherwise, your doctor may think that it was not effective and change your treatment unnecessarily.
Tell your doctor if you feel that Madopar is not helping your condition.
Tell your doctor if you suffer from sleepiness or sudden onset of sleep during daily activities.
Be sure to keep all of your appointments with your doctor so that your progress can be checked. Your doctor may perform regular tests while you are taking Madopar.
Things you must not do
Do not stop taking Madopar or change the dose without first checking with your doctor.
Do not let yourself run out of medicine over the weekend or on holidays.
Do not give Madopar to anyone else even if they have the same condition as you.
Do not use Madopar to treat any other complaints unless your doctor tells you to.
Do not take any other medicines whether they require a prescription or not without first talking to your doctor.
Things to be careful of
Be careful driving or operating machinery until you know how Madopar affects you.
Madopar is associated with excessive drowsiness and very rarely with excessive daytime drowsiness and sudden sleep onset episodes.
Tell your doctor if you experience these effects and do not drive or operate machinery until this has been resolved.
Side Effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Madopar.
Madopar helps most people with Parkinson's Disease but it may have unwanted side effects in some people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- abnormal involuntary jerking movements of the body. These are usually caused if your dose is too high and will lessen or disappear when your dose is reduced
- mental changes including paranoia, depression, mania, agitation and hallucinations (seeing, feeling or hearing things that are not there)
- fluctuations in the effect of Madopar known as freezing episodes. These can usually be helped by asking your doctor to adjust your dose
- nausea and vomiting
- loss of appetite
- weight gain
- constipation
- skin rash or itching
- confusion, tiredness, sleeplessness, or sudden onset of sleep episodes
- poor muscle tone, hiccups
- restlessness
- water retention, cramps
The above side effects are common, especially if you have just started Madopar.
- changes in sex drive or hypersexuality
- signs of compulsive behaviour (performing a repetitive purposeless activity)
- loss of taste
- your urine, mouth, saliva, tongue and teeth may be darker with a red tinge
- bleeding or bruising more easily
- frequent infections such as fever, severe chills
- sore throat or mouth ulcers
Tell your doctor as soon as possible if you notice any of the following:
- palpitations or changes in the rhythm or rate of the heartbeat, chest pain, dizziness on standing up
- diarrhoea
- confusion
These may be serious side effects and your doctor may need to change your Madopar dose.
This is not a complete list of all possible side effects. Others may occur in some people and there may be some side effects not yet known.
Tell your doctor if you notice anything else that is making you feel unwell, even if it is not on this list.
Ask your doctor or pharmacist if you don't understand anything on this list.
Do not be alarmed by this list of side effects. You may not experience any of them.
After taking Madopar
Storage
Keep your capsules or tablets in the bottle, with the cap tightly closed, until it is time to take them. If you take the capsules or tablets out of the bottle, or the cap is loose, they may not keep as well.
Keep Madopar capsules and tablets in a cool dry place where the temperature stays below 25°C.
Do not store it, or any other medicine, in a bathroom or near a sink.
Do not leave it in the car or on window sills. Heat and dampness can destroy this medicine.
Keep Madopar where young children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking Madopar, or the tablets/capsules have passed their expiry date, ask your pharmacist what to do with any that are left over.
Product Description
What Madopar looks like
There are eight Madopar products available to meet individual needs:
- Madopar 62.5 capsules are light grey at one end and powder blue at the other end, with 'ROCHE' printed on both ends.
- Madopar 125 capsules are flesh coloured at one end and powder blue at the other end, with 'ROCHE' printed on both ends.
- Madopar 250 capsules are caramel coloured at one end and powder blue at the other end, with 'ROCHE' printed on both ends.
- Madopar HBS 125 (slow-release) capsules are light blue at one end and dark green at the other end, with 'ROCHE' printed in red ink on both ends.
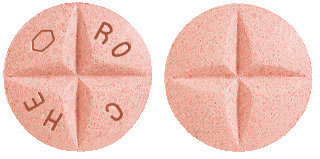
- Madopar 125 tablets are pale red in colour, cross-scored on both sides.
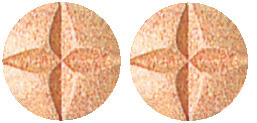
- Madopar 250 tablets are pale red in colour, imprinted on both sides with 'ROCHE and a hexagon on one side crossed-scored on both sides.
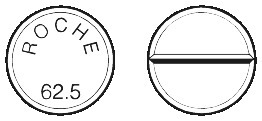
- Madopar Rapid 62.5 (dispersible) tablets are off-white in colour, scored on one side and printed with 'ROCHE 62.5' on the other.
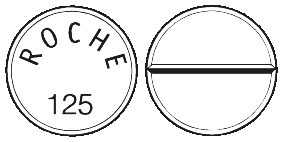
- Madopar Rapid 125 (dispersible) tablets are off-white in colour, scored on one side and printed with 'ROCHE 125' on the other.
All Madopar capsules and tablets come in bottles of 100.
Ingredients
Active ingredients:
Madopar Capsules:
Madopar 62.5 capsules contain levodopa 50 mg and benserazide 12.5 mg.
Madopar 125 capsules contain levodopa 100 mg and benserazide 25 mg.
Madopar 250 capsules contain levodopa 200 mg and benserazide 50 mg.
Madopar HBS 125 (slow-release) capsules contain levodopa 100 mg and benserazide 25 mg.
Madopar Tablets
Madopar 125 tablets contain levodopa 100 mg and benserazide 25 mg.
Madopar 250 tablets contain levodopa 200 mg and benserazide 50 mg.
Madopar Rapid (dispersible) Tablets:
Madopar Rapid 62.5 tablets contain levodopa 50 mg and benserazide 12.5 mg.
Madopar Rapid 125 tablets contain levodopa 100 mg and benserazide 25 mg.
Inactive ingredients:
Madopar Capsules:
Madopar 62.5, 125 and 250 capsules all contain:
microcrystalline cellulose, talc, povidone, magnesium stearate, indigo carmine (132), titanium dioxide (171), iron oxide (red, yellow or black - 172) and gelatin.
Madopar 62.5 capsules also contain mannitol.
Madopar HBS (slow-release) capsules contain:
mannitol, talc, povidone, magnesium stearate, calcium hydrogen phosphate, hypromellose, hydrogenated vegetable oil, indigo carmine (132), iron oxide yellow (172), titanium dioxide (171) and gelatin and TEKPRINT SW-1102 Red Ink used as a printing ink.
Madopar Tablets:
Madopar 125 and 250 tablets contain:
mannitol, microcrystalline cellulose, crospovidone, magnesium stearate, pregelatinised maize starch, ethylcellulose, anhydrous calcium hydrogen phosphate, colloidal anhydrous silica, docusate sodium and iron oxide red (172).
Madopar Rapid (dispersible) Tablets:
Madopar Rapid 62.5 and Rapid 125 tablets contain:
citric acid, starch- maize, microcrystalline cellulose and magnesium stearate.
Manufacturer
Madopar is distributed in Australia by:
Roche Products Pty Limited
ABN 70 000 132 865
Level 8, 30-34 Hickson Road
Sydney NSW 2000
Medical enquiries: 1800 233 950
Please check with your pharmacist for the latest Consumer Medicine Information.
Australian Registration Numbers
Madopar Capsules:
- Madopar 62.5 - AUST R 13744
- Madopar 125 - AUST R 13746
- Madopar 250 - AUST R 13742
- Madopar HBS - AUST R 13743
Madopar Tablets:
- Madopar 125 - AUST R 43517
- Madopar 250 - AUST R 13745
Madopar Rapid Tablets:
- Madopar Rapid 62.5 - AUST R 59577
- MADOPAR Rapid 125 - AUST R 59576
This leaflet was prepared on 23 November 2021.
Published by MIMS January 2022