What is in this leaflet
This leaflet answers some common questions about Mayzent.
It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
The information in this leaflet was last updated on the date listed on the final page. More recent information on the medicine may be available.
You should ensure that you speak to your pharmacist or doctor to obtain the most up to date information on the medicine. You can also download the most up to date leaflet from www.novartis.com.au. Those updates may contain important information about the medicine and its use of which you should be aware.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking Mayzent against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Mayzent is used for
Mayzent is used for the treatment of adult patients with secondary progressive multiple sclerosis (SPMS).
This medicine belongs to a group of medicines called sphingosine-1- phosphate (S1P) receptor modulators.
Mayzent binds selectively on two out of five receptors for S1P, namely S1P1 and S1P5. Mayzent can affect the ability of some white blood cells to move freely within the body and in this alter the way the body’s immune system works. Mayzent stops the cells that cause inflammation from reaching the brain and spinal cord and helps to fight against attacks of the immune system. This reduces nerve damage caused by MS and thus Mayzent helps to slow down the effects of the disease activity (such as worsening disability, brain lesions and relapses). Mayzent may also have a beneficial effect on certain brain cells (neural cells) involved in repairing or slowing down the damage caused by multiple sclerosis (MS).
In studies of patients with SPMS, Mayzent was seen to reduce attacks, disability progression and decline in thinking, and increase time without relapse.
Ask your doctor if you have any questions about why this medicine has been prescribed for you. Your doctor may have prescribed it for another reason.
This medicine is not addictive.
This medicine is available only with a doctor's prescription.
This medicine is not expected to affect your ability to drive a car or operate machinery.
It is not known whether Mayzent is safe and effective in children.
Before you take Mayzent
When you must not take it
Do not take Mayzent if you have an allergy to:
- any medicine containing siponimod
- any of the ingredients listed at the end of this leaflet.
- any other similar medicines (such as medicines of the same class or with a similar structure).
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Do not take Mayzent if:
- you have the CYP2C9 *3*3 genotype
- in the last 6 months, you have had a heart attack, angina, stroke or certain types of heart failure
- you have certain types of irregular or abnormal heartbeat (arrhythmia) and you do not have a pacemaker.
Do not take this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking this medicine, talk to your doctor.
Before you start to take it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Before initiation of treatment with Mayzent
Before initiation of treatment with Mayzent your CYP2C9 genotype should be determined. Your doctor will find this out with a blood test.
- If you have a CYP2C9 *3*3 genotype (less than 0.4 to 0.5% of the population), you should not take Mayzent.
Before you start taking Mayzent, you will have a blood test to check your white blood cells and to check your liver function unless your doctor has the results of a recent blood test.
Tell your doctor if you have or have had any of the following medical conditions:
- if you have an infection. Any infection that you already have may get worse. Infections could be serious and sometimes life-threatening. Before you start taking Mayzent, your doctor will confirm whether you have enough white blood cells in your blood.
- if you have a lowered immune response (due to a disease or medicines that suppress the immune system, see "Taking other medicines"). You may get infections more easily or an infection you already have may get worse. Mayzent lowers the white blood cell count (particularly the lymphocyte count). White blood cells fight infection. While you are taking Mayzent (and for up to 3 to 4 weeks after you stop taking it), you may get infections more easily.
- if you have no history of chickenpox or have not been vaccinated against varicella zoster virus.
Your doctor will test your status of the antibody against this virus and may decide to vaccinate you if you do not have antibodies to this virus. In this case, you will start Mayzent treatment one month after the full course of the vaccination is completed. - if you plan to receive a vaccine. You should not receive certain types of vaccines (called "live attenuated vaccines") during treatment with Mayzent and for up to 4 weeks after stopping treatment with Mayzent (see "Taking other medicines"). For the other vaccines, they can be less effective and your doctor may want you to stop Mayzent one week before the vaccination and for up to 4 weeks after vaccination.
- if you have or have had visual disturbances or other signs of swelling in the central vision area at the back of the eye (a condition known as macular oedema), if you have or have had inflammation or infection of the eye (uveitis) or diabetes. Your doctor will organise an eye examination before you start Mayzent and at regular intervals after the start of Mayzent treatment. The macula is a small area of the retina at the back of the eye which enables you to see shapes, colours, and details clearly and sharply (central vision). Mayzent may cause swelling in the macula and it usually happens during the first 3 to 4 months of Mayzent treatment. Your chance of developing macular oedema is higher if you have diabetes or have had an inflammation of the eye called uveitis. Macular oedema can cause some of the same vision symptoms as an MS attack (optic neuritis). Be sure to tell your doctor about any changes in your vision. This includes blurred vision or seeing shadows, or if you develop a blind spot in the centre of your vision, or if you have problems seeing colours or fine detail.
- if you have uncontrolled high blood pressure, if you have certain types of diseases related to blood vessels in the brain, if when you sleep you are severely affected by an inability to breathe (sleep apnoea that is not treated), if you are at risk for, or if you have heart rhythm disturbances (called abnormal ECG heart tracing). Your doctor may decide not to prescribe Mayzent if you have or have had one of these conditions or may refer you first to a cardiologist.
- if you are taking medicines for an irregular heartbeat such as quinidine, procainamide, amiodarone or sotalol, your doctor may decide not to prescribe Mayzent (see "Taking other medicines").
- if you suffer from a slow heart rate, if at the start of treatment with Mayzent you are taking medicines that slow your heart rate or if you have a history of sudden loss of consciousness (fainting). Your doctor may decide not to prescribe Mayzent or may refer you first to a cardiologist to switch you to medicines that do not slow your heart rate.
At the beginning of treatment, Mayzent can cause the heart rate to slow down. It cannot be excluded that Mayzent can also cause indirectly an irregular heartbeat, during the titration phase (first 5 days of treatment when the dose is gradually increased). Slow heart rate usually returns to normal within 10 days after treatment initiation. Irregular heartbeat usually returns to normal in less than one day. If your heart rate slows down during the titration phase, you may not feel anything or you may feel dizzy or tired. - if you have severe respiratory or breathing problems. Your asthma medicines will be checked by your doctor.
- if you have liver problems. Mayzent may affect your liver function. You will probably not notice any symptoms but if you notice yellowing of your skin or the whites of your eyes, abnormally dark urine or unexplained nausea, vomiting and tiredness during your treatment, tell your doctor straight away. Your doctor may carry out blood tests to check your liver function and may consider stopping Mayzent treatment if your liver problem is serious.
- If you have high blood pressure, your blood pressure will need to be checked regularly.
Tell your doctor straight away, if you get any of following symptoms or diseases during your treatment with Mayzent, because it could be serious:
- If you get an infection.
- If you believe your MS is getting worse (e.g. weakness or visual changes) or if you notice any new or unusual symptoms, because these may be the symptoms of a rare brain infection called progressive multifocal leukoencephalopathy (PML).
- If you get a fever, feel like you have a flu, or have a headache accompanied by stiff neck, sensitivity to light, nausea, and/or confusion as these may be symptoms of cryptococcal meningitis (caused by a fungal infection).
- If you have symptoms such as sudden onset of severe headache, confusion, seizures and vision changes, a condition called posterior reversible encephalopathy syndrome (PRES).
- If you develop a type of skin cancer called basal cell carcinoma (BCC) and other cutaneous neoplasms such as malignant melanoma. Symptoms of BCC may include skin nodules (e.g. shiny pearly nodules), patches or open sores that do not heal within weeks. Symptoms of other cutaneous neoplasms may include abnormal growth or changes of skin tissue (e.g. unusual moles) which may present as a change in colour, shape or size over time. Long-term exposure to the sun and a weak immune system can affect the risk of developing BCC or other cutaneous neoplasms. You should limit your exposure to the sun and UV rays by wearing appropriate protective clothing and regularly applying sunscreen with a high degree of UV protection. Phototherapy with UV radiation or PUVA photochemotherapy should be avoided during Mayzent treatment (it may increase your risk of developing skin cancer).
- If your MS is getting worse after you have stopped your treatment with Mayzent.
Children and adolescents (below 18 years)
Mayzent has not been studied in patients below 18 years.
Older people (65 years or above)
You can use Mayzent if you are aged 65 years or over at the same dose as for other adults.
Tell your doctor if you are pregnant or plan to become pregnant or are breast-feeding. Your doctor can discuss with you the risks involved. Mayzent may harm your unborn baby.
You should not breast-feed while you are taking Mayzent. Mayzent can pass into breast milk and there is a risk of serious side effects for a breast-fed baby. Talk with your doctor before breastfeeding while you take Mayzent.
Ask your doctor or pharmacist for advice before taking any medicine, if you are pregnant or breast-feeding.
You should avoid becoming pregnant while taking Mayzent and for at least 10 days after you stop taking it. Mayzent may harm your unborn baby. Female patients who might become pregnant should use effective birth control during treatment and for at least 10 days after stopping Mayzent. Ask your doctor about options of effective birth control.
If you become pregnant or think you are pregnant, tell your doctor right away. You and your doctor will decide what is best for you and your baby.
If you have not told your doctor about any of the above, tell him/ her before you start taking Mayzent.
Taking other medicines
Tell your doctor or pharmacist if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Mayzent may interfere with each other. These include:
- Medicines for an irregular heartbeat
such as, quinidine, procainamide, amiodarone or sotalol. Your doctor may decide not to prescribe Mayzent if you take these medicines due to a possible added effect on irregular heartbeat. - Medicines that slow down heartbeat
such as verapamil or diltiazem (called calcium channel blockers), ivabradine or digoxin. Your doctor may decide to refer you to a cardiologist to change your medicines due to a possible added effect on slowing down heartbeat during the first days you start Mayzent. For medicines such as atenolol or propranol (called beta-blockers), if your heart rate at rest is above 50 beats per minute, your doctor may ask you to start treatment with Mayzent without stopping your beta-blocker treatment. However if your heart rate at rest is below 50 beats per minute, your doctor may ask you to stop your beta-blocker treatment first for the heart rate to return above 50, then to start treatment with Mayzent. In this case, it is only after you have reached the usual daily dose of Mayzent that your doctor will ask you to restart your beta-blocker treatment. - Medicines that suppress or modulate the immune system including other medicines used to treat MS
such as beta-interferon, glatiramer acetate, natalizumab, mitoxantrone, dimethyl fumarate, teriflunomide, alemtuzumab or corticosteroids due to a possible added effect on the immune system. - Vaccines.
If you need to receive a vaccine, seek your doctor’s advice first. During treatment and for up to 4 weeks after stopping treatment with Mayzent, administration of some vaccines containing live virus (live attenuated vaccines) may result in infection. - Treatment with medicines such as carbamazepine
can lower the level of Mayzent.in your blood. Your doctor may give you further instruction or change your other medicines. - If you have the CYP2C9 *1*3 or *2*3 genotype,
treatment with medicines such as modafinil can lower the level of Mayzent in your blood. Your doctor may give you further instruction or change your other medicines. - If you have the CYP2C9 *2*2 genotype,
treatment with medicines such as fluconazole, can increase the level of Mayzent in your blood. Your doctor may give you further instruction, decide to reduce your daily dose to 1 mg Mayzent or change your other medicines.
Tell your doctor or a pharmacist if you are taking or have recently taken any other medicines, including medicines obtained without a prescription.
These medicines may be affected by Mayzent or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist have more information on medicines to be careful with or avoid while taking this medicine.
How to take Mayzent
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
Do not exceed the recommended dose prescribed by your doctor.
Your doctor will give you a Patient Reminder Card. Read it carefully and follow the instructions on it.
Always show the Patient Reminder Card to your doctor, pharmacist, or dentist when you see them or if you need to go to hospital.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
Treatment initiation with a starter pack:
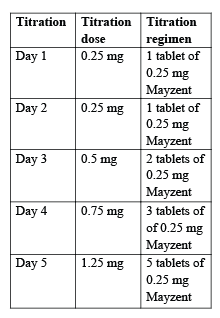
On day 6 switch to your prescribed treatment dose.
During the first 6 days of treatment the recommended daily dose should be taken once daily in the morning with or without food. If you missed a dose for more than one day during the first 6 days of treatment, the treatment should be re-started with a new starter pack.
The usual daily dose after the titration phase is 2 mg (one tablet of 2 mg Mayzent).
For some patients that have a CYP2C9 *2*3 or CYP2C9 *1*3 genotype the usual daily dose is 1 mg once daily. The same treatment initiation schedule (with a starter pack) applies as well for these patients.
How to take it
Take Mayzent once a day, with half a glass of water. You can take Mayzent with or without food.
When to take it
Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
It does not matter if you take this medicine before or after food.
How long to take it
Continue taking Mayzent every day for as long as your doctor tells you.
This is a long-term treatment, possibly lasting for months or years. Your doctor will regularly monitor your condition to check that the treatment is having the desired effect.
If you have questions about how long to take Mayzent, talk to your doctor or your pharmacist or healthcare provider.
If you forget to take it
If you missed a dose on one day during first 6 days of treatment, call your doctor before you take the next dose. Your doctor will need to prescribe a new starter pack. You will have to restart at Day 1 with a new starter pack.
Subsequently if you miss a dose, take it as soon as you remember, and then take the next tablet as usual.
If it is almost time for your next dose, skip the missed dose and continue as usual.
Do not take a double dose to make up for a forgotten tablet. Instead, wait until it is time for your next tablet.
Do not stop taking Mayzent or change your dose without talking with your doctor. If you stop taking Mayzent for 4 or more consecutive daily doses, treatment has to be re-started with the starter pack.
After Mayzent is stopped, symptoms of MS can return and may become worse compared to before or during treatment. Tell your doctor if you have worsening of MS symptoms after stopping Mayzent.
If you are a woman, see "Pregnancy and breast-feeding".
Mayzent will stay in your body for up to 10 days after you stop taking it. Your white blood cell count (lymphocyte count) may also remain low during this time and for up to 3 to 4 weeks and the side effects described in this leaflet may still occur.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much Mayzent. Do this even if there are no signs of discomfort or poisoning.
Your doctor may decide to observe you with heart rate and blood pressure measurements, to run electrocardiograms (ECGs) and he may decide to monitor you overnight.
While you are taking Mayzent
Things you must do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Mayzent.
Tell any other doctors, dentists, and pharmacists who treat you that you are taking this medicine.
If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine. It may affect other medicines used during surgery.
If you become pregnant while taking this medicine, tell your doctor immediately.
If you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
Things you must not do
Do not take Mayzent to treat any other complaints unless your doctor tells you to.
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop taking your medicine or lower the dosage without checking with your doctor.
Side effects
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking Mayzent.
This medicine helps most people with SPMS, but it may have unwanted side effects in a few people. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor or pharmacist if you notice any of the following and they worry you:
- Headache.
- High blood pressure with sometimes signs such as headache and dizziness (hypertension).
- Moles or nevi that appear recently: small spots or lumps of colours ranging from blue/dark to brown, pink to skin coloured (melanocytic naevus).
- Dizziness.
- Involuntary shaking of the body (tremor).
- Diarrhoea.
- Nausea.
- Pain in extremity.
- Swollen hands, ankles, legs or feet (peripheral oedema).
- Weakness (asthenia).
- Breathing difficulty.
The above list includes the more common side effects of your medicine. They are usually mild to moderate and will generally disappear after a few days to a week of treatment.
Tell your doctor as soon as possible if you notice any of the following:
- Rash of small fluid-filled blisters, appearing on reddened skin, signs of viral infection that can be potentially severe (herpes zoster).
- A type of skin cancer called basal cell carcinoma (BCC) which often appears as a pearly nodule, though it can also appear in other forms.
- Fever, sore throat or mouth ulcers due to infections (lymphopenia).
- Convulsion, fits (seizures).
- Shadows or blind spot in the centre of the vision, blurred vision, problems seeing colors or details (signs of swelling in the macular area of the retina at the back of the eye: macular oedema).
- Irregular heartbeat (atrioventricular block).
- Slow heartbeat (bradycardia).
Some side effects can only be found when your doctor does tests from time to time to check your progress. These include abnormal liver function test results that give information about the health of the liver (liver function test increased): high level of an enzyme called alanine aminotransferase (ALT), high level of an enzyme called gammaglutamyltransferase (GGT), high level of aspartate aminotransferase (AST).
Tell your doctor or pharmacist if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
Storage
Keep your tablets in the pack until it is time to take them. If you take the tablets out of the pack they may not keep well.
Keep your tablets in a refrigerator (2°C – 8°C). Do not freeze.
Do not store Mayzent or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Do not take this medicine after the expiry date, which is stated on the (carton)
Disposal
If your doctor tells you to stop taking this medicine or the expiry date has passed, ask your pharmacist what to do with any medicine that is left over.
Product description
What it looks like
Mayzent is supplied as film-coated tablets in blister packs.
Mayzent 0.25 mg tablets are: pale red, round, biconvex-beveled edged film-coated tablet with Novartis logo on one side and T on other side.
Mayzent 2 mg tablets are: pale yellow, round, biconvex-beveled edged film-coated tablet with Novartis logo on one side and II on other side.
Ingredients
Mayzent contains 0.25 mg or 2 mg of siponimod (as siponimod hemifumarate) as the active ingredient.
The other ingredients (excipients) of Mayzent are
Tablet core
- Lactose monohydrate
- microcrystalline cellulose
- crospovidone
- colloidal anhydrous silica
- glycerol dibehenate.
Each 0.25 mg tablet contains 62.2 mg lactose monohydrate.
Each 2 mg tablet contains 60.3 mg lactose monohydrate.
Tablet coating
- Polyvinyl alcohol
- titanium dioxide
- iron oxide yellow (2 mg tablet)
- iron oxide red (0.25 mg and 2 mg tablet)
- black iron oxide (0.25 mg tablet)
- purified talc
- lecithin (soya)
- xanthan gum.
This medicine does not contain sucrose, gluten, tartrazine or any other azo dyes.
Manufacturer/Distributor/ Supplier
Mayzent is supplied in Australia by:
Novartis Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone 1 800 671 203
Web site: www.novartis.com.au
™ = Registered Trademark
© Copyright 2021.
This leaflet was prepared in March 2021
MAYZENT 0.25 mg film-coated tablet blister pack AUST R 310498
MAYZENT 2 mg film-coated tablet blister pack AUST R 310499
(may100321c.doc based on PI may100321i.doc)
Published by MIMS May 2021




 During the first 6 days of treatment initiation the recommended daily dose should be taken once daily in the morning with or without food.
During the first 6 days of treatment initiation the recommended daily dose should be taken once daily in the morning with or without food.

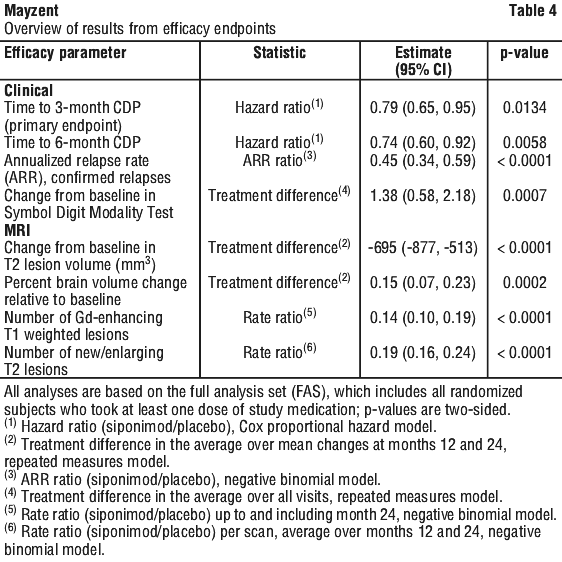
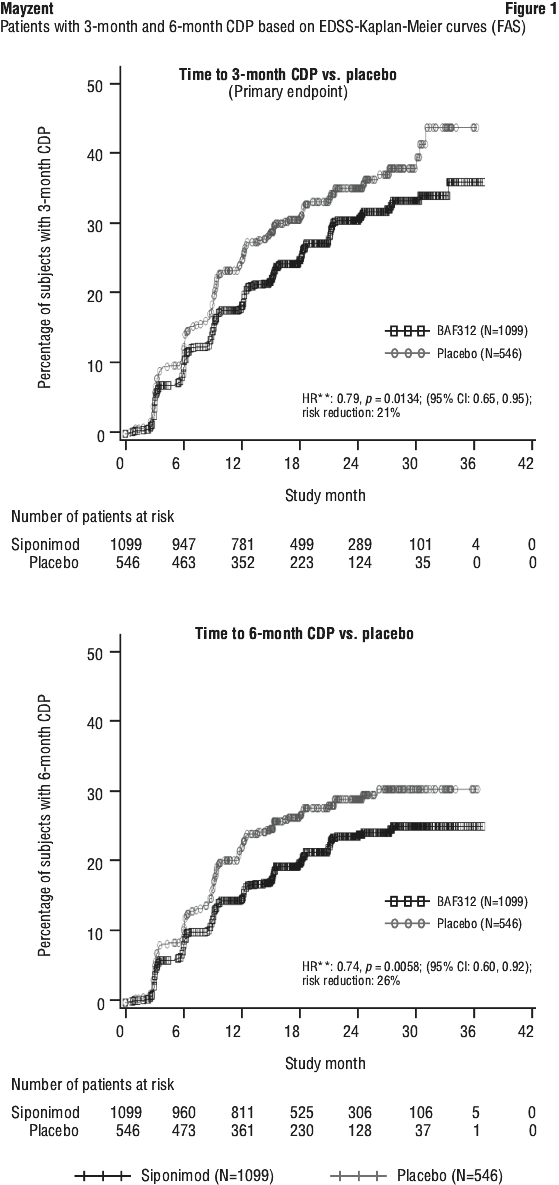 Mayzent did not significantly delay time to 3-month confirmed ≥ 20% deterioration in the T25FW compared to placebo (a numerical 6.2% risk reduction was observed).
Mayzent did not significantly delay time to 3-month confirmed ≥ 20% deterioration in the T25FW compared to placebo (a numerical 6.2% risk reduction was observed).
