1 Name of Medicine
Meningococcal (groups A, C, Y, W) polysaccharide tetanus toxoid conjugate vaccine.
2 Qualitative and Quantitative Composition
Each 0.5 mL dose of vaccine contains:
Meningococcal polysaccharide* Group A 10.0 microgram/dose;
Meningococcal polysaccharide* Group C 10.0 microgram/dose;
Meningococcal polysaccharide* Group Y 10.0 microgram/dose;
Meningococcal polysaccharide* Group W-135 10.0 microgram/dose.
* Each of the four polysaccharides is conjugated to tetanus toxoid (approximately 55 microgram/dose).
MenQuadfi is a sterile solution of Neisseria meningitidis (N. meningitidis) purified capsular polysaccharides of groups A, C, W-135, and Y, individually conjugated to tetanus toxoid protein prepared from cultures of Clostridium tetani. No preservative or adjuvant is added during manufacture.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection.
MenQuadfi is a clear, colourless, sterile, preservative-free solution.
4.1 Therapeutic Indications
MenQuadfi is indicated for active immunisation for the prevention of invasive meningococcal disease caused by Neisseria meningitidis serogroups A, C, W and Y.
The use of MenQuadfi should be in accordance with official recommendations.
4.2 Dose and Method of Administration
MenQuadfi should be administered as a 0.5 mL single dose injection by the intramuscular route only.
Primary vaccination.
Individuals 12 months of age and older receive a single dose.
Booster vaccination.
MenQuadfi may be given as a single booster dose to adolescents and adults who have previously been primed with meningococcal vaccine at least 4 years prior (see Section 5.1 Pharmacodynamic Properties).
Refer to official recommendations for further information regarding booster dosing.
Method of administration.
MenQuadfi should be administered as a single 0.5 mL injection by intramuscular route into the deltoid region or anterolateral thigh, depending on the recipient's age and muscle mass.
No data are available to establish safety and efficacy of the vaccine using intradermal or subcutaneous routes of administration.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions for concomitant administration with other vaccines.
The product is for single use only and must not be reused. Discard any remaining unused contents.4.3 Contraindications
MenQuadfi is contraindicated in anyone with a known systemic hypersensitivity reaction to any component of MenQuadfi or after previous administration of the vaccine or a vaccine containing the same components (see Section 2 Qualitative and Quantitative Composition; Section 6.1 List of Excipients).
4.4 Special Warnings and Precautions for Use
Appropriate observation and medical treatment should always be readily available in case of an anaphylactic event following the administration of the vaccine.
Protection.
As with any vaccine, vaccination with MenQuadfi may not protect all vaccine recipients.
MenQuadfi will not protect against N. meningitidis serogroup B disease.
Immunisation with MenQuadfi does not substitute for routine tetanus immunisation.
Waning of serum bactericidal antibody titres against serogroup A when using human complement in the assay (hSBA) has been reported for MenQuadfi and other quadrivalent meningococcal vaccines. The clinical relevance of this observation is unknown.
Intercurrent illness.
Vaccination should be postponed in individuals suffering from an acute severe febrile illness. However, the presence of a minor infection, such as cold, should not result in the deferral of vaccination.
Syncope.
Syncope can occur following or even before any vaccination as a psychogenic response to the needle injection. Procedures should be in place to prevent falling and injury and to manage syncope.
Altered immunocompetence.
Reduced immune response.
Some individuals with altered immunocompetence, including some individuals receiving immunosuppressant therapy, may have reduced immune responses to MenQuadfi.
Complement deficiency.
Individuals with certain complement deficiencies and individuals receiving treatment that inhibits terminal complement activation (for example, eculizumab) are at increased risk for invasive disease caused by Neisseria meningitidis, including invasive disease caused by serogroups A, C, W, and Y, even if they develop antibodies following vaccination with MenQuadfi.
Use in the elderly.
Safety and efficacy of MenQuadfi administration in individuals older than 56 years of age have been established. See Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties for more information.
Paediatric use.
Safety and efficacy of MenQuadfi administration in individuals less than 12 months of age have not been established.
Effects on laboratory tests.
No data available.4.5 Interactions with Other Medicines and Other Forms of Interactions
Use with other vaccines.
MenQuadfi should not be mixed with any other vaccine in the same vial or syringe.
If MenQuadfi needs to be given at the same time as another injectable vaccine(s), immunisation should be carried out on separate limbs.
MenQuadfi can be given concomitantly with any of the following vaccines:
Measles-mumps-rubella vaccine (MMR) and varicella vaccine (V).
Combined diphtheria - tetanus - acellular pertussis (DTPa) vaccines, including combination DTPa vaccines with hepatitis B, inactivated poliovirus or Haemophilus influenzae type b (HBV, IPV or Hib) such as DTPa-IPV-HB-Hib vaccine.
13-valent pneumococcal polysaccharide conjugate vaccine (PCV13).
Diphtheria, Tetanus, Pertussis (acellular, component) Vaccine (adsorbed, reduced antigen(s) content) (dTpa).
Human Papillomavirus Vaccine (Recombinant, adsorbed) (HPV).
Meningococcal serogroup B vaccine (see Section 5.1 Pharmacodynamic Properties, Concomitantly administered vaccines).
The anti-pertussis responses following dTpa administered concomitantly with MenQuadfi and HPV versus dTpa administered concomitantly with HPV did not meet non-inferiority for the FHA, PRN, and FIM antigens. Because there are no established serological correlates of protection for pertussis, the clinical implications of the observed pertussis antigen responses are unknown.
(See Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties, Concomitantly administered vaccines for safety and immunogenicity data).
Use with systemic immunosuppressive medicinal products.
It may be expected that in individuals receiving immunosuppressive treatment or individuals with immunodeficiency, an adequate immune response may not be elicited.4.6 Fertility, Pregnancy and Lactation
Effects on fertility.
A developmental and reproductive toxicity study was performed in female rabbits. The animals were administered a full human dose (0.5 mL) of MenQuadfi intramuscularly on two occasions before mating and three occasions during gestation. There were no effects on mating performances or female fertility. No study was conducted on male fertility.
(Category B1)
Limited data are available on the use of MenQuadfi in pregnant women. However, no conclusions can be drawn regarding whether or not MenQuadfi is safe for use during pregnancy.
A developmental and reproductive toxicity study was performed in female rabbits. The animals were administered a full human dose of MenQuadfi (0.5 mL) intramuscularly on two occasions before mating and three occasions during gestation. The study showed no adverse effects on embryo-fetal development (including an evaluation of teratogenicity) or early post-natal development.
MenQuadfi should be used during pregnancy only if the potential benefits to the mother outweigh the potential risks, including those to the fetus.
There are no available data on the presence of MenQuadfi in human milk, milk production, or the effects on the breastfed infant. No conclusions can be drawn regarding whether or not MenQuadfi is safe for use during breastfeeding.
MenQuadfi should be used during breastfeeding only if the potential benefits to the mother outweigh the potential risks, including those to the breastfed child.4.7 Effects on Ability to Drive and Use Machines
No studies on the effects of MenQuadfi on the ability to drive or use machines have been performed.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials.
The safety of MenQuadfi in individuals 12 months of age and older is based on 7 pivotal clinical studies in which participants received either MenQuadfi alone (5,327 participants), MenQuadfi concomitantly with other vaccines (981 participants), the concomitant vaccines without MenQuadfi (590 participants), or a comparator meningococcal vaccine (2,898 participants).
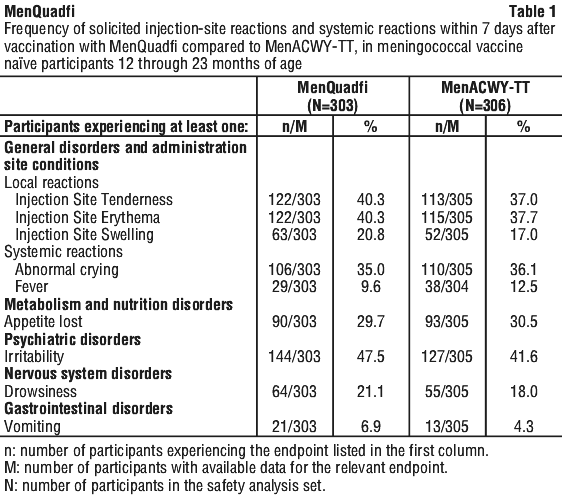
Participants 12 through 23 months of age.
The safety of MenQuadfi in participants 12 months through 23 months of age who were either meningococcal vaccine naive or who had received monovalent meningococcal C conjugate (MenC) vaccination during infancy was evaluated in a randomised, active-controlled, modified double-blind trial (MET51).
The rates of the solicited adverse reactions that occurred within seven days following MenQuadfi compared with a licensed quadrivalent meningococcal groups A, C, W-135 and Y Tetanus Toxoid conjugate vaccine (MenACWY-TT) are presented in Table 1.
Unsolicited injection-site reactions at the site of MenQuadfi injection included bruising, haematoma, induration, pruritus, and rash (0.3% each). Unsolicited systemic adverse events assessed as vaccine-related by the investigator more than once among recipient of MenQuadfi and which occurred at a rate of at least 1% during the 30 days post-vaccination included diarrhea (MenQuadfi 7.6%, MenACWY-TT 5.2%).
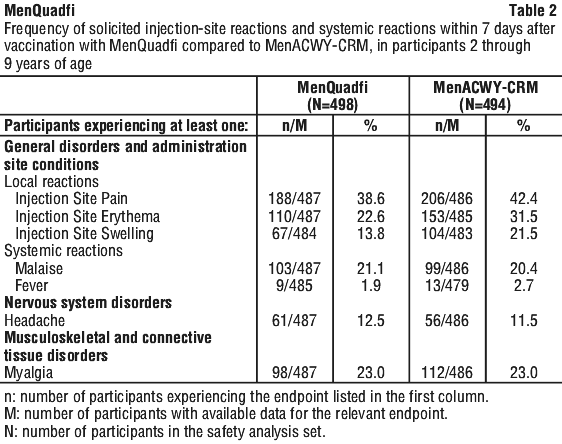
Participants 2 through 9 years of age.
The safety of MenQuadfi in participants 2 years through 9 years of age was evaluated in a randomised, active-controlled, modified double-blind trial (MET35).
The rates of the solicited adverse reactions that occurred within seven days following MenQuadfi compared with a licensed quadrivalent meningococcal groups A, C, W-135, and Y CRM197 protein conjugate vaccine (MenACWY-CRM) are presented in Table 2. Most adverse reactions were mild to moderate in severity.
The rates of local and systemic reactions among MenQuadfi recipients within 7 days after vaccination were generally comparable between the age subgroups 2-5 years and 6-9 years, with the exception of headache, which occurred more frequently among the older children (2-5 years 7.0%, 6-9 years 18.0%).
Unsolicited injection-site reactions at the site of MenQuadfi injection included bruising (0.4%), induration (0.2%), and warmth (0.2%). Unsolicited systemic adverse events assessed as vaccine-related by the investigator more than once among recipient of MenQuadfi and which occurred at a rate of at least 1% during the 30 days post-vaccination included vomiting (MenQuadfi 2.4%, MenACWY-CRM 2.2%) and stomach pain (MenQuadfi 1.4%, MenACWY-CRM 1.0%).
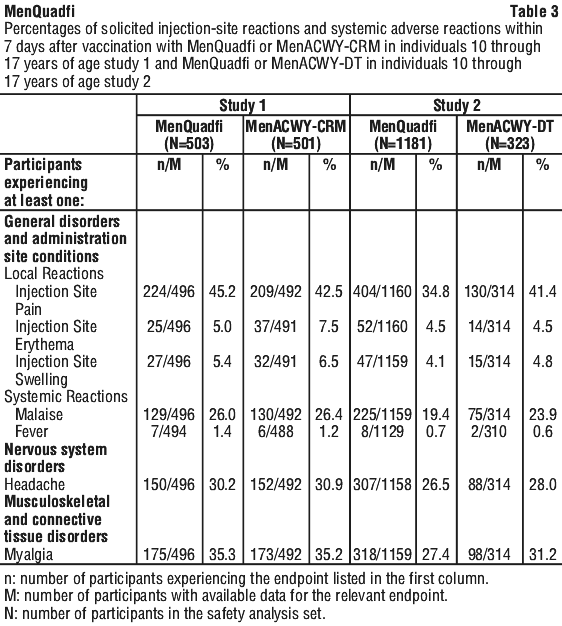
Participants 10 through 17 years of age.
The safety of MenQuadfi in participants 10 years through 17 years of age was evaluated in two clinical trials (MET43 and MET50): Study 1 (MET43) was a randomised, active-controlled, modified double-blind trial; Study 2 (MET50) was a randomised, controlled, open-label (the laboratory technicians were blinded to group assignment) concomitant trial. In the concomitant trial, MenQuadfi was given with dTpa and HPV. The comparator meningococcal vaccine was either MenACWY-CRM (501 participants) or Meningococcal Groups A, C, Y, and W-135 Polysaccharide conjugated to Diphtheria Toxoid (MenACWY-DT) (323 participants).
The rates of the solicited adverse reactions that occurred within seven days following MenQuadfi alone compared with MenACWY-CRM and MenACWY-DT are presented in Table 3 respectively. Most adverse reactions were of mild to moderate severity.
Unsolicited injection-site reactions at the site of MenQuadfi injection when given alone and which occurred at a rate of at least 0.1% in either study MET50 or study MET43, included pruritus (0.6% and 0.7%), rash (0.2% and 0.2%), warmth (0.8% and 0.5%), bruising (0.2% and < 0.1%) and induration (0.0% and 0.2%). There were no unsolicited systemic adverse events assessed as vaccine-related by the investigator any more than once among recipients of MenQuadfi and which occurred at a rate of at least 1% during the 30 days post-vaccination.
A few participants experienced dizziness or syncope within 30 minutes following vaccination (MenQuadfi 0.2% [dizziness], MenACWY-CRM 0.2% [syncope], MenACWY-DT 0.0%). These events were non-serious and spontaneously resolved on the same day.
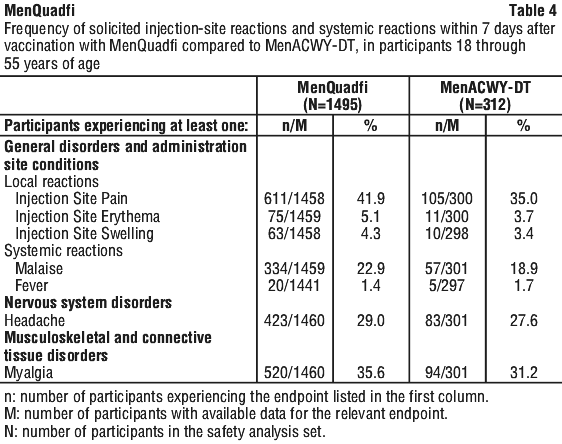
Participants 18 through 55 years of age.
The safety of MenQuadfi in participants 18 years through 55 years of age was evaluated in a randomised, active-controlled, modified double-blind trial (MET43).
The rates of the solicited adverse reactions that occurred within seven days following MenQuadfi compared with MenACWY-DT are presented in Table 4. Most adverse reactions were mild to moderate in severity.
Unsolicited injection-site reactions at the site of MenQuadfi injection with a frequency of at least 0.1% included pruritus (0.8%), warmth (0.3%), and mass (0.1%). There were no unsolicited systemic adverse events assessed as vaccine-related by the investigator more than once among recipient of MenQuadfi and which occurred at a rate of at least 1% during the 30 days post-vaccination.
A few participants experienced dizziness within 30 minutes following vaccination (MenQuadfi 0.3%, MenACWY-DT 0.3%). These events were non-serious and spontaneously resolved on the same day.
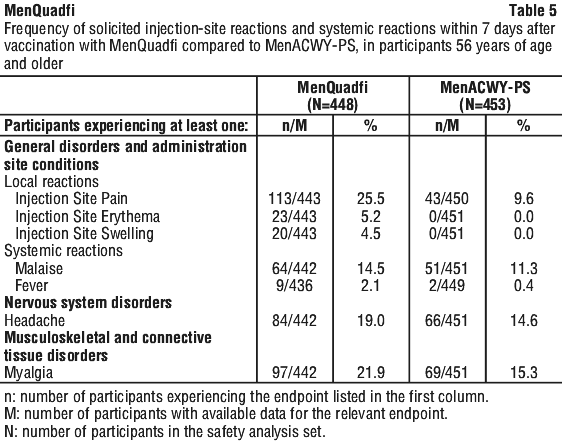
Participants 56 years of age and older.
The safety of MenQuadfi in participants 56 years of age and older was evaluated in clinical trial (MET49).
The rates of the solicited adverse reactions that occurred within seven days following MenQuadfi compared with Meningococcal Polysaccharide Vaccine, Groups A, C, Y, and W combined (MenACWY-PS) in study MET49 are presented in Table 5. Most adverse reactions were mild to moderate in severity.
The rates of local and systemic reactions among MenQuadfi within 7 days after vaccination were generally higher in the 56-64 year age subgroup compared with the 65 years of age and older subgroup.
Unsolicited injection-site reactions at the site of MenQuadfi injection included pruritus (1.8%), warmth (0.2%) and ecchymosis (0.2%). There were no unsolicited systemic adverse events assessed as vaccine-related by the investigator more than once among recipient of MenQuadfi and which occurred at a rate of at least 1% during the 30 days post-vaccination.
Individuals 15 years of age and older who had been previously vaccinated with either MenACWY-DT or MenACWY-CRM.
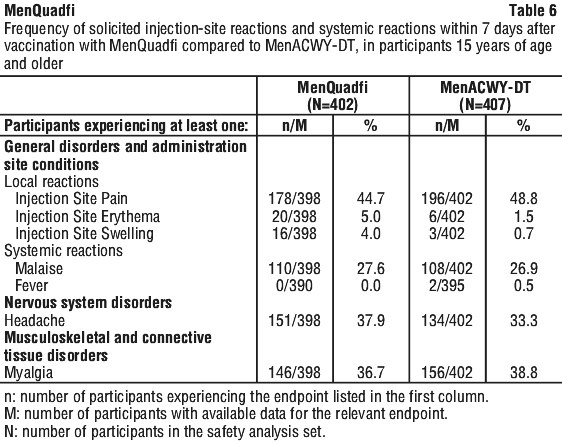
The safety of MenQuadfi in previously vaccinated participants 15 years of age and older was evaluated in a randomised, active-controlled, modified double-blind (MET56) trial. Participants had received a quadrivalent meningococcal conjugate vaccine 4 to 10 years previously.
The rates of the solicited adverse reactions that occurred within seven days following MenQuadfi compared with MenACWY-DT are presented in Table 6. Most adverse reactions were mild to moderate in severity.
Concomitant use with MMR and V for ages 12-23 months.
The safety of MenQuadfi administered concomitantly with MMR and V was evaluated in a randomised, controlled, open-label (the laboratory technicians were blinded to group assignment) trial (MET57).
The rates of local reactions at each of the injection sites were comparable when MenQuadfi was given concomitantly with MMR and V, MenQuadfi was given alone, and MMR and V were given without MenQuadfi.
The overall rates of solicited systemic reactions reported for participants receiving MenQuadfi + MMR+V (46.6%) were comparable to rates among participants who received MMR+V without MenQuadfi (43.2%), or MenQuadfi alone (54.3%). In the three groups the most common solicited systemic reactions were irritability (MenQuadfi + MMR+V, 23.8%; MMR+V, 26.3%; MenQuadfi alone, 24.5%), abnormal crying (MenQuadfi + MMR+V, 18.5%; MMR+V, 18.9%; MenQuadfi alone, 27.7%), and appetite lost (MenQuadfi + MMR+V, 21.2%; MMR+V, 13.7%; MenQuadfi alone, 23.4%).
Concomitant use with PCV13 for ages 12-23 months.
The safety of MenQuadfi administered concomitantly with PCV13 as evaluated in a randomised, open-label (the laboratory technicians were blinded to group assignment) trial (MET57).
The rates of local reactions at the PCV13 injection sites tended to be higher when MenQuadfi was given concomitantly with PCV13 compared with PCV13 given without MenQuadfi.
The overall rates of solicited systemic reactions reported for participants receiving MenQuadfi + PCV13 (20.0%) were comparable to rates among participants who received MenQuadfi alone (19.0%). The overall rate of solicited systemic reactions was lower for participants receiving PCV13 without MenQuadfi (10.1%). In the three groups the most common systemic reactions were irritability (MenQuadfi + PCV13, 13.0%; PCV13, 9.1%; MenQuadfi alone, 16.0%), appetite lost (MenQuadfi + PCV13, 9.5%; PCV13, 7.1%; MenQuadfi alone, 12.0%), and drowsiness (MenQuadfi + PCV13, 12.5%; PCV13, 4.0%; MenQuadfi alone, 6.0%).
Concomitant use with dTpa and HPV for ages 10-17 years.
The safety of MenQuadfi administered concomitantly with dTpa and HPV was evaluated in a randomised, controlled, open-label (the laboratory technicians were blinded to group assignment) trial (MET50).
The overall rate of solicited systemic reactions was higher when MenQuadfi was given concomitantly with dTpa and HPV (70.6%) than when MenQuadfi was given alone (52.0%) and comparable to when dTpa and HPV were given without MenQuadfi (65.9%). In the three groups the most common solicited systemic reactions were myalgia (MenQuadfi + dTpa + HPV, 61.3%; dTpa +HPV, 55.4%; MenQuadfi alone, 35.3%) and headache (MenQuadfi + dTpa + HPV, 33.8%; dTpa + HPV, 29%; MenQuadfi alone, 30.2%). The rates of local reactions at each of the injection sites were comparable when MenQuadfi was given concomitantly with dTpa and HPV, MenQuadfi was given alone, and dTpa and HPV were given without MenQuadfi.
Concomitant use with dTpa-IPV and 9vHPV for ages 10-17 years.
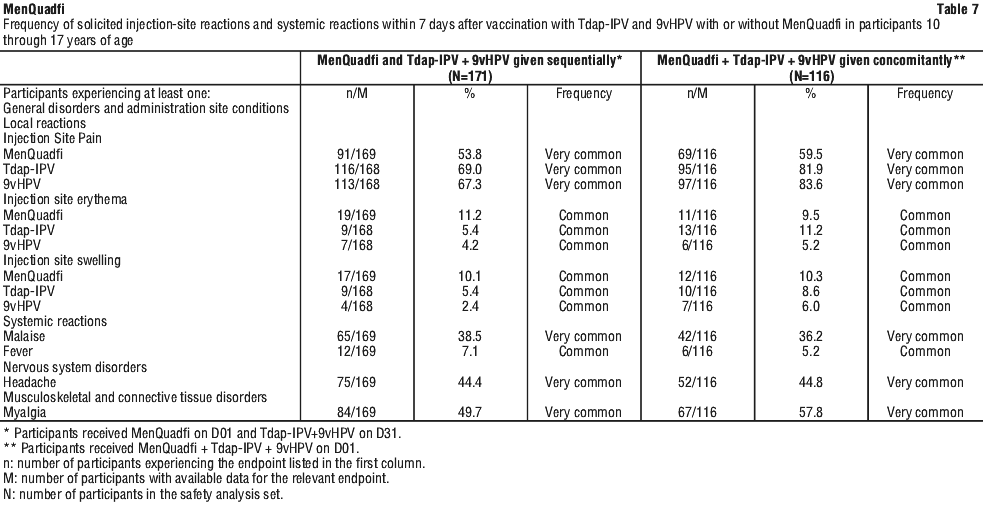
The safety of MenQuadfi administered concomitantly with dTpa-IPV and 9vHPV was evaluated in a randomised, active controlled, partially observer-blind (open-label for one of the study groups) trial (MEQ00071). The safety analysis set included 458 participants who received MenQuadfi alone (171 participants), MenQuadfi concomitantly with Tdap-IPV and 9vHPV (116 participants), or a comparator meningococcal vaccine (171 participants, MenACWY-TT). The participants 10 years through 17 years of age who received MenQuadfi alone were a mean age of 12.4 years and 12.5 years for those who received MenQuadfi concomitantly with Tdap-IPV and 9vHPV.
The rates of systemic reactions were comparable between all groups. The most common solicited systemic reactions were myalgia, headache and malaise.
The most common solicited injection site reaction following MenQuadfi vaccination was pain. The rates of pain at the Tdap-IPV and 9vHPV injection site were numerically higher when given concomitantly with MenQuadfi compared to when Tdap-IPV and 9vHPV were given alone.
Majority of solicited reactions were grade 1 or 2 and resolved within 3 days after vaccination.
No SAEs occurred following administration with MenQuadfi alone or concomitantly with Tdap-IPV and 9vHPV during the entire study period. See Table 7.
Concomitant use with meningococcal serogroup B (MenB) vaccines for ages 13-26 years.
In one additional clinical study (MET59), adolescents and adults 13-26 years of age primed with MenQuadfi 3-6 years previously received MenQuadfi co-administered with meningococcal serogroup B (MenB) vaccine, Trumenba (N=93) or Bexsero (N=92).
Rates and intensity of systemic reactions within 7 days following vaccination tended to be higher when MenQuadfi was given concomitantly with MenB vaccine than when MenQuadfi was given alone. The most common solicited systemic reaction was myalgia, of mild intensity, which was experienced more frequently in adolescents and adults who received MenQuadfi and MenB vaccine concomitantly (Trumenba, 65.2%; Bexsero, 63%) compared to those who received MenQuadfi alone (32.8%).
Post marketing.
In addition to the adverse events observed during the clinical trials, the following events have been reported during the post marketing use of MenQuadfi. The frequency is qualified as "not known" (cannot be estimated from available data).
Immune system disorders.
Hypersensitivity including anaphylaxis.
Nervous system disorders.
Convulsions with or without fever.
Reporting suspected adverse effects.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems (Australia).4.9 Overdose
For general advice on overdose management, contact the Poisons Information Centre, telephone number 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: meningococcal vaccine, ATC code: J07AH08.
Mechanism of action.
Invasive meningococcal disease (IMD) is caused by the bacterium N. meningitidis, a gram-negative diplococcus found exclusively in humans. The presence of bactericidal anti-capsular meningococcal antibodies has been associated with protection from IMD. MenQuadfi induces the production of bactericidal antibodies specific to the capsular polysaccharides of N. meningitidis serogroups A, C, W, and Y.
Clinical trials.
The immunogenicity of a single dose of MenQuadfi for primary vaccination in toddlers (12 - 23 months of age), children and adolescents (2 - 17 years of age), adults (18 - 55 years of age) and older adults (56 years and above) was assessed in six pivotal studies and in one additional study (MEQ65) in toddlers (12 - 23 months of age); the immunogenicity of a single dose of MenQuadfi for booster vaccination (ages 15-55 years of age) was assessed in one pivotal study. In addition, antibody persistence after primary vaccination and immunogenicity of a booster dose was assessed in three studies in children (4-5 years of age), adolescents and adults (13-26 years of age), and older adults (≥ 59 years of age). Ten out of eleven studies were randomised (one partially randomised), parallel-group, multi-centre studies. Nine out of eleven studies were active controlled. Clinical study comparators were MenACWY-TT, MenACWY-CRM, MenACWY-DT, MenC-TT and MenACWY-PS. Five out of eleven studies were open-label. The other six studies were modified, double-blind.
Primary immunogenicity analyses were conducted by measuring serum bactericidal activity (SBA) using human serum as the source of exogenous complement (hSBA). Rabbit complement (rSBA) data are available in subsets in all age groups and generally follows the trends observed with human complement (hSBA) data. In addition, all participants were assessed for primary immunogenicity measured by hSBA and rSBA for serogroup C in MEQ00065 study.
Clinical data on the persistence of antibody response ≥ 3 years after primary vaccination with MenQuadfi in children (4-5 years of age), adolescents and adults (13-26 years of age), and older adults (≥ 59 years of age) are available. Clinical data on booster vaccination with MenQuadfi in those participants are also available (MET62).
Immunogenicity in toddlers 12 to 23 month of age. Immunogenicity in participants 12 through 23 months of age was evaluated in three clinical studies (MET51, MET57 and MEQ00065).
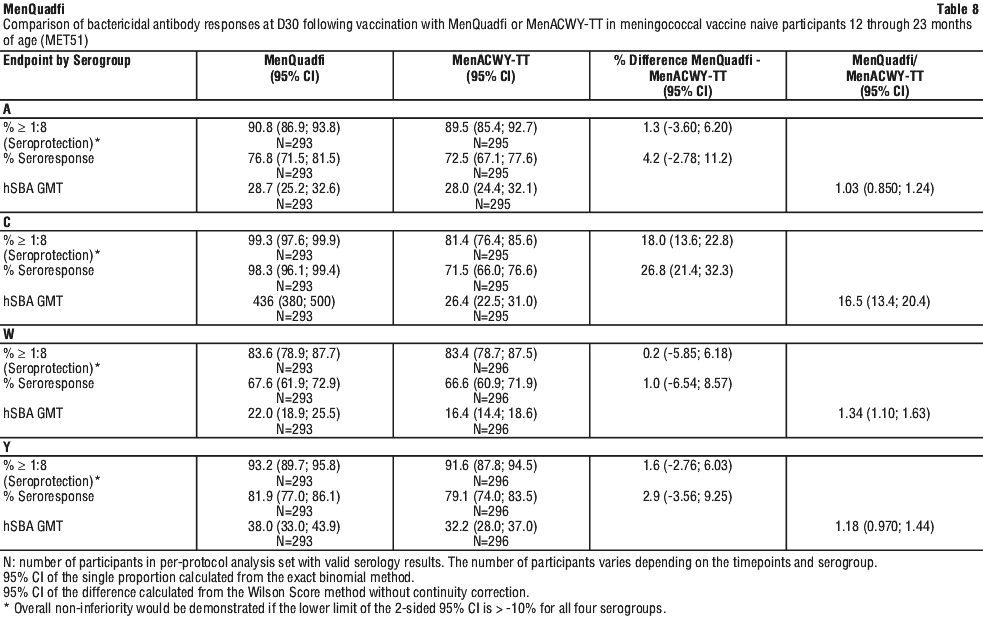
MET51 was conducted in participants who were either meningococcal vaccine naive or had been primed with monovalent meningococcal C vaccines (MenC-TT or MenC-CRM) in the first year of life.
Non-inferiority of immune response, based on percentage of participants achieving a post-vaccination hSBA titre ≥ 1:8 at Day 30 regardless of their meningococcal vaccine background, was demonstrated for MenQuadfi versus MenACWY-TT vaccine for all serogroups.
Non-inferiority of immune response, based on percentage of participants achieving a post-vaccination hSBA titre ≥ 1:8 at Day 30 in meningococcal vaccine naive toddlers, was demonstrated for MenQuadfi versus MenACWY-TT vaccine for all serogroups (see Table 8).
The point estimates of the immune response endpoints (with corresponding 95% confidence intervals [CIs]) and the differences or ratios observed between the two vaccines administered (with corresponding 95% CIs) in naive toddlers are summarised in Table 8.
 MET57 was conducted in meningococcal vaccine naive toddlers 12 through 23 months of age to assess the immunogenicity and safety of concomitant administration of MenQuadfi with paediatric vaccines (MMR+V, DTPa-IPV-HB-Hib or PCV). Overall, the postvaccination hSBA seroprotection rates in participants who received MenQuadfi was high for all serogroups (between 88.9% and 100%), and GMTs were higher for serogroup C than for serogroups A, W and Y.
MET57 was conducted in meningococcal vaccine naive toddlers 12 through 23 months of age to assess the immunogenicity and safety of concomitant administration of MenQuadfi with paediatric vaccines (MMR+V, DTPa-IPV-HB-Hib or PCV). Overall, the postvaccination hSBA seroprotection rates in participants who received MenQuadfi was high for all serogroups (between 88.9% and 100%), and GMTs were higher for serogroup C than for serogroups A, W and Y.
MEQ00065 study was conducted in meningococcal vaccine naïve toddlers 12 through 23 months of age to assess the immunogenicity of serogroup C using hSBA and rSBA assays following administration of a single dose of MenQuadfi compared to MenACWY-TT or to MenC-TT.
Superiority of MenQuadfi was demonstrated in comparison to MenACWY-TT vaccine for the hSBA seroprotection (titers ≥ 1:8) rate and hSBA and rSBA GMTs to meningococcal serogroup C. Noninferiority was demonstrated for the rSBA seroprotection rate to meningococcal serogroup C.
Superiority of MenQuadfi was also demonstrated in comparison to MenC-TT vaccine for the rSBA and hSBA GMTs to meningococcal serogroup C and non-inferiority was demonstrated for the rSBA and hSBA seroprotection rates to meningococcal serogroup C (see Table 9).
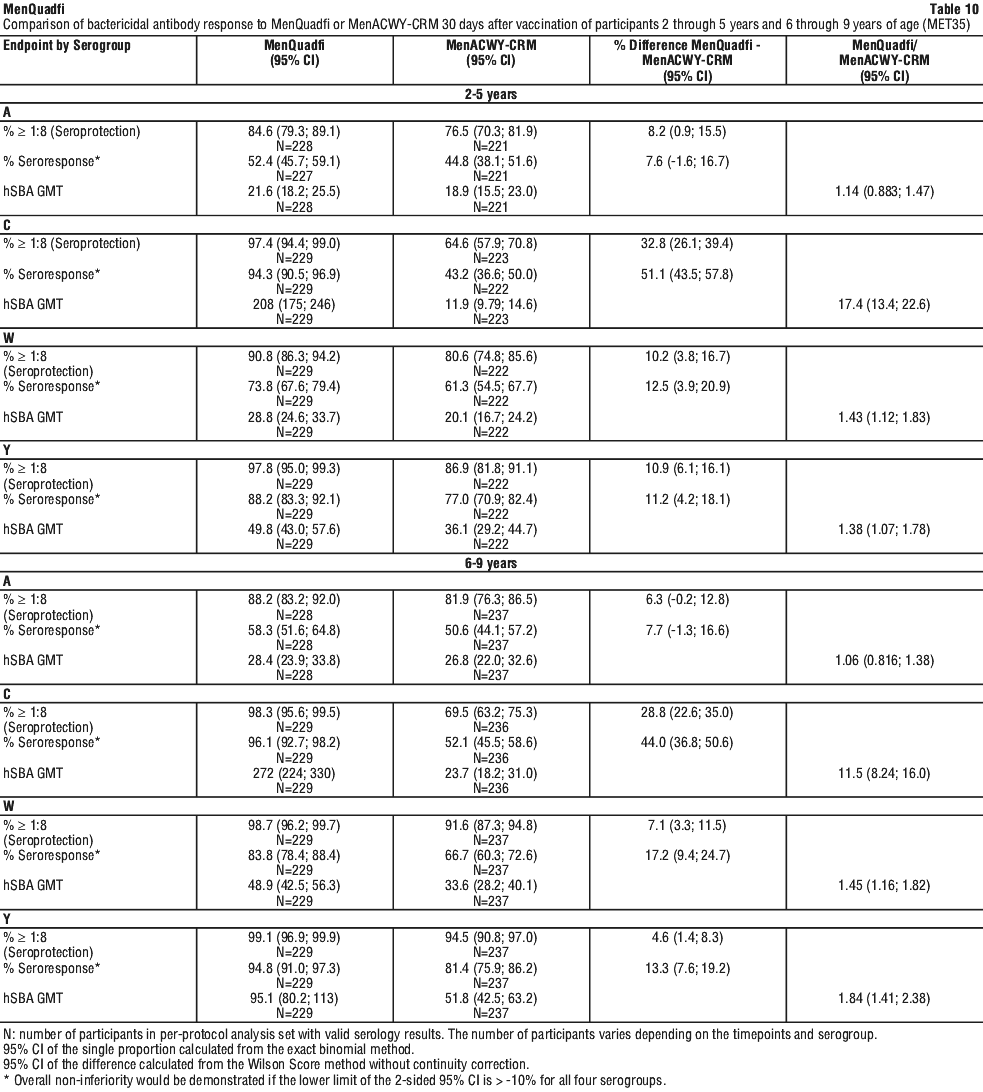
 Immunogenicity in children 2 through 9 years of age. Immunogenicity in participants 2 through 9 years of age was evaluated in study MET35 (stratified by ages 2 through 5 and 6 through 9 years) comparing seroresponses following administration of either MenQuadfi or MenACWY-CRM.
Immunogenicity in children 2 through 9 years of age. Immunogenicity in participants 2 through 9 years of age was evaluated in study MET35 (stratified by ages 2 through 5 and 6 through 9 years) comparing seroresponses following administration of either MenQuadfi or MenACWY-CRM.
Overall for participants 2 through 9 years of age, immune non-inferiority, based on hSBA seroresponse, was demonstrated for MenQuadfi as compared to MenACWY-CRM for all four serogroups. The post vaccination hSBA seroprotection rates and GMTs for serogroups C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-CRM. For Serogroup A, the post vaccination hSBA seroprotection rates and GMTs were similar in participants who received MenQuadfi than those who received MenACWY-CRM. The point estimates of the immune response endpoints (with corresponding 95% confidence intervals [CIs]) and the differences or ratios observed between the two vaccines administered (with corresponding 95% CIs) in naive children are summarised in Table 10.
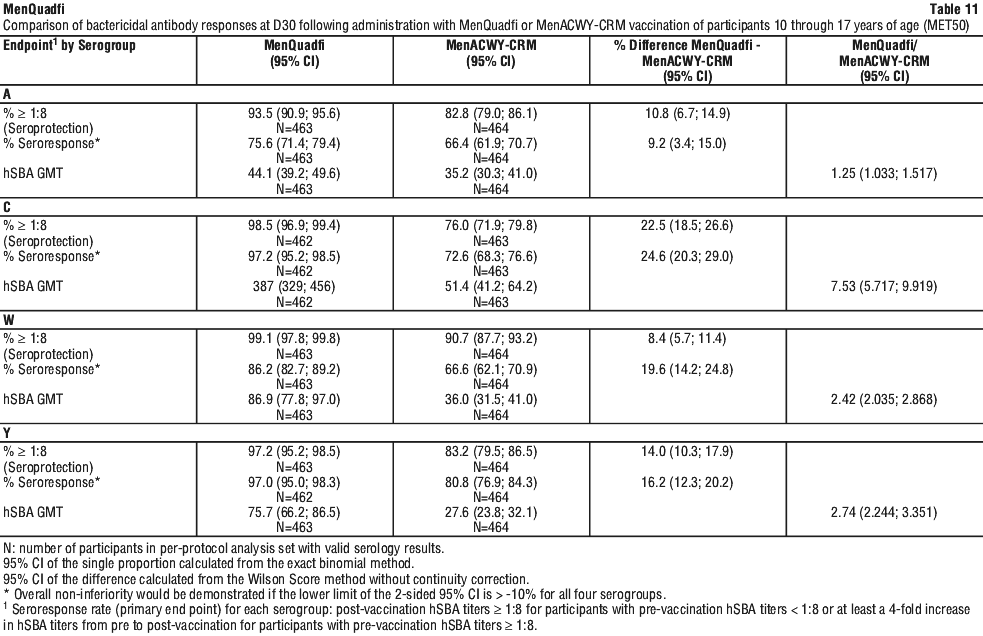
 Immunogenicity in children and adolescents 10 through 17 years of age. Immunogenicity in participants aged 10 through 17 years of age was evaluated in two studies comparing seroresponses following administration of MenQuadfi with either MenACWY-CRM (MET50) or MenACWY-DT (MET43) and in one study comparing seroprotection following administration of MenACWY-TT MEQ00071.
Immunogenicity in children and adolescents 10 through 17 years of age. Immunogenicity in participants aged 10 through 17 years of age was evaluated in two studies comparing seroresponses following administration of MenQuadfi with either MenACWY-CRM (MET50) or MenACWY-DT (MET43) and in one study comparing seroprotection following administration of MenACWY-TT MEQ00071.
MET50 was conducted in meningococcal vaccine naive participants and evaluated seroresponses following administration with either MenQuadfi alone; MenACWY-CRM alone; MenQuadfi co-administered with dTpa and HPV; or dTpa and HPV alone.
Overall immune non-inferiority, based on hSBA seroresponse, was demonstrated for MenQuadfi as compared to MenACWY-CRM for all four serogroups. The post vaccination hSBA seroprotection rates for serogroups A, C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-CRM. The post vaccination hSBA GMTs for serogroups C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-CRM and comparable for serogroup A. The point estimates of the immune response endpoints (with corresponding 95% confidence intervals) and the differences or ratio observed between the two vaccines administered (with corresponding 95% confidence intervals) in naive adolescents is summarised in Table 11.
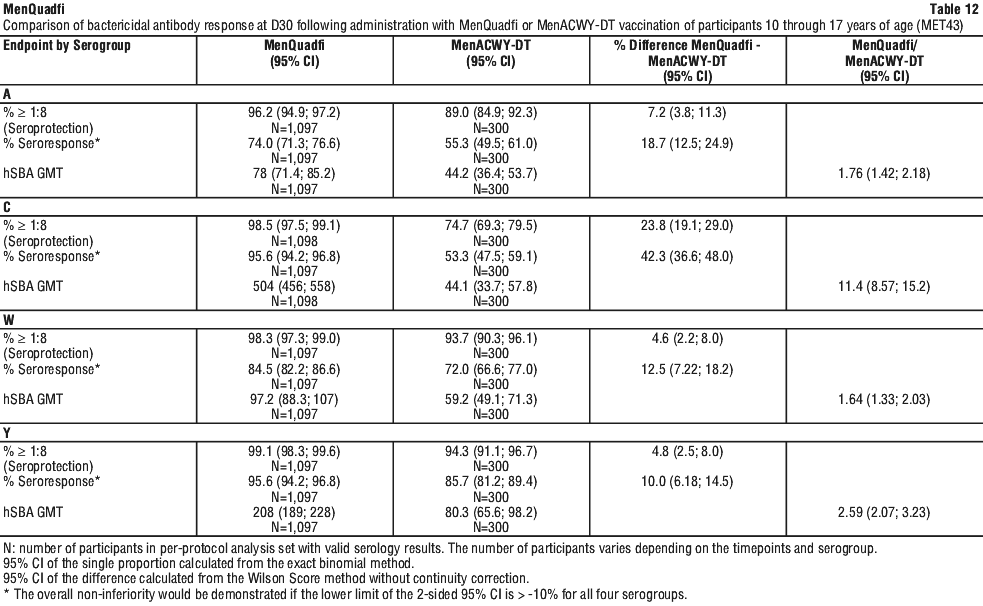
 Study MET43 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-DT in meningococcal vaccine naïve participants 10 through 55 years of age.
Study MET43 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-DT in meningococcal vaccine naïve participants 10 through 55 years of age.
In MET 43, immune non-inferiority, based on hSBA seroresponse, was demonstrated for MenQuadfi as compared to MenACWY-DT for all four serogroups. The post vaccination hSBA seroprotection rates and GMTs for serogroups A, C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-DT. The point estimates of the immune endpoints (with corresponding 95% confidence intervals) and the differences or ratio observed between the two vaccines administered (with corresponding 95% confidence intervals) in naive adolescents is summarised in Table 12.
 MEQ00071 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-TT in participants 10 through 17 years of age.
MEQ00071 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-TT in participants 10 through 17 years of age.
MEQ00071 was conducted in participants who were either meningococcal vaccine naïve or had been primed with monovalent MenC vaccines (MenC-TT or MenC-CRM) before two years of age. The participants were randomized to receive either a single dose of MenQuadfi alone, a single dose of the licensed MenACWY-TT vaccine, or a single dose of MenQuadfi given concomitantly with Tdap-IPV and 9vHPV.
Non-inferiority of immune response 30 days following vaccination, based on the percentages of participants with hSBA titers ≥ 1:8 (seroprotection rates), was demonstrated for MenQuadfi versus MenACWY-TT vaccine for all serogroups (see Table 13).
The post vaccination hSBA seroresponse rates were higher in participants who received MenQuadfi than those who received MenACWY-TT for all serogroups. The post vaccination hSBA GMTs for serogroups C, W, and Y were higher in participants who received MenQuadfi versus those who received MenACWY-TT and comparable for serogroup A (see Table 13).
Immune response was also measured 6 days following vaccination in a subset of participants who received MenQuadfi concomitantly with Tdap-IPV and 9vHPV (N=60). A rapid and robust immune response was observed, with hSBA GMTs increasing mainly within the first 6 days after vaccination. hSBA GMTs ranged from 2.36 to 9.23 before vaccination, from 22.2 to 2224 six days after vaccination and from 39.5 to 2358 thirty days after vaccination.

Response in participants according to MenC vaccination status.
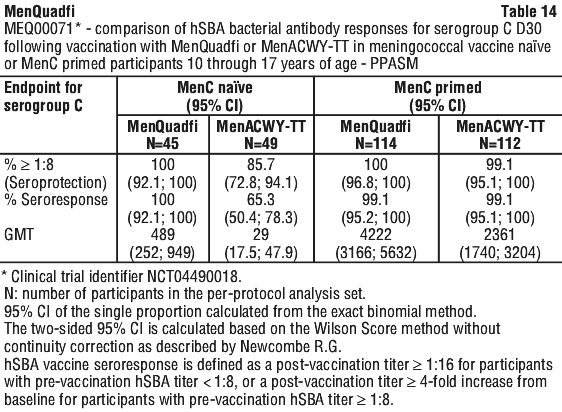
The immunogenicity of serogroup C following a single dose of MenQuadfi compared to a single dose of MenACWY-TT 30 days after vaccination was assessed in both meningococcal vaccine naïve and MenC primed (before two years of age) participants (MEQ00071).
Overall, the post vaccination hSBA seroprotection rates, hSBA seroresponse rates, and hSBA GMTs were higher in meningococcal vaccine naïve participants who received MenQuadfi than those who received MenACWY-TT. In MenC primed participants, the post vaccination hSBA seroprotection and seroresponse rates against meningococcal serogroup C were comparable between both study groups and the hSBA GMTs tended to be higher in participants who received MenQuadfi than those who received MenACWY-TT (see Table 14).
 Immunogenicity in adults 18 through 55 years of age. Immunogenicity in participants from 18 through 55 years of age was evaluated in study MET43 comparing MenQuadfi to MenACWY-DT. Immune non-inferiority, based on hSBA seroresponse, was demonstrated for MenQuadfi as compared to MenACWY-DT for all four serogroups. The post vaccination hSBA seroprotection rates and GMTs for serogroups A, C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-DT. The point estimates of the immune endpoints (with corresponding 95% confidence intervals) and the differences or ratio observed between the two vaccines administered (with corresponding 95% confidence intervals) in naive adults is summarised in Table 15.
Immunogenicity in adults 18 through 55 years of age. Immunogenicity in participants from 18 through 55 years of age was evaluated in study MET43 comparing MenQuadfi to MenACWY-DT. Immune non-inferiority, based on hSBA seroresponse, was demonstrated for MenQuadfi as compared to MenACWY-DT for all four serogroups. The post vaccination hSBA seroprotection rates and GMTs for serogroups A, C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-DT. The point estimates of the immune endpoints (with corresponding 95% confidence intervals) and the differences or ratio observed between the two vaccines administered (with corresponding 95% confidence intervals) in naive adults is summarised in Table 15.
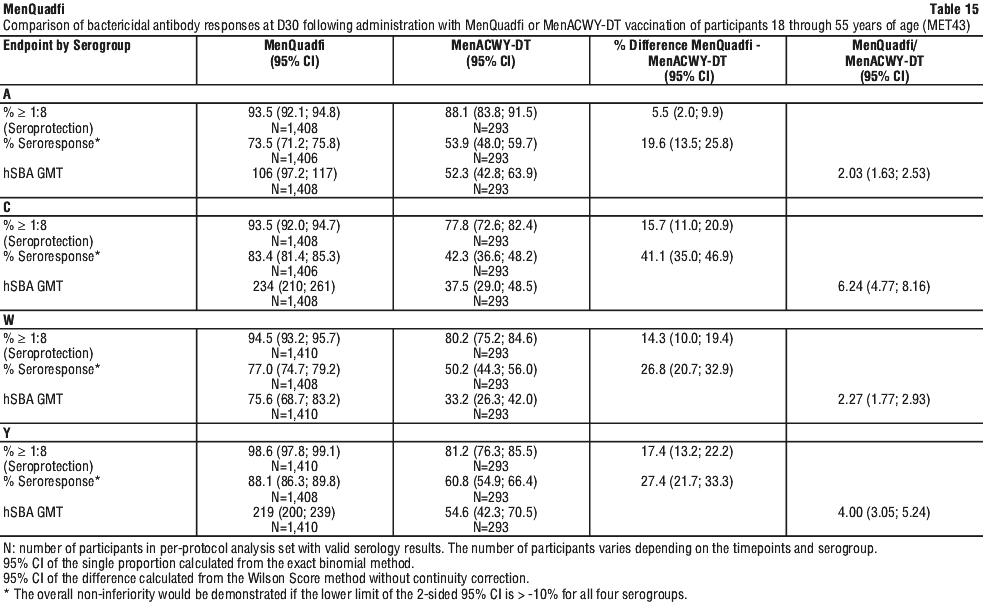 Immunogenicity in adults 56 years of age and above. Immunogenicity in adults ≥ 56 years of age was assessed in study MET49 comparing the immunogenicity of MenQuadfi to MenACWY-PS.
Immunogenicity in adults 56 years of age and above. Immunogenicity in adults ≥ 56 years of age was assessed in study MET49 comparing the immunogenicity of MenQuadfi to MenACWY-PS.
In study MET49, the overall mean age of participants who received MenQuadfi was 66.9 years. The age range of participants was 56 through 89.8 years of age. The immune response to MenQuadfi based on hSBA seroresponse was non-inferior to that of MenACWY-PS for all four serogroups. The percentages of participants with hSBA titres ≥ 1:8 increased from baseline for all serogroups and in both groups (see Table 16).
In participants 56 through 64 years of age, participants ≥ 65 years, participants 65 through 74 years and participants ≥ 75 years of age, seroprotection rates were comparable between MenQuadfi and MenACWY-PS for serogroup A and higher for serogroups C, Y and W in participants who received MenQuadfi than those who received MenACWY-PS. In participants 56 through 64 years of age and ≥ 65 years the GMTs were higher for all serogroups in those who received MenQuadfi than those who received MenACWY-PS. In participants 65 through 74 years of age, the GMTs were higher for serogroups C, Y and W, and comparable for serogroup A in those who received MenQuadfi than those who received MenACWY-PS. In participants ≥ 75 years of age the GMTs were higher for serogroup C, and comparable for serogroups A, Y and W in those who received MenQuadfi than those who received MenACWY-PS.
Overall for adults ≥ 56 years of age, immune non-inferiority, based on hSBA seroresponse, was demonstrated for MenQuadfi as compared to MenACWY PS for all four serogroups. The post vaccination hSBA GMTs for serogroups A, C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-PS. The post vaccination hSBA seroprotection rates for serogroups C, W, and Y were higher in participants who received MenQuadfi than those who received MenACWY-PS. The point estimates of the immune endpoints (with corresponding 95% confidence intervals) and the differences or ratio observed between the two vaccines administered (with corresponding 95% confidence intervals) in naive older adults is summarised in Table 16.
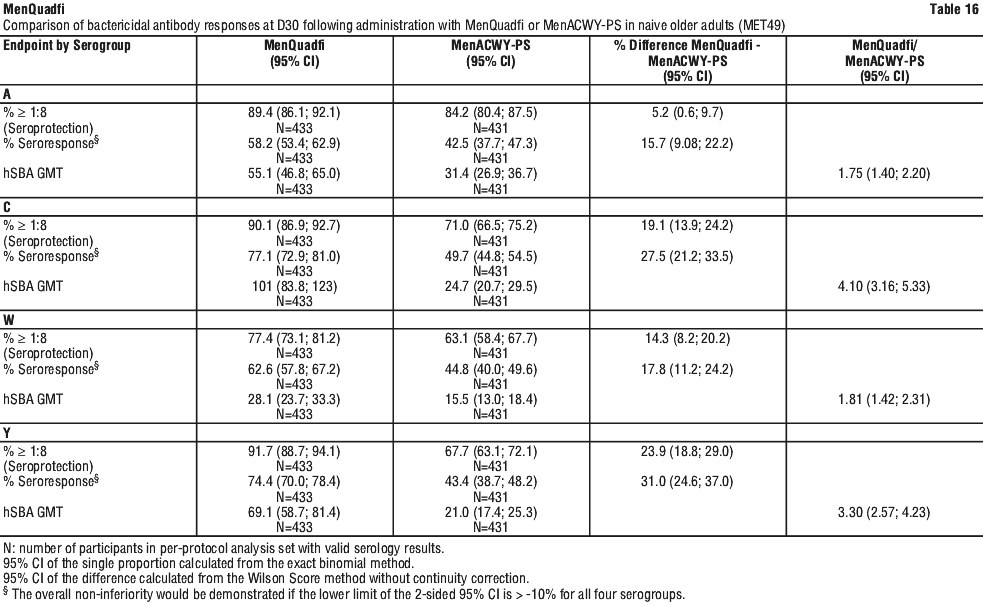 Persistence of immune response and MenQuadfi booster response. Antibody persistence after primary vaccination and immunogenicity of a MenQuadfi booster dose was assessed in four studies in children (4-5 years of age, and 6-7 years of age), adolescents and adults (13-26 years of age), and older adults (≥ 59 years of age).
Persistence of immune response and MenQuadfi booster response. Antibody persistence after primary vaccination and immunogenicity of a MenQuadfi booster dose was assessed in four studies in children (4-5 years of age, and 6-7 years of age), adolescents and adults (13-26 years of age), and older adults (≥ 59 years of age).
Persistence of immune response and MenQuadfi booster response in children 4 through 5 years of age. MET62 evaluated the antibody persistence of a primary dose, immunogenicity and the safety of a booster dose of MenQuadfi in children approximately 4 years of age. These children were primed with a single dose of MenQuadfi or MenACWY-TT 3 years before as part of the phase II study MET54 when they were 12-23 months old. The antibody persistence prior to the MenQuadfi booster dose and the booster immune response were assessed according to the vaccine (MenQuadfi or MenACWY-TT) children had received 3 years ago (see Table 17).
For all serogroups, hSBA GMTs were higher at D30 post-primary dose than at D0 pre-booster dose for MenQuadfi or MenACWY-TT. The pre-booster GMTs were higher than the pre-primary dose, indicative of long-term persistence of immune response.
After the booster dose, seroprotection rates were nearly 100% for all serogroups in children primed with MenQuadfi.
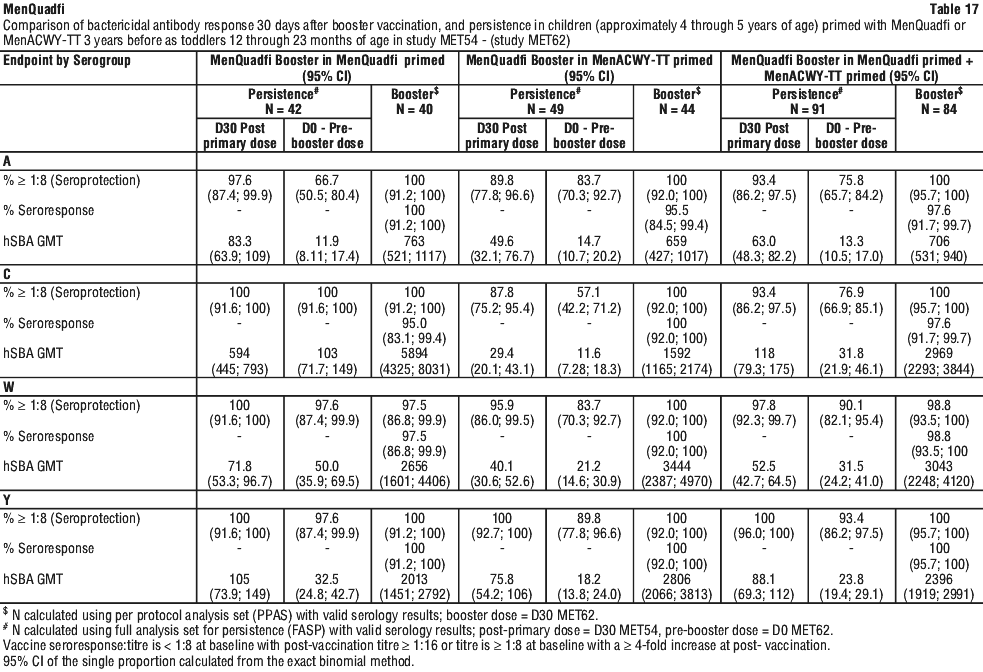 Persistence of immune response and MenQuadfi booster response in children 6 through 7 years of age. MEQ00073 evaluated the antibody response of a booster dose of MenQuadfi in children 6 through 7 years of age who had previously received a primary dose of MenQuadfi 5 years earlier as part of study MET51 when they were 12 through 23 months of age and were either meningococcal vaccine naive or primed with a MenC vaccine in their first year of life.
Persistence of immune response and MenQuadfi booster response in children 6 through 7 years of age. MEQ00073 evaluated the antibody response of a booster dose of MenQuadfi in children 6 through 7 years of age who had previously received a primary dose of MenQuadfi 5 years earlier as part of study MET51 when they were 12 through 23 months of age and were either meningococcal vaccine naive or primed with a MenC vaccine in their first year of life.
For all serogroups, 5Y post-primary (pre-booster) GMTs were higher than the pre-primary GMTs, indicative of long-term persistence of immune response.
At D30 post-booster dose, the proportion of individuals exhibiting ≥ 1:8 seroprotection rates were as follows: 98.9% for serogroup A, 97.7% for serogroup C, and 100% for serogroups W and Y in children primed with MenQuadfi (see Table 18).
The antibody responses against serogroup C at D30 post-booster dose of MenQuadfi were comparable regardless of MenC vaccination status during the first year of life before priming with MenQuadfi 5 years earlier.
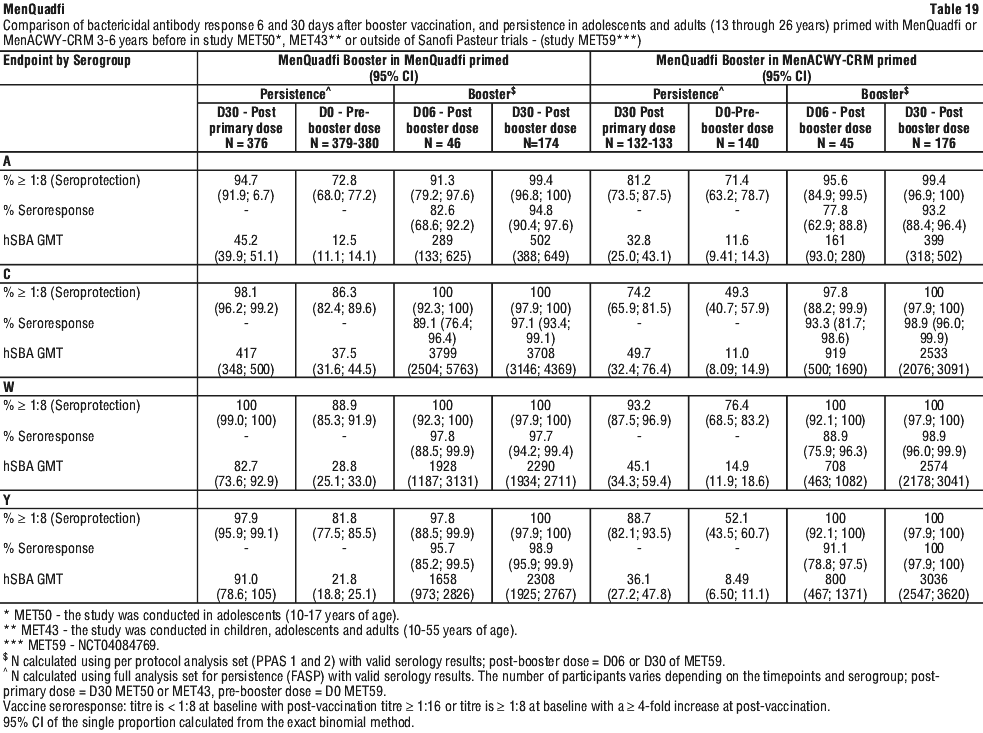
 Persistence of immune response and MenQuadfi booster response in adolescents and adults 13 through 26 years of age. MET59 evaluated the antibody persistence of primary dose, immunogenicity and safety of a booster dose of MenQuadfi in adolescents and adults 13 through 26 years of age who had received a single dose of MenQuadfi in study MET50 or MET43 or MenACWY-CRM in study MET50 or outside of Sanofi Pasteur trials 3-6 years prior. The antibody persistence prior to the MenQuadfi booster dose and the booster immune response were assessed according to the vaccine (MenQuadfi or MenACWY-CRM) participants had received 3-6 years previously (see Table 19).
Persistence of immune response and MenQuadfi booster response in adolescents and adults 13 through 26 years of age. MET59 evaluated the antibody persistence of primary dose, immunogenicity and safety of a booster dose of MenQuadfi in adolescents and adults 13 through 26 years of age who had received a single dose of MenQuadfi in study MET50 or MET43 or MenACWY-CRM in study MET50 or outside of Sanofi Pasteur trials 3-6 years prior. The antibody persistence prior to the MenQuadfi booster dose and the booster immune response were assessed according to the vaccine (MenQuadfi or MenACWY-CRM) participants had received 3-6 years previously (see Table 19).
For all serogroups, hSBA GMTs were higher at D30 post-primary dose than at D0 pre-booster for MenQuadfi and MenACWY-CRM primed participants. The pre-booster GMTs were higher than the pre-primary dose, indicative of long-term persistence of immune response.
After the booster dose, seroprotection rates were nearly 100% for all serogroups in adolescents and adults primed with MenQuadfi.
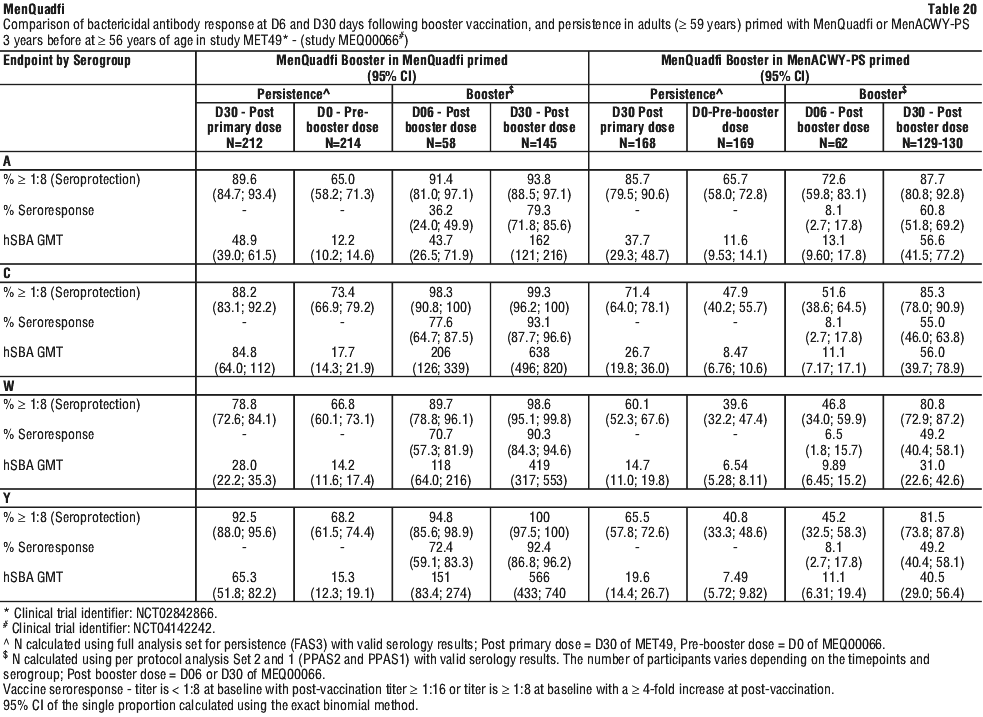
 Persistence of immune response and MenQuadfi booster response in adults 59 years of age and older. MEQ00066 evaluated the antibody persistence of primary dose, immunogenicity, and safety of a booster dose of MenQuadfi in older adults who had received a single dose of MenQuadfi or MenACWY-PS ≥ 3 or 5 years previously in study MET49 or MET44.
Persistence of immune response and MenQuadfi booster response in adults 59 years of age and older. MEQ00066 evaluated the antibody persistence of primary dose, immunogenicity, and safety of a booster dose of MenQuadfi in older adults who had received a single dose of MenQuadfi or MenACWY-PS ≥ 3 or 5 years previously in study MET49 or MET44.
3 year persistence.
The antibody persistence prior to the MenQuadfi booster dose and the booster immune response were assessed according to the vaccine (MenQuadfi or MenACWY-PS) participants had received 3 years previously at ≥ 56 years of age in MET49 (see Table 20).
For all serogroups, hSBA GMTs were higher at D30 post-primary dose than at D0 pre-booster dose for both MenQuadfi-primed and MenACWY-PS-primed adults. In addition, for both primed groups, the pre-booster GMTs were higher than the pre-primary dose for serogroups C, W and Y (indicative of long-term persistence of immune response for these serogroups) and were comparable for serogroup A.
5 year persistence.
A subset of participants who were assessed for antibody persistence at 3 years and did not receive the MenQuadfi booster dose were re-assessed for antibody persistence at 5 years at which time they received a booster dose of MenQuadfi.
In MenQuadfi-primed subjects, hSBA GMTs for serogroups C, W and Y 5Y post-primary dose were higher than the pre-priming GMTs (and were comparable for serogroup A), indicating long-term persistence of immune response.
Following the MenQuadfi booster dose, seroprotection rates were 100% for serogroups A, C, and Y, and 95.0% for serogroup W in subjects primed with MenQuadfi and 87.5%, 62.5%, 87.5% and 68.8% for serogroups A, C, W and Y, respectively, for those primed with MenACWY-PS.
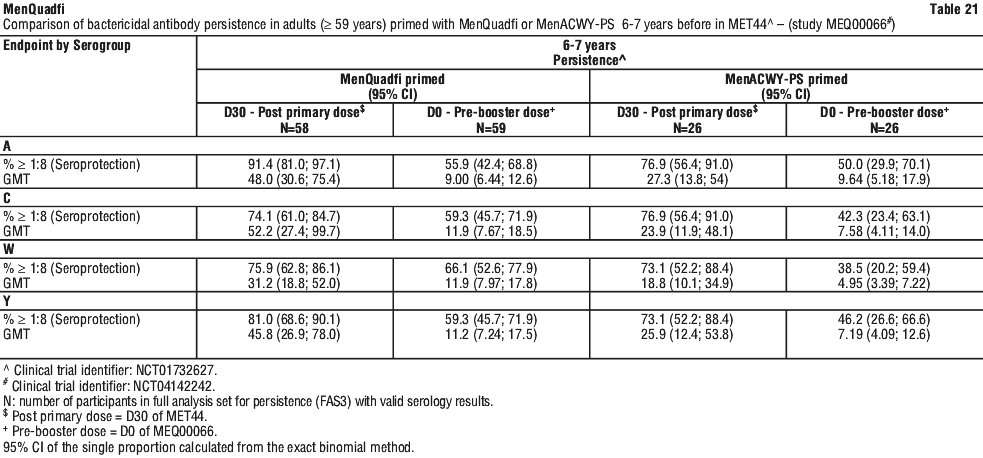
6-7 year persistence.
The antibody persistence was assessed according to the vaccine (MenQuadfi or MenACWY-PS) participants had received 6-7 years previously in study MET44 (see Table 21).
For all serogroups, hSBA GMTs were higher at D30 post-primary dose than at D0 pre-booster dose for MenQuadfi-primed adults. The pre-booster GMTs were higher than the pre-primary dose for serogroup C, W, and Y in MenQuadfi-primed adults, indicative of long-term persistence of immune response for these serogroups, and were comparable for serogroup A.
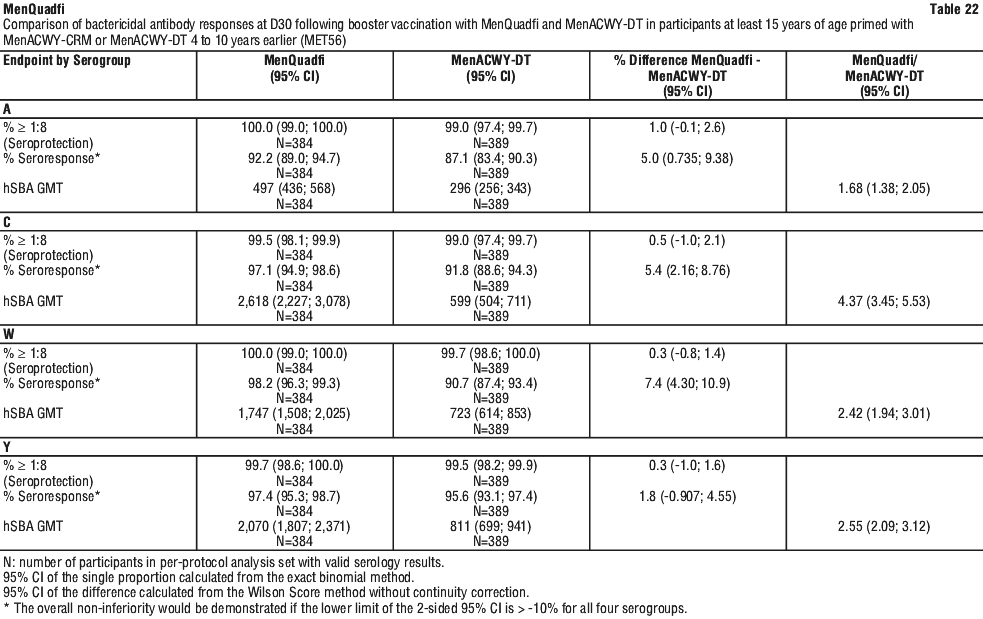
 Booster response in adolescents and adults at least 15 years of age primed with other MenACWY vaccines. Study MET56 compared the immunogenicity of a booster dose of MenQuadfi to a booster dose of MenACWY-DT in participants at least 15 years of age and primed with quadrivalent meningococcal conjugate vaccine (MCV4; MenACWY-CRM or MenACWY-DT) 4 to 10 years earlier.
Booster response in adolescents and adults at least 15 years of age primed with other MenACWY vaccines. Study MET56 compared the immunogenicity of a booster dose of MenQuadfi to a booster dose of MenACWY-DT in participants at least 15 years of age and primed with quadrivalent meningococcal conjugate vaccine (MCV4; MenACWY-CRM or MenACWY-DT) 4 to 10 years earlier.
At baseline, hSBA seroprotection and GMT were similar for serogroups A, C, W, and Y.
The hSBA seroresponse following a booster dose of MenQuadfi was non-inferior to that following a booster dose of MenACWY-DT for all four serogroups.
The percentages of participants with hSBA titres ≥ 1:8 increased from baseline for all serogroups and in both groups. The percentages were comparable in MenQuadfi and MenACWY-DT for all serogroups (see Table 22).
 Concomitantly administered vaccines.
Concomitantly administered vaccines. Immunogenicity of a single dose of MenQuadfi when given concomitantly with routine paediatric vaccines or when routine paediatric vaccines were given alone in meningococcal naïve infants 12 through 23 months of age - Study MET57.
A Phase III study MET57 was performed in meningococcal vaccine naive toddlers to evaluate the efficacy of MenQuadfi concomitantly administered with MMR, V, PCV13, and DTPa-IPV-HB-Hib and showed no clinically relevant interference on antibody responses to each of the antigens. Overall, the immunogenicity profile of MenQuadfi administered alone was comparable to the MenQuadfi administered concomitantly with licensed paediatric vaccines (MMR+V, DTPa-IPV-HB-Hib, or PCV13).
Overall, the immunogenicity profile of licensed paediatric vaccines (MMR+V, DTPa-IPV-HB-Hib, or PCV13) administered alone without MenQuadfi was comparable to that of the licensed paediatric vaccines administered concomitantly with MenQuadfi.
Immunogenicity of a single dose of MenQuadfi given alone, MenQuadfi given concomitantly with Tdap and HPV or Tdap and HPV given alone in meningococcal vaccine naïve adolescents 10 to 17 years of age - Study MET50.
A Phase II Study (MET50) was performed in meningococcal naive children and adolescents to evaluate the efficacy of MenQuadfi administered concomitantly with dTpa and HPV vaccines.
The antibody responses to MenQuadfi and to HPV, tetanus and diphtheria antigens were similar in both study groups. The anti-pertussis responses of the dTpa administered concomitantly with MenQuadfi and HPV versus dTpa administered concomitantly with HPV only were non-inferior for the PT antigen and did not meet non-inferiority for the FHA, PRN, FIM antigens. Vaccine response rates were robust and comparable across both groups. This trend is in line with the data available with the existing quadrivalent meningococcal conjugate vaccines. Because there are no established serological correlates of protection for pertussis, the clinical implications of the observed pertussis antigen responses are unknown.
Concomitant administration of MenQuadfi with MenB vaccine (Trumenba, N=90 or Bexsero, N=89) in adolescents and adults 13 through 26 years of age was evaluated in MET59. There was no suggestion of interference in MenQuadfi hSBA seroresponse rates when the vaccine was coadministered with MenB vaccine. The potential impact of MenQuadfi on MenB vaccine immune response was not assessed.
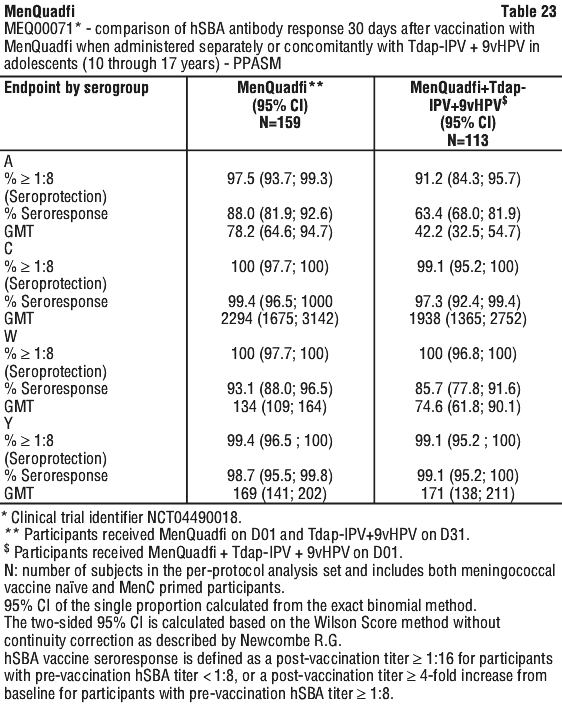
Immunogenicity of a single dose of MenQuadfi when given separately or concomitantly with Tdap-IPV and 9vHPV in meningococcal vaccine naïve or MenC primed (before two years of age) adolescents 10 to 17 years of age - Study MEQ00071.
MEQ00071 was performed to evaluate the antibody responses of MenQuadfi when given alone compared to that of MenACWY-TT and when MenQuadfi was given concomitantly with tetanus, diphtheria, acellular pertussis with inactivated poliomyelitis [Tdap-IPV] vaccine and human papillomavirus 9-valent [9vHPV] vaccine in participants 10 through 17 years of age.
Participants were randomised to receive one of the following study regimens: MenQuadfi alone (N=173), MenACWY-TT alone (N=173) or MenQuadfi + Tdap-IPV + 9vHPV (N = 117).
The post vaccination hSBA seroprotection rates for each serogroup were comparable in participants who received MenQuadfi alone or concomitantly with Tdap-IPV and 9vHPV. The post vaccination hSBA GMTs were higher for serogroups A and W in participants who received MenQuadfi alone compared to those who received MenQuadfi concomitantly with Tdap-IPV + 9vHPV and comparable for serogroups C and Y between study groups. The post vaccination hSBA seroresponse rates for serogroup A were higher in participants who received MenQuadfi alone compared to those who received MenQuadfi concomitantly with Tdap-IPV + 9vHPV and comparable for serogroups C, Y and W between study groups (see Table 23).
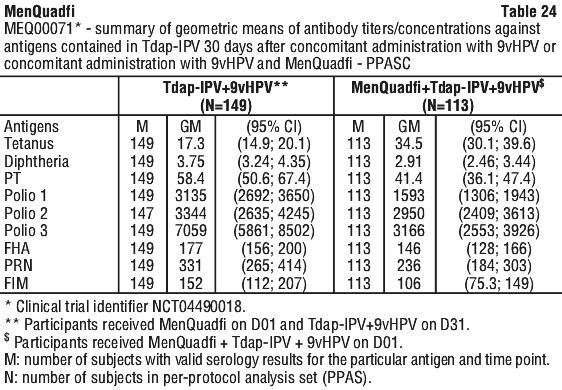
The anti-diphtheria, -polio type 2, -FHA and -FIM geometric means (GMs) were similar when Tdap-IPV + 9vHPV were given concomitantly with MenQuadfi or when Tdap-IPV + 9vHPV were given separately from MenQuadfi.
Anti-PT, -polio type 1 and 3 and -PRN GMs were lower when Tdap-IPV + 9vHPV were given concomitantly with MenQuadfi than when given separately from MenQuadfi. Higher GMs were observed to tetanus when Tdap-IPV + 9vHPV were given concomitantly with MenQuadfi than when Tdap-IPV + 9vHPV were given separately from MenQuadfi (see Table 24).
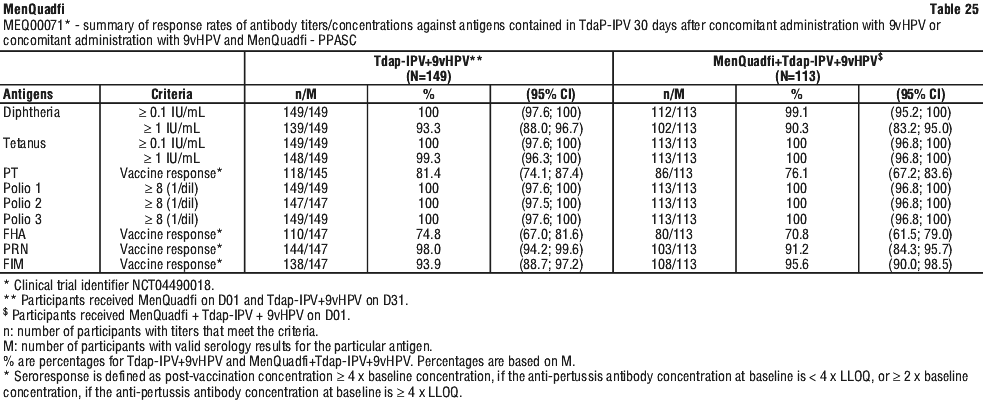
The response rates of antibody titers/concentrations against antigens contained in Tdap-IPV were comparable when given concomitantly with 9vHPV + MenQuadfi compared to when Tdap-IPV + 9vHPV were given separately from MenQuadfi (see Table 25).
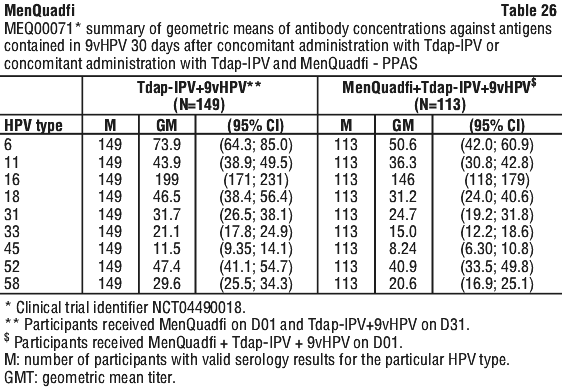
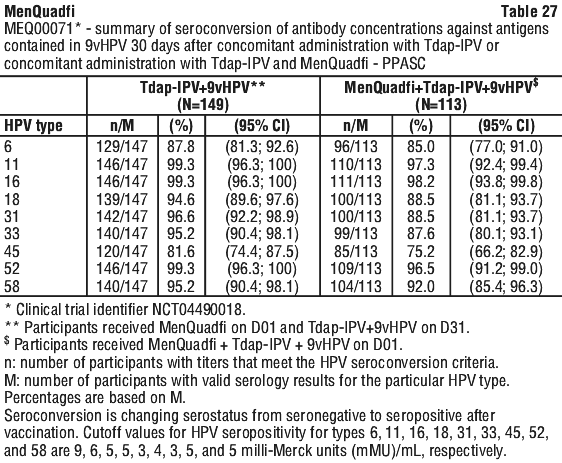
All anti-HPV antibodies, as measured by GMs, before and after vaccination were similar between study groups except anti-HPV type-6 and type-58, for which the GMs were lower in participants who received Tdap-IPV + 9vHPV concomitantly with MenQuadfi compared to those who received Tdap-IPV + 9vHPV separately from MenQuadfi. The proportions of participants who achieved seroconversion were comparable between both study groups (see Table 26 and Table 27).
Although antibody responses, when assessed by GMs, tended to be lower when MenQuadfi was given concomitantly with Tdap-IPV + 9vHPV compared to when Tdap-IPV + 9vHPV were given separately from MenQuadfi, no clinically relevant differences were observed.
5.2 Pharmacokinetic Properties
No pharmacokinetic studies have been performed.
5.3 Preclinical Safety Data
Genotoxicity.
MenQuadfi has not been evaluated for genotoxic potential.
Carcinogenicity.
MenQuadfi has not been evaluated for carcinogenic potential.6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, sodium acetate and water for injections.
6.2 Incompatibilities
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal products.
6.3 Shelf Life
48 months when stored at 2°C to 8°C.
Stability data indicate that the vaccine components are stable at temperatures up to 25°C for 72 hours. At the end of this period, MenQuadfi must be used or discarded. It must not be returned to storage. These data are intended to guide healthcare professionals in case of temporary temperature excursion only.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze).
6.5 Nature and Contents of Container
Pack of 1 or 10 single dose (0.5 mL) vials.
Vial stopper is not made with natural latex.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
After use, any remaining vaccine and container must be disposed of safely, according to locally acceptable procedures.
6.7 Physicochemical Properties
No data available.
7 Medicine Schedule (Poisons Standard)
S4 Prescription Only Medicine.
Summary Table of Changes









 MET57 was conducted in meningococcal vaccine naive toddlers 12 through 23 months of age to assess the immunogenicity and safety of concomitant administration of MenQuadfi with paediatric vaccines (MMR+V, DTPa-IPV-HB-Hib or PCV). Overall, the postvaccination hSBA seroprotection rates in participants who received MenQuadfi was high for all serogroups (between 88.9% and 100%), and GMTs were higher for serogroup C than for serogroups A, W and Y.
MET57 was conducted in meningococcal vaccine naive toddlers 12 through 23 months of age to assess the immunogenicity and safety of concomitant administration of MenQuadfi with paediatric vaccines (MMR+V, DTPa-IPV-HB-Hib or PCV). Overall, the postvaccination hSBA seroprotection rates in participants who received MenQuadfi was high for all serogroups (between 88.9% and 100%), and GMTs were higher for serogroup C than for serogroups A, W and Y.

 Study MET43 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-DT in meningococcal vaccine naïve participants 10 through 55 years of age.
Study MET43 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-DT in meningococcal vaccine naïve participants 10 through 55 years of age. MEQ00071 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-TT in participants 10 through 17 years of age.
MEQ00071 was performed to evaluate the efficacy of MenQuadfi compared to MenACWY-TT in participants 10 through 17 years of age.














